Abstract
Background
Neonatal jaundice affects 85% term and most of the preterm infants. Unconjugated bilirubin is bound by albumin, which promotes its transit and lessens bilirubin toxicity, thereby serves as early determinant for the risk of pathological hyperbilirubinemia among neonates. Present study was undertaken to know the utility of cord blood bilirubin (CBB), cord blood albumin (CBA), and their ratio at time of birth for the development of significant neonatal hyperbilirubinemia.
Objectives
To evaluate CBB/CBA ratio at the time of delivery as potential predictive factor for the development of significant neonatal hyperbilirubinemia and compare this with CBB and CBA alone.
Study Design
Prospective observational analytical study.
Participants
Total 178 inborn term neonates delivered during 3-month time period (22nd October, 2022–22nd January, 2023).
Interventions
Cord blood sample was collected at birth for CBA, CBB, and blood grouping/phenotyping. Transcutaneous bilirubin (TCB) measurements were done 12 hourly in all babies for the first 7 days of life/till discharge, and also in the case of development of significant clinical jaundice. For TCB values above cut-off, serum total bilirubin was done and need for phototherapy (PT) decided based on cut offs suggested in AAP 2004 nomograms.
Results
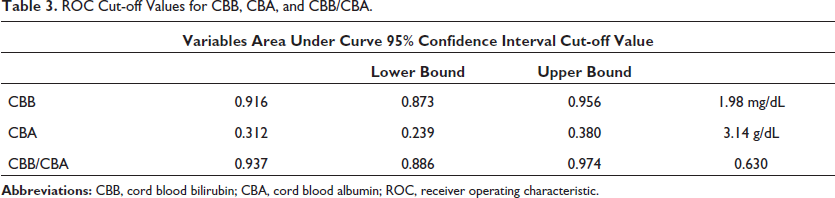
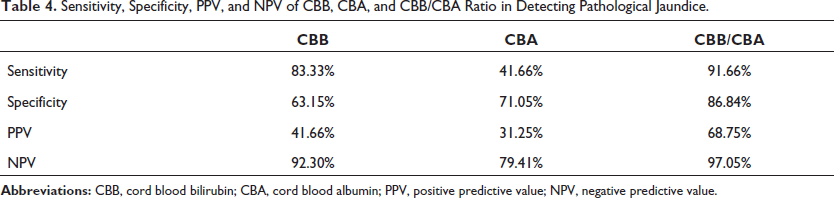
Cut-off values of CBB and CBA as obtained by the receiver operating characteristic (ROC) curves were 1.98 mg/dL and 3.14 g/dL, respectively. Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) for CBB were 83.33%, 63.15%, 41.66%, and 92.30%, while for CBA, 41.66%, 71.05%, 31.25%, and 79.41%. CBB/CBA cutoff ratio was 0.630 with 91.66% sensitivity, 86.84% specificity, 68.75% PPV, and 97.05% NPV.
Conclusions
Compared to CBB and CBA alone, CBB/CBA ratio has greater sensitivity, specificity, PPV, and NPV and it may serve as a better indicator for prediction of pathological hyperbilirubinemia in neonates.
Introduction
The most frequent issue all through the first week of life is neonatal jaundice. 1 Jaundice affects around two-thirds of newborns. 2 Most preterm newborns and over 85% of term infants are affected. 3 Up to 78% of neonatal hospital admissions during the first week of life are caused by it. 4 In neonatal period, it is also a frequent reason for re-admission of the infant, 5 which places a financial and socioeconomic hardship on families in developing and impoverished countries.
Neonatal hyperbilirubinemia either has a self-limiting course or is easily treated with phototherapy (PT) when it is discovered early in the course of the illness. This prevents the development of its most terrifying consequence, chronic bilirubin encephalopathy (Kernicterus), which is linked to high morbidity and long-term neurological abnormalities like dyskinetic cerebral palsy. 6 Additionally, neonatal jaundice is a significant clinical trait because it may indicate the presence of an underlying illness, such as hemolytic anemia, an infection, an inborn metabolic mistake, or liver disease. 7
The American Academy of Pediatrics (AAP) recommended that infants who are discharged within 48 h should have a follow-up appointment within 2–3 days in order to detect serious jaundice and other related issues. The guidelines of AAP-2004 stated that infants discharged before 24 h of age should be seen at 72 h of age, discharged between 24 and 48 h of age at 96 h of age, and those discharged between 48 and 72 h of age should be seen at the 5th day of life. 6
Because there are few community follow-up resources in underdeveloped nations, this advice cannot be implemented. If the neonates who are at risk of hyperbilirubinemia are recognized at birth, then a suitable care strategy can be suggested for follow-up. The first step in preventing neonatal hyperbilirubinemia and a step toward shielding neonates from difficulties later in life is the early identification of risk factors. Additionally, PT for jaundice is a quick, easy, and affordable treatment option.
