Abstract
Background
Ketamine, a commonly used anesthetic, plays a significant role during surgery, but its potential risk for neurotoxicity has drawn considerable attention.
Objectives
This study was aimed to investigate the effects of methylprednisolone (MP) loaded onto mesoporous silica nanoparticles (MSN) based on chitosan solution (CS) in ketamine overdose-induced neurotoxicity, focusing on its effects on neural function recovery and cellular damage. The goal was to provide new strategies and a theoretical foundation for the treatment of neuro injury.
Materials and Methods
MSN/CS/MP collagen (MP col) was prepared using the sol-gel method and characterized for its drug loading capacity, encapsulation efficiency, and release performance. Forty Sprague-Dawley rats were divided into five groups: control group (CG), ketamine group (KG), low concentration group (LG), medium concentration group (MG), and high concentration group (HG)). Except for the CG, the remaining groups were subjected to a ketamine-induced nerve injury model and received different concentrations of MSN/CS/MP col injections. Subsequently, the expression of key hippocampal proteins, apoptosis levels, and spatial memory were evaluated.
Results
The drug loading capacity of MSN/CS/MP col was 23.01 ± 0.51%, and the encapsulation efficiency was 95.24 ± 2.36%. Compared to CG, the escape latency was prolonged and exploratory time was shortened in the KG, LG, MG, and HG. The levels of p-Akt/Akt, Bcl-2, and PSD-95 were reduced, while the levels of Cas-3/Cas-3 and Bax were increased in the KG. The density of apoptotic cells was elevated in the KG and HG (
Conclusion
MSN/CS/MP col promotes functional recovery by regulating key proteins and reducing neuronal apoptosis, offering a novel approach for the treatment of nerve injury.
Introduction
Anesthetics play an essential role in surgical procedures, making the process safer and more comfortable. However, the use of anesthetics has also raised some concerns. Ketamine, as a widely used anesthetic drug, has played an obvious role in surgery but has attracted attention due to its potential risk of neural injury (Abram et al., 2022). Studies have found that excessive ketamine use can lead to abnormal changes in neurons, especially affecting the developing nervous system more clearly (Zhang et al., 2021). Research has indicated that ketamine overdose induces hippocampal neuronal injury in developing rats, leading to cognitive dysfunction (Barreto Chang et al., 2022; Cichon et al., 2023). However, there is still no ideal treatment to address this issue.
Methylprednisolone (MP) has been shown to protect and promote the repair and regeneration of neural tissue through its anti-inflammatory, immunosuppressive, and anti-allergic pharmacological effects in neural injury (Dubert et al., 2020; Li et al., 2022). However, the clinical use of MP is often limited by poor drug release and targeting, which restricts the effectiveness of its therapeutic impact. The application of nanotechnology in the medical field is gaining increasing attention, especially for its potential in drug delivery systems (Rabiee et al., 2021). Nanoparticles, as nano drug carriers, have shown great promise in drug delivery to the nervous system and neuroprotection due to their tiny particle size and unique surface properties (Feng et al., 2022). Studies have indicated that mesoporous silica nanoparticles (MSN) can carry and release drugs, and influence cell survival, proliferation, and function through interactions with cells, playing a role in neural function repair (Farshchi & Hasanzadeh, 2020). However, researchers have developed MSN due to potential cytotoxicity when used alone (Abdali et al., 2022; Sharma et al., 2023). Mesoporous silica nano/collagen particles, as drug delivery carriers, provide an ideal microenvironment for cell attachment and proliferation, and promote the repair and regeneration of neural tissue through biocompatibility and biodegradability (Berger et al., 2021; Sirkkunan et al., 2021). The application of nano drug-loaded particles in drug delivery offers new possibilities for the treatment of neurofunctional disorders. It brings a new perspective to the exploration of neuroprotective and reparative mechanisms (Mihaiescu et al., 2012). Chitosan solution (CS) is a natural polymer material that forms microparticles typically ranging from 1 to 1,000 nm. Due to its unique physicochemical properties, chitosan nanoparticles have attracted considerable attention in drug delivery. When combined with materials such as MSN to construct composite drug delivery systems, CS can enhance the biocompatibility of the system, as well as enable controlled drug release, targeted delivery, and reduction of toxic side effects (Poznanski et al., 2023; Zhou et al., 2023). Therefore, this study considers the use of CS as one of the components for constructing composite nanomaterials.
