Abstract
Background
In Alzheimer’s disease (AD), β-amyloid protein and the blood–brain barrier (BBB) are important factors in its pathogenesis. Research has found that resveratrol has a certain therapeutic effect on AD. It inhibits the expression of macrophage inflammatory protein-1α (MIP-1α), thereby reducing the number of white blood cells crossing the BBB and protecting the integrity.
Objectives
This article will explore the role of resveratrol in AD.
Materials and Methods
The amyloid precursor protein (APP)/presenilin 1B (PS1) dementia mouse model was successfully constructed. Morris water maze test, total superoxide dismutase (SOD) activity detection, catalase (CAT) activity detection, glutathione reductase activity detection, lipid oxidation detection, Western blot detection of Aβ and CCR5 protein expression, and polymerase chain reaction (PCR) detection of MIP-1α and CCR5.
Results
The APP/PS1 mouse model successfully mimicked the pathological characteristics of AD patients in terms of amyloid plaque formation and cognitive function decline based on the established APP/PS1 mouse model. After adding various doses of resveratrol and culturing for a period of time, it was detected that resveratrol affected the expression of β-amyloid protein (Aβ) in the APP/PS1 mouse model and reduced Aβ protein deposition, thereby regulating MIP-1α expression and stabilizing blood circulation. Brain barrier, further studies have found that Aβ expression is proportional to the increase in MIP-1α. Still, due to the decrease in the expression of BBB-related proteins, the BBB function will be weakened to a certain extent. By comparing the Aβ and resveratrol groups injected into the hippocampus of normal mice, in the Aβ1-42 group, MIP-α and CCR5 were highly expressed, and occludin and claudin-5 were low in protein expression.
Conclusion
Aβ can increase the expression of MIP-α, downregulate occludin and claudin-5 proteins, thereby destroying BBB stability.
Keywords
Introduction
Alzheimer’s disease (AD) is partly caused by the deposition of β-amyloid peptide, which causes neuronal death and cognitive decline (Swanson et al., 2021). The blood–brain barrier (BBB) is an important structure that protects the brain. The occurrence of AD leads to its destruction. Therefore, protecting BBB integrity is one of the important strategies for treating AD (Qiu et al., 2021). Resveratrol has a certain protective effect on nerves, and macrophage inflammatory protein-1α (MIP-1α) is involved in inflammation and immune responses (Rudnicka et al., 2021). Studies have shown that resveratrol can improve the cognitive function of AD rats to a certain extent (Wong et al., 2020). Therefore, this study aimed to explore the mechanism of resveratrol in regulating MIP-1α to stabilize the BBB in AD mice through the β-amyloid peptide.
Resveratrol not only has the functions of an antioxidants, scavenging free radicals, and protecting and delaying aging; it can also regulate the level of cholesterol in the blood and reduce the risk of cardiovascular disease. In addition, it can also exert anti-inflammatory and anti-tumor effects (Meng et al., 2021). Resveratrol can exert anti-cancer effects by inhibiting tumor cell proliferation and has inhibitory effects on breast cancer, lung cancer, and colon cancer (Feng et al., 2022); it can also lower blood lipids, inhibit platelets, and help prevent the occurrence of cardiovascular diseases (Przybylska & Tokarczyk, 2022). In addition, it has also been studied in AD. Studies believe that resveratrol can play a certain preventive role in immune diseases through anti-inflammatory and antioxidant mechanisms (Chen & Holtzman, 2022). Studies have found that Aβ not only promotes the expression of inflammatory factors such as monocyte chemoattractant protein-1 (MCP-1), cyclooxygenase-2 (COX-2), and intercellular adhesion molecule-1 (ICAM-1) but also activates the inflammatory response and promotes the release of inflammatory mediators, such as MCP-1 or COX-2. The production and infiltration of inflammatory cells (Yang et al., 2020) and pathways such as ERK, JNK, and p38 promote the release of cytochrome C by activating caspases, Bax, Bak, and other apoptotic proteins, thereby triggering the apoptosis process (Cheng et al., 2020). In addition, the activation of growth factor receptors such as epidermal growth factor receptor (EGFR) and insulin-like growth factor receptor 1 (IGFR-1) can not only affect the proliferation of nerve cells and other biological behaviors but also lead to neurodegenerative diseases to a certain extent (Zhang et al., 2022). In this process, the participation of toll-like receptor (TLR), peroxisome proliferator-activated receptor (PPAR), and NOD-like receptor pyrin domain-containing protein (NLRP) signaling pathways regulates the expression of MIP-1α and the inflammatory response to a certain extent (Qiang et al., 2021). It not only induces the expression of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), and other antioxidant enzymes but also inhibits interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), MIP-1α, and others, thereby playing an important role in anti-oxidation (Barnes, 2020). It further protects cells from oxidative stress damage and maintains normal cell functions (Jiao et al., 2021).
