Abstract
Background:
Risperidone and methylphenidate are commonly used drugs to manage problem behaviors in individuals with autism spectrum disorder (ASD). Studies report that these 2 drugs may be helpful in improving core symptoms of ASD as well.
Aim:
To assess and compare the efficacy and safety profile of risperidone and methylphenidate for problem behaviors and core symptoms in children and adolescents with ASD.
Setting and Design:
An 8-week, prospective, interventional, randomized, and open-label trial carried out in a tertiary care hospital of northern India.
Materials and Methods:
Forty subjects with a diagnosis of ASD according to Diagnostic and Statistical Manual of Mental Disorders, 5th Edition were randomized to risperidone and methylphenidate groups. Childhood Autism Rating Scale, Second Edition, Indian Scale for Assessment of Autism, and Nisonger Child Behavior Rating Form were used for assessments. Adverse effects, physical, and laboratory parameters were monitored regularly.
Statistical Analysis:
Tests applied include chi-square test, repeated measures Analysis of Variance, and Student t test, and nonparametric tests for skewed data. Data was analyzed using SPSS Version 22.0. Level of significance was set at 0.05.
Results:
Both drugs improved inattention, hyperactivity, and stereotypies. Methylphenidate reduced self-isolation, and risperidone improved emotional dysregulation, aggression, and self-injury. Adverse effects were seen in 55% of methylphenidate group (commonly decreased appetite and irritability) and 40% of risperidone group (commonly sedation and increased appetite). Improvement in stereotypy with both drugs and self-isolated behavior with methylphenidate indicates a possible role in improving core symptoms of ASD.
Conclusion:
Both drugs were beneficial in the management of behaviors associated with ASD. Further research to establish their role in reducing core symptoms and long-term safety is warranted.
Introduction
Autism spectrum disorder (ASD) is a neurodevelopmental disorder characterized by impairment of social skills, communication, and restricted and repetitive behavior, which manifests in the first few years of life. 1 These make up the core symptoms domains of ASD. However, ASD can present with various other emotional and behavioral problems, collectively labelled as “problem behaviors.” 2 These include irritability, violent outbursts, temper tantrums, self-injurious behaviors, noncompliance, hyperactivity, and inattention. 3 The management of ASD entails comprehensive assessment and extensive interventions started early in life targeted to reduce impairment due to the core deficits and other associated symptoms. Management includes both nonpharmacological and pharmacological interventions, with the latter considered second line to the former. 4
Risperidone was found to be useful in managing impulsivity, aggression, and repetitive behavior in children aged 5 years to 18 years (mean aged 10.2 years) at a mean dose of 1.8 mg/day in an open label trial. Sedation and weight gain were the common side effects reported. 5 Similar findings were seen in another trial of children between ages 7 and 14 years with a mean dose of 2.7 mg/day. 6 An Indian study comparing risperidone with fluoxetine found that risperidone improved symptoms of irritability, aggression, and hyperactivity. 7 The Research Units on Pediatric Psychopharmacology (RUPP) trial also found a decrease in irritability, aggression, tantrums, self-injury, and stereotypies with risperidone at a mean dose of 1.8 mg/day in children aged 5 years to 17 years. 8 Social relationships and social withdrawal also improved in these children. However, this was attributed to the fact that reduction in tantrums, self-injury, and aggressive behavior allows the subject to be available for social interaction. A placebo-controlled trial of subjects aged 2 years to 9 years with 1 mg/day risperidone dosage carried out in India as well as a 21-month follow-up study of the RUPP trial found improvement in the core symptom domains of ASD.9,10
Stimulants are used to manage hyperactivity in children with ASD, but have been reported to worsen behavioral functioning and induce or worsen stereotypies.11,12 Methylphenidate (administered at dosages 20 mg/day and 40 mg/day in children aged 7 years to 11 years for 1 month) has been found to improve inattention/hyperactivity, but not social withdrawal and communication deficits in some studies. Usual adverse effects of sleep disturbance, irritability, and decreased appetite are also documented.13,14 A placebo-controlled trial done in children aged 5 years to 11 years found methylphenidate (used in the dose of 0.3 mg and 0.6 mg/kg) useful in improving stereotypy and inappropriate speech, associated with core autistic features. 15 In the RUPP methylphenidate trial, subjects aged 5 years to 14 years showed improvement in inattention, distractibility, hyperactivity, and impulsivity, but not in core symptoms of ASD. Methylphenidate was dosed at 3 levels: 0.125, 0.250, and 0.500 mg/kg/dose given 3 times a day. 16 The secondary analysis concluded that methylphenidate may have positive effects on social behaviors.
Both risperidone and methylphenidate are commonly used drugs in individuals with ASD and help in managing problem behaviors associated with ASD. Risperidone, while reducing irritability and aggression, is also reported to show improvement in socialization. Methylphenidate is usually prescribed for inattention and hyperactivity, but has a potential role in improving social communication. Keeping in mind this background, this study was planned to assess and compare the efficacy and safety of risperidone and methylphenidate in children and adolescents with ASD.
