Abstract
Background
Resveratrol (RV) plays a crucial part in regulating the development of the nervous system. Tet methylcytosine dioxygenase 2 (TET2) has an important physiological significance in the nervous system. TET2 pertains to the deoxyribonucleic acid (DNA) hydroxylase family. Sirtuin 1 (SIRT1) plays a part in the development of Parkinson’s disease (PD) in patients.
Objectives
This study will verify the effect of RV on SIRT1 activity and analyze whether this effect is related to changes in TET2 expression levels, further exploring its potential molecular mechanisms.
Materials and Methods
The SH-SY5Y cells were divided into control, RV treatment, 1-methyl-4-phenylpyridinium (MPP+) treatment, and RV + MPP+ combination treatment groups. Neuron TET2 expression was tested in SH-SY5Y cells treated with SRT1720 or EX-527, and SIRT1 agonists and inhibitors were used to detect TET2 messenger ribonucleic acid (mRNA) levels. After treatment with EX-527 or RV, endogenous acetylated TET2 was measured by coimmunoprecipitation (CO-IP). After treatment with dissolved in dimethyl sulfoxide (DMSO) or EX-527 for 24 h, cells were collected after adding MG-132 to the culture medium to detect protein levels of polyubiquitination.
Results
SIRT1 activity is negatively linked to TET2. RV activates endogenous SIRT1 and decreases TET2 levels and cyclin-dependent kinase inhibitor 2A (CDKN2A) DNA hypermethylation, thereby upregulating pRb. TET2 inhibitors have inhibitory effects on endogenous TET2 enzyme activity.
Conclusion
RV alleviates MPP+-induced cell damage by activating SIRT1, confirming a negative correlation between SIRT1 activity and TET2 protein levels, and affects its stability by promoting TET2 deacetylation and ubiquitination degradation, regulating DNA methylation. In addition, SIRT1 regulates TET2 to inhibit cell cycle arrest, affecting neuronal growth and apoptosis. These findings provide a theoretical basis for new treatment methods based on the SIRT1/TET2 pathway, demonstrating the potential of RV in the treatment of PD and bringing new hope for improving patients’ quality of life.
Introduction
Parkinson’s disease (PD) is a complex neurological disorder, whose pathogenesis includes mitochondrial dysfunction and oxidative stress damage (Eguchi et al., 2021; Koga et al., 2021). Natural polyphenols have multiple functions to improve bodily functions. Resveratrol (RV) is a natural polyphenol that not only has antioxidant properties, regulates Sirtuin 1 (SIRT1) activity, affects tet methylcytosine dioxygenase 2 (TET2) expression and its related deoxyribonucleic acid (DNA) methylation status, but also inhibits neuroinflammation and other mechanisms closely related to the pathogenesis of PD. There are many health benefits of anti-aging, including improving metabolism, protecting the nervous system, and preventing cancer (Corpas et al., 2019; Meng et al., 2020). Although the potential of RV in neuroprotection has been confirmed in multiple in vitro studies, research on its specific effects and mechanisms in nerve cells is still limited (Rasheed et al., 2020).
The process of DNA demethylation is crucial for neural development, and the TET enzyme family plays an important role in this process (Jiang, 2020; Zhang et al., 2020). Recent studies have shown that TET2 expression is upregulated in PD patients, and TET2 deficiency is associated with changes in cytosine modifications, which is opposite to the characteristics of PD neurons (Kharat et al., 2020; Zhang et al., 2017). Therefore, regulating TET2 expression or activity may become a new strategy for treating PD.
On the other hand, SIRT1, as a histone deacetylase, participates in the regulation of various cellular functions, such as inflammation, cell proliferation, and differentiation. The latest research reveals that SIRT1 can deacetylate TET2, and this process is particularly important in progenitor cells (Senyildiz et al., 2017). In addition, SIRT1 has been shown to regulate synaptic plasticity and memory formation (Jiang et al., 2020). Given that RV is an effective activator of SIRT1, this study explores how RV downregulates TET2 expression by activating SIRT1, which is of great significance for the development of PD, by providing a new treatment method.