Studies have found that MIP-1α not only aggravates neuronal damage by promoting the aggregation of Aβ but also causes damage to the BBB (Renthal et al., 2020). Resveratrol can inhibit the expression of MIP-1α to a certain extent and reduce the promotion of Aβ aggregation by MIP-1α, thereby reducing the damage to neurons and the destruction of the BBB in patients (Hanin et al., 2023); in addition, there are studies that believe that resveratrol further inhibits the inflammatory response by inhibiting MIP-1α induced by Aβ, thus protecting the BBB integrity and alleviating AD symptoms to a certain extent (Hecker et al., 2022). Therefore, multiple signaling pathways are involved in the mechanism of resveratrol regulating MIP-1α to stabilize the BBB in Alzheimer’s mice through β-amyloid peptide. Although there have been relevant studies, further research is needed. Therefore, the present study aims to explore the specific mechanism of resveratrol on AD to provide more approaches for the treatment of AD.
Materials and Methods
Experimental Materials
Instruments, Reagents, and Animals
Resveratrol (Purity: >99%; No.: 501-36-0) were purchased from Henan Alpha Chemical Ltd.; real-time polymerase chain reaction (RT-PCR) primers, reverse transcription (RT) kits, and real-time reagents were purchased from the American Axygen Company; Western blot (WB)-related chemical reagents were purchased from Shanghai Bolson Biotechnology Ltd.; enhanced chemiluminescence (ECL) reagents were purchased from Beijing Biotech Ltd.; Qinglian double antibody was purchased from HyClone Company, USA; and the rabbit anti-human pERK1/2 monoclonal antibody was purchased from Cell Signaling Technology (Danvers, MA, USA).
Animals
Forty double transgenic mice and 40 C57 wild-type mice were purchased from Jiangsu Synergy Pharmaceutical Bioengineering Ltd. (Nanjing, Jiangsu, China).
Establishment of Amyloid Precursor Protein (APP)/Presenilin 1B (PS1) AD Mouse Model and Group Intervention
Construction of APP/PS1 AD Mouse Model
The test sample of double transgenic mice (APP/PS1) was raised normally, and after 1 week, they were crossed with C57 wild-type mice to obtain genetic offspring (C57 APP/PS1). The model construction method is consistent with the research report of Ma et al. (2024). The APP/PS1 dementia mouse model established by the above method was divided into a control group (C57 wild type) and a model group (C57 APP/PS1). All mice underwent brain PET/CT imaging and behavioral observation after the successful modeling.
APP/PS1 AD Mice Were Divided into Groups
After successful modeling, 9-month-old AD models were selected and randomly divided into the Res group and the model group. Mice in the Res group were further divided into a high-dose group (ResH, 0.5 µmol/L of resveratrol injected intraperitoneally, 200 µg/kg) and a low-dose group (ResL, 0.5 µmol/L of resveratrol injected intraperitoneally, 100 µg/kg), once a day. Mice in the model group were intraperitoneally injected with the same dose of phosphate-buffered saline (PBS). All mice were treated for 5 weeks. After administration, behavioral and related indices were further tested.
APP/PS1 Positron Emission Tomography (PET)/Computed Tomography (CT) Imaging of Mouse Brain
After the completion of the drug administration intervention, the mice were subjected to a fasting intervention on the day before the experiment. Gas anesthesia was administered with 2% isoflurane, and (200 ± 10) µCi 18F-FDG was injected into the tail vein. PET scan and CT scan were performed successively (the PET scan was in dynamic mode, and the head scan time was 10 min; the CT scan was performed in the conventional mode). Avatar software was used for image reconstruction, and the CT image was reconstructed using the Feldkamp–Davis–Kress (FDK) algorithm, and the reconstructed PET image was imported into PMOD software to obtain the average standardized uptake value (SUV mean) of each brain region.
Behavioral Testing of the APP/PS1 Mouse Model
Opening Experiment
An open field test (Seibenhener & Wooten, 2015) was used to observe the behavior and activity of mice in an unfamiliar environment and their exploratory behavior in the unfamiliar environment. First, the mice were placed in the center of the test box (96 × 96 × 50 cm, divided into nine equal squares). After the mice had adapted to free movement for 2 min, the monitoring camera system recorded the activities of the mice within 3 min. The total distance of the area traveled by the mouse was recorded through the opening experiment system, and alcohol was used to clean the test box after the test.
Shuttle Box Experiment
On the first day of the experiment, the mice were placed in a shuttle box (25 × 16 × 30 cm) for adaptation. On the second day, the mice were put into the shuttle box in a certain order for measurement. During the experiment, if the mouse ran to the other side of the shuttle box due to stimulation, it was an active escape; if it ran away after clicking, it was a passive escape. Each animal was subjected to 50 cycle tests, recording the activity track for 5 min, with an interval of 40 s each time. After each mouse test, clean the shuttle box.