This study explored the application effects of MSN/CS/methylprednisolone-collagen (MP-col) (MSN/CS/MP-col) in neural injury caused by ketamine overdose, especially its impact on neural function recovery and the cellular damage mechanism. By systematically evaluating the role of this nano drug delivery system in a ketamine overdose neural injury model, it helps reveal its potential therapeutic mechanisms and provides new ideas and evidence for the intervention of neural injury after ketamine overdose.
Materials and Methods
Preparation of MSN/CS/MP-col
The method for synthesizing MSN: A total of 0.5 g of cetyltrimethylammonium bromide (Sigma–Aldrich, USA), 4 g of NaOH (Sigma–Aldrich, USA), and 240 g of deionized water was added to a flask and stirred at 300 rpm for 2 h at 80°C to ensure complete dissolution of the chemicals. Subsequently, 2.5 mL of tetraethyl orthosilicate (Sigma–Aldrich, USA) was rapidly introduced, and stirring was continued for 2 h. The resulting mixture was then filtered and washed three times with ethanol (Sigma–Aldrich, USA). Next, a 10 mg/mL NH4NO3-ethanol solution (Jining Fangyu Chemical Co., Ltd., China) was added, and the mixture was stirred at 80°C and 300 rpm for 6 h. The precipitate was collected and washed, yielding the MSNs.
A total of 0.675 g of MSN was added to 5 mL of CS (Sigma–Aldrich, USA) and ultrasonically dispersed for 5 min. Under a nitrogen atmosphere, 2 mL of methacrylic acid (Sigma–Aldrich, USA) was added, and the mixture was stirred at 300 rpm for 30 min. The temperature was then rapidly increased to 80°C, and 8 mg of potassium persulfate (Sigma–Aldrich, USA) was introduced, followed by stirring at 300 rpm for 2 h. Afterward, the temperature was adjusted to 50°C, and 0.1 mL of glutaraldehyde (Sigma–Aldrich, USA) was added, with stirring maintained at 300 rpm for 2 h. The resulting precipitate was collected and washed, yielding the MSN/CS composite.
Next, 5 mg of MSN/CS was added to 2 mL of a 1 mg/mL MP solution (Sigma–Aldrich, USA) and stirred at 800 rpm at room temperature for 24 h. The precipitate was collected after centrifugation at 1,000 rpm for 10 min, resulting in the MSN/CS/MP composite.
A total of 3 g of dry powdered collagen (Shanghai Guduo Biotechnology Co., Ltd., China) was gradually added to 40 mL of a 2% acrylic acid solution (Sigma–Aldrich, USA), and the mixture was stirred at 100 rpm until it was completely dissolved. The pH of the resulting solution was adjusted to neutral (pH = 7), and an appropriate amount of MSN/CS/MP was added. The final concentrations of the mixture were adjusted to 10, 25, and 50 µg/mL based on the mass of MSN. The solution was then transferred to lyophilization vials, rapidly frozen, and thoroughly ground. The resulting powder was re-dissolved in sterile physiological saline (Beijing Institute of Biological Products Co. Ltd., China), filtered, and the 10, 25, and 50 µg/mL MSN/CS/MP-col solutions were obtained. The solutions were sterilized and stored at 4°C.
Characterization of MSN, MSN/CS, and MSN/CS/MP-col
The particle size and morphology were analyzed using scanning electron microscopy (SEM, JEOL Ltd., Japan) to observe the morphology and particle size distribution of MSN and MSN/CS composites. The samples were dispersed in ethanol, dropped onto copper grids, and allowed to air dry at room temperature before testing.