Materials and Methods
Experimental Materials
Resuscitation SH-SY5Y neuroblastoma cells (purchased from Shanghai Qiming Biotechnology Ltd., item number QMS-h1237), cryopreserved in our laboratory, were developed in Dulbecco’s modified medium (DMEM, Gibco) containing 10% fetal bovine serum (Gibco, Brazil). They maintained the atmospheric oxygen level and a 5% growth in a CO2 incubator. The concentration of 1-methyl-4-phenylpyridinium (MPP+) (Sigma–Aldrich) is 25 mM. They are divided into the MPP+ group and the phosphate-buffered saline (PBS) control group. RV is prepared and dissolved in dimethyl sulfoxide (DMSO).
SRT1720 (0.1 mM) (MedChemExpress) and EX-527 (0.2 mM) (MedChemExpress) were treated with SH-SY5Y cells for the experiment.
Real-time Quantitative Polymerase Chain Reaction (PCR)
Total ribonucleic acid (RNA) was extracted using the RNeasy kit, and 2 µg of purified RNA was reverse transcribed into complementary DNA (cDNA), and quantitative reverse transcription polymerase chain reaction (qRT-PCR) was performed using the ABI7500 real-time PCR quantitative SYBR Green PCR kit. The qRT-PCR cycle was pre-denatured at 95°C for 3 min, then denatured at 95°C for 5 s and annealed at 60°C for 30 s, for 35 cycles, and finally analyzed at 60°C–95°C. The upstream primer of TET2 is 5′-ATTGATGCGGAGTAG-3′, and the downstream primer is 5′-AGTGGGTCTCCTTTCTAAC-3′; the upstream primer of SIRT1 is 5′-CCAAGCUAAUTT-3′, and the downstream primer is 5′-AUUACUCUUGGTT-3′; the upstream primer of glyceraldehyde 3-phosphate dehydrogenase (GAPDH) is 5′-CGTGGAAACCTGCAAGTATG-3′, and the downstream primer is 5′-GGTGAAGAATGGGAGTCT-3′. The qRT-PCR results were quantified using the 2−∇∇Ct method. Choose GAPDH as the reference gene. All gene expression levels are based on GAPDH measured in parallel.
Analysis of the Influence of Cell Proliferation
In the quantitative proliferation experiment, the number of cells is planted in a regular medium. The cells in each group were treated for 12, 24, and 48 h, respectively, after treatment according to each action condition. The cell proliferation evaluation was analyzed on the 6th day using an automatic cell counter, and the absorbance was analyzed using a spectrometer to record the value at 495 nm.
Cell Ubiquitination Detection
Cells were planted in a six-well plate. Lipofectamine 3000 was used to transfect the hemagglutinin-tagged ubiquitin (HA-Ub) plasmid into cells. The supernatant was incubated with anti-rabbit TET2 (Abcam), and protein A/G Sepharose beads were added overnight for Western blot detection.
Cell Cycle Detection
Use a specific concentration of the chemotherapy group to treat the cells for a specified time. After the cells were washed, propidium iodide (PI) (50 µg/mL) was added, and staining was performed at 37°C for 30 min.
Apoptosis Measurement
Use a specific concentration of the experimental group to treat the cells for a specified time. Collect cells and adherent cells, stain with fluorescein isothiocyanate (FITC, 2.5 mg/mL) and PI (100 µg/mL), protect from light at 4°C for 30 min, and use the average of three independent experiments for apoptosis; the value ± standard deviation (SD) is expressed. Flow cytometry reagents were purchased from Miltenyi Biotec, Germany.
Western Blot
The cells were cultured in a cell plate, and radioimmunoprecipitation assay (RIPA) buffer (Shanghai Shenggong Biological Co., Ltd.) was used to lyse the total cell protein, and the buffer (Shanghai Shenggong Biological Co., Ltd.) was used to prepare a 1:1,000 primary antibody (anti-SIRT1, anti-TET2, anti-GAPDH) incubate in an incubator at 4°C overnight. The GAPDH antibody (CST Biotech Co., Ltd., USA) was used as an internal control. The next day, after the secondary antibody was incubated for 1 h, the membrane was washed again and then colored. Finally, use the imaging system for band analysis.