Morris Water Maze Experiment
The Morris Water Maze detection device is a circular pool with a diameter of 100 cm and a height of 50 cm. The pool is divided into four quadrants, and a white platform is placed in the center of either quadrant. Add (23°C ± 1°C) constant temperature water to the pool, so that the water level is 1 cm higher than the platform. At the same time, add edible coloring to bleach the water and hide the platform in the level. Each mouse was tested four times, and the interval between each experiment was not less than 20 min. Finally, the useful trajectories of mice were recorded and analyzed.
Total SOD Activity Detection
After the behavioral tests, the mice were killed by neck anesthesia, and brain tissue was collected. The obtained mouse brain tissue samples and water-soluble tetrazolium (WST) working solution were inoculated into 96 wells, mixed, and incubated, respectively. The absorbance was measured to calculate SOD activity.
CAT Activity Detection
After measuring the A520 standard curve, take the mouse brain tissue sample, put it into a centrifuge tube, and add CAT detection buffer. At the same time, a certain amount of CAT detection buffer and peroxidase should be added to the blank control substance. Mix the hydrogen solution to react. After terminating the reaction, add the CAT detection buffer and the terminated reaction system to another centrifuge tube, mix them, and inoculate them into a 96-well plate, and then add the color development working solution for color development. Finally, after 15 min, the absorbance value was measured with a spectrophotometer at A520. Record the values and calculate the CAT activity of brain tissue samples.
Glutathione Reductase Activity Detection
Add the mouse brain tissue sample, GPx detection buffer, and GPx detection working solution in order and mix, followed by adding the peroxide reagent. Set the spectrophotometer temperature to 25°C. A340 was measured every 30 s, recorded continuously for 3 min, and six data points were obtained. Calculate GPx activity in the sample.
Lipid Oxidation Detection
Add PBS buffer to the blank control, draw standard curves of different concentrations, and inoculate them in a 96-well plate. Then, add the malondialdehyde (MDA) detection working solution, mix well, and then perform the heating treatment. After cooling, the supernatant was inoculated into a 96-well plate. The absorbance was further measured using a spectrophotometer, and the MDA content was calculated.
Congo Red Staining
After the frozen sections are hydrated, they are washed three times with PBS and then placed in Congo red staining solution for staining and then put into staining differentiation solution to keep them from fading; finally, they are further treated with hematoxylin and hydrochloric acid alcohol, and after dehydration, the slides were mounted, and their morphological changes were observed through a light microscope.
Western Blot Detection of Aβ and CCR5 Protein Expression
When using the bicinchoninic acid (BCA) method to measure the expression of related proteins, first wash the sample cells with PBS and then lyse them. Then, load the sample, transfer it to a polyvinylidene difluoride (PVDF) membrane, and block it. Add the primary antibodies MIP-1α (ab229900, 1:1,000), CCR5 (ab287959), 1:1,000), occludin (ab216327, 1:1,000), and claudin-5 (ab131259, 1:1,000). The secondary antibody (goat anti-rabbit 1:2,000) was incubated overnight. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) served as an internal reference. After interaction with ECL reagents, the blots are analyzed by software.
Unbiased Stereological Quantitative Analysis of Aβ Plaques, Microglia, and Astrocyte Cluster Areas in the Hippocampus
Counting is done randomly and only by one person. After completion, another researcher randomly selects at least two animal slices for verification through blind selection. When the error of the verification result is less than 10%, it is judged to be valid, and the total volume parameter = total area × average thickness of the slice. Optical double sectioner, “x” constitutes a measurement technology, and the distance between “x” and “x” can be adjusted to a certain extent by the detection structure size. Finally, through computer control, after the counting is completed, a related result is obtained.
Real-time Quantitative PCR (RT-qPCR) Detection
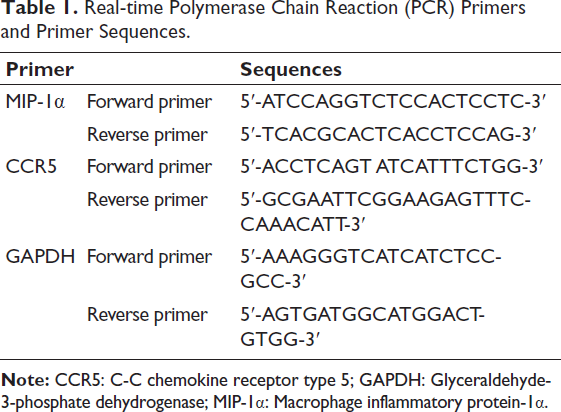
Ribonucleic acid (RNA) was extracted by the TRIzol method, and complementary DNA (cDNA) was reverse transcribed at the same time. The results of this method were verified using SYBR fluorescence PCR technology. GAPDH is an intrinsic control between rho-associated protein kinase (ROCK)/myosin light chain kinase (MLCK) and miRNA, and the MIP-1α, CCR5, and GAPDH primers are listed in Table 1. Relative levels were estimated using the 2−∇∇Ct method.