The specific surface area and pore size distribution were determined by nitrogen adsorption-desorption isotherms (Brunauer–Emmett–Teller (BET) method, Micromeritics, USA) to measure the specific surface area, pore volume, and pore size distribution of MSN and MSN/CS composites.
The drug loading and encapsulation efficiency of MP in the MSN/CS/MP-col were obtained. The initial mass of MP added was determined. The mass of MP in the supernatant during the preparation of the MSN/CS/MP-col was obtained by centrifugation; the optical density (OD) at 254 nm was subjected to measurement using an ultraviolet (UV) spectrophotometer (Agilent Technologies, USA), and the mass of MP in the supernatant was calculated based on the standard curve.
An appropriate amount of MSN/CS/MP-col was added to phosphate-buffered saline (PBS) (Moregate Biotech, Australia) containing a small amount of type I collagenase (Shanghai Guduo Biotechnology Co., Ltd., China) at pH = 7.2. The mixture was incubated at 37°C and 72 rpm/min under constant temperature and oscillation. Samples were taken at different time points, and the OD at 254 nm was subjected to measurement using a UV spectrophotometer. The release of MP was calculated based on the standard curve.
Animals and Grouping
Forty male specific pathogen-free (SPF)-grade Sprague–Dawley (SD) rats (Chengdu Dasu Laboratory Animal Co., Ltd., China) were prepared, aged 7 days at the start of the experiment, and with an approximate body weight of 10–15 g. Rats at 7 days of age were selected because, during this period, the central nervous system is undergoing rapid development, making it ideal for observing the effects of drugs on neurodevelopment. Their mothers raised the rats under standard breeding conditions (23°C ± 2°C, relative humidity at 50% ± 5%, dim light with a light exposure time of 12 h/day, well-ventilated, and noise below 80 decibels) before the experiment. They were fasted and water-deprived for 6 h before the experiment. This experiment was reviewed and approved by the ethics committee of the hospital.
The 40 rats were randomly grouped, with eight rats in each group. They were designated as the control group (CG), ketamine group (KG), low concentration MSN/CS/MP-col group (LG), medium concentration MSN/CS/MP-col group (MG), and high concentration MSN/CS/MP-col group (HG). Except for the CG, the other four groups were prepared with a rat model of hippocampal neuronal injury caused by excessive ketamine.
CG: 0.1 mL of physiological saline was injected at the same time; KG: Ketamine (Fujian Gutian Pharmaceutical Co., Ltd., China) was administered at a dose of 50 mg/kg via intraperitoneal injection. LG, MG, and HG: 10, 25, and 50 µg/mL of MSN/CS/MP-col and 0.1 mL of 50 mg/kg ketamine mixture were injected via the tail vein.
Impact of MSN/CS/MP-col on Cells
Human neuroblastoma SH-SY5Y cells (Kunming Cell Bank of the Chinese Academy of Sciences, China) were subjected to culture in Dulbecco’s modified Eagle medium (DMEM) (Sigma–Aldrich, USA) having 15% fetal bovine serum (FBS) (Gibco, USA) and 1% penicillin and streptomycin, at 37°C, 5% CO2 (Shenzhen RWD Life Technology Co., Ltd., China). When the cells grew to about 80%, the supernatant was discarded, washed once with PBS, and trypsin containing ethylene diamine tetraacetic acid (Thermo Fisher Scientific, USA) was applied, centrifuged in a KS50R low-speed (Hunan Kaida Scientific Instrument Co., Ltd., China) at 1,800 r/min for 5 min. The supernatant was discarded, and DMEM with 10% FBS was applied. Cells in the log phase were collected, centrifuged (1,800 r/min for 5 min), and divided into four groups, which were stored at 4°C. 10, 25, and 50 µg/mL of MSN/CS/MP-col solutions and an equal volume of physiological saline (CG) were added, cultured in a 37°C, 5% CO2 incubator for 24 and 72 h. Methylene blue (Sigma–Aldrich, USA) was used for hematoxylin and eosin (H&E) staining, and the morphology was observed under a light microscope.