The two most critical indicators of neonatal hyperbilirubinemia are cord blood albumin (CBA) and cord blood bilirubin (CBB). The liver is where albumin is synthesized. 7 Albumin competes with tissues for bilirubin by binding to unconjugated bilirubin and aiding in its transit, and hence lowering bilirubin toxicity in the tissues. 8 As a result, it plays an important role in determining development of neonatal hyperbilirubinemia. Along with total serum bilirubin level, the AAP advised using ratio of total serum bilirubin and albumin (TSB/A) in the treatment of newborn jaundice. This study evaluated CBB with CBA and their ratio at delivery to predict the possibility of substantial newborn hyperbilirubinemia.
Aims and Objectives
To evaluate the ratio of CBB to albumin at delivery as a potential predictive factor for the development of significant neonatal hyperbilirubinemia.
To compare predictive accuracy of CBB/CBA ratio with that of CBB and CBA alone.
Methods
The present prospective observational analytical study was carried out from 22nd October 2022 to 22nd January 2023 in Neonatal Intensive Care Unit, Department of Pediatrics, Narayana Medical College, Nellore, Andhra Pradesh following approval by the Institutional Ethics Committee on 22nd October 2022 (Ref. No. IEC/NMC/22.10.22_1). The sample size was 178, which obtained by convenience sampling method. Subjects were recruited from amongst the inborn healthy term neonates irrespective of gender and delivery mode after taking informed as well as written consent from the parents.
Inclusion Criteria
Inborn term neonates (>37 completed weeks of gestation) of either sex.
Birth weight >2500 g.
Exclusion Criteria
Parents who are unwilling to take part in the study.
Neonates with congenital anomalies.
ABO/Rh incompatibility.
Neonates with significant illness such as neonatal sepsis, RDS, MAS, birth asphyxia, or hemodynamic instability.
Parents of subjects who meet the requirements for eligibility were recruited to participate in the study and provided with information about it. The newborn was enrolled after receiving written informed consent from a parent. The predesigned Performa had a complete history, including prenatal and perinatal information.
Two samples of cord blood—one in an EDTA tube (0.5 ml) and the other in a plain tube (2 ml) were taken under strict aseptic conditions after the baby was delivered. Plain sample tube was sent to biochemistry facility to estimate serum albumin using a semi-automated analyzer and serum bilirubin using the diazo technique and a standard photospectrometric bilirubin analyzer. The hematology lab received an EDTA sample tube to determine the ABO-Rh blood type. The modified New Ballard scoring method was used to determine the newborn’s gestational age.
For infants delivered via LSCS, transcutaneous bilirubin (TCB) measurements (using DRAGER JM105) were taken up to 7 days of life, every 12 h. For infants delivered vaginally and discharged earlier, the measurements were taken every 12 h until they were discharged, and later also on follow-up if they had significant neonatal jaundice. Follow-up with babies born vaginally was done within 4 to 5 days of discharge. Any infant whose TCB value was higher than the threshold for PT had serum total bilirubin performed, and the need for PT was determined using hour-specific percentile nomograms developed by the AAP. If PT was required, the newborn was then shifted to the NICU.
Measurement
Transcutaneous bilirubinometer was used to measure 12th hourly bilirubin levels. It works by directing light into the skin of neonates and measuring the intensity of a specific wavelength. New generation devices have built-in algorithms to account for skin pigmentation and dermal thickness. Preferred sites of measurement are the forehead, sternum, and thigh. Three serial measurements were taken, and the average of the three values was taken as the final. Measurement against bruises, subcutaneous hematomas, hairy areas, and birthmarks was avoided. Results are presented in units of mg/dl or µmol/L that are clinically suitable. The advantage of using a transcutaneous bilirubinometer is that it is non-invasive and it reduces the need for blood sampling by 30%. 9
Laboratory Measurements
Blood samples were taken through venipuncture from infants whose TCB readings were higher than the cut-off as per hour-specific nomograms. The amount of bilirubin was calculated using a photo spectrometric bilirubin analyzer (BiliMicro Meter, Kohsoku Denki Co Ltd, Tokyo, Japan). 10 The blood sample was centrifuged in a microcapillary tube at 12,000 rpm for five minutes to separate the serum. In order to make sure that the serum column stretched, the entire length of the slit through which the light flows, the processed micro-capillary tube was secured to the holder of the spectrometric bilirubin analyzer. The total bilirubin value is shown digitally as a result of a microprocessor converting the photodetector’s measured light intensity into a number. This simple bilirubin measurement technique requires only 50–70 µL of blood and does not require any special equipment.
