Abstract
Background
A restrictive red blood cell (RBC) transfusion guideline was established in 2019 in a neonatal intensive care unit (NICU), suggesting a hemoglobin concentration threshold of 7 g/dL.
Objectives
(a) To determine its impact on the number of RBC transfusions and donor exposure; (b) to characterize RBC-transfusion determinants and justifications.
Study Design
Single-center retrospective historical control study comparing all neonates consecutively admitted to the NICU during two five-month periods: 401 patients in 2013 before and 402 patients in 2021 after restrictive guideline implementation. Possible determinants were assessed via logistic regressions and justifications via a questionnaire.
Results
In 2021, 9.2% of neonates received at least one RBC transfusion compared to 13.5% in 2013 (p = .075). Adherence to protocol thresholds was 50%. Implementation of a restrictive transfusion protocol had some impact on determinants related to neonatal morbidity and illness severity and some impact on justifications being mainly based on hemoglobin value.
Conclusion
Our study demonstrates that the implementation of a restrictive RBC transfusion protocol tended to decrease transfusion rates and donor exposure in the NICU, but the trend was not statistically significant. Future work should focus on improving protocol adherence.
Introduction
Premature newborns rank among the most frequent recipients of transfusions, with as many as 90% of babies weighing less than 1,000 g needing at least one red blood cell (RBC) transfusion while in the Neonatal intensive care unit (NICU). 1 In 2013, a study carried out among the NICU patients at the Centre Hospitalier Universitaire (CHU) Sainte-Justine examined the patterns and influential factors related to RBC transfusions in critically ill newborns. This study revealed a significant disparity in transfusion approaches among neonatologists. 2 The potential misuse or abuse of blood transfusions, possibly stemming from unclear guidelines, may put at risk vulnerable newborns, exposing them to potential adverse events or reactions from transfusions, often without clear benefits.3, 4 Following these results and based on literature suggesting that RBC transfusions in neonates might be associated with worse outcomes for mortality and morbidity,5–8 the NICU of CHU Sainte-Justine established local restrictive RBC transfusion thresholds in 2019. Moreover, recent randomized controlled trials have backed this local approach, demonstrating that employing conservative blood transfusion strategies for newborns weighing less than 1,000 g, as opposed to more liberal thresholds, did not elevate the risk of death or neurodevelopmental issues after approximately two years of corrected age. 9
The primary objective of the study was to determine the impact of the implementation of a local restrictive hemoglobin thresholds guideline on the number of RBC transfusions and donor exposure in the NICU of CHU Sainte-Justine. The secondary objectives were comparing the determinants and justifications behind RBC transfusions in neonatology before and after the protocol was introduced. We hypothesized that implementing these guidelines would lead to a reduction in RBC transfusions among neonates admitted to the NICU.
Methods
Design
Every patient admitted to the CHU Sainte-Justine NICU between January 18 and June 22, 2021, was included in this historical control study, with the sole exclusion criterion being the use of extracorporeal membrane oxygenation. The comparison cohort was gathered over a five-month duration in 2013 from the same NICU. Information for both the pre- and post-intervention groups was gathered prospectively. Approval for this study was granted by the Research Ethics Board of the Sainte-Justine Hospital, which waived the need for consent.
Setting
The NICU of CHU Sainte-Justine in Montreal, Canada, is a tertiary care (level 3B) and serves as a tertiary care (level 3B) university-affiliated center dedicated to mother-child health. It admits around 1,000 patients annually with a capacity of 65 beds.
RBC Transfusions and Local Practice
Attending clinicians hold the authority to make decisions concerning blood product transfusions. All blood products, sourced from the Héma-Québec blood bank, underwent leukoreduction via pre-storage filtration. Neonates weighing ≤1,200 g received gamma-irradiated products. Further processing, such as washing, was determined by the medical team’s discretion. The recommended volume for RBC transfusion stood at 15 mL/kg.
Generation and Application of the Protocol
In 2019, a team of neonatologists and providers in neonatal care collaborated to develop the local transfusion protocol. This protocol was formulated by drawing upon existing literature and consolidating the consensus among local experts in the field. Prior to 2019, there were no local RBC transfusion guidelines. The local RBC transfusion thresholds implemented in April 2019 were:
1 week post-natal (days 1–7): hemoglobin threshold = 100 g/L. 2 weeks post-natal (days 8–14): hemoglobin threshold = 85 g/L. ≥3 weeks post-natal (≥15 days): hemoglobin threshold = 75 g/L.
