Abstract
Background
Parkinson’s disease (PD) is a prevalent age-associated neurodegenerative disorder, primarily characterized by dopaminergic neuronal loss, oxidative stress, and microglia-mediated neuroinflammation. Current therapies offer limited relief and are often associated with adverse effects, necessitating the development of safer, more effective alternatives.
Objectives
This study aimed to investigate the neuroprotective effects of trifolirhizin (TF), a natural flavonoid, in a mouse model of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced PD.
Materials and Methods
Male C57BL/6 mice were administered MPTP to induce PD-like symptoms, followed by treatment with TF. Behavioral assessments (wire hang and open field tests) evaluated motor function. Biochemical assays measured oxidative stress markers malondialdehyde (MDA), monoamine oxidase-A (MAO-A) activity, and antioxidant enzyme levels. Pro-inflammatory cytokines (interleukin (IL)-1β, tumor necrosis factor (TNF)-α, IL-6) were quantified, and histopathological analysis of the substantia nigra was performed.
Results
TF-treated mice showed a 48% improvement in rotarod performance and a significant increase in exploratory behavior compared to MPTP-only controls. MDA levels were reduced by 41%, while MAO-A activity was suppressed by 35%. Antioxidant enzymes (superoxide dismutase (SOD), catalase (CAT)) were markedly upregulated. Notably, the TF treatment led to a greater than 50% reduction in pro-inflammatory cytokines and preserved the structural integrity of dopaminergic neurons, as confirmed by histological evaluation.
Conclusion
TF significantly improves motor function, mitigates oxidative damage, suppresses neuroinflammation, and protects dopaminergic neurons in MPTP-induced PD mice. These findings underscore TF’s potential as a promising therapeutic candidate for slowing PD progression and alleviating its symptoms.
Keywords
Introduction
Parkinson’s disease (PD) is among the most common neurodegenerative diseases, following Alzheimer’s disease, impacting 1% of people aged 60 or older (Koziorowski et al., 2021; Lorente-Picón & Laguna, 2021). The characteristic features of PD pathogenesis are defined by the gradual loss of dopaminergic (DA) neurons within the substantia nigra pars compacta (SNpc), resulting in a decrease of DA levels of the striatum, leading to irreversible motor defects like resting tremor, bradykinesia, stiffness, and instability of posture (Hayes, 2019; Lotankar et al., 2017). Non-motor symptoms that typically accompany motor symptoms include olfactory and urinary dysfunction, postural hypotension, pain, constipation, forgetfulness, depression, and sleep difficulties (Tolosa et al., 2021). Elevated oxidative stress and inflammatory mechanisms have been linked with the pathophysiology of PD overall (Masato et al., 2021). Modern PD therapies, such as the most well-known levodopa, carbinoxamine, and carbidopa, can manage symptoms and slow down the disease’s progression. Still, they are associated with an array of side effects that can negatively affect a patient’s health, including stress, anxiety, and fatigue (Marino et al., 2020; Tambasco et al., 2018). Hence, research is necessary for evaluating novel substances that might influence neuroinflammation as well as the oxidative imbalance, which triggers neurodegeneration in PD patients.
A widely recognized Parkinson-inducing animal model is established via intraperitoneal injection of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), which is metabolized by monoamine oxidase type B into its active form, 1-methyl-4-phenylpyridinium (MPP+) (Heng et al., 2022). MPP+ demonstrates a strong affinity for the dopamine transporter and is internalized by dopamine neurons, where it disrupts respiration by inhibition of the mitochondrial complex. This leads to a heightened generation of reactive oxygen species (ROS). ROS induce cell death by generating oxidatively damaging chemicals, including hydroxyl and superoxide radicals, leading to protein and lipid peroxidation (LPO). The impacted DA neurons may ultimately undergo degeneration by either necrosis or apoptosis (Aktas, 2023; Cuenca-Bermejo et al., 2021).