Methods
Trial Design
This study was designed as a prospective, interventional, randomized, open-label trial to assess and compare the efficacy and safety of risperidone and methylphenidate in children and adolescents with ASD. Approval from the Institutional Ethics Committee was taken before initiating recruitment. Each participant was studied for a duration of 8 weeks. Participant intake was carried out over a period of 14 months, from March 2018 to April 2019.
Participants
Children and adolescents attending the child guidance clinic of a tertiary care hospital, and attending outpatient services/enrolled at an associated institute for intellectual disability, were assessed for eligibility. Those having a diagnosis of ASD according to the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5) criteria, age between 6 and 16 years, parent/guardian giving written informed consent were included. 17
In subjects 7 years and older who were able to understand and write, assent was taken. Those with major medical illness (seizure disorder, heart disease, juvenile diabetes), mental illness (mood disorder, psychotic illness), multiple disability (except mild intellectual disability), meeting DSM-5 criteria for attention-deficit hyperactivity disorder (ADHD), and those already on psychotropic medication were excluded. Out of the 11 participants excluded for not fulfilling inclusion criteria, 4 were already on psychotropic medication. Two of these were on risperidone, and 1 each on olanzapine and aripiprazole. Only drug naïve participants were included. Since methylphenidate is commonly used for ADHD, a comorbid diagnosis of ADHD along with ASD would confound the results. Hence, ADHD was excluded after detailed clinical interview and examination by all the authors.
Interventions
Duration of intervention was 8 weeks. Methylphenidate was started at a dose of 0.125 mg/kg body weight with thrice daily dosage (morning, noon, and late afternoon) in tablet form. Dose was incremented by 2.5 mg/day as clinically indicated and tolerated, up to the maximum possible dose in 2 weeks. Risperidone (oral solution) was initiated to reach a dose of 0.25 mg or 0.5 mg/day by the end of the first week, according to weight of the child. It was titrated at 0.25 mg to 0.5 mg at 2 weeks or more. The target dose was 0.5 mg for participants weighing less than 20 kg, and 1 mg/day for those weighing more than or equal to 20 kg. Participants were assessed every week for the first 4 weeks, and then every 2 weeks for the next 4 weeks.
Outcomes
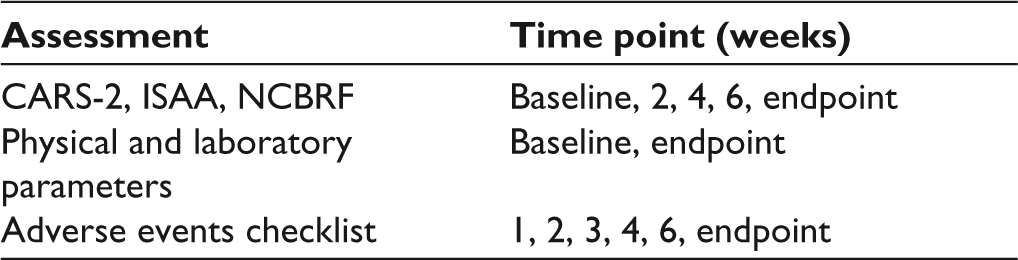
The tools used for assessment of symptoms of ASD were Childhood Autism Rating Scale, Second Edition (CARS-2), and Indian Scale for Assessment of Autism (ISAA).18,19 CARS-2 is an internationally used diagnostic and assessment tool for ASD, while ISAA is a validated and culturally adapted tool in the Indian context for rating the severity of ASD symptoms. Nisonger Child Behavior Rating Form (NCBRF), used to assess behaviors in children with intellectual or developmental disabilities, was applied to assess for behavior problems in the subjects. 20 These were applied at baseline, and then every 2 weeks. For monitoring side effects, exhaustive adverse effects checklists for both the drugs were prepared for the specific purpose of this study. Adverse effects for both drugs were rated every week for the initial 4 weeks, and then every 2 weeks for the remaining 4 weeks. Physical parameters including weight, height, and Body Mass Index (BMI) and laboratory investigations including complete blood count, electrolytes and renal function tests, liver function tests, lipid profile, fasting blood sugar, and electrocardiogram were assessed at baseline and then at study end-point. The schedule of assessment for various parameters is given in Table 1.
Schedule of Assessments of Outcome Parameters
Sample Size
In order to estimate the sample size, records of the outpatient clinic and associated institute were surveyed. After assessing the approximate population proportion to be 2.7%, with a 2-tailed α of 0.05 and confidence interval of 95%, appropriate sample size for the study was calculated to be 40.
Randomization
Participants fulfilling the inclusion and exclusion criteria were randomly assigned to the methylphenidate and risperidone groups using computer-generated random number tables.
Allocation Concealment Mechanism
Sequentially numbered opaque sealed envelopes were used for allocation concealment. Blinding was not done.
Implementation
Generation of random allocation sequence, participant enrolment, and assignment of participant interventions was done by the first author.