The hospital’s neonatology division ensured the widespread distribution of the protocol. It was shared with all team members, including fellows and nurses, not just once but repeatedly, particularly for new staff members. Additionally, the guidelines were accessible on the hospital’s secure protocols website for easy reference. Presentations were given to the department describing the new guidelines.
We considered that compliance with the protocol was adequate if the RBC transfusion was prescribed according to the protocol thresholds. The compliance rate was determined by dividing the number of RBC transfusions administered within the recommended protocol threshold by the total number of RBC transfusions given.
Data Collection
Data abstraction and entry were conducted using a validated case report form. Baseline patient characteristics were gathered at admission to the NICU, marked as time zero for each individual. For patients who received transfusions, daily monitoring of potential factors influencing RBC transfusions began at admission (time zero) and concluded upon their first transfusion. For those who didn’t receive transfusions, daily monitoring ceased at either patient discharge, death, or when they reached 44 weeks of corrected age, whichever came first. Upon admission, validated scores, including the Clinical Risk Index for Babies (CRIB)-II score, were recorded to assess the severity of illness in NICU patients 10 and the Score for Neonatal Acute Physiology Perinatal Extension (SNAPPE)-II. 11
Determinants of RBC Transfusions
In the 2013 prospective cohort study, a compilation of potential factors influencing RBC transfusions in neonates was utilized. This comprehensive list originated from a review of literature and insights from local experts in transfusion medicine and neonatology. It encompasses patients’ baseline characteristics along with information collected from their admission day and throughout their stay. The same list was used in 2021 to allow for a direct comparison of RBC transfusion determinants among both cohorts.
Justifications of RBC Transfusions
Within 48 hours of each initial RBC transfusion, the justifications behind these transfusions were documented using an online validated questionnaire developed based on consensus among local experts (refer to supplemental information). This standardized questionnaire, identical in both 2013 and 2021 studies, facilitated a direct comparison. The clinician responsible for requesting the transfusion could select one or multiple justifications from a predefined list and arrange them based on their relevance.
Statistical Analysis
Descriptive statistics included the number and proportion of patients (%) and mean ± standard deviation (SD). To compare the 2013 and 2021 cohorts, the Fisher exact test evaluated categorical variables, while the Student t-test examined continuous variables. For potential determinants distinguishing transfused from non-transfused patients, univariate analysis was employed. Logistic regression and odds ratios with a 95% confidence interval were utilized. Continuous variables were stratified into categories based on clinically significant thresholds or quartiles for analysis. All statistical assessments were conducted using SPSS statistical software version 26, with a significance level set at 5%.
Results
Baseline Characteristics
A total of 402 infants consecutively admitted to the NICU were included during the five-month study period. None were excluded. Neonates in this cohort were born with a mean gestational age (GA) ± SD of 34.9 ± 4.2 weeks and birth weight (BW) of 2.5 ± 1.0 kg. Table 1 summarizes the demographic data of the 2013 and 2021 cohorts. Both cohorts shared similarities, yet notable statistical differences emerged. Specifically, these variances were observed in the mean BW ± SD (2.3 ± 1.0 kg in 2013 compared to 2.5 ± 1.0 kg in 2021; p = .039) and the proportion of bacterial infection as the admission diagnosis (74/401 (18.5%) in 2013 vs. 41/402 (11.0%) in 2021; p < .001). BW was deemed clinically similar between cohorts and the statistical difference was attributed to the sample size. In Table 2, RBC-transfused patients from both cohorts were compared. These cohorts had statistically significant differences in the proportions of admission diagnoses of respiratory disease (44/54 (81.5%) in 2013 vs. 20/37 (52.1%) in 2021; p = .006) and bacterial infection (20/54 (37.0%) in 2013 vs. 4/37 (10.8%) in 2021; p = .007).
Baseline Characteristics of Cohorts in 2013 and 2021.
