Abstract
Background
Astragalus polysaccharide (APS), a component of traditional Chinese medicine, has recently shown promise in treating sarcopenia.
Objectives
This study investigates APS’s potential to reduce reactive oxygen species (ROS) and promote intramuscular protein levels in sarcopenic mice by activating the AKT-mammalian target of rapamycin (mTOR) pathway.
Materials and Methods
By constructing a mouse model of sarcopenia and dividing it into a blank model group, a low, medium and high concentration group, a model group as well as a control group, the therapeutic effect and mechanism of Astragali polysaccharides on sarcopenia were observed through animal experiments, obtaining gastrocnemius muscle to carry out in vitro cellular experiments, respectively and polymerase chain reaction (PCR), Sirius staining, immunofluorescence microscopy, Western blot (WB) experiments, and so on, combined with the simulation of the oxidative environment of ROS in vitro, to observe the level of AKT-mammalian target of rapamycin (mTOR) markers, superoxide dismutase 2 (Sod2) gene level and level of each protein in mouse muscle cells.
Results
(a) The animal model of myasthenia gravis was successfully established. (b) The effect of APS in the treatment of myasthenia gravis was outstanding and concentration dependent. (c) Compared with the model group, AKT was upregulated in the Astragali polysaccharide group, the proliferation rate of muscle cells was increased, and Sod2 messenger ribonucleic acid (mRNA) was elevated, while ROS was reduced. (d) Compared with the Astragali polysaccharide group, AKT was upregulated in + SC79 group, while muscle cell proliferation rate increased, Sod2 mRNA expression was significantly elevated, and ROS content was reduced. (e) Sod2 mRNA and matrix metallopeptidase 1 (MMP-1) protein expression in the Astragali polysaccharide + N-acetylcysteine (NAC) group were significantly higher, while ROS, collagen-1, and TIMP metallopeptidase inhibitor 1 (TIMP-1) levels were lower. (f) Compared with Astragali polysaccharide + SC79 group, collagen-1, TIMP-1, p38 mitogen-activated protein kinase (p38MAPK) protein expression in cells of Astragali polysaccharide + SC79 + NAC group was significantly reduced, while MMP-1 content was elevated. (g) Compared with the Astragali polysaccharide + SC79 + NAC group, cellular collagen-1, TIMP-1, and p38MAPK were elevated, and MMP-1 content was decreased in the Astragali polysaccharide + SC79 + NAC + hydrogen peroxide group.
Conclusion
APS treatment improves muscle cell proliferation and enhances sarcopenia mice’s antioxidant capacity by activating phosphoinositide 3-kinase (PI3K)/AKT to inhibit ROS, effectively maintaining the intracellular redox balance.
Introduction
Sarcopenia is a skeletal muscle disease featuring skeletal muscle mass loss (Weber, 2021). This condition mainly occurs in the elderly, but it can occasionally manifest early in life under certain conditions (Millozzi et al., 2023). Sarcopenia can lead to severe complications such as fractures, physical disability, and even death (Pickhardt et al., 2022). Proteins play a crucial role in cell structure and function (Araiso et al., 2022), and during sarcopenia, declining skeletal muscle function impacts intracellular protein levels, disrupting the dynamic balance within cells (Cheng et al., 2023). Therefore, this study aims to explore more effective treatment options for patients with sarcopenia.
Astragalus polysaccharide (APS) is derived from the Chinese herb Astragalus membranaceus. It boosts the body’s immunity by promoting immune cell differentiation, which helps in combating pathogens (Li et al., 2022); it also scavenges a significant number of free radicals and maintains cellular function (Zheng et al., 2020). Additionally, APS exhibits various pharmacological effects, including anti-tumor properties, liver function regulation, and promotion of liver cell regeneration (Li et al., 2020). Research has found that APS can induce apoptosis by inhibiting high mobility group box 1 (HMGB1), toll-like receptor 4 (TLR4), and nuclear factor kappa-B (NF-κB), which aids in repairing damage caused by hypoxia in cardiomyocytes (Xu et al., 2023); it can also promote cell proliferation by activating the mitogen-activated protein kinase (MAPK) pathway, thereby maintaining intracellular protein balance (Dong et al., 2020). Furthermore, APS influences the mammalian target of rapamycin (mTOR) pathway by activating phosphoinositide 3-kinase (PI3K) and AKT kinases, enhancing protein synthesis, increasing intracellular protein levels, and supporting muscle growth and repair (Li et al., 2023). However, the detailed mechanism by which APS acts in the treatment of sarcopenia requires further investigation.
Activating the AKT-mTOR pathway can enhance protein synthesis and increase protein levels in muscle cells, playing a vital role in managing sarcopenia (Tewari et al., 2022). APS has been shown not only to inhibit 4E-BP1 activity but also to stimulate protein synthesis via the AKT-mTOR pathway, promoting neuron regeneration (Wei et al., 2023). It can also regulate proteins like Bax, Bcl-2, and Beclin-1, providing anti-apoptotic benefits (Yang et al., 2019). Moreover, APS increases the expression of LC3 through the AKT-mTOR pathway, promoting cell autophagy, which helps eliminate harmful cellular substances, protects cells from damage, and enhances cell viability. The specific mechanisms through which APS activates the AKT-mTOR pathway in treating sarcopenia merit further exploration.