In recent times, several studies have documented the positive benefits of medicinal plant-derived treatment on a range of illnesses, including neurological diseases (Corona, 2018; Rahman et al., 2021; Siracusa et al., 2021). It has been demonstrated that natural compounds can reduce tremors and enhance cognitive function (Atiq et al., 2023). PD has been successfully treated by employing plants like ashwagandha, flannel weed, black henbane, and velvet bean. Additionally, high-antioxidant containing plants, including berries, green tea, and maidenhair trees, have also been employed to treat PD and slow down its progression (Yu et al., 2021). The plant Sophora flavescens is employed for the treatment of a number of illnesses in traditional Chinese medicine. Its root is utilized for dealing with inflammation, asthma, bacterial infections, and psychological disorders. Trifolirhizin (TF), a pterocarpan flavonoid, which is extracted from S. flavescens roots, has a number of medicinal as well as pharmacological characteristics, like antioxidant, anti-inflammatory, anti-cancer, anti-bacterial, and hepatoprotective actions (Jaiswal & Lee, 2025). The key objective of this work is to elucidate the protective impact of TF against MPTP-stimulated Parkinsonian condition in mouse models. Moreover, further studies are necessary to understand the precise underlying molecular mechanisms by which TF exhibits therapeutic properties.
Materials and Methods
Experimental Plan
Male C57BL/6 mice 7–8 weeks old (22–25 g) were procured from an institutional animal facility for this study. Animals were kept on a 12 h light/dark cycle in a controlled temperature (25°C ± 2°C) and relative humidity (50%–70%) for a minimum of 3 days before experiments. All experiments were approved by the Animal Ethics Committee of China, Department of Neurology, First Hospital of Shanxi Medical University. Animals were allocated into four experimental groups (n = 6) as follows: as a control, Group 1 mice received intraperitoneal injections of normal saline (0.9%). Group 2 mice were given two doses of MPTP (30 mg/kg body weight) by intraperitoneal injection at 16-h intervals. Following two MPTP doses, starting from the next day, the Group 3 animals were orally fed with TF (15 mg/kg body weight) for 3 weeks. Following two MPTP doses, starting from the next day, Group 4 mice were orally administered Tidomet Plus as a positive control for 21 days.
Chemicals
The primary chemicals, including Trifolirhizin, MPTP, and others, were purchased from Sigma–Aldrich, USA. The diagnostic kits for the estimation of biochemical markers were procured from Elabscience, Abcam, and MyBioSource, USA, respectively.
Tissue Preparation
At the end of the experimental period, intracardiac blood samples were obtained under ketamine/xylazine anesthesia, and the mice were killed by cervical dislocation. Brain tissues were immediately taken out, rinsed with cold normal saline (0.9%), and stored in a freezer at −80°C. The samples of brain tissue were thawed and suspended in cold phosphate buffer. Parameters such as LPO and MAO-A levels were all assessed using the supernatant.
Animal Behavioral Examination
Before carrying out the motor activity, all rodents involved were acclimatized, given food and water, and habituated to an independent room.
Open Field Test
The open-field apparatus (40 × 40 × 40 cm) was split into 16 equal-sized squares. The animals being studied were positioned in the center of the field. A 30-min period began after placing the mouse in the middle of the box and allowing it to move around the open field setup. A SMART video monitoring system was employed to record both the overall distance covered and the time spent in the center (Lee et al., 2021).
Wire Hang Examination
The rodents were mounted on a wire positioned horizontally 20 cm above the ground level. The animals were introduced, and they gripped the wire with their hands and forepaws. The delay from when the mice fell was noted. The trials were performed 10 times, and the average findings were noted. The animals were permitted to relax in between sessions (Saleem et al., 2021). Blinded experimenters conducted all behavioral tests to reduce bias.