Real-time Polymerase Chain Reaction (PCR) Primers and Primer Sequences.
6E10 Immunohistochemical Staining
Tissue section preparation: first, cut the tissue to be tested into thin slices and fix them on a glass slide, then heat the antigen retrieval solution to expose the antigen, then use a blocking solution to block endogenous peroxidase, and finally, the 6E10 antibody was incubated at the same time and allowed to bind to the amyloid protein. After the color reaction, the protein was labeled, and the deposition of amyloid protein was observed through a microscope.
Statistical Analysis
The data obtained in each of the above experiments were analyzed using Statistical Package for the Social Sciences (SPSS) 21.0 and GraphPad Prism software (version 8.1.2, GraphPad Software Inc.). If there are no special requirements, p < .05 is used as the test standard.
Results
Changes in Behavior, Pathological Morphology, and Antioxidant Levels in APP/PS1 Mouse Model Brain
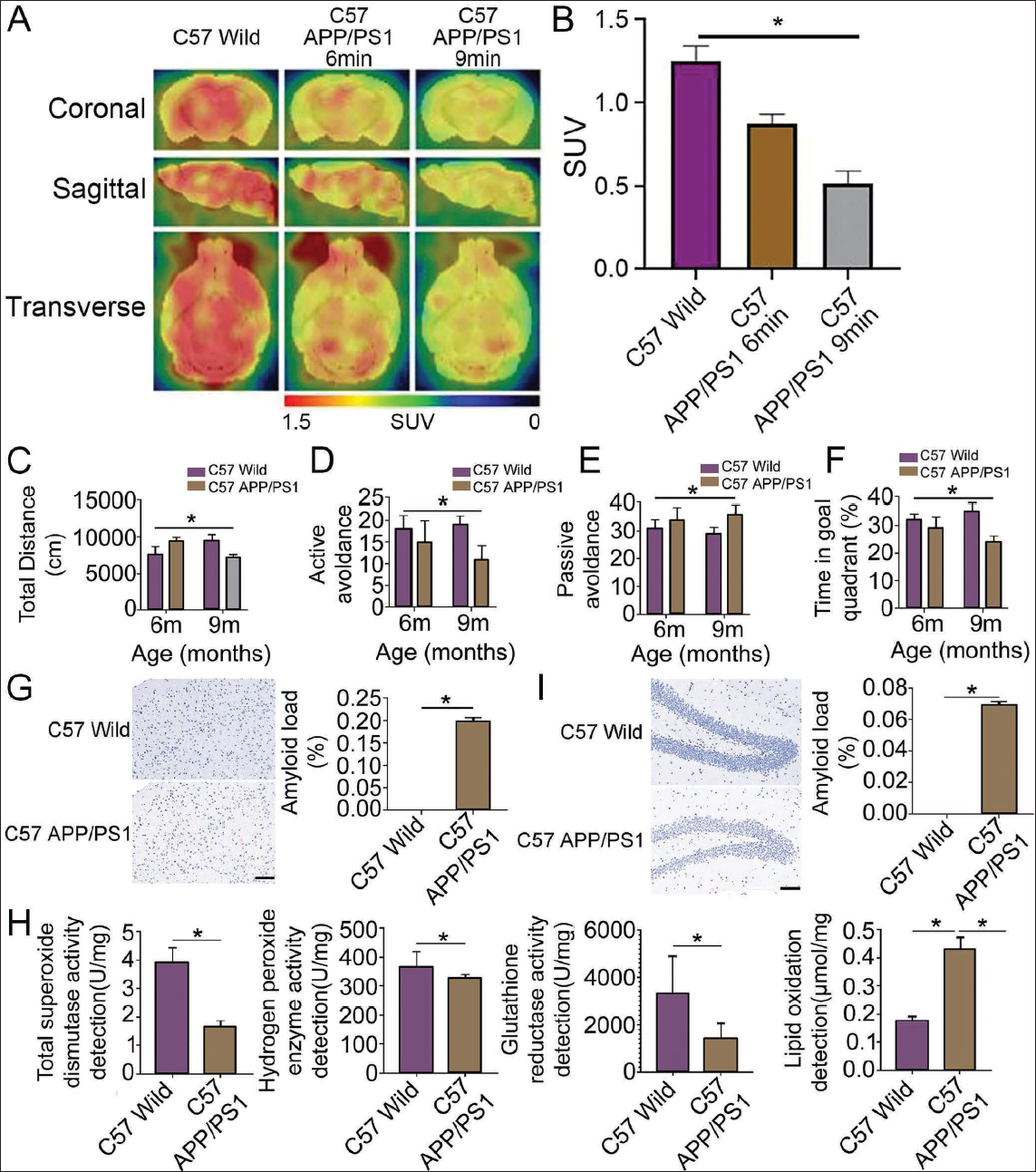
All mice survived after feeding, with a survival rate of 100%. We first observed the uptake of 18F-FDG in the brain tissues of mice in each group by PET/CT. The results showed that the cerebral blood perfusion imaging in the frontal and temporal lobes of APP/PS1 mice at 6 months and 9 months of age was significantly asymmetric. The uptake of 18F-FDG was significantly reduced compared with that of the C57 wild group. The intake was significantly reduced (Figure 1A and 1B). We conducted group testing on the APP/PS1 mouse model. We found that in the mouse behavior testing experiment, the autonomous activity distance of the mice in the 6-month-old and 9-month-old C57 APP/PS1 groups and the number of passive avoidances in the shuttle box test were higher than those of the C57 wild group (p < .05) (Figure 