Statistical Methods
The statistical analysis included chi-square test for qualitative data, and repeated measures Analysis of Variance for quantitative data. Student t test was applied to compare individual assessments. Data was analyzed using SPSS Version 22.0, and represented in mean and Standard Deviation. 21 In case of skewed data, nonpara-metric tests (Wilcoxon Signed Ranks Test and Mann-Whitney U Test) were applied. Level of significance was set at 0.05.
Results
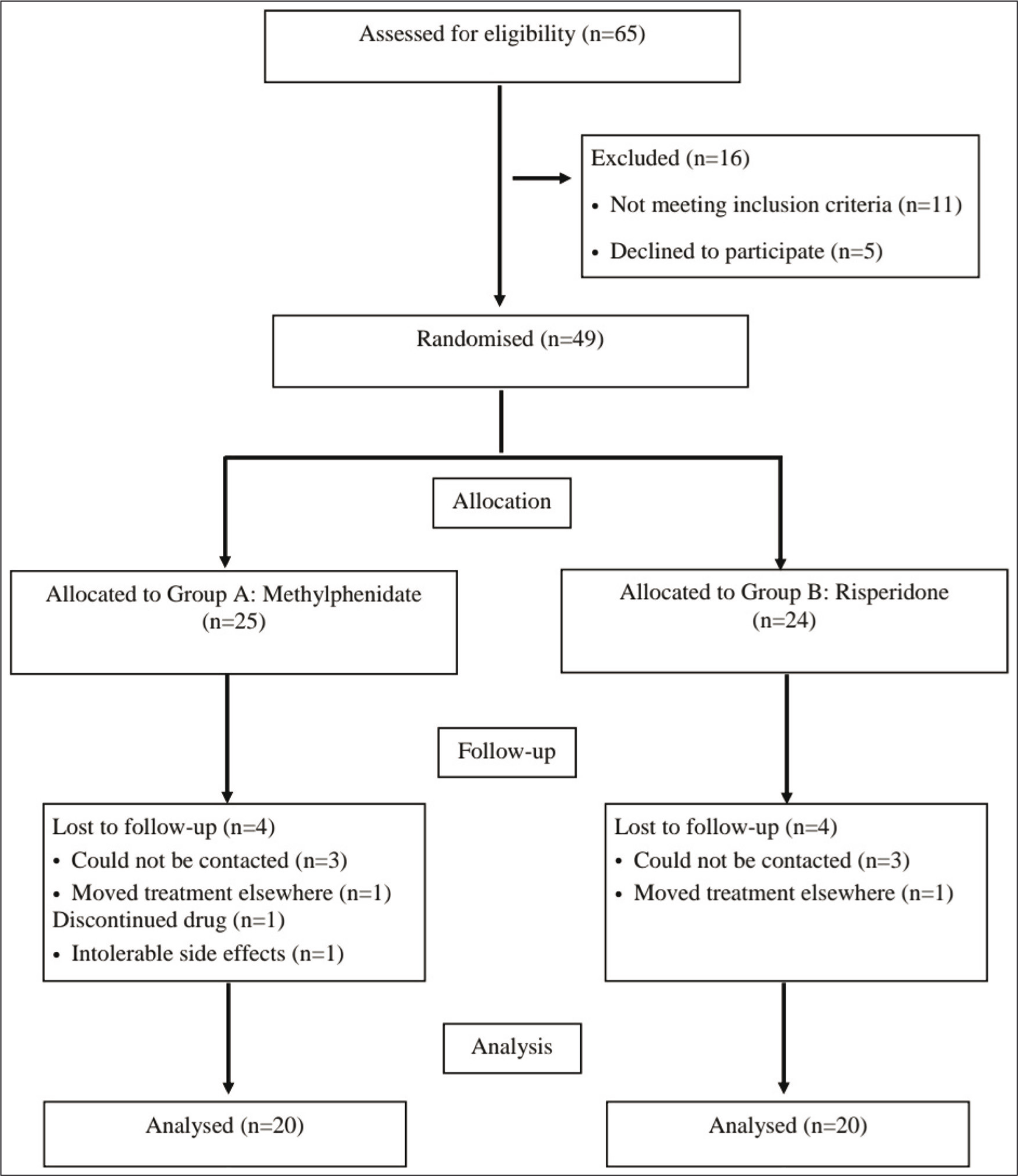
A total of 65 cases of ASD were assessed for eligibility, out of which 49 fulfilled eligibility criteria and agreed to participate. Of these 49, 40 subjects completed the study, with 20 each in the risperidone and methylphenidate groups. The participant flow is outlined in Figure 1. Drug therapy was initiated after baseline assessments and investigations, and the subjects were followed up regularly according to the schedule of assessments.