During the pathological process of sarcopenia, a substantial amount of reactive oxygen species (ROS) is produced, which exacerbates muscle damage (Antuña et al., 2022). Furthermore, studies have shown that activating the AKT-mTOR pathway boosts the activity of antioxidant enzymes as well as stimulates the synthesis of antioxidant substances. This dual action reduces ROS production and diminishes DNA damage caused by oxidative stress, thereby protecting cell membranes from lipid peroxidation and preserving mitochondrial function (Zhang et al., 2019). Therefore, this study aimed to explore the potential interaction between ROS, AKT-mTOR pathway, and APS in sarcopenic mice. This evidence might provide new methods for treating sarcopenia and provide novel insights into Traditional Chinese medicine.
Materials and Methods
Reagents and Materials
APS (Beijing Shengtai Technology Co., Ltd., CAS number: 89250-26-0; Purity: 98%) is an extract of A. membranaceus. It enhances the body’s immunity, resists pathogens, scavenges free radicals, and reduces oxidative damage, thus maintaining cellular function. Other reagents included real-time polymerase chain reaction (PCR) reagent (Shanghai Daixuan Biotechnology Co., Ltd.), Western blot related chemical reagents (Beijing Solebao Technology Co., Ltd.), type II collagenase (Shanghai Mambo Biomedicine), fetal bovine serum (FBS, Nanjing Aofan Biotechnology Co., Ltd.), rabbit anti-human ERK1/2 monoclonal antibody and secondary antibody (U.S. cell signaling company); ELISA kit (U.S. R&D Company), dexamethasone reagent (Chengdu Tiantaishan Pharmaceutical Co., Ltd.), SC79 (MedChemExpress Inc.); N-acetylcysteine (Shanghai Biyuntian Biotechnology Co., Ltd.), hydrogen peroxide (Lilkang Medical Science and Technology Co., Ltd.), wortmannin (Nanjing Baxindeno Bio-technology Co., Ltd.).
Animals
A total of 29 IVC grade SD male mice were purchased from Kaisheng Biotechnology Co., Ltd.
Animal Experiments
Four out of 29 randomly selected IVC class SD-aged mice (n = 4) were used as a control group, and the remaining 25 mice were injected with dexamethasone (20 mg/kg/d) intramuscularly for Sarcopenia modeling. In contrast, the control mice were injected with an equal amount of saline intramuscularly for 42 days. During the experiments, the environment was maintained at a constant temperature and humidity, the cages were cleaned, and the bedding was changed at a fixed time every week. During the experiment, the mice were kept in cages with free access to water and food, and the environment was maintained at a constant temperature and humidity; the cages were cleaned, and the bedding was changed at a fixed time every week, in addition to the simulation of a 12-h light cycle with alternating light and dark.
After modeling, mice were examined for changes in gastrocnemius muscle mass index, lean body mass, and grip strength. Modeling success was indicated by a gastrocnemius muscle mass index that was two times the standard deviation below that of control mice and a decrease in lean body mass and grip strength. Gastrocnemius muscle mass index = bilateral gastrocnemius muscle mass/body mass, and gastrocnemius muscle mass was measured by dual-energy X-ray absorptiometry (DXA).
The mice, after successful modeling, were randomly divided into a model group (n = 5), blank model group (n = 5), low concentration group (100 mg/kg for 6 weeks) (n = 5), medium concentration group (200 mg/kg for 6 weeks) (n = 5) and high concentration group (400 mg/kg for 6 weeks) (n = 5). The model group received an equal amount of 5% dextrose intraperitoneally.
Dual-energy X-ray Absorptiometry (DXA)
Under anesthesia, with mice placed on the examination bed, mouse body weight, and lean mass were accurately scanned using a dual-energy X-ray absorptiometry instrument and recorded.
Measurement of Mouse Forelimb Grasping Strength
An electronic grasp force meter was used to measure the grasping force of the mouse’s forelimbs. By lifting the mouse by the tail and placing its forelimbs on the sensing crossbar, the maximum grip force was recorded as the mouse was pulled until the forelimbs were released.
Hematoxylin and Eosin (H&E) Staining and Sirius Red Staining
After the mice were sacrificed, their gastrocnemius muscles were fixed in 4% paraformaldehyde for 24 h, embedded, and sectioned. The sections were then stained using Weigert iron hematoxylin, differentiated with an acidic solution, rinsed, and then stained with Sirius red for 1 h. The slides were rinsed again to remove excess dye and then sealed.
Western Blot
Total protein concentration was determined, separated, and transferred to the polyvinylidene difluoride (PVDF) membrane, which was incubated with primary antibodies (matrix metallopeptidase 1 (MMP-1), TIMP metallopeptidase inhibitor 1 (TIMP-1), COL-1) and then with secondary antibodies. The results were visualized using enhanced chemiluminescence (ECL) detection and quantified using ImageJ software.
Determination of ROS Content of Gastrocnemius Muscle Tissue
Freshly isolated gastrocnemius muscle tissue was cut with scissors and washed with pre-cooled PBS three times; after discarding the washing solution, it was put into a grinding tube, 1 mL of pre-cooled PBS was added as well as the grinding beads, and the grinding was done for 5–10 s and then rested for 50 s followed by centrifugation at 100 × g for 5 min. Take a 96-well plate, add 100 µL of supernatant, as well as 2 µL of dissolved dihydroethidium (DHE) solution, incubate for 90 min away from light, and then turn on the fluorescence microscope to preheat, select the green excitation light, and observe the red fluorescence under the microscope.