1C–1E). The time of the 6-month-old and 9-month-old C57 APP/PS1 group in the water maze experiment was significantly lower than that of the C57 wild group (p < .05) (Figure 1F). In the mouse pathological morphology detection experiment, C57 APP/PS1 mice had a small number of red Congolese plaques in the cortical area, while the C57 wild group had no obvious red plaques. Further comparative image analysis results showed a significantly higher number and area of amyloid protein in C57 APP/PS1 mice than in the C57 wild group (Figure 1G). The results were consistent with the Congo red staining results of the cortical area (Figure 1H). In the experiment detecting antioxidant levels in the mouse brain, the total SOD activity in the brain tissue of model mice in the C57 wild group was higher than that of the C57 wild group (p < .05). In the APP/PS1 group, interestingly, the activities of CAT and glutathione reductase also showed the same trend. But, contrary to this trend, the level of lipid oxidation, which showed that the C57 APP/PS1 group was higher than the C57 wild group (Figure 1I). Therefore, the APP/PS1 mouse model successfully simulates the pathological characteristics of AD patients in terms of amyloid plaque formation and cognitive function decline. It is stable and reliable, providing help for subsequent in-depth research.
Resveratrol Affects β-Amyloid (Aβ) Expression in APP/PS1 Mouse Model
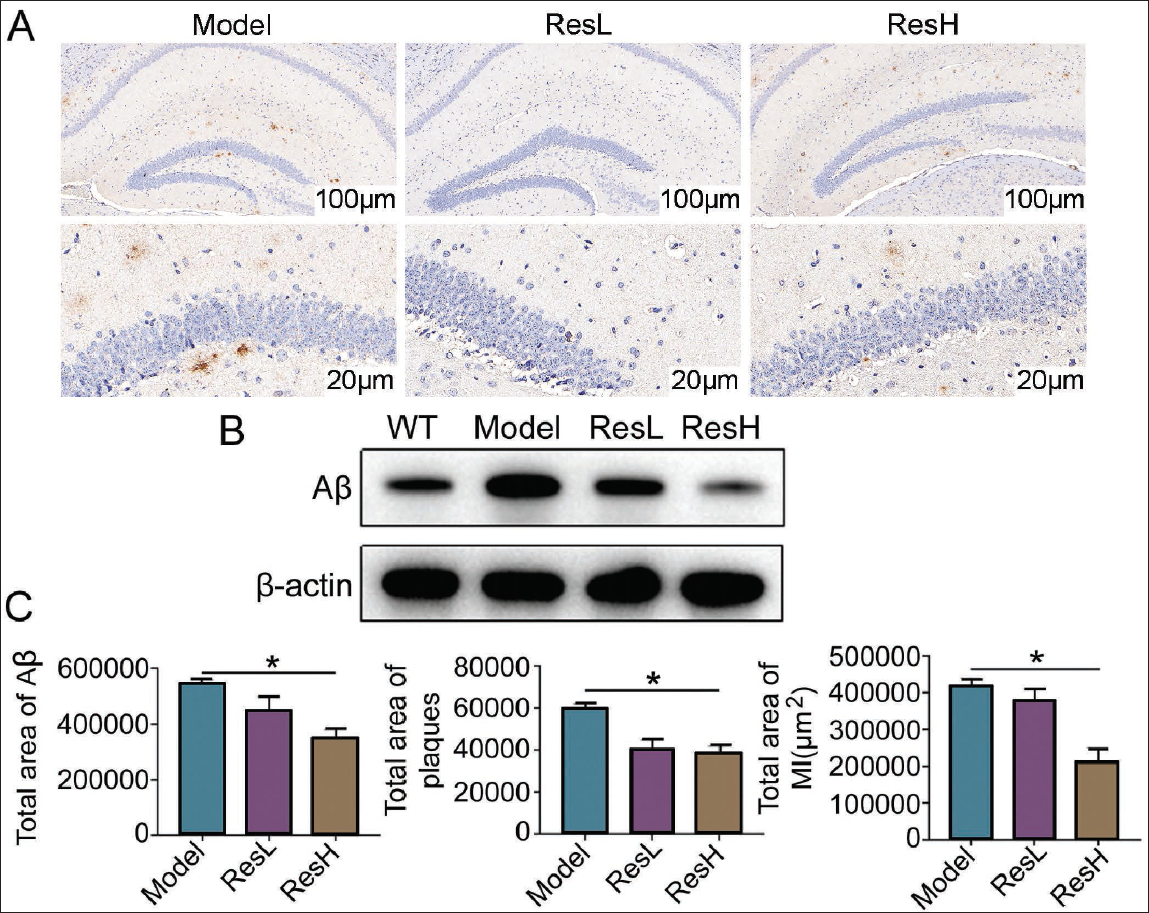
In order to explore resveratrol’s effect on AD, we added various doses of resveratrol to the established APP/PS1 mouse model. We cultured them for a period of time before conducting tests. It was found that after resveratrol intervention, the pathological characteristics of AD mice were significantly improved. The results of 6E10 immunohistochemical staining