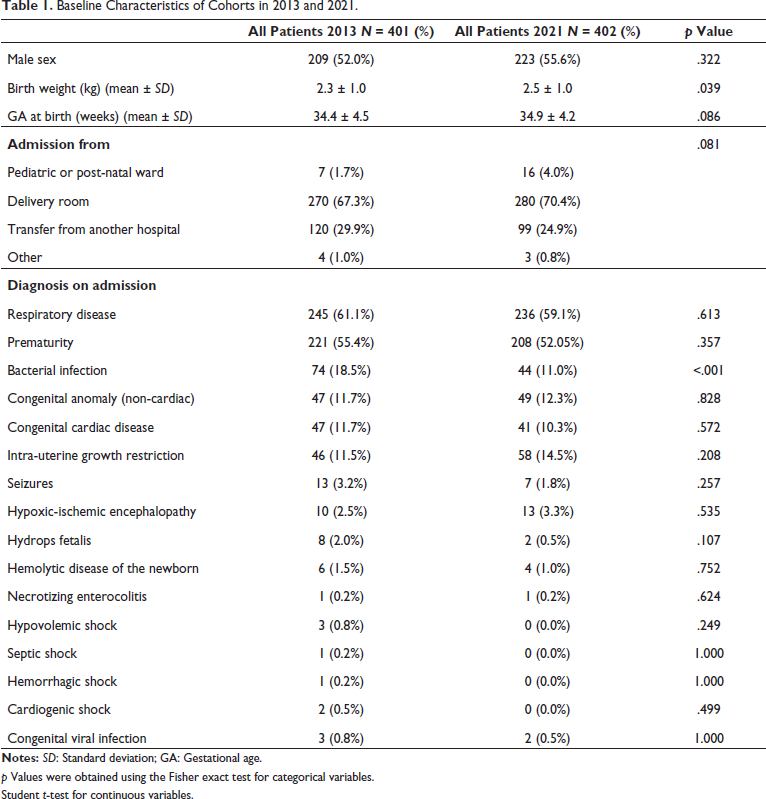
p Values were obtained using the Fisher exact test for categorical variables.
Student t-test for continuous variables.
Baseline Characteristics of pRBC-transfused Neonates in 2013 Versus 2021.
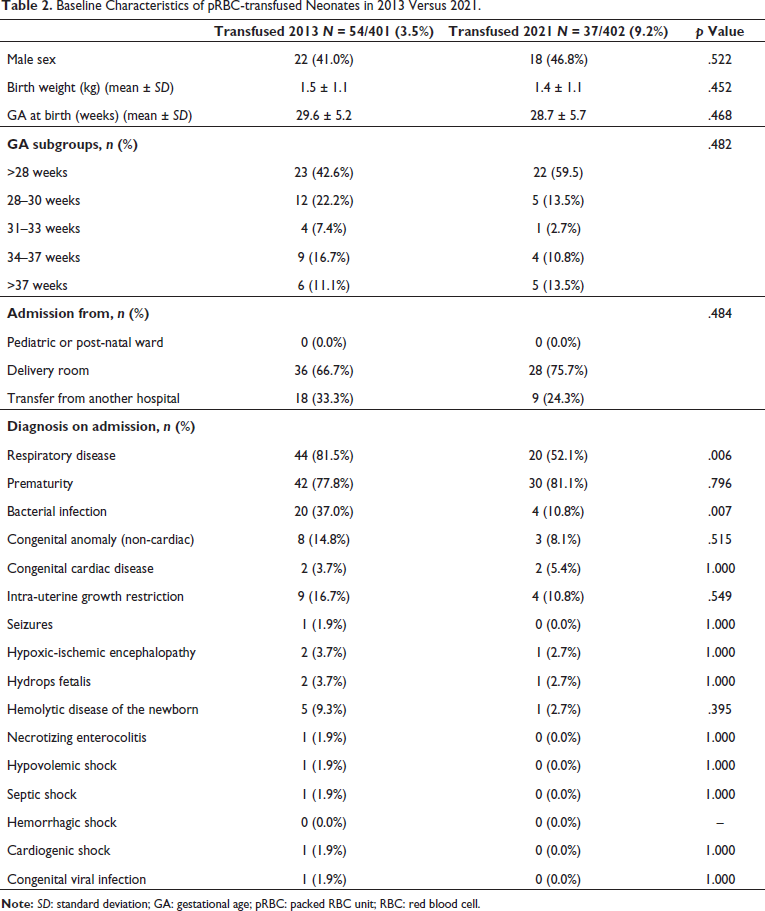
RBC Transfusion Data
Table 3 presents a comparison of transfusion data from the two cohorts. In 2021, at least one RBC transfusion was given to 37/402 patients (9.2%) during their stay in 2021, while in 2013, 54/402 patients (13.5%) (p = .075)received an RBC transfusion. Among transfused patients, 16/402 (43.2%) received a single transfusion in 2021 compared to 15/401 (27.8%) in 2013 (p = .079). 2/402 (5.4%) transfused patients received 5 or more RBC transfusions in 2021 compared to 16/401 (29.6%) in 2013 (p = .079). 20/402 transfused patients (54.1%) were exposed to a single donor in 2021 compared to 38/401 (70.4%) in the comparison group (p = .412).
Comparison in pRBC Transfusions Between the 2013 and 2021 Cohorts.