Cell Experiments
Primary Culture, Cell Differentiation, and Processing Methods of Mouse Muscle Cells
The gastrocnemius muscles of mice in the model, control group, and high-concentration group were obtained. After being cleaned by sterile Hank’s solution (Shanghai Xinyu Biotechnology Co., Ltd.), the tissues were cut into approximately 1 mm pieces and digested with type I collagenase (Shanghai Yudo Biotechnology Co., Ltd.) under the aseptic operating table. The tissues were washed and centrifuged with DMEM high-glucose medium containing 10% fetal bovine serum (FBS, Wuhan Punuosai). The cells were then resuspended in a cell culture medium, seeded into 6-well plates, and incubated. Medium changes and cell morphology observations were conducted on the 3rd, 4th, and 5th days post-inoculation. Cell passaging was performed when myoblast fusion exceeded 90%, and myoblast differentiation experiments were performed with adult muscle differentiation medium after 5 days, and myotube formation, as well as cell proliferation ability, was observed under the microscope. The AKT agonist SC79, the AKT inhibitor wortmannin, the ROS inhibitor N-acetylcysteine (NAC), and the ROS agonist hydrogen peroxide were added to the model group and APS group, respectively, to intervene and observe the changes in cellular ROS content and various proteins.
Detection of Intracellular ROS
ROS levels were determined using a kit from Shanghai Qiyuan Biotechnology Co., Ltd. Cells were evenly seeded, and A 2′,7′-dichlorofluorescein diacetate (DCFH-DA) probe was added and incubated in the dark. After washing, intracellular ROS levels were subsequently observed under a fluorescence microscope.
Real-time Quantitative Reverse Transcription PCR (RT-qPCR)
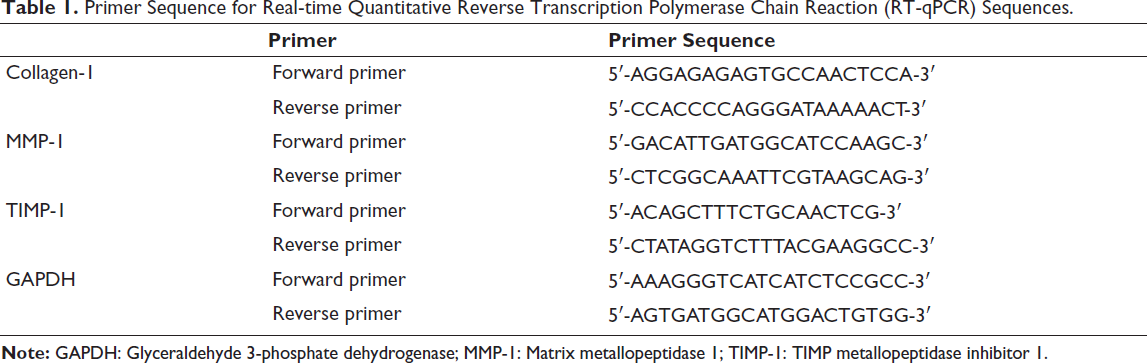
Total RNA was extracted and reverse transcribed into cDNA, followed by the measurement of gene levels using SYBR Green-based real-time PCR. The primers for collagen-1, MMP-1, TIMP-1, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH), used in the RT-qPCR, are detailed in Table 1.
Primer Sequence for Real-time Quantitative Reverse Transcription Polymerase Chain Reaction (RT-qPCR) Sequences.
Cell Counting Kit-8 (CCK-8) Assay
The proliferation ability of muscle cells was assessed using a CCK-8 kit (Guangzhou Saiku Biotechnology Co., Ltd.). First, cells were seeded and fully adhered to the wall. Then, CCK-8 reagent was added and incubated. The absorbance (A) was then measured with a microplate reader to determine cell proliferation at various time points.
Effect of APS on Muscle Cell Protein Expression
Cells were lysed using radioimmunoprecipitation assay buffer (RIPA) buffer, and protein was quantified by bicinchoninic acid assay (BCA) protein assay kit (Beijing Kangwei Century Biotechnology Co., Ltd.). Proteins (20 µg per sample) were separated by electrophoresis and transferred to a PVDF membrane. After blocking, the membrane was incubated overnight with primary antibodies against GAPDH (ab8245, 1:1,000), Atrogin-1 (ab168372, 1:1,000), phosphorylated AKT (p-AKT, ab8805, 1:1,000), phosphorylated mTOR (p-mTOR, ab109268, 1:1,000) and mTOR (ab134903, 1:1,000), p38 mitogen-activated protein kinase (p38MAPK) (MD345, 1∶1,000). The following day, a secondary antibody (1:10,000, Proteintech, Cat: SA00001-) was added and incubated before washing with TBST. The analysis was conducted using a Western blot gel analyzer and ImageJ software.
Statistical Processing
Data from the experiments were analyzed using SPSS 21.0 and GraphPad Prism software. p < .05 refers to a significance.
Results
Modeling of Sarcopenia
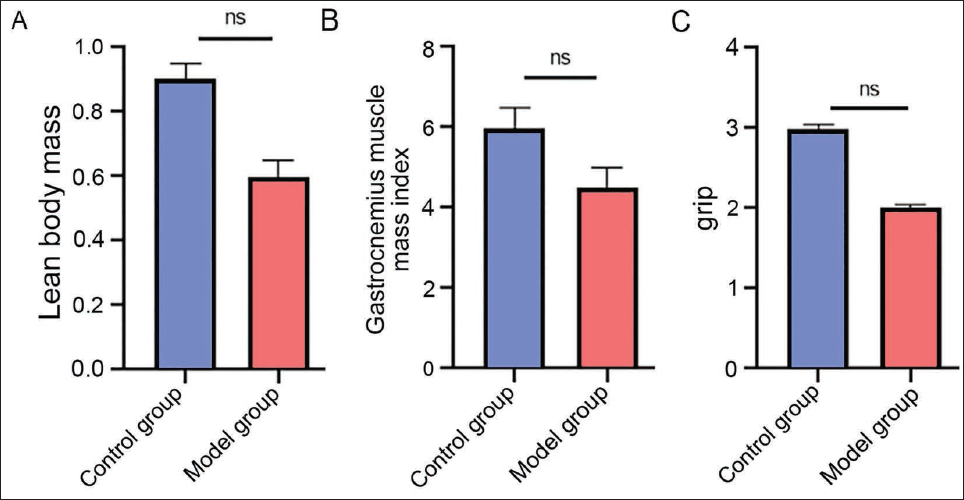
We constructed a mouse model of sarcopenia, and we found that after modeling, the percentage of lean mass, gastrocnemius muscle mass index, and grip strength were reduced in the model group (vs. control group, Figure 1A–1C), and the modeling was successful in all modeled mice.