showed that the ResH group had fewer Aβ plaques, and the area of a single Aβ plaque was also smaller. In the model group, the number of Aβ plaques was not only greater, but also the area of each one was larger; in the ResL group, the Aβ plaque situation was between the ResH group and the model group (Figure 2A). Aβ protein level in the resveratrol intervention group was significantly reduced (p < .05) (vs. the model group). Further observation found that the Aβ protein level of the resveratrol high-dose group (ResH) was the lowest, even lower than that of the wild-type non-transgenic (WT) mice (p < .05) (Figure 2B). The further unbiased stereological quantitative analysis found that after resveratrol intervention, the various total areas showed a downward trend dose-dependently (Figure 2C). Therefore, we believe that resveratrol can downregulate Aβ protein in the APP/PS1 mouse model and reduce the formation of Aβ amyloid plaques, thereby improving the pathological characteristics of the AD model.
Resveratrol Can Reduce Aβ Protein Deposition and Regulate MIP-1α Expression to Stabilize the BBB
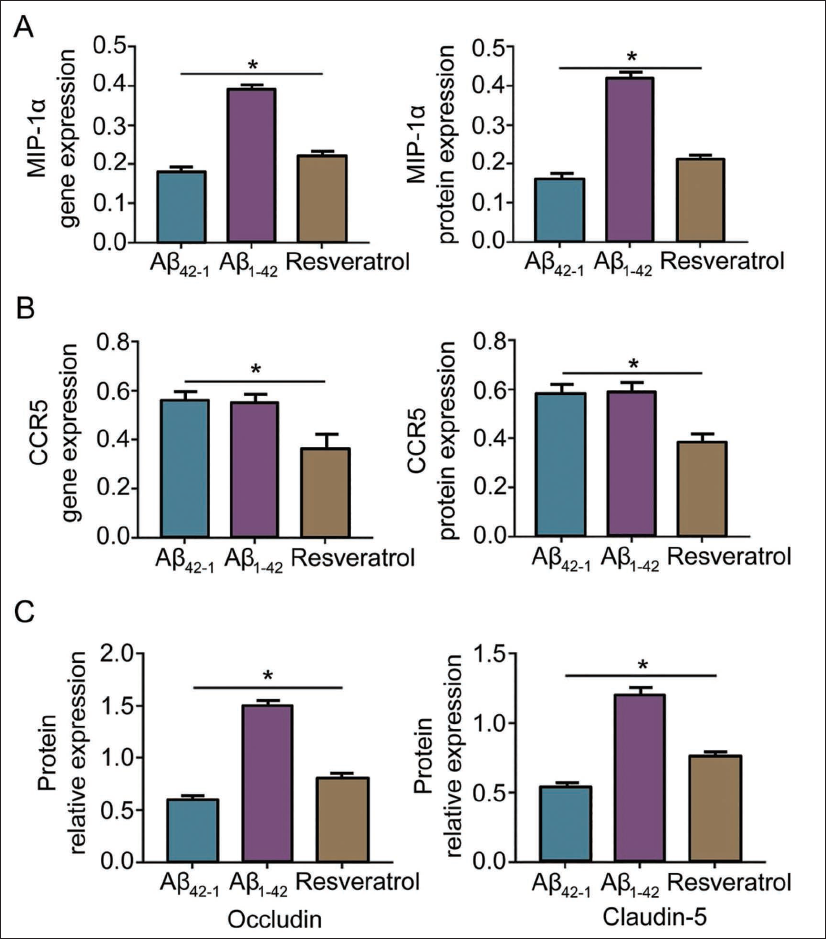
In order to explore whether Aβ protein can affect the expression of MIP-1α and thereby affect the BBB, we injected Aβ into the hippocampus of normal mice. It was found that the expressions of MIP-1α and CCR5 genes and proteins in the resveratrol group were decreased (vs. the Aβ1-42 group). By detecting the protein expression of occludin and claudin-5, the expression of these proteins was the lowest in the Aβ1-42 group (p < .05). At the same time, it was increased in the resveratrol group (p < .05) (vs. Aβ1-42 group) (Figure 3A–3C). From this, we can see that resveratrol may stabilize the BBB by reducing Aβ protein content and thereby reducing the expression of MIP-1α.