Protein Ubiquitination Reaction
Lipofectamine R 3000 was added to the cells in the six-well plate culture dish to make the cells transfected with the HA-Ub plasmid. It was eluted after incubation, adding a buffer at 95°C for 5 min and then centrifuge. The supernatant was then used for Western blotting (WB).
Statistical Analysis
All values are expressed as the mean ± standard error of the mean (SEM) of at least three separate experiments. The Student’s t-test was used to compare the two groups. All data are expressed as the Mean ± SD of at least three separate experiments. p < .05 indicates that the difference is statistically significant.
Results
RV Reduces the Damage Induced by MPP+
Studies have shown that SIRT1 decreases markedly after 24 h of MPP+ induction (Figure 1A). By detecting the cell viability, it was found that after 12 h of RV pretreatment, apoptosis induced by MPP+ was markedly reduced (Figure 1B, p < .05). After treating SH-SY5Y cells with RV and EX-527, SIRT1 protein levels were examined. The results stated that RV and EX-527 did not affect SIRT1 protein levels (Figure 1C, p < .05). Research has shown that SIRT1 protein levels are significantly reduced after MPP+ treatment, while RV pretreatment can significantly reduce cell apoptosis. This suggests that RV may counteract MPP+-induced neuronal damage by activating SIRT1. Specifically, RV can enhance the function of SIRT1, thereby protecting neurons from the effects of PD-related toxins.
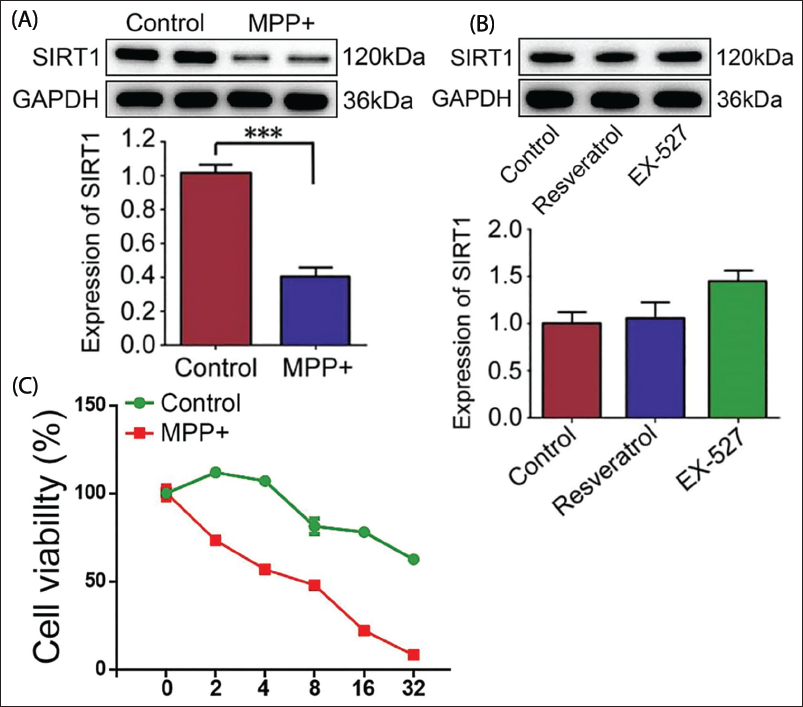
SIRT1 Enzyme Activity Is Negatively Correlated with TET2 Protein
They were separated into control, RV, MPP+, and RV + MPP+ combination groups. The results stated that TET2 expression was upregulated in MPP+ (Figure 2A). SIRT1 agonists and inhibitors do not influence TET2 messenger ribonucleic acid (mRNA) (Figure 2B, p > .05). After treatment with different concentrations of SRT1720, TET2 protein expression was remarkably reduced (Figure 2C). This result indicates a negative correlation between SIRT1 activity and TET2 expression, suggesting that increasing SIRT1 activity can alleviate symptoms of PD by downregulating TET2 expression. This is of great significance for exploring the mechanism of action of the SIRT1/TET2 pathway in PD.