APS has Outstanding Therapeutic Effects in Sarcopenia
The percentage of lean body mass, gastrocnemius muscle mass index, and grip strength of mice intervened with different concentrations of astragaloside were improved (vs. model group, Figure 2A–2C). At the same time, the performance was more obvious in high concentration (vs. medium- and low-concentration groups, Figure 2A–2C). It indicated that APS could have a certain therapeutic effect on sarcopenia, and it was concentration dependent.
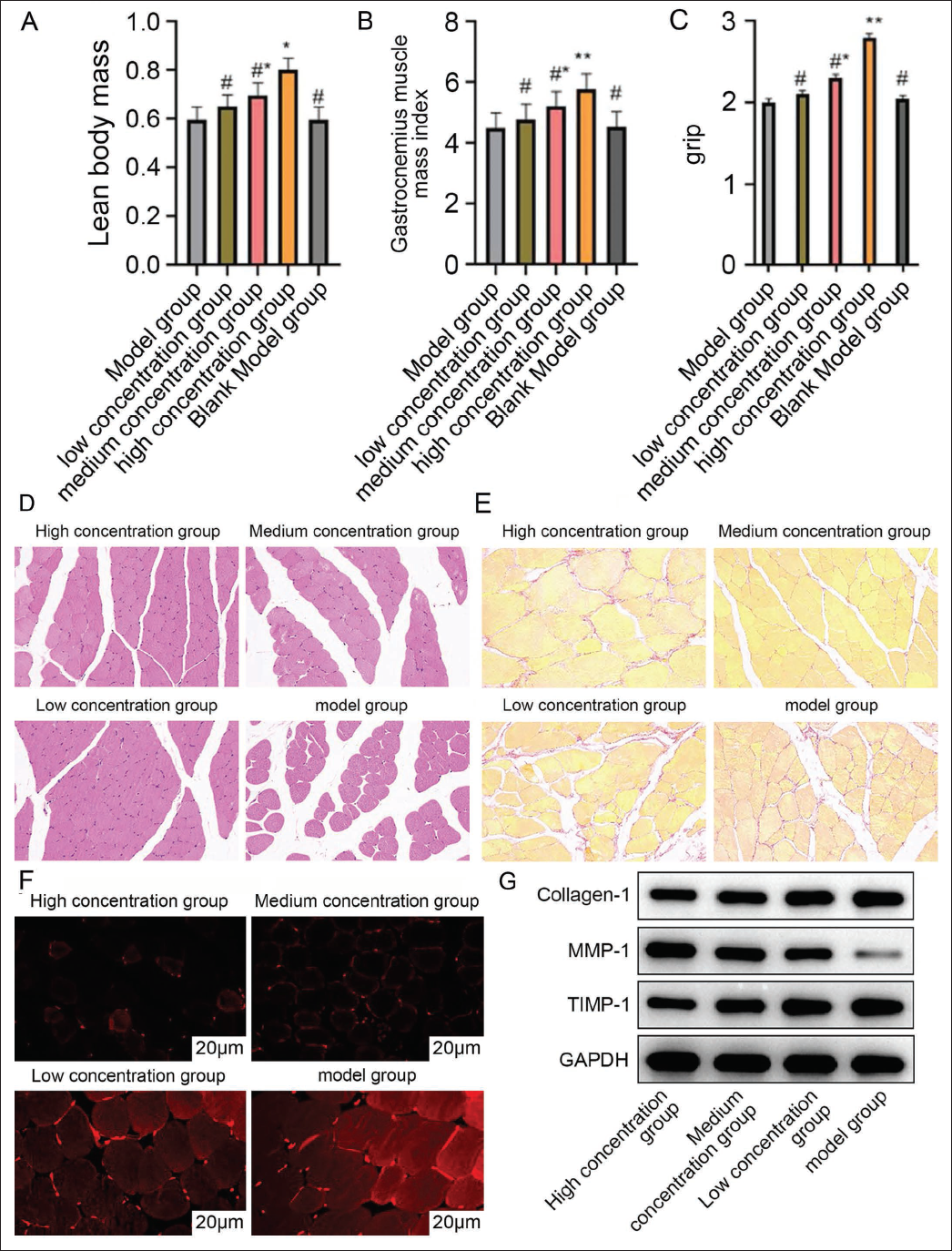
Further observation of H&E staining results showed that the cross-sectional area of muscle fibers in the high-concentration group was larger, the gap between myocytes was narrowed, the cell morphology was more regular, and the boundary was clear. The proliferation of collagen fibers in the muscle tissues of the mice was reduced in the Sirius red staining (vs. the medium-and low-concentration groups, Figure 2D and 2E), which indicated that Astragali polysaccharides were able to improve the degree of fibrosis of muscle tissues. The effect of Astragali polysaccharides was concentration dependent as well. In the high-concentration group, ROS, collagen-1, and TIMP-1 protein levels were significantly reduced. The content of MMP-1 was increased (vs. medium- and low-concentration groups, Figure 2F and 2G), indicating that Astragali polysaccharides were able to reduce the expression of collagen and inhibit the production of ROS, and that the therapeutic effect of high-concentration Astragali polysaccharides was the most significant in myasthenia gravis.