Estimation of MAO-A Levels in the Mice Groups
For inhibition of MAO-A activity, the suspensions were treated with either tyramine (500 µM) or pargyline (500 nM). The test mixture’s chromogenic solution included vanillic acid (1 mM), peroxidase (4 U/mL), and 4-aminoantipyrine (500 mM). The levels of MAO-A activity have been identified using spectrophotometric analysis by measuring the optical density (OD) at 425 nm. These levels were measured in units/mg protein, with one unit representing 1 µmol of product produced per minute.
Estimation of Malondialdehyde (MDA) Levels
MDA, the byproduct of LPO, the supernatant (0.2 mL) was blended with 20% acetic acid (1.5 mL), 0.8% thiobarbituric acid (1.5 mL), and 8.1% sodium dodecyl sulphate (0.2 mL), and then combined with processed tissue samples (0.1 mL) before being heated at 100°C for 60 min. The resulting mixture was allowed to cool, and 500 µL of n-butanol-pyridine (15:1) and 100 µL of distilled water were combined and vortexed. After 10 min of centrifugation at 4,000 rpm, the organic layer was removed, and its OD at 532 nm was determined with a ultraviolet (UV) spectrophotometer.
Examination of Superoxide Dismutase (SOD), Glutathione (GSH), and Catalase (CAT) Enzyme Level in the Mice Groups
The SOD enzyme present in the brain homogenate was tested for its capacity to scavenge superoxide radicals produced by the automated oxidation of pyrogallol in an alkaline media. The reaction mixture contained potassium phosphate buffer (2.8 mL), pyrogallol solution (0.1 mL), and tissue homogenate (0.1 mL). For 5 min, an increase in OD was measured every 30 s at 325 nm. The quantity of enzymes necessary to inhibit pyrogallol auto-oxidation by 50% per mL of the test solution constituted one unit of SOD.
The activity of the CAT enzyme was measured using the protocol described by Hadwan and Ali (2018), with minor adjustments. The homogenized tissue (0.1 mL) was treated with 10 mM H2O2 (2.9 mL) suspended in potassium phosphate buffer (50 mM; pH 7) for 2 min. The rate of reduction in absorbance at 240 nm was monitored for 3 min. The outcome was presented as CAT activity units per milligram of protein.
Reduced GSH levels were measured using 5,5′-dithiobis(2-nitrobenzoic acid) (DTNB, Ellman’s reagent). Tissue homogenate (0.1 mL) was mixed with 2.9 mL of DTNB in potassium phosphate buffer (pH 7.4) and incubated for 15 min at room temperature. The absorbance was measured at 415 nm. The results are expressed as µmol GSH/mg protein. This method selectively quantifies reduced GSH, as no reducing agents or enzymes (e.g., GSH reductase) were included to convert glutathione disulfide (GSSG) to GSH.
Estimation of Tumor Necrosis Factor (TNF)-α, Interleukin (IL)-6, and IL-1β Cytokine Activity
The concentration of IL-6, TNF-α, and IL-1β was estimated with an enzyme-linked immunosorbent assay (ELISA) kit. Brain tissues were placed in 96-well plates with corresponding antibodies (IL-6, IL-1β, or TNF-α) and left for incubation at 4°C overnight. Following that, bovine serum albumin (BSA) (1%) was incorporated into the block for 1 h at 37°C. Later, horseradish peroxidase (50 µL) was added to each well and left for 45 min. Finally, the OD of the plate was estimated at 450 nm, and the data were represented as pg/mg of protein.
Histopathological Analysis of Brain Tissues
Brain tissues from all mice groups were fixed in formalin (10%). The sections of tissues were coated in paraffin wax and sliced at a 5–6 µm thickness. They were stained with hematoxylin and eosin for photomicroscopic study of the brain architecture.
Statistical Analysis
All experimental data were analyzed using Statistical Package for the Social Sciences (SPSS) software version 20.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism version 9.0 (GraphPad Software Inc., San Diego, CA, USA). Data are presented as mean ± standard deviation (SD). Statistical comparisons between groups were made using one-way analysis of variance (ANOVA), followed by Tukey’s post hoc test for multiple comparisons. A p value of less than .05 was considered statistically significant.