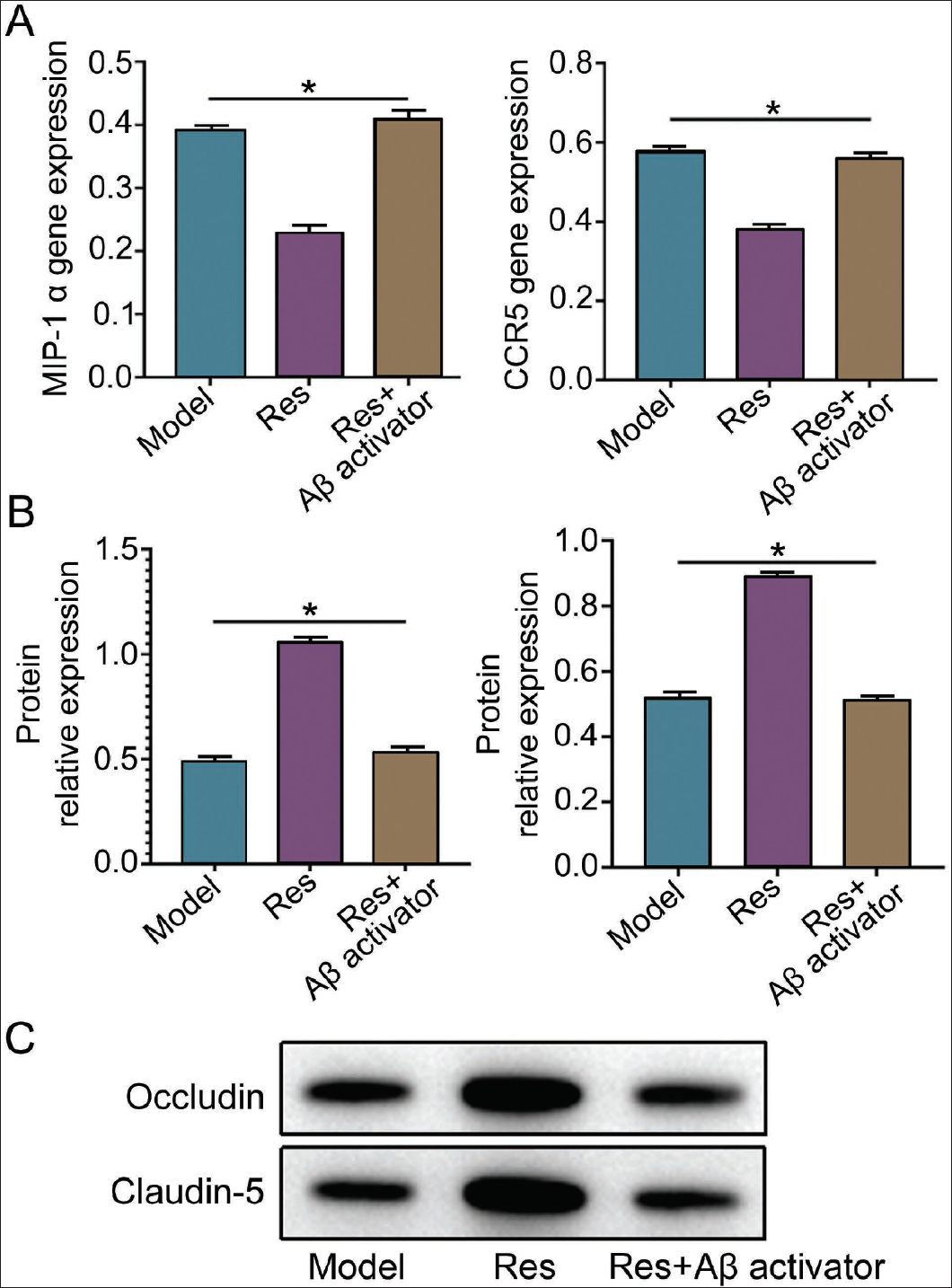
Use Aβ Activator (APP) to Verify That Resveratrol (Res) Can Reduce the Deposition of Aβ Protein, Downregulate MIP-1α, and Stabilize BBB
In order to increase the rigor, we added APP on the basis of the Res. We found that the MIP-1α and CCR5 gene levels increased significantly after adding APP (vs. Res group) without differences compared with the model group (p < .05) (Figure 4A). BBB-related proteins, occludin and claudin-5 levels, were further detected, and it was found that the protein expression levels decreased significantly after adding APP (p < .05), and there was still no difference compared with the model group (Figure 4B and 4C). Therefore, we can see that the expression of MIP-1α also increases after the expression of Aβ increases. Still, at this time, the expression of BBB-related proteins decreases, and the BBB function weakens.