Participant Flow
Sample Characteristics
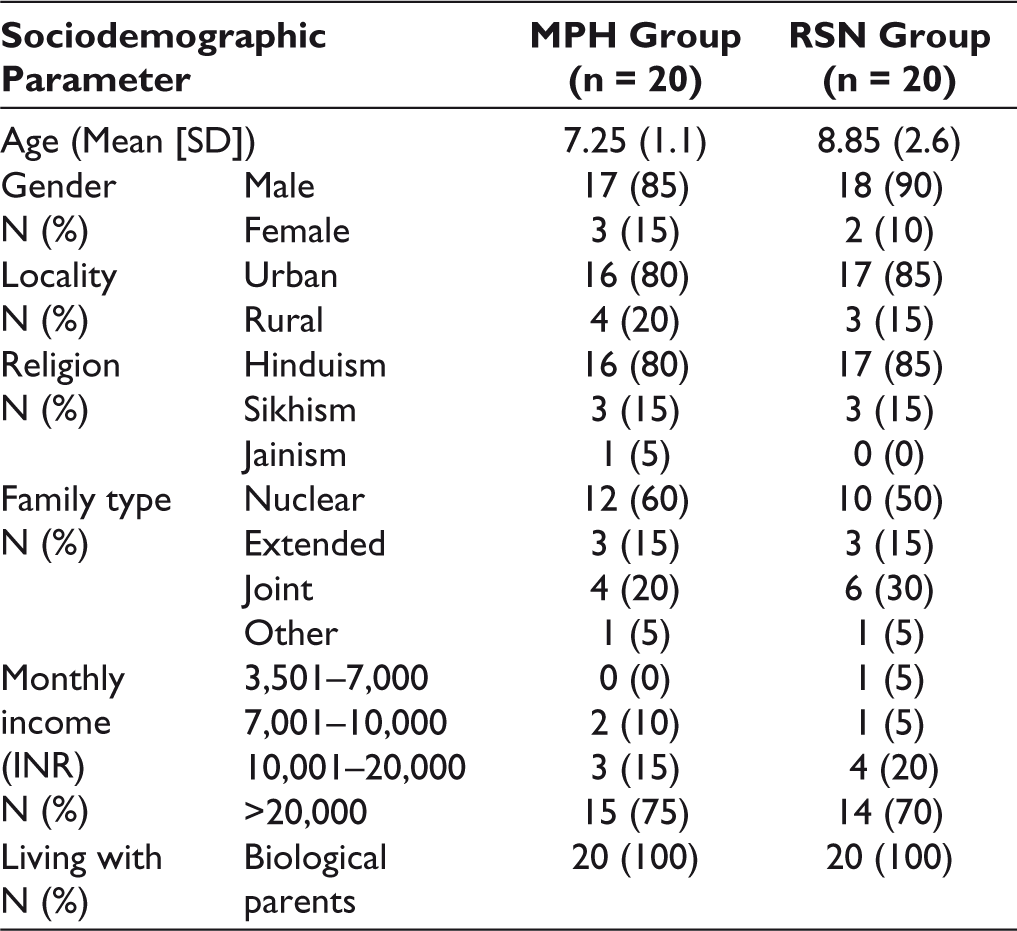
A total of 40 subjects completed the study. The mean age of study sample was 8.05 ± 2.31 years (range: 6–15 years). The mean age of subjects was 7.25 ± 2.0 years in the methyphenidate (MPH) group (n = 20) and 8.85 ± 2.6 years in the risperidone (RSN) group (n = 20). Males comprised of 87.5% (n = 35) of the total study subjects, with 85% (n = 17) males in the MPH group and 90% (n = 18) males in the RSN group. Table 2 gives the sociodemographic characteristics of the study sample. Twenty subjects out of 40 (50%) also had mild intellectual disability. Both the groups were comparable in all respects at the beginning of the study, with no statistically significant difference between them on all parameters of the outcome measures.
Sociodemographic Characteristics of Both Groups
Efficacy Parameters
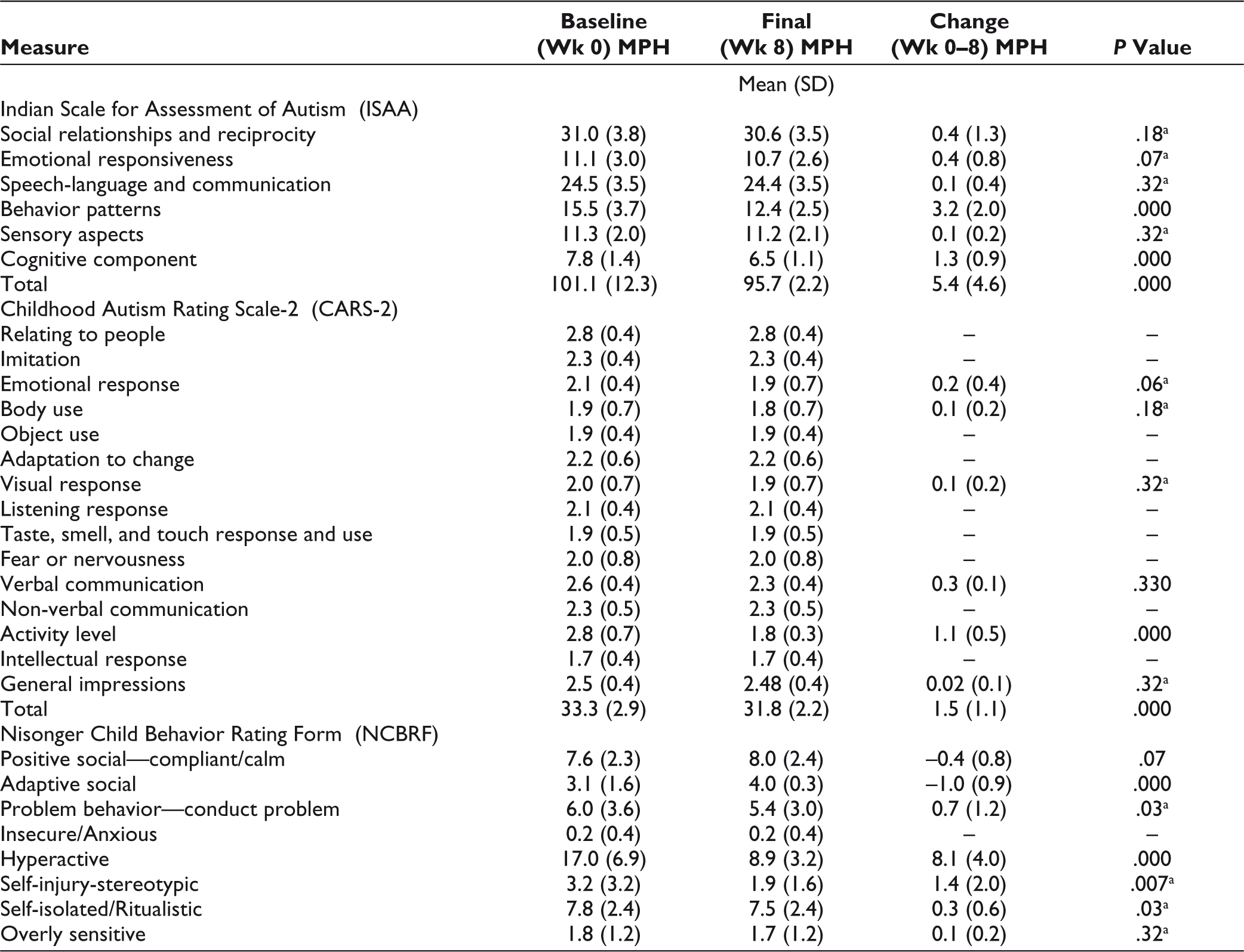
The mean final dose of methylphenidate being received by study subjects in MPH group was 19.13 ± 7.0 mg/day. In this group, significant improvement was seen on the behavior patterns and cognitive component subscales of ISAA, the Activity level item of CARS-2, and the Conduct problem, Hyperactive, Self-injury/stereotypic, and Self-isolated/ritualistic subscales of NCBRF. Improvement was also