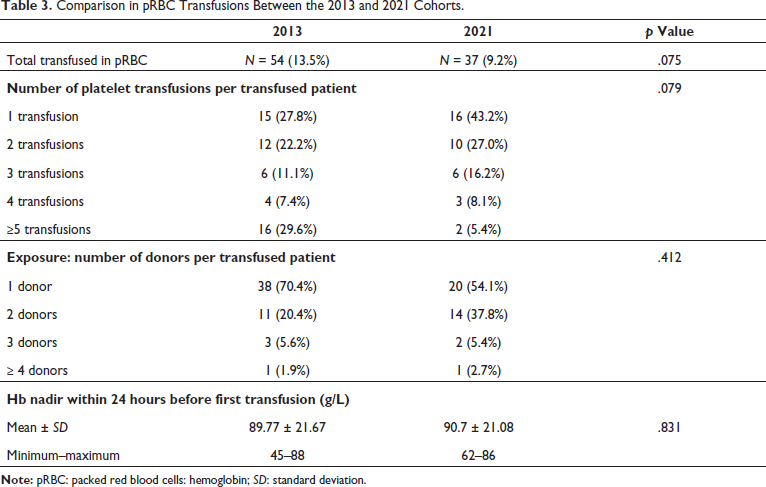
The mean hemoglobin nadir within 24 hours before the first transfusion was similar between the two cohorts: 91.42 ± 18.21 g/L in 2013 versus 90.7 ± 21.1 g/L in 2021 (p = .878). The after-protocol cohort shares similar RBC transfusion rates and characteristics compared to the 2013 transfusion protocol cohort (Table 3).
Protocol Adhesion
The proportion of RBC transfusions given in concordance with the established protocol was 50% in the 2021 cohort.
Determinants of RBC Transfusions
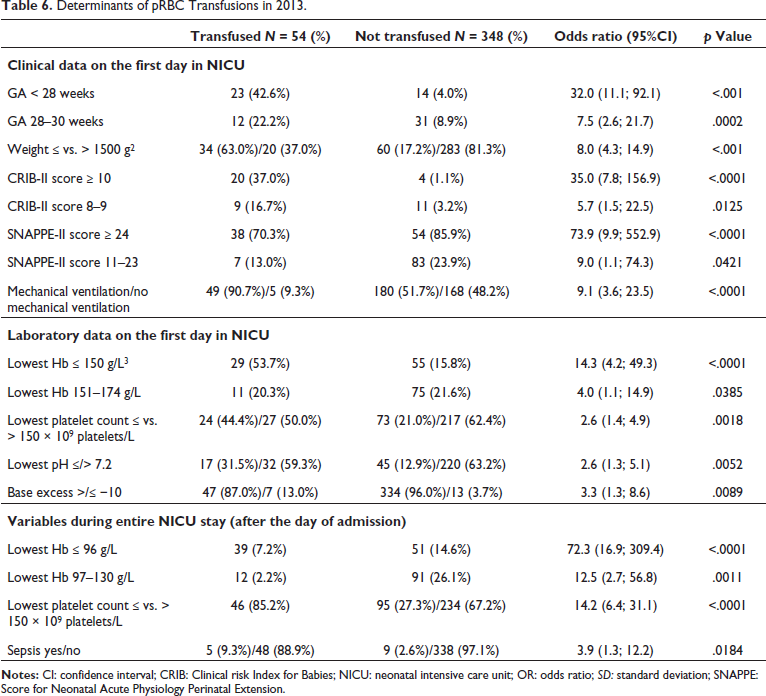
Table 4 presents the determinants of RBC transfusions in the 2021 cohort. Characteristics that predicted RBC transfusions in the 2021 cohort are listed in Table 5. Determinants of RBC transfusions in 2013 are listed in Table 6.
Possible Determinants of pRBC Transfusions in 2021.