After the end of the culture, the culture medium in the culture dish was poured off, and then 20 µL of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution (Aladdin, USA) was applied to immerse the cells fully. The dish was placed at 37°C, 5% CO2 for 4 h. The MTT solution in the culture dish was aspirated using a pipette (Eppendorf, Germany), and 200 µL of dimethyl sulfoxide (DMSO) (OriGen, USA) was added. 180 µL of the sample solution was added to the culture plate, and the OD at 570 nm was measured using an HBS-ScanX microplate reader (Nanjing Detie Experimental Equipment Co., Ltd., China) to assess the survival and proliferation of the cells.
Morris Water Maze (MWM) Test
When the rats reached 30 days of age, a circular MWM (Beijing Keyue Huacheng Technology Co., Ltd., China) (diameter 1.5 m, water depth 30 cm, 22°C–24°C) was prepared, with a transparent platform placed in the pool for the rats to escape onto. Multiple positioning marks were placed on the pool walls to help the rats recognize different positions. The rats were placed into the pool from different quadrants (excluding the quadrant with the platform), and the time (s) it took to climb onto the platform, that is, the escape latency, was recorded. The test was conducted for 5 consecutive days. On the 6th day, the platform was removed, and the time (s) the rats spent in the quadrant where the platform was located within 2 min, the exploration time, was recorded.
Neurological Injury Indicators (Immunoblotting Detection)
At 24 h following the experiment, the selected rats were intraperitoneally injected with 50 mg/kg pentobarbital. The hippocampal tissues of the rats were isolated on ice, cut into blocks, and fixed with 4% paraformaldehyde (Thermo Barnstead, USA) for 24 h, and stored at 4°C for later use.
The sample tissues were rinsed using PBS, and 100 µL of radio-immunoprecipitation assay lysis solution (Beijing Solarbio Technology Co., Ltd., China) was applied (4°C, half an hour). The cells or tissues were broken using a cell scraper to release the proteins. The bicinchoninic acid detection kit was used for protein quantification. 15% gel was selected for sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), and proteins were loaded into the gel according to the sample concentration, with molecular weight markers added to determine the size of the proteins, and electrophoresis separation was performed. The separated proteins were transferred from the gel to the polyvinylidene fluoride (PVDF) membrane using a semi-wet transfer apparatus (in 5% skim milk powder for blocking to avoid non-specific binding). Diluted at 1:1,000, anti-p-Akt antibody (Shanghai Sig Biology Technology Co., Ltd., China), anti-Akt antibody (Shanghai Sig Biology Technology Co., Ltd., China), anti-activated Cas-3 antibody, anti-Cas-3 antibody, anti-Bax antibody, anti-Bcl-2 antibody, anti-PSD-95 antibody (Abcam, UK), and diluted at 1:500, anti-activated Cas-3 antibody, and anti-β-actin antibody were added, and incubated at 4°C overnight. The membrane was rinsed three times with PBS having Tween-20 (Sigma–Aldrich, USA), each time for 5–10 min, to remove unbound antibody. Horseradish peroxidase-labeled corresponding biotinylated secondary antibody (Sigma–Aldrich, USA) diluted at 1:5,000 was added, and incubated for 2 h. The membrane was rewashed three times. The proteins on the membrane were visualized using the enhanced chemiluminescence (ECL) reagent (Thermo Barnstead, USA), and the luminescent signals of the protein bands were observed. The APX100 digital imaging system (Olympus, Japan) was used for imaging, and the Image J software was used to quantitatively determine the OD of each protein band, which was corrected with molecular weight markers. The expression of p-Akt/Akt, anti-activated Cas-3 antibody/anti-Cas-3 antibody, anti-Bax antibody, anti-Bcl-2 antibody, and anti-PSD-95 antibody was statistically compared with the reference protein.