Discussion
BBB destruction is a cause of AD (Alahmari, 2021). As a special cellular structure, the BBB can not only isolate harmful substances from invading the brain but also maintain the stability of the brain’s internal environment to a certain extent, maintaining the central nervous system (Xie et al., 2019). Disruption of the BBB can lead to extravasation of plasma components, edema formation, inflammatory cell infiltration, and massive extravasation of immunoglobulins (Qiu et al., 2021). Resveratrol comes from peanuts, grapes, and other plants. It prevents blood clots and plays an anti-inflammatory role. In-depth research has found that it also has various pharmacological effects, such as scavenging free radicals and anti-aging (Hecker et al., 2022; Meng et al., 2021). Resveratrol has shown the potential to maintain BBB integrity. Relevant experimental studies have shown that resveratrol can inhibit occludin and claudin-5 expression, which are important components of the BBB, and inhibit pro-inflammatory cytokines, nitric oxide synthase (NOS) and interleukin-1 (IL-1), through its anti-inflammatory and antioxidant activities expression (Pan et al., 2022), upregulating the expression of the anti-inflammatory gene arginase1 and the cytokine interleukin-10 (IL-10), thereby achieving the purpose of promoting BBB integrity (Shi et al., 2022). At the same time, some experimental studies have suggested that resveratrol can effectively inhibit Aβ deposition formation, which is a hallmark lesion of AD (Dong et al., 2023). In addition, Aβ in AD patients can promote the high expression of MIP-α in peripheral blood T cells, thereby promoting the transendothelial migration of T cells and upregulating MIP-α receptor CCR5 (Yamakawa et al., 2023). Therefore, it is feasible and of great significance to explore how resveratrol regulates MIP-α to stabilize the BBB in AD rats through β-amyloid peptide.
In the experiment, by establishing a model of mice and injecting various doses of resveratrol, on the basis of the established model, it was found that 6E10 immunohistochemical staining showed that the pathological state of mice in the resveratrol group was significantly improved. Further observation found that the expression of Aβ in each dose of the resveratrol group was reduced, and the distribution and area of Aβ plaques were reduced. This suggests that resveratrol can downregulate the expression of Aβ and reduce Aβ deposition, thereby alleviating AD to a certain extent. The reason may be that resveratrol inhibits the neuroinflammatory response caused by Aβ and downregulates the nuclear factor kappa B (NF-kB) inflammatory signaling pathway, thereby reducing the release of inflammatory factors, achieving the purpose of alleviating the inflammatory response, and ultimately alleviating the pathological state of the model mice. According to relevant studies, the reduction of Aβ plaques may be related to the BBB transport and clearance mechanisms (Edavettal et al., 2022). Aβ can be transported across the BBB through cerebral microvessels, and the deposition of Aβ also indicates the occurrence of BBB disorders. This suggests that resveratrol can maintain BBB stability by affecting Aβ.
MIP-α can further aggravate local damage by binding to inflammatory factors (Kochumon et al., 2020). Relevant studies have shown that the Aβ-dependent promotion of T lymphocytes across the BBB may be related to the overexpression of MIP-α (Costa-Madeira et al., 2022). Further in-depth research found that overexpressed MIP-α interacts with the CCR5 receptor on the cell membrane, causing the ROCK signaling pathway to be activated, and leading to the disorder of the tight junctions of the BBB (Kumar et al., 2023). Therefore, effectively reducing the expression of MIP-α can maintain the stability of the BBB to a certain extent.
Conclusion
In this animal experiment, we found that in the Aβ1-42 group, MIP-α and CCR5, all showed high expression, and occludin and claudin-5 had the lowest expression. It can be seen that Aβ can increase the expression of MIP-α and downregulate occludin and claudin-5 proteins, thereby destroying BBB stability. In summary, resveratrol may stabilize the BBB by inhibiting the production of Aβ, anti-inflammatory and antioxidant mechanisms, and it may also regulate the expression of MIP-1α, thereby promoting the tight junctions of endothelial cells. The recovery further maintains BBB integrity to a certain extent. Although resveratrol has shown some potential in treating AD, further clinical trials are needed to evaluate its safety.
Footnotes
Abbreviations
AD: Alzheimer’s disease; APP: Amyloid precursor protein; BB: Blood–brain barrier; CCR5: C-C chemokine receptor type 5; MIP-1α: Macrophage inflammatory protein-1α; PS1: Presenilin 1B.
Acknowledgments
The authors gratefully acknowledge the Inner Mongolia Medical University Jinshan Campus School of Basic Medicine Laboratory for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethics committee of Inner Mongolia Medical University, Jinshan Campus, School of Basic Medicine.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