seen on the Adaptive/Social subscale of positive behaviors of the NCBRF. There was an overall significant improvement in the final scores of ISAA (P = <.001) and CARS-2 (P = <.001). This is depicted in Table 3.
Comparison of Change in Scores in MPH Group
Three of the 20 subjects (15%) in the RSN group were administered 0.5 mg, while the remaining 17 (85%) were given 1 mg/day. In this group, significant improvement was seen on the emotional responsiveness, behavior patterns, and cognitive component subscales of ISAA, the Activity level item of CARS-2, and the Conduct problem, Hyperactive and Self-injury/stereotypic subscales of NCBRF. There was an increase in positive behaviors as evidenced by the Compliant/Calm and Adaptive Social sub scales of the NCBRF. There was a significant decrease in the total scores of ISAA (P = <.001) and CARS-2 (P = <.001). The results are depicted in Table 4.
Comparison of Change in Scores in Risperidone Group
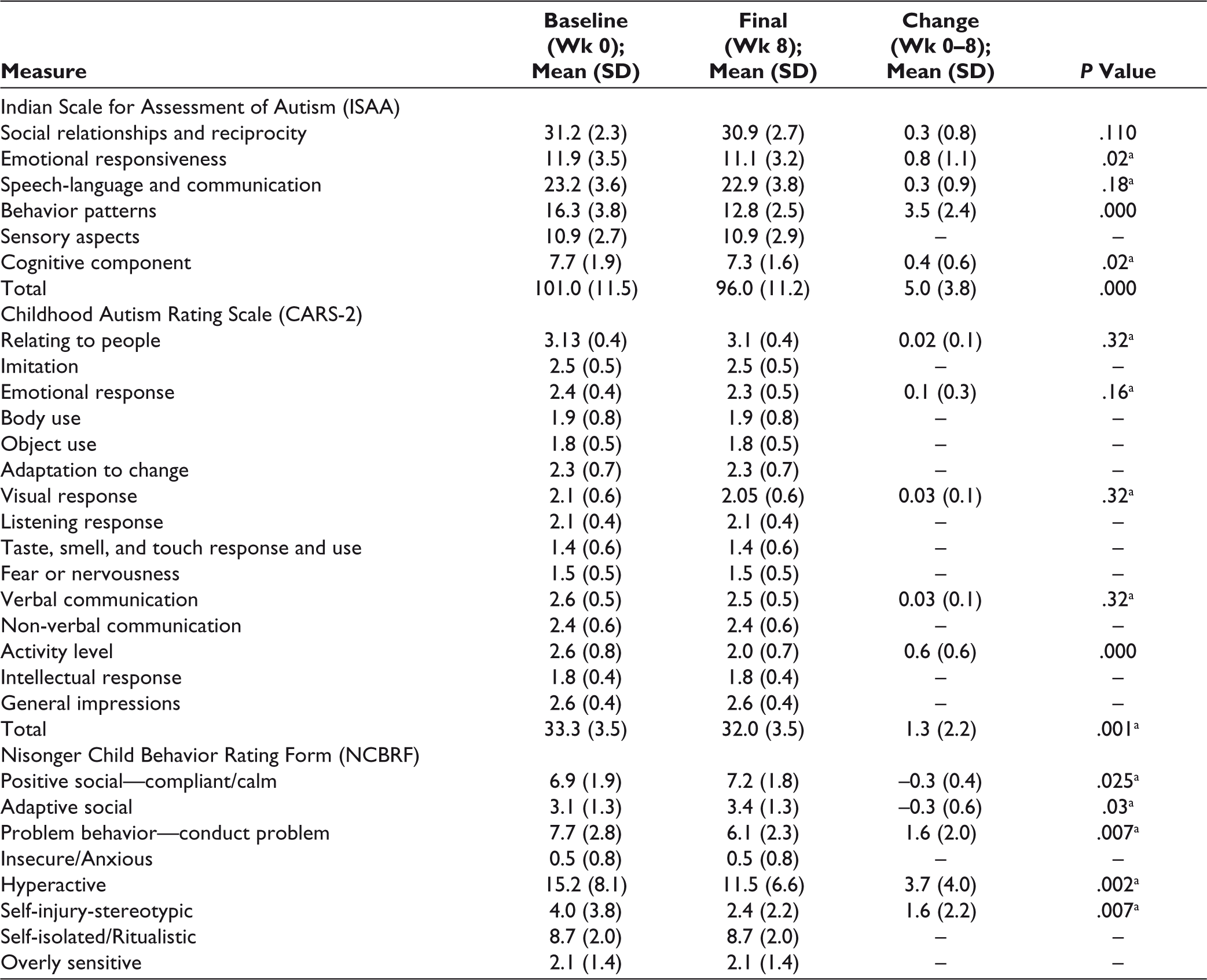
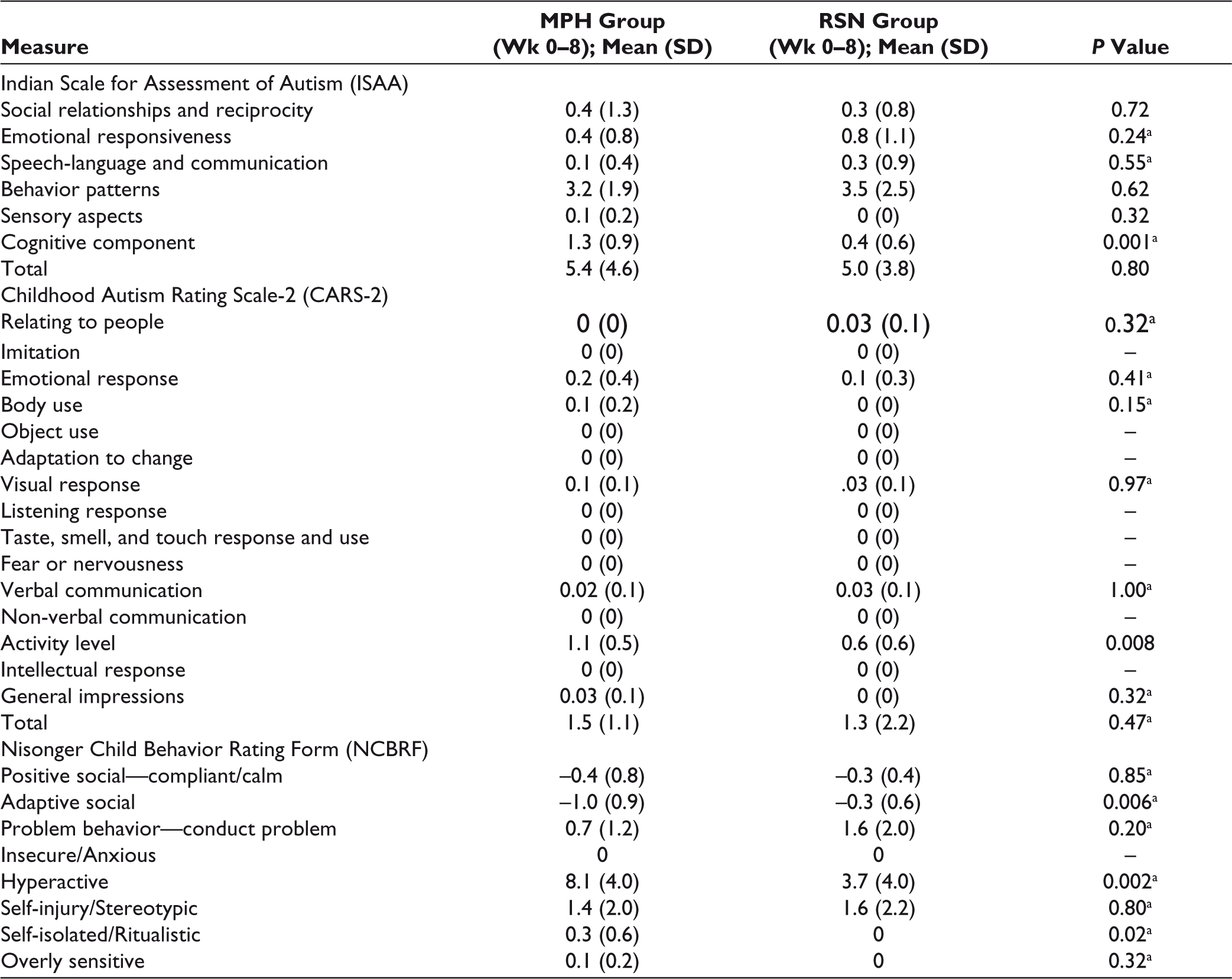
On comparing the change in scores with both drugs over 8 weeks, methylphenidate showed a significantly greater improvement on the Cognitive component subscale of ISAA, Activity level item of CARS-2, and the Hyperactive and Self-isolated/ritualistic subscales of NCBRF. It also showed