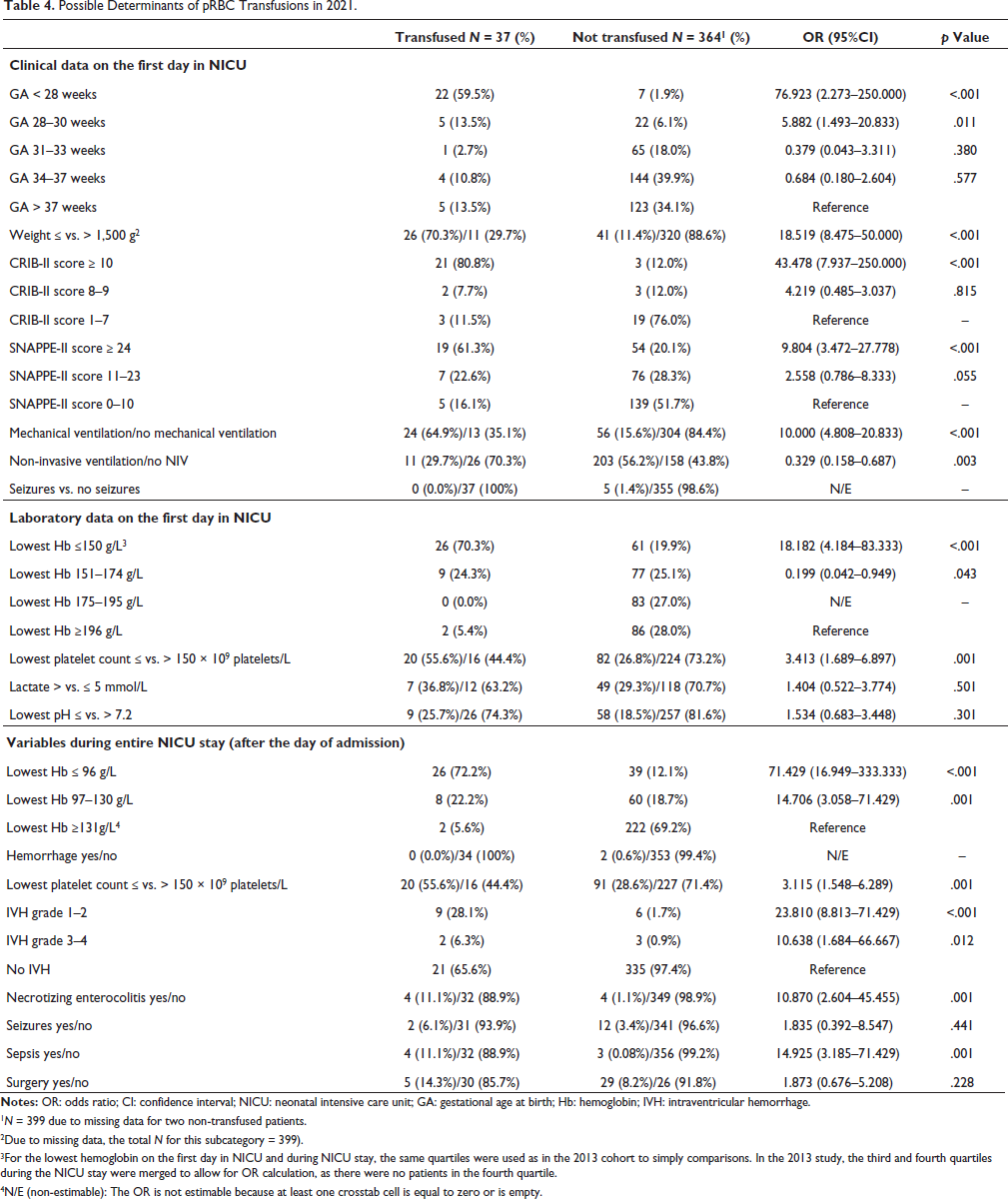
1N = 399 due to missing data for two non-transfused patients.
2Due to missing data, the total N for this subcategory = 399).
3For the lowest hemoglobin on the first day in NICU and during NICU stay, the same quartiles were used as in the 2013 cohort to simply comparisons. In the 2013 study, the third and fourth quartiles during the NICU stay were merged to allow for OR calculation, as there were no patients in the fourth quartile.
4N/E (non-estimable): The OR is not estimable because at least one crosstab cell is equal to zero or is empty.
Determinants of pRBC Transfusions in 2021.