Results
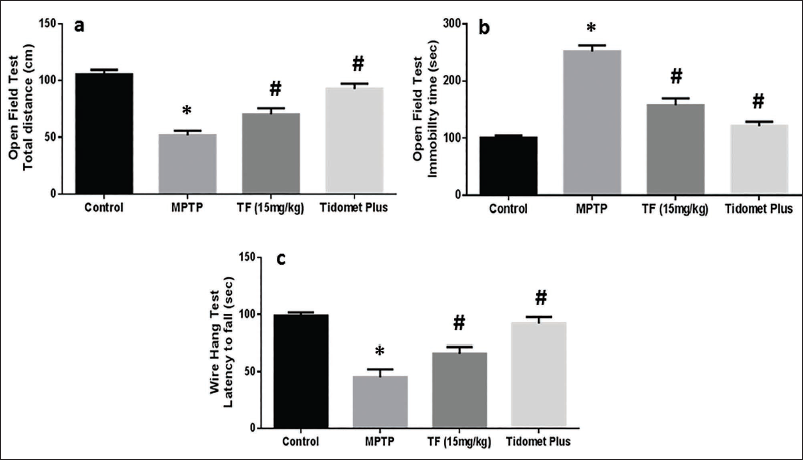
Impact of TF on the Behavioral Analysis of the Mice Groups
Standard approaches for assessing medicine efficacy in Parkinson’s animals include the open field and wire hang tests. As a result, these two experiments were performed. Figure 1a and 1b show that TF (15 mg/kg) improved the total covered distance and reduced the immobility time in MPTP-induced PD animals (p < .05). Tidomet Plus substantially improved the total distance covered and decreased the immobility time of the animals compared to MPTP alone (p < .05). Figure 1c, with respect to the wire hang test, the delay period of the falling of the animals improved upon treatment with TF and Tidomet Plus.
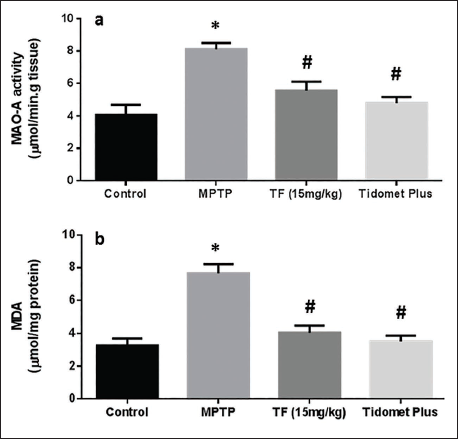
Impact of TF on the MDA and MAO-A Levels in the Mice Groups
To examine the mechanism of the protective effect of TF, the levels of MDA and MAO-A were estimated. The MAO-A activity and MDA levels in the brains of MPTP-induced animals were substantially (p < .05) elevated when compared with those in the TF and Tidomet Plus groups. However, TF administration reduced both the MDA levels as well as the MAO-A activity (Figure 2a and b). Additionally, the administration of TF decreased the pathological consequences of MPTP as effectively as Tidomet Plus.