greater improvement on the Adaptive Social subscale of the NCBRF. This can be potentially attributed to decrease in inattention and hyperactivity, which allowed the subjects to participate better in activities and stay on task. These results are depicted in Table 5.
Comparison of Change in Scores Between Both Groups
Safety Parameters
Side effects were experienced by 55% (n = 11) subjects in the MPH group, with the commonest ones being decreased appetite and irritability. Appetite decreased to a mild severity in 2 (10%) subjects, and moderately in another 3 (15%) subjects. Irritability was seen in 5 (25%) subjects, but this was mild and tolerable. Sleep disturbance of mild intensity was seen in 3 (15%) patients. In the RSN group, side effects were reported by 40% (n = 8) subjects. The common adverse effects with risperidone were sedation and increased appetite. Six (30%) subjects reported mild sedation during the day, with 1 subject’s sedation subsiding over a 2-week period. Three (15%) subjects experienced mild increase in appetite, with 2 (10%) of these also reporting mild weight gain. Only 1 subject developed hand tremors at 4 weeks, but these were mild in intensity and subsided by 6 weeks. Subjects in the RSN group showed a higher mean weight gain (0.35 kg) than those in the MPH group (0.13 kg). Height gains in both groups were minimal and comparable (0.003 m). The RSN group had a mean BMI increase of 0.19 kg/m2, while MPH group showed a mean decrease of 0.89 kg/m2. No significant changes were seen in the laboratory parameters of any subjects.
Discussion
This study was carried out to assess and compare the efficacy and tolerability of methylphenidate and risperidone in children and adolescents with ASD. The sample comprised of 40 subjects, with an average age of 8.05 ± 2.31 years. Most trials in the literature have studied children of a similar age range and mean age. Out of the 40 subjects studied, 35 (87.5%) were males and 5 (12.5%) were females. It is common knowledge that ASD is more common in males, and this proportion is consistent with other studies which have commented on their gender distribution and also with the male-to-female ratio of 4:1 mentioned in DSM-5. 6 The baseline subscale and total scores on all assessment tools, namely CARS-2, ISAA, and NCBRF, had no significant difference, indicating a normal distribution. Baseline CARS-2 scores were 33.3 ± 2.9 in the MPH group and 33.3 ± 3.5 in the RSN group. Baseline ISAA scores were 101.1 ± 12.3 in the MPH group and 100.95 ± 11.5 in the RSN group. These scores indicated mild to moderately severe symptoms of ASD in the 2 groups.