AKT-mTOR Pathway is Involved in APS-promoted Muscle Cell Proliferation with Reduced ROS Production
The study intervened with Astragali polysaccharide and AKT agonist SC79 on mouse muscle cells and found that Astragali polysaccharide Akt expression, as well as the growth and value-added rate of the cells, were increased. At the same time, the p-AKT messenger ribonucleic acid (mRNA) level decreased (vs. model group, Figure 3A and 3B), and the SC79 group showed the same trend (vs. model group, Figure 3A and 3B). In contrast, the wortmannin group cells exhibited the opposite trend (vs. model group, Figure 3A and 3B). This suggested that APS could promote accelerated muscle cell growth, and the process was associated with the activation of the AKT-mTOR pathway.
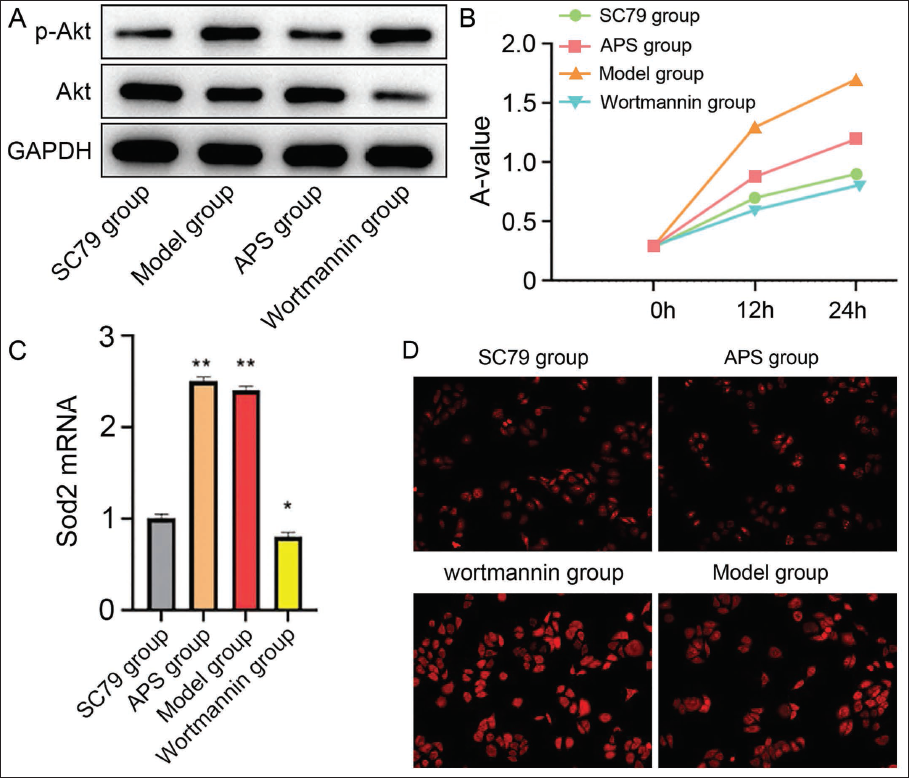
Further observation of superoxide dismutase 2 (Sod2) mRNA expression and ROS content revealed that Sod2 mRNA expression was significantly higher. ROS content was lower in Astragali polysaccharide group and SC79 group (vs. model group, Figure 3C and 3D), and the changes were more significant in the Astragali polysaccharide group (vs. SC79 group, Figure 3C and 3D). This suggests that APS can significantly increase the antioxidant capacity and downregulate the ROS level in mouse muscle cells.
APS activates the AKT-mTOR pathway for the treatment of sarcopenia, and ROS are involved in protein expression in mouse muscle cells by APS. Further intervention with the addition of SC79 to the cells in the APS group (APS + SC79 group) revealed that the cellular Sod2 mRNA expression level was further elevated and the ROS content was reduced (vs. APS group, Figure 4A and 4B) and the addition of the ROC inhibitor NAC to the cells in the APS group (APS + NAC group) exhibited the same trend (vs. APS group, Figure 4A and 4B). Whereas the addition of the AKT inhibitor wortmannin to the APS group (APS + wortmannin group) attenuated the ability of APS to upregulate the expression of Sod2 mRNA and increased the ROS content in the gastrocnemius muscle (vs. APS group, Figure 4A and 4B), the addition of the ROS agonist hydrogen peroxide to the APS + hydrogen peroxide group similarly showed an action inhibitory effect. In further protein level expression assays, it was found that collagen-1 and TIMP-1 expression was significantly reduced, and MMP-1 content was elevated in the APS + SC79 group as well as the APS + NAC group (vs. APS group, Figure 4C), while the APS + wortmannin group as well as the APS + hydrogen peroxide group showed the opposite trend (vs. APS group, Figure 4C). This result suggests that APS enhances the antioxidant function of muscle cells and exerts a therapeutic effect on sarcopenia by activating the AKT-mTOR pathway and that ROS influences this process.