Statistical Analysis
Data were entered into a Performa spreadsheet that had been previously created, appropriate statistical analysis was performed, and percentile curves were produced for the observed values. For the purpose of predicting neonatal hyperbilirubinemia, a Receiver Operative Characteristic (ROC) curve was made using the statistical software (SPSS version 26) for CBB, CBA, and CBB/CBA values. Each was given sensitivity, specificity, and positive and negative predictive values (PPV and NPV), respectively. The ROC curves for CBB, CBA, and their ratio CBB/CBA were generated. An ideal cut-off point was chosen through identifying area on the ROC Curve that is most closely associated with the left upper corner of the curve, where the predicted benefit is greatest.
Results
Among the neonates delivered during the study period, 254 babies met the eligibility criteria. Of them, 76 babies were excluded. Out of excluded babies, 49 neonates were admitted to NICU, one neonate had ABO/Rh incompatibility, 12 had ABO setting, and four neonates were born with congenital anomalies. Ten babies were excluded in view of the denial of consent (Figure 1).
Flow of Participants.
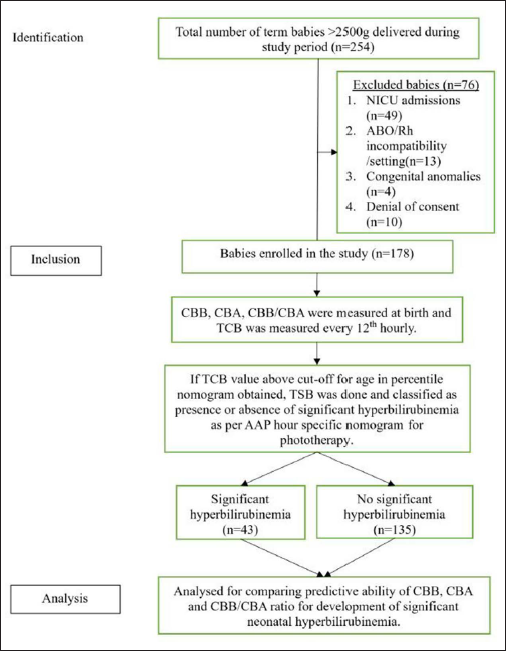
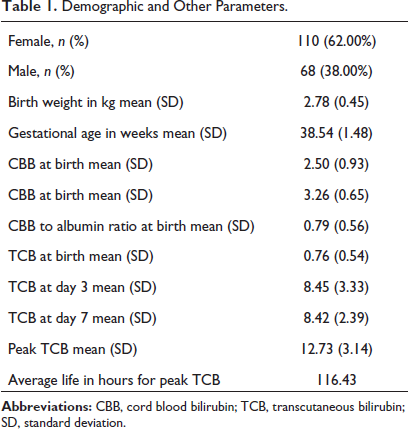
Finally, 178 neonates were included in the study. Of those, 43 neonates had significant jaundice (values above the cut-off in AAP percentile nomograms) and 135 neonates did not develop significant jaundice. Data from all 178 participants were collected and analyzed, and no subjects were missed for follow-up. The study included 178 newborns, 110 (62%) of whom were female, and 68 (38%) were male. The neonates in our research had a birth weight ranging from 2.55 to 4.15 kg and the average birth weight was 2.78 (0.45) kg. Additional characteristics are shown in Table 1 for your reference. Exchange transfusion was not required for any of the neonates with pathological hyperbilirubinemia. The specified guidelines were followed while using PT. Out of the 178, 43 (24%) had a significant hyperbilirubinemia. Using a cut-off of 2.50 mg/dL, we split the population into two groups depending on the levels of CBB (as seen in prior study). 11 Of 178 newborns, 86 (48.31%) had readings ≥2.50 mg/dL, and 92 (51.68%) had levels that were less than that. Of the 43 newborns with pathological hyperbilirubinemia, only 7 (16.28%) had CBB levels under 2.50 mg/dL, whereas the other 36 (83.72%) had CBB levels above or equal to this threshold. The CBB levels of 85 (63.15%) of the 135 (76%) newborns without pathological hyperbilirubinemia were below 2.50 mg/dL, whereas the remaining 50 (36.85%) had levels higher than that.