The mean final dose of methylphenidate was 19.13 ± 7.0 mg/day. The dose of methylphenidate is in accordance with the dose recommended for use in children and also with that being used in earlier studies.13,16 Children who weighed less than 20 kg were given 0.5 mg/day risperidone, and all others were given 1 mg/day. Western literature supports use of higher doses in the range of 1 mg/day to 6 mg/day.22,23,24 However, Indian studies have used comparable dose of risperidone as in the present study.7,9
The endpoint score of ISAA showed significant improvement with both drugs, with a mean decrease of 5.5 ± 4.6 points in the methylphenidate group, and 5.0 ± 3.8 points in the risperidone group, with no group showing significantly higher improvement than the other. Both drugs showed improvement its behavior patterns and cognitive component subscales, as seen in earlier literature.7–9,13–15 In the RSN group, improvement was also seen on the Emotional Responsiveness subscale, owing to a decrease in agitation and exaggerated emotions, similar to previous studies.7,9,10
The total score of CARS-2 showed significant improvement with both drugs, with a mean decrease of 1.5 ± 1.1 points in the methylphenidate group and 1.3 ± 2.2 in the risperidone group. However, on the 15 items of this scale, no statistically significant change was seen except for in the activity level, which led to significant improvement in the total score. Myriad studies of methylphenidate and risperidone as cited in literature use the first edition of CARS as an assessment tool and this may be leading to the findings in the present study. Additionally, most studies which observed improvement on other items of CARS with risperidone had a longer duration or used higher doses or both.6,9,22
Both drugs showed an improvement on the Conduct problem, Hyperactive and Self-injurious/stereotypic subscales of the NCBRF. Risperidone has been observed to improve self-injury stereotypic behavior earlier.5,8,25 Some studies have found methylphenidate to induce or worsen existing stereotypic behavior in ASD.14,25 Some studies mention that it did not cause stereotypies or worsen existing ones.16,26,27 Interestingly, there is 1 study wherein it led to improvement in stereotypies. 15 Additionally, methylphenidate showed a significant reduction on the Self-isolated/Ritualistic subscale of NCBRF. This might be due to greater reduction in hyperactivity and inattention, leading to better involvement and interaction with the environment. Methylphenidate showed significantly higher improvement in inattention and hyperactivity, as evidenced by changes in all the assessment scales. Earlier literature has shown efficacy of risperidone in improving these symptoms, 28 but there is overwhelming evidence in the existing literature for methylphenidate in this domain, which explains these findings.14,15,26
Both risperidone and methylphenidate showed significant reduction in symptoms like inattention and hyperactivity, with the latter being superior to the former in this respect. Methylphenidate was superior in reduction of hyperactivity and attention problems, and contributed to improvement in cognitive areas and adaptive behavior. It also led to a decrease in stereotypic behaviors, which contradicts the usual findings of worsened stereotypy in literature.13,25 Risperidone was effective in decreasing emotional dysregulation, aggression, and self-injurious behavior, and increasing compliance, calmness, and adaptive behavior, similar to existing literature.5,24,27,29
Common side effects of methylphenidate included decreased appetite, irritability, and sleep disturbance, which were mild to moderate in severity and were tolerable. This was in line with previously available studies of methylphenidate.13,14,16 Side effects due to risperidone included sedation and increased appetite, with 1 subject showing transient hand tremors. This side effect profile was similar to earlier studies.5,6,9 Risperidone also showed greater weight and BMI gain than methylphenidate, which was actually favored by and was not problematic for the parents/guardians of these subjects.
Both drugs brought about an increase in positive behaviors and aided in improving social and adaptive behavior, but risperidone seemed to have higher efficacy in this aspect. The improvement in stereotypic behavior due to both drugs is noteworthy, as stereotypy is a part of core symptom domains of ASD. Moreover, methylphenidate also showed efficacy in decreasing self-isolated behavior, which is an indicator of improvement of core symptomatology. This is in line with findings from some earlier studies.14,30
The prospective and randomized study design is the major strength of the index study. Additionally, the 2 groups evaluated as a part of this study were comparable on all assessment tools at baseline, thus heterogeneity was in a way controlled. The use of validated tools, that is, ISAA (indigenous tool validated for use in Indian children) and CARS-2, which has been used in Western studies, makes this study relevant to Indian population as well as renders it comparable to the Western literature. Frequent assessments were done mainly to assess side effects and avoid any untoward major adverse effect. However, short duration of the study is a limiting factor while interpreting the results of the study. Blinding could not be done and sources of bias cannot be ruled out. Nonpharmacological treatment being received by some participants was not accounted for, and is another limitation of this study. No standardized tool was used to exclude ADHD. Most participants had mild ASD, so results cannot be generalized to moderate-severe disorder. Only immediate release preparation of methylphenidate was used; hence, results with other longer-acting formulations cannot be commented upon. Additionally, the study did not include postpubertal subjects, hence the findings cannot be generalized to that population.
Conclusion
This study supports the finding that both drugs are useful in the management of ASD in terms of decreasing problematic behaviors, promoting positive behaviors, and aiding in social functioning. Though efficacy of these drugs in core symptom domains of ASD cannot be established unequivocally in the present study, there are findings which are definitively indicative of this. This can possibly be attributed to the short duration of the study and lower dose range used. Further, it adds to the Indian literature on the subject, as to the best of our knowledge no Indian study has so far studied the parameters used in the index study and at the same time compared the efficacy and safety of 2 important molecules used frequently in children and adolescents with ASD.
Future Directions
Research on management of ASD, especially pharmacological and combination strategies, still remains inadequate in the Indian population. There are no definitive guidelines available on these drug therapies, which is a major lacuna in the available literature. A combination of behavioral therapy and drug treatment with risperidone or methylphenidate could be a useful strategy for managing the core symptoms of ASD, and further research is warranted in this area. Additionally, further studies are required to establish the long-term usefulness of these drugs, and also the role of intelligence quotient in moderating the response of these drug therapies. All these are significant areas of potential enquiry which would open new avenues in the management of ASD.
Footnotes
Author Contributions
Conception of the study was by PA, and designing of the work was by PA with inputs from NC. Data collection was carried out by SM, and data analysis and interpretation was by all authors. The article was drafted by SM, with critical revisions from PA and NC. Final approval was by all authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