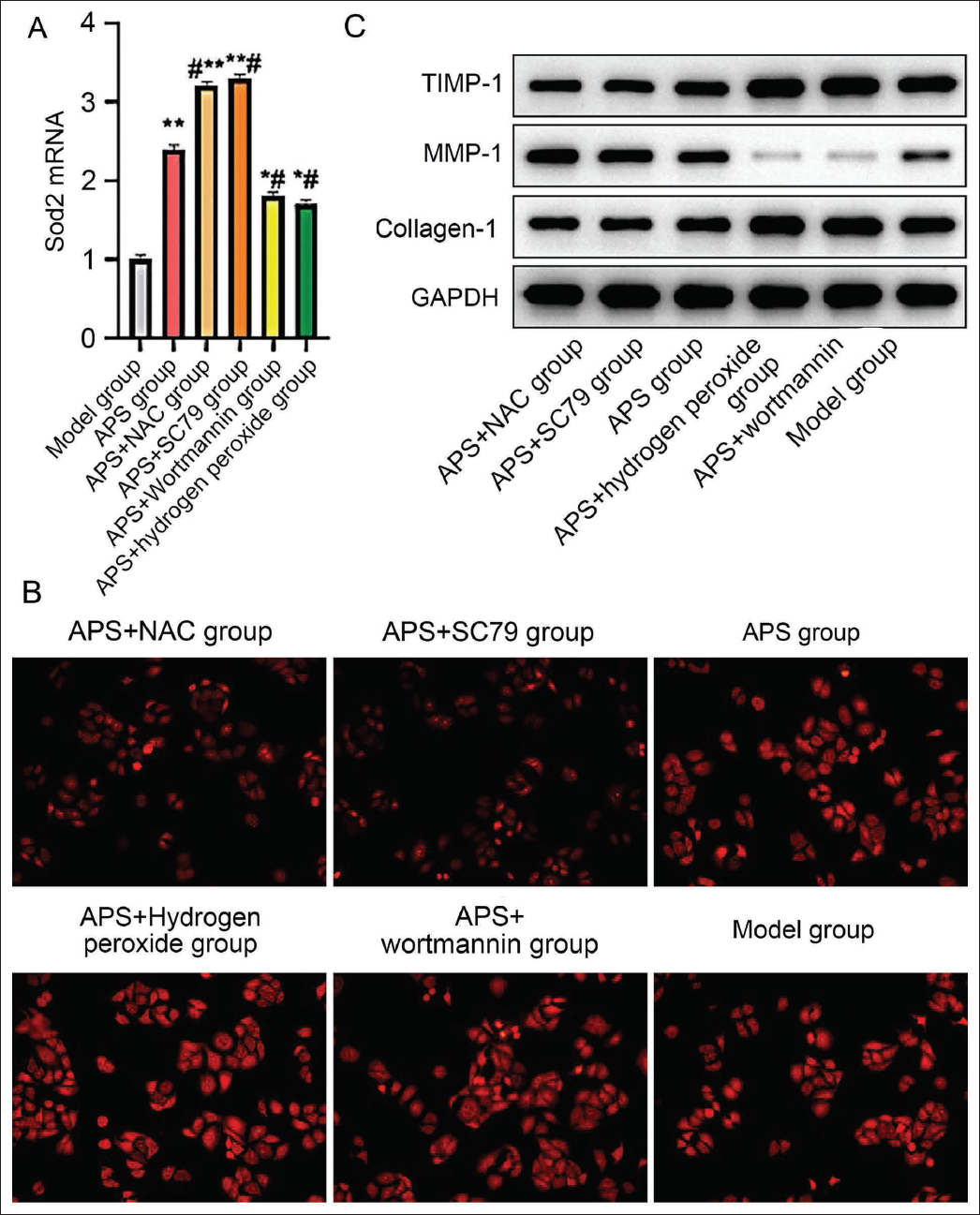
Activation of the AKT-mTOR Pathway by APS Inhibits ROS Production in the Treatment of Sarcopenia
In order to further verify that Astragali polysaccharide plays a therapeutic role in myasthenia gravis by activating the AKT-mTOR pathway to inhibit ROS, the study was further filled with ROS agonists and inhibitors on the basis of Astragali polysaccharide + SC79 group. It was found that, in Astragali polysaccharide + SC79 + NAC group, the cellular expression of collagen-1, TIMP-1, and p38MAPK proteins was significantly decreased, and MMP-1 content increased (vs. Astragali polysaccharide + SC79 group, Figure 5). In contrast, the Astragali polysaccharide + SC79 + hydrogen peroxide group showed the opposite trend (vs. Astragali polysaccharide + SC79 group, Figure 5). Continued addition of hydrogen peroxide intervention to the Astragali polysaccharide + SC79 + NAC group revealed elevated cellular collagen-1, TIMP-1, and p38MAPK protein expression. It reduced MMP-1 content in the Astragali polysaccharide + SC79 + NAC group (vs. Astragali polysaccharide + SC79 + NAC group).