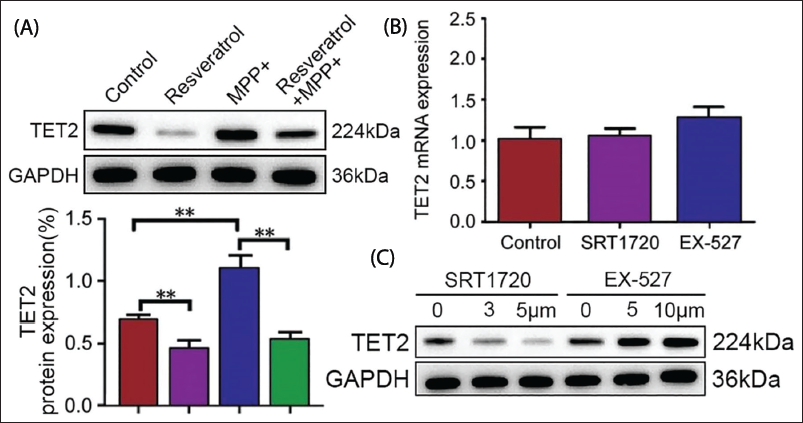
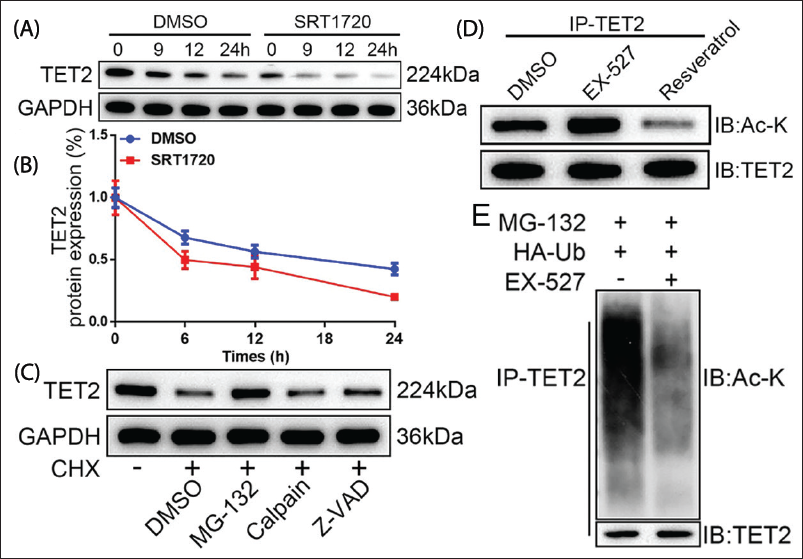
TET2 Degradation Was Monitored by SIRT1-induced TET2 Deacetylation
It can be seen that SIRT1 cannot influence the transcription of TET2, but affects its protein level. Cycloheximide (CHX) was used to inhibit protein synthesis, and the results showed that TET2 was stably expressed, and a small amount of protein was still present (Figure 3A). TET2 was almost degraded within 24 h (Figure 3B, p < .05), indicating that the increase in SIRT1 enzyme activity made the TET2 protein unstable. Our results show that TET2 is stabilized by the proteasome inhibitor MG-132, not by calpain or caspase inhibitors (Figure 3C). With the participation of MG-132 and SIRT1, the abundance of TET2 protein disappeared. EX-527 treatment increased TET2 acetylation (Figure 3D), while markedly reducing TET2 ubiquitination (Figure 3E). This result indicates that increased SIRT1 activity leads to the instability of TET2 protein, and TET2 stability can be restored through the proteasome inhibitor MG-132. EX-527 treatment increased TET2 acetylation while reducing TET2 ubiquitination. Therefore, SIRT1 not only affects the expression level of TET2 but also regulates its protein stability and degradation process. Especially, SIRT1-mediated TET2 deacetylation promotes TET2 ubiquitination and degradation, which is crucial for regulating DNA methylation status.
Sirtuin 1 (SIRT1)-induced Tet Methylcytosine Dioxygenase 2 (TET2) Deacetylation Promotes TET2 Degradation Through the Ubiquitin-proteasome Pathway. (A) The Western-blot Detection of TET2 Protein Expression. (B) TET2 Protein (C) The Western-blot Detection of TET2 Protein Expression Under Different Conditions. (D) EX- SH-SY5Y Cells Were Treated with 527 or Resveratrol for 24 h to Coimmunoprecipitation (CO-IP) to Detect Endogenous Acetylated TET2. (E) Western Blot to Detect the Level of TET2 Protein Polyubiquitination.