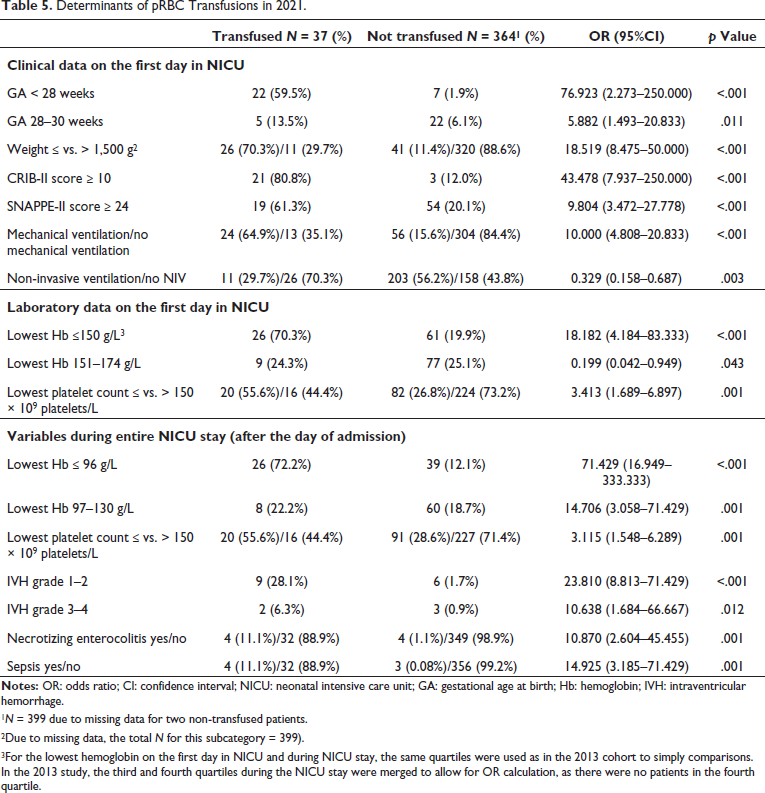
1N = 399 due to missing data for two non-transfused patients.
2Due to missing data, the total N for this subcategory = 399).
3For the lowest hemoglobin on the first day in NICU and during NICU stay, the same quartiles were used as in the 2013 cohort to simply comparisons. In the 2013 study, the third and fourth quartiles during the NICU stay were merged to allow for OR calculation, as there were no patients in the fourth quartile.
Determinants of pRBC Transfusions in 2013.
Justifications for the First pRBC Transfusion in NICU Patients Before (2013) and After (2021) the Implementation of a Restrictive Transfusion Protocol.
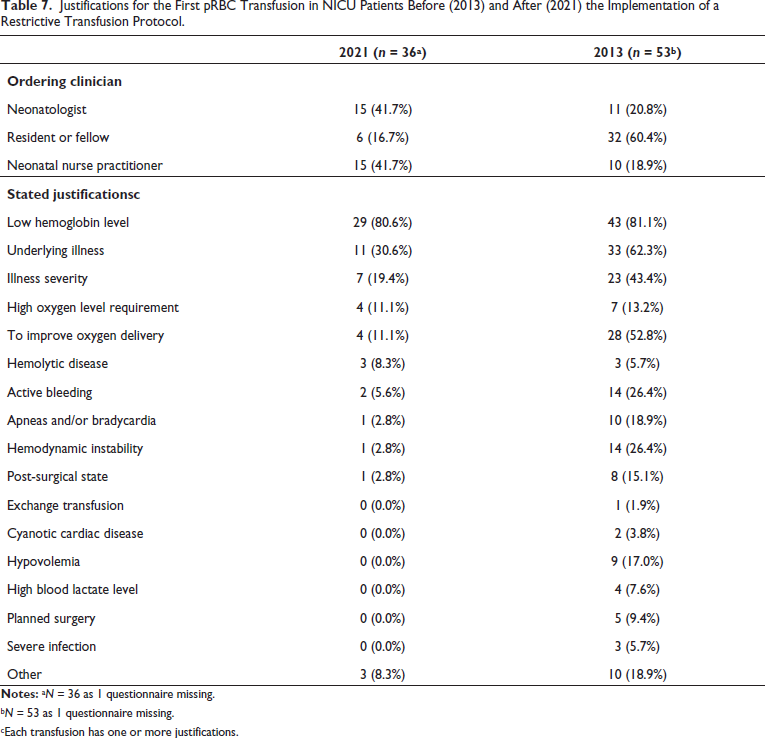
bN = 53 as 1 questionnaire missing.
cEach transfusion has one or more justifications.
Justifications of RBC Transfusions
Table 7 presents the primary reasons for requesting the first RBC transfusion within the 2021 cohort. The most commonly cited justifications included low hemoglobin levels (80.6%), underlying illness (30.6%), and the severity of illness (19.4%). The most frequent underlying illness of transfused patients was “extreme prematurity.” Suspicion of hemolytic disease, sepsis, and hypoxic-ischemic encephalopathy were the other illnesses that justified ordering transfusions. Among the 54 transfusions within the 2013 cohort, the primary reasons cited for prescribing transfusions were a low concentration of hemoglobin (81.1%), perceived underlying severity of illness (62.3%), and the goal to enhance oxygen delivery (52.3%). The stated justifications to prescribe a transfusion even though the hemoglobin counts were above hemoglobin thresholds in the protocol were: low hemoglobin (n = 12), underlying disease (hemolytic anemia and twin-twin transfusion syndrome) (n = 2), disease severity (n = 1), hemodynamic instability (n = 1), apnea and/or bradycardia (n = 1), post-operative state (n = 1) and removal of veinous umbilical cord catheter (n = 1).