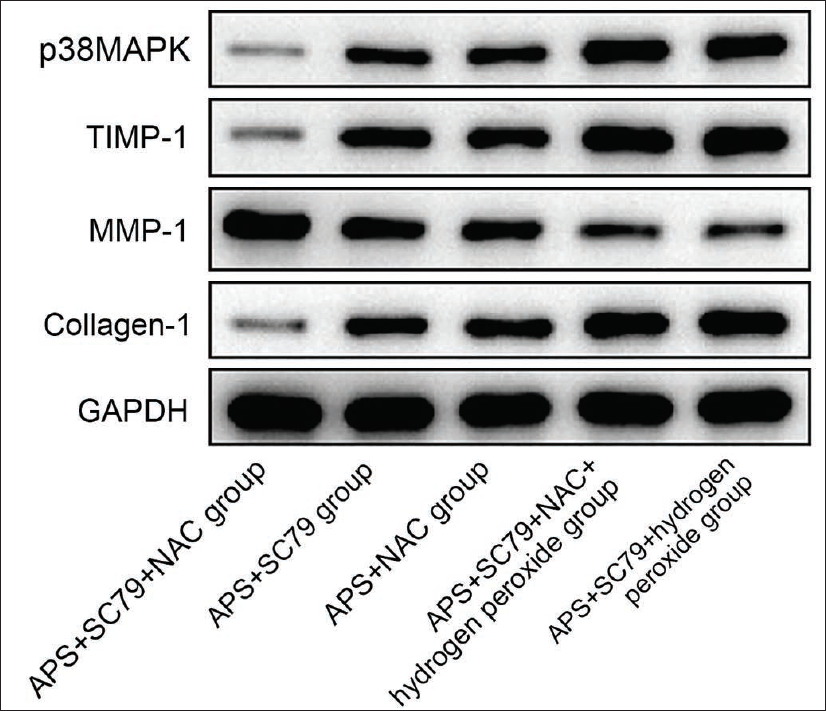
Discussion
Sarcopenia has garnered attention from the medical community in recent years. This condition not only impairs an individual’s ability to exercise but is also linked to various chronic diseases, such as cardiovascular disease, diabetes, and osteoporosis (Cho et al., 2022). Intracellular protein levels are crucial to the pathological process of sarcopenia, and changes play a key role (Tandon et al., 2021). Proteins, as the core components of muscle tissue, maintain a balance between synthesis and degradation, which is essential for the proper functioning of muscle tissue (Ravindran et al., 2023). This study successfully established a sarcopenia mouse model and observed significant reductions in grip strength and muscle mass in the model group. These declines may result from various factors, such as oxidative stress and inflammatory responses, which often lead to decreased intracellular protein levels and subsequent loss of muscle mass and strength. As a natural active ingredient, APS is highlighted for its role in regulating immunity and reducing inflammation. In this study, treatment with APS led to improvements in sarcopenia-related indicators in the model mice. This evidence suggests APS’s critical role in modulating immune and inflammatory responses, suggesting its potential utility in promoting muscle growth and repair. This offers new perspectives for the treatment of sarcopenia.
Studies have demonstrated that the pathogenesis of neurodegenerative and neuromuscular diseases can be influenced by factors such as oxidative stress and apoptosis. APS inhibits the signal transducer and activator of transcription 1 (STAT1)/I kappa B (IκB)/NF-κB pathways through interferon gamma (IFN-γ), thereby reducing NO production. This reduction has a therapeutic effect on neurodegenerative diseases associated with microglial inflammatory activation and suggests that APS might regulate mechanisms similar to those of neuromuscular diseases. Moreover, some studies have shown that APS can decrease the BCL-2/BAX ratio (You et al., 2022) and enhance the activation of caspase-3 by inhibiting AKT and NF-κB, leading to apoptosis in gastric cancer MGC-803 cells, indicating APS’s broad anti-tumor activity (Li et al., 2019).
Based on the above previous studies, we further used different concentrations of APS in the sarcopenia model. We found that: under the conditions of low, medium, and high concentrations, the percentage of lean mass, gastrocnemius muscle mass index, and grip strength of sarcopenic mice gradually increased; gastrocnemius muscle cell morphology and fibrosis degree gradually improved; collagen-1 and TIMP-1 expression was gradually decreased; and the MMP-1 content gradually increased, showing concentration dependence. In this study, the therapeutic effect of APS on sarcopenia was the best under the condition of high concentration. Extracellular matrix deposition occurs throughout the pathogenesis of sarcopenia, and a large amount of collagen-1 deposition suggests a decrease in skeletal muscle compliance and a weakening of the contractile ability of muscle fibers (Holwerda & van Loon, 2022). In contrast, MMP-1/TIMP-1 is an important pathway that regulates extracellular matrix metabolism, and TIMP-1 regulates MMP-1 action to activate collagen-1 collagenase and reduce collagen-1 deposition (Cabral-Pacheco et al., 2020). Additionally, the pathogenesis of sarcopenia is related to ROS. When ROS are overproduced, it leads to increased oxidative stress in the body, leading to cellular oxidative damage, activating lysosomes, and upregulating protease activity, which results in increased proteolysis in muscle tissue, leading to sarcopenia. In our study, high concentrations of Astragali polysaccharides were able to reduce the ROS content in gastrocnemius muscle tissues to a certain extent and led to decreased collagen-1 and TIMP-1 and increased MMP-1. This suggests that Astragali polysaccharide can regulate the balance of the MMP-1/TIMP-1 system to reduce collagen-1 deposition for the treatment of sarcopenia, and Astragali polysaccharide can also reduce ROS content in muscle cells to lower the level of oxidative stress.
ROS are closely related to the AKT-mTOR pathway, which can activate cellular autophagy to remove extracellular ROS and reduce apoptosis. And AKT-mTOR pathway is related to the pathogenesis of diabetes-related sarcopenia, it was found that Apelin-13 can promote PI3K and p-AKT expression in skeletal muscle, which makes PDK1 bind to AKT and then phosphorylates AKT’s Thr308 site, leading to partial activation of AKT, at the same time, mTORC2 phosphorylates Ser473, which further causes its fully activated (Zheng et al., 2021) leading to the development of sarcopenia. We found that AKT expression in muscle cells and the proliferation level of mouse muscle cells were increased after further intervention with the AKT agonist SC79 on the cells of the model group. At the same time, APS showed the same trend. This suggests that APS promotes muscle cell proliferation and antioxidant capacity through activation of PI3K/AKT signaling (Sha et al., 2023).