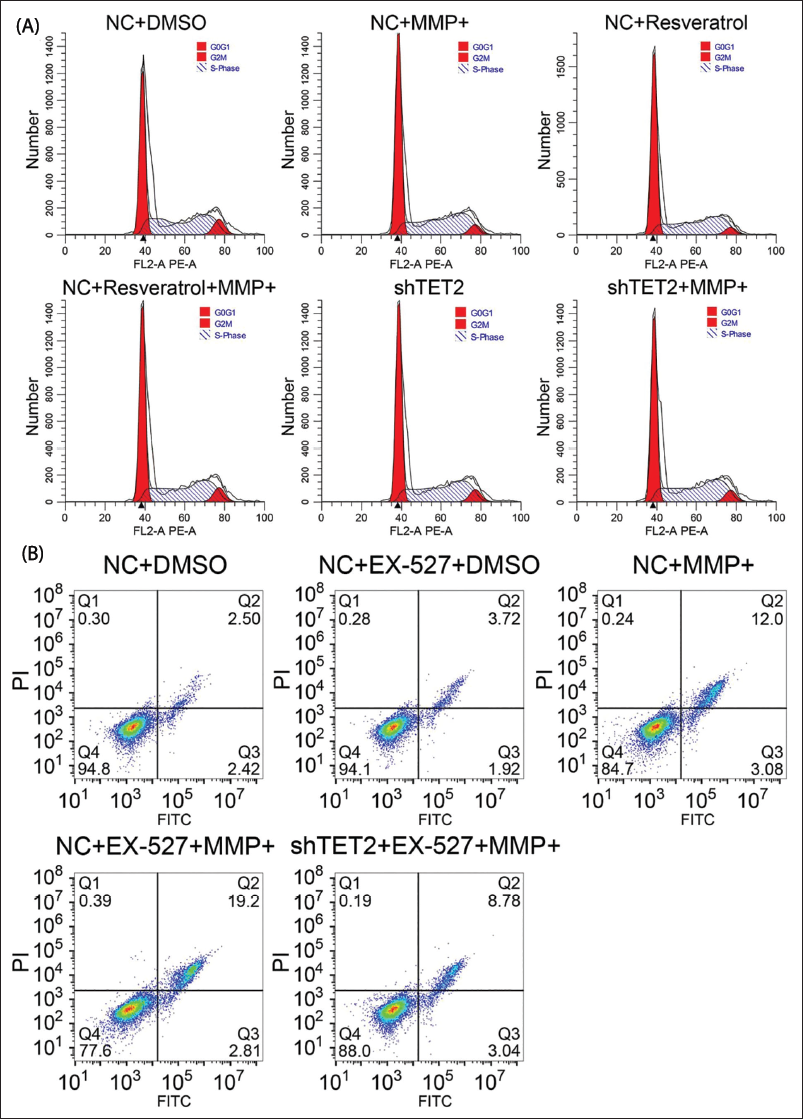
SIRT1 Affects Neuron Growth and Apoptosis Through TET2
MPP+ affects the cell cycle progression; S-phase cells are significantly reduced, and G0/G1- and G2/M-phase cells increase. In addition, after RV treatment and TET2 knockout, this effect of MPP+ was significantly reduced (Figure 4A). When MPP+ treated EX-527 inhibited SIRT1 enzyme activity, the proportion of S-phase cells was less than that of MPP+ treated alone. In addition, the number of cells increased remarkably. The activity of SIRT1 was inhibited, and at the same time, TET2 was reduced, and the exacerbation of apoptosis caused by EX-527 was increased (Figure 4B). This result indicates that MPP+ treatment leads to cell cycle arrest, especially in the case of the reduced number of S-phase cells, while this effect is significantly weakened after RV treatment and TET2 knockout. In addition, when EX-527 is used to inhibit SIRT1, the proportion of S-phase cells further decreases, accompanied by more severe cell apoptosis. Therefore, SIRT1 may control cell cycle progression by regulating TET2, thereby affecting neuronal survival. Specifically, activating SIRT1 or inhibiting TET2 can promote neuronal health and prevent cell death caused by PD-related cell damage.