Demographic and Other Parameters.
Based on the levels of CBA, the newborns were subsequently separated into three groups. CBA was divided into three categories: ≤2.99 g/dL, 3 to 3.29 g/dL, and ≥3.30 g/dL. 3 Of the 178 neonates, 53 (29.80%) had levels ≥3.30 g/dL, 68 (38.20%) had values between 3 and 3.29 g/dL, and 57 (32%) had levels ≤2.99 g/dL. 10
The CBA values for the 43 newborns with pathological hyperbilirubinemia were as follows: 18 (41.66%) had values ≤2.99 g/dL, 11 (25%) between 3 and 3.29 g/dL, and 14 (33.33%) had values ≥3.30 g/dL (Table 2).
Incidence of Pathological Hyperbilirubinemia in Relation to CBB and CBA Levels.
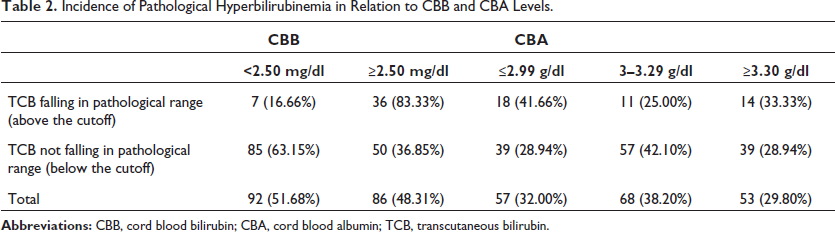
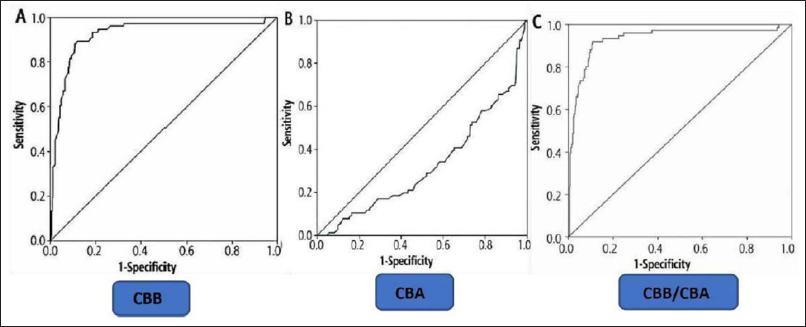
Figure 1 and Table 3 demonstrate that cut-off value for CBB was 1.98 mg/dL as determined by ROC curve, which had a sensitivity of 83.33%, a specificity of 63.15%, a PPV of 41.66%, NPV of 92.30%, and standard error of 0.020 under nonparametric assumption. Under nonparametric assumption, the cut-off value for CBA was found to be 3.14 g/dL and had a sensitivity of 41.66%, specificity of 71.05%, PPV of 31.25%, NPV of 79.41%, and standard error of 0.036. According to ROC curve, cutoff value for the CBB to albumin ratio was 0.630 and this value was shown to have 91.66% sensitivity, 86.84% specificity, a PPV of 68.75%, NPV of 97.05%, and standard error of 0.019 under non-parametric assumption.
ROC Cut-off Values for CBB, CBA, and CBB/CBA.