Zhang et al. (2022) suggest that AKT not only regulates Bad and caspase-9, affecting the cell apoptosis process, but also activates IKK and interacts with the NF-κB pathway to further regulate inflammatory and immune responses. During muscle protein degradation, NF-κB activation can trigger muscle atrophy and protein degradation. It can also interact with the AKT-mTOR pathway, impacting the balance of protein synthesis and degradation (Peng et al., 2022). Activation of NF-κB further decreased protein synthesis in muscle cells by inhibiting the AKT-mTOR pathway, thus exacerbating muscle atrophy. Therefore, inhibiting the NF-κB pathway and activating the AKT-mTOR pathway could effectively reduce the degradation of muscle proteins, thereby preserving muscle mass and function. The Sod2 gene is an important member of the superoxide dismutase family, which is an important factor involved in ROS metabolism, and its expression is related to the antioxidant capacity of cells. In this experiment, the expression of the Sod2 gene in APS group was higher than in the model group, and ROS was reduced; this result implies that APS can improve the antioxidant capacity of mouse muscle cells in the model of sarcopenia to a certain extent and reduce the level of oxidative stress.
In the present study, after the addition of the ROS inhibitor NAC to the cells in the APS group, the expression of Sod2 mRNA in mouse muscle cells was further increased, while the ROS content was reduced and collagen-1 and TIMP-1 expression was significantly reduced and the content of MMP-1 was elevated, which suggests that the therapeutic effect of APS on sarcopenia is amplified when ROS is inhibited. This indicates that when ROS is inhibited, the therapeutic effect of APS on sarcopenia is amplified, suggesting that ROS are involved in the regulation of the MMP-1/TIMP-1 system by APS. ROS is believed to act as a “second messenger” by activating the p38MAPK signaling pathway, which leads to the upregulation of activator protein-1 (AP-1) expression and further upregulation of MMPs, resulting in an increase in collagen-1 synthesis (Hajam et al., 2022). In our study, the addition of NAC to the Astragali polysaccharide + SC79 group revealed that the cellular collagen-1, TIMP-1 and p38MAPK protein expression was further reduced, while the MMP-1 content was further increased and the addition of the ROS agonist hydrogen peroxide revealed that the PI3K/AKT pathway activated by Astragali polysaccharides was blocked, resulting in an impaired intracellular ROS The scavenging mechanism was impaired and the ROS level increased. This suggests that the ameliorative effect of APS on sarcopenia is indeed related to the activation of the AKT-mTOR pathway and reduction of ROS. In other words, the inhibition of AKT diminishes the protective effects of APS, leading to increased intracellular ROS and reduced antioxidant capacity. Sod2, which efficiently removes superoxide anion free radicals from cells and protects against oxidative damage, also saw reduced mRNA expression following wortmannin application. This reduction in Sod2 mRNA levels signifies a diminished intracellular antioxidant capacity, underscoring the pivotal role of the AKT pathway in maintaining intracellular redox balance (Gan et al., 2021; Hou et al., 2023) that cells possess multiple negative feedback mechanisms. When AKT/GSK-3B is overactivated, astragaloside IV (AS-IV) increases the antioxidant capacity of cells and inhibits the production or promotes the dephosphorylation of p-AKT and p-GSK-3β proteins. It also enhances TRPC6 expression and opens Ca2+ channels, significantly increasing intracellular free calcium concentration and providing a regulatory and protective effect against sarcopenia. Additionally, other traditional Chinese medicine extracts have shown efficacy in improving sarcopenia. For instance, naringin reduces ROS levels by activating AKT, thereby promoting muscle cell proliferation and playing a crucial role in treating disuse sarcopenia (Kma & Baruah, 2022); in the context of sarcopenia treatment, Migu Capsule was reported to alleviate muscle protein degradation by inhibiting the NF-κB pathway and activating AKT-mTOR, thus mitigating muscle atrophy and promoting protein synthesis in muscle cells (Huang et al., 2021). These findings further validate the results of our research and highlight the potential of traditional Chinese medicine in the treatment of sarcopenia.
Conclusion
In summary, this study found that Astragali polysaccharide has a significant proliferation-promoting effect on mouse muscle cells, which mainly plays a role by activating the AKT-mTOR pathway, decreasing ROS to enhance its antioxidant capacity, downregulating the level of oxidative stress, and regulating the balance of MMP-1/TIMP-1 to reduce the deposition of collagen-1. These findings not only elucidate the potential mechanism of APS in treating sarcopenia but also highlight its broad application prospects for improving this condition. We aim to assess the mechanisms of APS and other traditional Chinese medicines in the treatment of sarcopenia in the future. Future research may also explore combining APS with other traditional Chinese medicine extracts or treatment methods to enhance therapeutic outcomes.
Footnotes
Abbreviations
AKT: Protein kinase B; APS: Astragalus polysaccharide; MMP-1: Matrix metallopeptidase 1; mTOR: Mammalian target of rapamycin; PCR: Polymerase chain reaction; p38MAPK: p38 mitogen-activated protein kinase; PI3K: Phosphatidylinositol 3-kinase; ROS: Reactive oxygen species; SOD2: Superoxide dismutase 2.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethics committee of the Naval Medical Center of PLA.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