Sirtuin 1 (SIRT1) Affects Neuron Growth and Cell Apoptosis Through Tet Methylcytosine Dioxygenase 2 (TET2). (A) The Flow Cytometry Detection of Cell Cycle. (B) The Flow Cytometry Detection of Cell Apoptosis.
Discussion
It took a long time to explore the treatment for Parkinson’s. The pathogenesis of PD has not yet been fully elucidated. Drugs and deep brain stimulation cannot stop the progression of PD (Etchegaray et al., 2015; Wang et al., 2020). The relationship between methylation and acetylation is not clear in PD. In our exploration, we found that RV activates endogenous SIRT1 to downregulate TET2 degradation through ubiquitination.
A neuron is a cell after mitosis. It is defined as a mature cell that has stopped replicating and left the cell cycle permanently (Herrup & Yang, 2007; Rashidian et al., 2007). However, under certain conditions, cells after mitosis can re-enter the cell cycle, for example, neurons after damage to the central nervous system, including early Parkinson’s (Bang et al., 2021). Restricting cell cycle re-entry seems to help DNA repair. Polyploid neurons also appear to be functionally active, which suggests that they may be helpful in preventing apoptosis and may have significant advantages (Nagy et al., 1997; Xu et al., 2018). The beneficial influence of SIRT1 on Parkinson’s neurons involves many aspects. TET2 remarkably attenuates cell cycle arrest and apoptosis (Li et al., 2020). Our study showed the pathogenesis of TET2 in Parkinson’s. It was found that these TET2 markedly reduced the expression of cyclin-dependent kinase inhibitor 2A (CDKN2A). The number of cells increased when treated with MPP+. In addition, after RV treatment and TET2 knockout, the effect of MPP+ was significantly reduced. Decreasing TET2 basically alleviates the exacerbation of cell cycle arrest. It has been probed that RV and TET2 knockdown can upregulate Bcl-2. p16INK4a reduces Bcl-2 protein and mRNA after incubation for 48 h, suggesting that p16INK4a recombination at least partly plays a role in changing the expression of Bcl-2 mRNA (Obexer et al., 2009).
As we all know, RV or other SIRT1 activators have been shown to activate enzymes through allosteric sites near the catalytic domain directly. The neuroprotective effect of RV has been confirmed, but it may also attack non-SIRT1 to take part in biological activity (Gomes et al., 2018; Maugeri et al., 2018; Sarubbo et al., 2018; Zhang et al., 2019). In addition, it was stated that the downregulation of TET2 also leads to an increase in SIRT1. Therefore, our findings indicate that MPP+ reduces SIRT1, promotes TET2 hyperacetylation, and increases TET2 stability. The TET2 protein induces DNA demethylation to inhibit the elevated protein level of p16INK4a, thereby downregulating the pRb protein, which allows SH-SY5Y cell cycle arrest and induces apoptosis. Therefore, TET2 regulation of SIRT1 is a promising treatment for PD. It was further shown that SIRT1 can take part in the cell cycle by regulating TET2 in PD progression. Also, our study focused on in vitro exploration, but more mouse models are needed to verify whether in vivo studies will have consistent conclusions.
Conclusion
This study revealed that the activation of SIRT1 by RV can downregulate TET2 acetylation through ubiquitination. The SIRT1/TET2 pathway can be a promising intervention for PD.
Footnotes
Abbreviations
CDKN2A: Cyclin-dependent kinase inhibitor 2A; SIRT1: Sirtuin 1; TET2: Tet methylcytosine dioxygenase 2.
Acknowledgments
This work was supported by the Huanggang Science and Technology Bureau (No. YBXM20230018-3).
Authors Contribution
All authors participated in the completion of the manuscript. Yulong Xiong and Yuangao Liao were responsible for drafting the manuscript, while Xuebin Xia and Jiajun Wang provided the overarching direction and guidance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethnic committee of Huanggang Central Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
This study is a cell experiment and did not involve patient samples, so patient consent is not applicable.