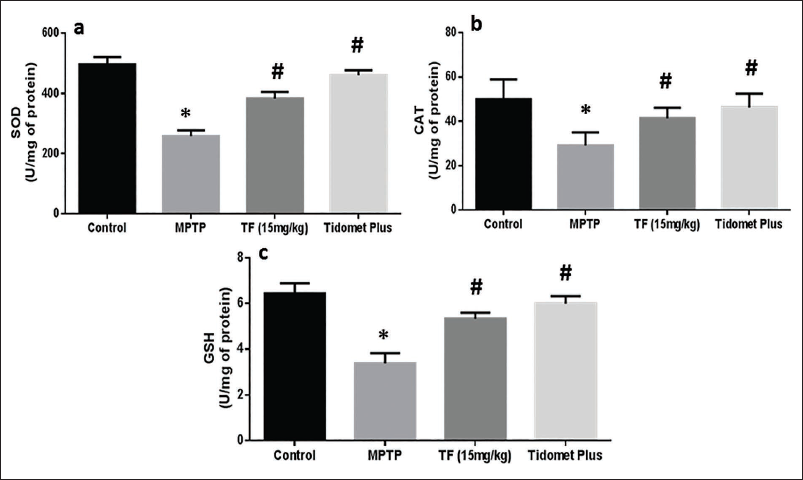
Impact of TF on the SOD, GSH, and CAT Levels in the Mice Groups
To detect the specific cellular impairment triggered by oxidative stress, the CAT, SOD, and GSH levels were measured, which are the most crucial constituents in the natural antioxidant defense mechanism (Figure 3a–c). The SOD, CAT, and GSH activity levels were substantially diminished in MPTP-induced mice compared to normal mice, suggesting that MPTP induction led to the formation of oxidative stress (p < .05). These levels were higher in the TF and Tidomet Plus-treated groups than in the MPTP group. Interestingly, the cellular damage caused by MPTP was reversed upon treatment with TF.
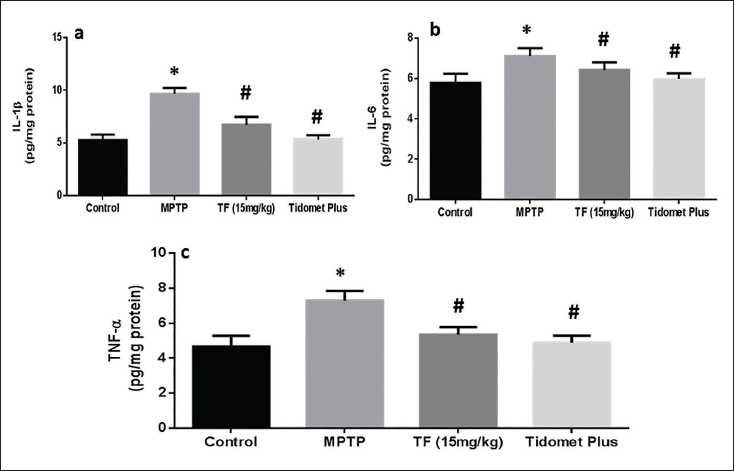
Impact of TF on the Inflammatory Cytokine Levels of TNF-α, IL-6, and IL-1β
In comparison with the normal mice, the TNF-α, IL-6, and IL-1β levels were elevated in the MPTP-stimulated rodents considerably (p < .05) (Figure 4). These levels were reduced in the TF and Tidomet Plus-treated groups than in the MPTP group. Moreover, the cytokine levels in the Tidomet Plus-treated groups were comparable to those of the control group mice.
Histopathological Examination of Mice Organs
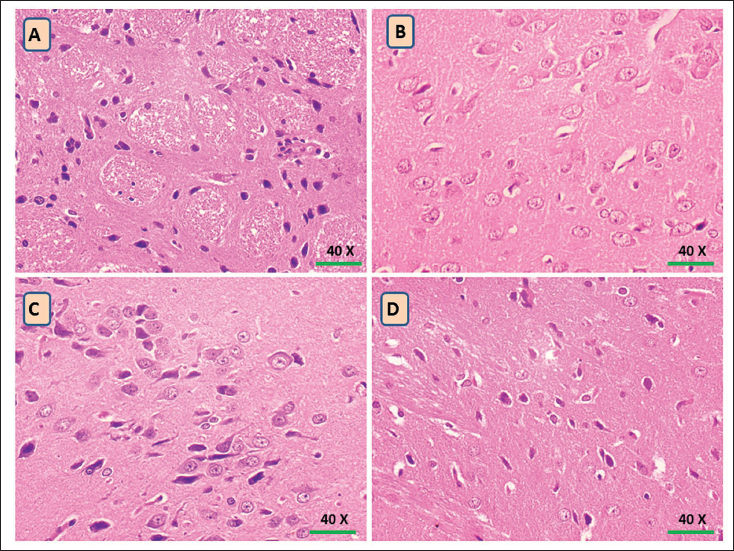
The histopathological examination of the animal’s brain in the normal group revealed typical substantia nigra, no neuronal loss, and the absence of Lewy bodies. The MPTP group displayed unique characteristics, like vacuolated areas, pyknotic nuclei, neuronal loss, and Lewy bodies. The TF and Tidomet Plus-treated group exhibited a minor midbrain enhancement, with a minimal vacuolated area, minor neuronal loss, certain pyknotic nuclei, and no Lewy bodies. TF administration significantly improved neuronal appearance and Lewy bodies in contrast to the MPTP group (Figure 5).