Discussion
In countries with limited resources and high patient-to-bed ratios, early identification of hyperbilirubinemia aids with prompt discharge, prevent re-hospitalization, and decrease the amount of time of newborns’ and mothers’ hospital stay.12–15 Therefore, a marker that can accurately anticipate the onset of severe jaundice in a newborn is always needed. In our study, we observed that 24% of newborns had neonatal hyperbilirubinemia, which is close to Bernaldo et al. 16 that reported a somewhat comparable frequency of 19.86%. Though few studies claim high incidence rate ranging from 34% up to 54.3%, others17– reported a lesser incidence of 10.70%, 12.80%, 10.30%, 10.60%, and 9.50%. These variations may result from various inclusion or exclusion criteria taken into account by various research, varying racial and ethnic groups, or both. As determined by the ROC curve analysis, the cut-off CBB level in our study (1.98 mg/dL) was lower than those reported in similar studies by Knudsen et al., 15 Taksande et al., 22 Sun et al., 23 and Ahire et al., 26 that reported cut-offs of 2.30, 2, 2, and 3 mg/dL. The cutoff level of CBA as determined by ROC curve analysis was 3.14 g/dL, compared to levels in earlier investigations 24 of 2.60, 3, 2.75, 2.75, and 2.95 g/dL. This disparity seen might be a result of these studies’ limited sample and also inclusion of ill and preterm infants (Figure 2 and Table 4).
Sensitivity, Specificity, PPV, and NPV of CBB, CBA, and CBB/CBA Ratio in Detecting Pathological Jaundice.
A total of 418 neonates were enrolled in the study by Gupta et al., 25 and 17 of them needed PT. CBB had a cut-off value of 1.67 mg/dL with a sensitivity of 82% and a specificity of 99% for predicting the incidence of hyperbilirubinemia necessitating PT. Our study’s cut-off value was 1.98 mg/dl. In neonates receiving PT, the mean CBB level was 2.4 0.9 mg/dL. When blood group, CBB level, DC, gender, and mode of delivery were assigned as possible risk factors, multivariate analysis showed ABO, Rh incompatibility, and CBB level increased the risk of PT requirement.
Cord serum albumin 2.8 gm/dL had sensitivity 71.8%, specificity 65.1%, PPV 38.9%, and NPV 88.2%, according to a hospital-based study by Sun G et al., 23 on 165 healthy term newborns. The area under the curve (AUC) for the ROC curve was 0.684. In the study by Rajpurohit et al., 31 the CBA level cut-off of 2.6 gm/dL with a high sensitivity (80%) and specificity (86.67%) was selected. At this stage, the predictive values for the good and negative outcomes were 40% and 97.5%, respectively. This study also supports a higher test’s NPV.
We chose to employ those combined as the CBB to CBA ratio so as to achieve a greater sensitivity, specificity, NPV, and PPV than either CBB or CBA individually. Cut-off points of the CBB/CBA ratio attained in other studies27–30 for the prediction of the development of significant hyperbilirubinemia were 0.60, 0.78, and 0.98. ROC curve estimation on the CBB/CBA ratio showed a cut-off point of 0.630 with a sensitivity of 91.66%, specificity of 86.84%, PPV of 68.75%, and NPV of 97.05%. Variance in sample size and bilirubin estimate techniques used in other studies may be the cause of this variation in cut-off value. By using ROC curve estimation, AUC for the CBB/CBA ratio is 0.937, which is also greater than the AUC for just CBB and CBA alone.
Based on the foregoing discussion, it can be concluded that, when compared to CBB and CBA alone, the CBB/CBA ratio is a superior predictor of the development of neonatal hyperbilirubinemia with greater specificity, sensitivity, NPV, and PPV.
Strengths of the Study
A good sample number, adequate follow up for development of pathological hyperbilirubinemia and the findings recorded by a single observer (which offered us a more consistent approach and no interobserver bias) are the merits of this study. The combination of the CBB and CBA approaches in the form of a ratio provides a new parameter that is more sensitive and specific than CBB or CBA alone.
Limitations
Our study has a few drawbacks, including the exclusion of late preterm infants, extremely low-birth-weight infants and the usage of a transcutaneous bilirubinometer, which tends to exaggerate bilirubin values.
However, the issue is somewhat mitigated by the fact that bilirubinometers are more accurate in term infants.
Conclusion
In comparison to CBB and CBA alone, the CBB/CBA ratio has greater sensitivity, specificity, PPV, and NPV and it is a better indicator for the early detection of the development of significant neonatal hyperbilirubinemia.
Footnotes
What is Already Known?
Albumin serves as an early determinant for the development of significant neonatal Hyperbilirubinemia. 31
What this Study Adds?
CBB/CBA ratio has greater sensitivity, specificity, PPV, and NPV, and it is a better indicator of early detection of the development of significant neonatal hyperbilirubinemia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Institutional Ethical committee on 22/10/2022 (Ref. No. IEC/NMC/22.10.22_1).
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The participant has consented to the submission of the article to the journal.