Discussion
In this single-center study, a prospective historical control design was employed to compare two consecutive groups of neonates admitted to the same NICU over distinct five-month periods. The comparison was between the period preceding and the period following the implementation of a restrictive RBC transfusion protocol. The study demonstrated that the implementation of restrictive RBC transfusion thresholds did not significantly decrease transfusion rates, nor did it find a significant decrease in transfusions and donor exposure per transfused patient. However, we observed a low protocol adherence rate of 50% which limits the potential impact of the protocol. The determinants of RBC transfusion are similar between the two cohorts. The introduction of a more restrictive transfusion protocol influenced the reasons given by clinicians for administering RBC transfusions.
Our study reports similar transfusion rates and Hb nadir values before the first transfusion between the two cohorts. Both cohorts share comparable baseline characteristics. An exception was a significantly lower bacterial infection rate as a diagnosis of admission in the 2021 cohort compared to the 2013 cohort. The transfusion rates could be influenced by the association between sepsis and RBC transfusions in neonates. 12 There is scant data regarding transfusion practices in the NICU involving neonates of all GA. However, a cohort study conducted in our NICU in 2013 determined that the prevalence of transfusions in neonates of GA < 30 weeks was 43.7%. 2 In a study conducted in Canada, the reported rate of RBC transfusions in this specific patient population was 56%. 13 Our lower transfusion rate suggests that our initial practice was already quite restrictive. Several studies have contrasted RBC transfusion methods and outcomes in NICUs, examining the impacts of restrictive versus liberal transfusion protocols. One such study demonstrated that adopting a more conservative protocol resulted in a decrease in RBC transfusions among preterm neonates. The prevalence of these infants receiving a blood transfusion dropped from 37.5% to 32.7% (p = .040). 14 The absence of statistical significance between the two cohorts of our study could also be explained by a limited sample size. Additionally, patient protocol compliance was 50%, lower than in similar studies. The TOP trial reports an adherence rate of 77% in its low hemoglobin threshold cohort 15 while the ETTNO trial reports an adherence rate of 81% to the restrictive protocol. 9 Among the 18 transfusions prescribed outside the protocol’s recommendation in our study, most clinicians exerted clinical judgment while aware of their deviation from the guidelines. Six were given due to the velocity of decrease of hemoglobin and two were given with a declining hemoglobin value superior to the threshold to benefit from the peripherally inserted central catheter line before its removal the following day. Four transfusions were given based on hemoglobin values obtained from arterial blood gas. Choosing not to add a complete blood count to laboratory studies to determine the value of hemoglobin contributes to reducing iatrogenic blood loss. A study found that the combined iatrogenic blood losses throughout the hospital stay for very low BW neonates were a key predictor for requiring RBC transfusions. 16 Limiting blood sampling should contribute to reducing RBC transfusions. One way to decrease the number of blood samples would be to use hemoglobin measurement not only done in the hematology laboratory but also on blood gases, which is considered reliable by some authors. 17 In our study, if we add hemoglobin measured in the hematology laboratory and on blood gas analyses, our protocol adherence rate would be 66%. Protocol deviations could be attributed to factors like unawareness of the protocol’s existence or its location, potentially influenced by the frequent rotation of residents and fellows within the NICU. The use of computer programs to order transfusions in the pediatric population has proven to increase adherence rates. A study conducted in the pediatric NICU found that employing an electronic order entry system for RBC transfusion enhanced protocol adherence. 18 A multicenter study demonstrated that introducing an electronic transfusion ordering and monitoring system in the NICU increased compliance to transfusion guidelines from 65% to 90%. 19
The two cohorts shared similar determinants of RBC transfusion. Lowest blood pH ≤7.2 and base excess level <−10 were no longer determinants in 2021. This shift could translate the prescription of RBC transfusion based more on the hemoglobin concentration rather than on perfusion and oxygenation markers. In 2021, both the CRIB-II and SNAPPE-II scores on admission showed a tendency toward increased RBC transfusions with higher scores. A low hemoglobin nadir value was associated with an increase in transfusion risk in both cohorts but was significant at a lower value in 2021 than in 2013. These findings could indicate a tendency toward more restrictive transfusion practices as neonates with higher scores of predicted mortality and lower hemoglobin values had an increased transfusion risk compared to the transfusion practice before the implementation of the protocol. IVH was not a determinant of RBC transfusion in 2013 while neonates under the restrictive protocol in 2021 suffering from IVH were more likely to receive a transfusion. With the velocity of hemoglobin decrease being an important