Histopathological Modifications in the Mice’s Brain Displayed Neuronal Degeneration. (A) Group I: Control; (B) Group II: MPTP-stimulated Mice; (C) Group III: MPTP Mice Administered with Trifolirhizin; (D) Group IV: MPTP Mice Administered with Tidomet Plus (Positive Control).
Discussion
PD is marked by behavioral defects, motor deficiencies, and deterioration in cognition as well as memory, resulting from progressive neurodegeneration. There is no appropriate therapy or treatment option to stop or reverse the illness symptoms. MPTP treatment in rodents can induce PD symptoms, resulting in experimental animal models with behavioral alterations and pathophysiological characteristics associated with neurodegenerative conditions such as oxidative stress, DA neurodegeneration, as well as inflammation-facilitated glial cell stimulation (Blesa et al., 2015). Previous research additionally demonstrated that MPTP treatment of mice and primates induces DA neuron death in the SNpc and causes reduced DA levels, which ultimately results in serious motor impairment (Zhang et al., 2019).
To minimize adverse consequences and improve patient-specific assistance, novel and safer medications with an array of treatment techniques are necessary; natural medications obtained from plants are thus a desired choice to manage and prevent PD (Atiq et al., 2023; Pariyar et al., 2022). In the current investigation, we examined the neuroprotective nature of TF in MPTP-stimulated dopamine neuron degeneration. The positive impact of TF is well understood through the improvement in behavioral defects, decreased oxidative stress, and inhibition of the inflammatory cascade in the experimental animal groups administered with TF.
Standard approaches for assessing the efficacy of TF included the open field and wire hang tests. MPTP toxicity is known to promote motor impairments and bradykinesia in rodents. Moreover, MPTP-stimulated animals have been reported to demonstrate impaired neuromuscular coordination and a considerable decrease in retention time (Park et al., 2018). TF treatment was shown to improve the total distance covered and reduce the immobility period in MPTP-induced PD animals. With respect to the wire hang test, the delay period of the falling of the animals improved upon treatment with TF, which implies the restoration of motor impairments by the compound.
As oxidative stress, particularly stress triggered by hydroxyl radicals (•OH), is an essential aspect of PD advancement, levels of MDA in the experimental mice were measured. Typically, the enzymatic breakdown of DA by mitochondrial MAO enzymes generates its metabolites and hydrogen peroxide, leading to substantial amounts of •OH buildup through the Fenton reaction. In the present work, the goal was to reduce dopamine breakdown to restrict the MAO-based process of dopamine removal. MAO-A activity was found to be reduced in the TF-administered groups as compared to the MPTP-induced group. Consequently, the extract could possibly be able to protect the DA neurons from enzyme inhibition. Oxidative stress is a significant risk indicator for PD, owing to its growing impact on the aging process. MPTP treatment induced free radical production as well as oxidative imbalance in the substantia nigra, resulting in DA neuron degeneration (Jiang et al., 2016). The indicators of oxidative imbalance, including SOD, CAT, and GSH levels, were measured before and after TF treatment. These levels were substantially diminished in MPTP-induced animals in comparison with normal animals, suggesting that MPTP induction led to the formation of oxidative imbalance. Interestingly, the cellular damage caused by MPTP was reversed upon treatment with TF. The antioxidant activity of TF has been confirmed by several studies conducted in the past (Jaiswal & Lee, 2025).