consideration in the decision to transfuse a neonate, a low hemoglobin value could be anticipated sooner in patients suffering from IVH, thus, increasing the risk of RBC transfusion. In 2021, patients suffering from necrotizing enterocolitis (NEC) had a higher probability of receiving a transfusion. In 2013, NEC could not be analyzed as a potential determinant since there were only six cases of NEC in the cohort and all of them were transfused. A study conducted among VLBW infants determined that the likelihood of receiving one or more transfusions increased in infants suffering from NEC. 20 In a prospective cohort study involving preterm VLBW infants in Brazil, a multiple logistic regression model identified several factors significantly linked to RBC transfusions. These factors were GA, SNAPPE-II score, occurrences of apnea and pulmonary hemorrhage, oxygen requirement at 28 days of life, instances of clinical sepsis and NEC, severe IVH, mechanical ventilation, use of an umbilical catheter, reliance on parenteral nutrition, and hospitalization for more than 60 days. 12 In a Brazilian study, NEC among VLBW infants, GA and SNAP score, 3 lower initial hemoglobin concentration, male sex, and low BW 20 were reported as determinants for the need for RBC transfusion. The duration of mechanical ventilation used as an indicator of illness severity, was highlighted as an independent factor linked to a higher probability of requiring a transfusion. 20 Our study’s determinants of RBC transfusions are similar to the results reported in the literature.
The two most common justifications stated by clinicians for RBC transfusions (hemoglobin value and underlying disease) were identical between the two cohorts. The implementation of the restrictive protocol shifted clinical practices to have hemoglobin values as the main consideration when transfusing NICU patients compared to oxygenation and perfusion markers as well as hemodynamic state being important justifications in 2013. To our knowledge, no other study has taken interest in the clinician-stated-justifications of RBC transfusions in the NICU population.
This study’s strengths lie in its prospective design, which reduces the potential for information bias, and its approach of universal inclusion of consecutively admitted patients, limiting selection bias and improving overall generalizability. The study took place in the same NICU in similar timeframes, employing identical case report forms and questionnaires, ensuring high comparability between settings and cohorts. Additionally, the NICU represents a typical North American multidisciplinary unit, caring for both preterm and term surgical and medical patients, thus increasing its generalizability.
The study’s external validity was constrained due to its single-center design. Physicians were conscious that their transfusion practices were under observation during the research, which could have impacted their approach. Additionally, the reasons provided for transfusions might have been influenced by the format of the multiple-choice questionnaire. Protocol adhesion was mediocre, limiting the potential impact of the implementation of the restrictive protocol. Although the study was conducted in the same NICU, potential changes in practices during the eight-year gap between the two cohorts such as the routine use of delayed cord clamping which has not been accounted in our variables. Furthermore, the difference in patients admitted with a diagnosis of sepsis between both cohorts indicates a possible confounding variable.
Conclusion
Our study demonstrates that the implementation of a restrictive RBC transfusion protocol did decrease transfusion rates and donor exposure in the NICU, but the trend was not statistically significant. The implemented thresholds modestly impacted the transfusion determinants and justifications. Nonetheless, the discrepancies between both cohorts demonstrate a shift in transfusion practices based more importantly in Hb values than perfusion and oxygenation as well as RBC transfusions being more likely to be given to sicker neonates with higher predicted mortality scores and lower hemoglobin values. The interpretation of our results needs to consider the poor protocol adherence rate which limits the potential impact of the protocol. Future efforts should focus on additional education to reinforce guideline adherence through presentations, workshops, clinical audits, and electronic transfusion ordering systems to contribute to optimizing blood product use in a vulnerable population.
Footnotes
Acknowledgments
We thank Karine Fondrouge for her help in data collection, Miguel Chagnon and Justine Zehr for statistical analysis as well as the patients and their families for contributing to this study.
Author Contributions
AZ contributed to the study design, data analysis, data interpretation and drafted the manuscript. LC contributed to data acquisition and drafted the manuscript. JL contributed to the study conception and design. AV contributed to study conception, design, data analysis, and data interpretation. All authors critically reviewed the manuscript for intellectual context and agreed to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the CHU Sainte-Justine Ethics Committee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was supported by a bursary from the Centre d’excellence en soins intensifs du nouveau-né de la Fondation du CHU Sainte-Justine as well as the PREMIER program from the Université de Montréal.
Informed Consent
Not applicable