Neuroinflammation is a prominent attribute of a number of progressive neurodegenerative illnesses, notably PD (Jayaram & Krishnamurthy, 2021; Wang et al., 2015). In the progressive stages of PD, neuroinflammation occurs primarily through the stimulation of microglia, the brain’s immune cells. Their activation may negatively impact DA neurons by overproducing pro-inflammatory cytokines like nitric oxide, TNF-α, IL-1β, and IL-6. Thus, eliminating inflammation by regulating the pro-inflammatory molecules might be a valuable approach for minimizing the advancement of neuroinflammation (Zhang et al., 2019). Data from previous work imply that blocking inflammatory processes driven by microglial activation is an appropriate approach to slow the advancement of the condition (Cordaro et al., 2022). In the last decade, it has been reported that natural substances exert positive impacts on health, with a majority of these products acting on aging as well as on neurodegeneration (Siracusa et al., 2020). Ursolic acid has been established to exhibit a substantial anti-inflammatory effect by inhibiting the DA-neuron degeneration in MPTP-stimulated Parkinson’s rats (Rai et al., 2019). A recent study found that Apium graveolens extract improved behavioral activity by facilitating protection against MPTP-stimulated PD through its antioxidant properties and a rise in DA neurons (Chonpathompikunlert et al., 2018).
The present investigation identified that MPTP exhibits a significant impact on microglia, triggering them and leading to modifications in their shape and phenotype, which contributes to the release of elevated ROS levels. This is consistent with a prior investigation showing elevated inflammatory marker levels, like IL-1β, IL-6, and TNF-α. ROS activates the NF-κB pathway, which phosphorylates a kappa inhibitor and increases the generation of pro-inflammatory cytokines, including TNF-α, IL-1β, and IL-6 (Ahmed & Kadhim, 2024). TF treatment successfully resulted in the suppression of TNF-α, IL-1β, and IL-6 levels, which was also noticed in previous studies (Rong et al., 2020; Zhang et al., 2022).
Histopathological illustrations of the effect of MPTP on DA neurons indicated that the tissues exhibited a decline in DA neurons, which resulted in a reduction in dopamine production by these neurons. This outcome coincides with research demonstrating that MPTP lesioning causes elevated glutamate release and hyperglutamatergic activity in the subthalamic nucleus. These consequences could be linked to diminished dopamine synthesis and, as a result, the loss of striatal dopamine due to the degradation of DA neurons in PD brain tissue (Moraes et al., 2016).
Conclusion
In summary, our in vivo study demonstrates that TF exerts significant neuroprotective effects in a mice model of MPTP-induced PD. TF treatment effectively alleviated motor deficits, reduced oxidative stress, inhibited MAO-A activity, and suppressed pro-inflammatory cytokine levels, thereby preserving dopaminergic neurons in the substantia nigra. These findings highlight TF’s potential as a promising therapeutic agent for the prevention or management of PD. Further studies are warranted to elucidate the precise molecular mechanisms underlying its neuroprotective effects and to evaluate its clinical applicability.
Footnotes
Abbreviations
BSA: Bovine serum albumin; CAT: Catalase; DA: Dopaminergic; GSH: Glutathione; IL-1β: Interleukin-1β; IL-6: Interleukin-6; MAO-A: Monoamine oxidase-A; MDA: Malondialdehyde; MPP+: 1-Methyl-4-phenylpyridinium; MPTP: 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine; PD: Parkinson’s disease; ROS: Reactive oxygen species; SNpc: Substantia nigra pars compacta; SOD: Superoxide dismutase; TNF-α: Tumor necrosis factor-α.
Data Availability
Data will be made available on request.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the Department of Neurology, First Hospital of Shanxi Medical University.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The participant has provided informed consent for the submission of the article to the journal.