Apoptosis Detection
The same tissue samples used for immunoblotting detection were fixed, dehydrated in a gradient, and the hippocampal tissues were gradually soaked in ethanol solutions to remove moisture. The hippocampal tissues were embedded in paraffin, and 5-µm-thick sections were obtained using a microtome. After sectioning, the sections were deparaffinized. PBS was used for washing, and the labeling mixture was added at 37°C for 60 min, followed by another PBS wash, and the conversion solution was added at 37°C for half an hour, followed by another PBS wash. The 5-bromo-4-chloro-3-indolyl phosphate (BCIP)/nitro blue tetrazolium (NBT) alkaline phosphatase chromogenic reagent kit (Shanghai Enzyme-linked Biotechnology Co., Ltd., China) was used for H&E staining, followed by dehydration with an ethanol solution and clarification. The sections were placed on slides under a BX51 microscope (Olympus, Japan) for observation and pathological morphological assessment. ImageJ was used to compute the density of apoptotic cells.
Statistical Processing
Statistical Package for the Social Sciences (SPSS) 26.0 was employed. Measurement data were presented as mean ± standard deviation (SD) (
Results
Characterization of MSN and MSN/CS Composites
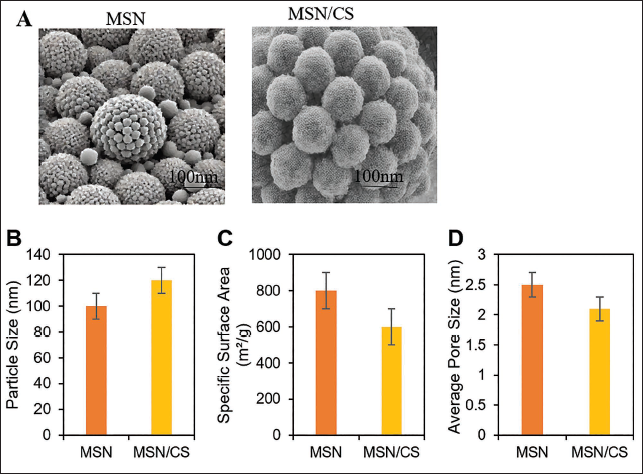
SEM images revealed that MSN particles were spherical with a uniform size distribution, approximately 100 nm in diameter. The MSN/CS composite exhibited a uniformly coated shell on the surface, with a slight increase in particle size to approximately 120 nm (Figure 1A). The surface area of MSN was relatively high (around 800 m2/g), with an average pore size of approximately 2.5 nm. After CS coating, the specific surface area decreased to 600 m2/g, indicating that CS partially filled the pores (Figure 1B).
Scanning Electron Microscopy (SEM) Images (A), Average Particle Size (B), Specific Surface Area (C), and Average Pore Size (D).
Drug Loading and Release Amount of MSN/CS/MP-col
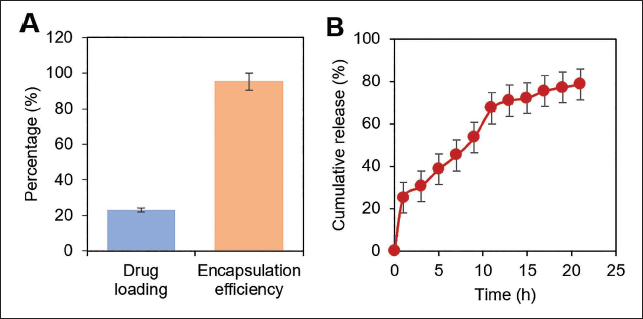
The drug loading and encapsulation efficiency of MP in the MSN/CS/MP-col were 23.01% ± 0.51% and 95.24% ± 2.36%, respectively. The
Drug Release Effect of Mesoporous Silica Nanoparticles (MSN)/Chitosan Solution (CS)/Methylprednisolone-collagen (MP-col). (A) Drug Loading and Encapsulation Efficiency of MSN/CS/MP-col and (B) In Vitro MP Release Curve of MSN/CS/MP-col.
Impact of MSN/CS/MP-col on SH-SY5Y
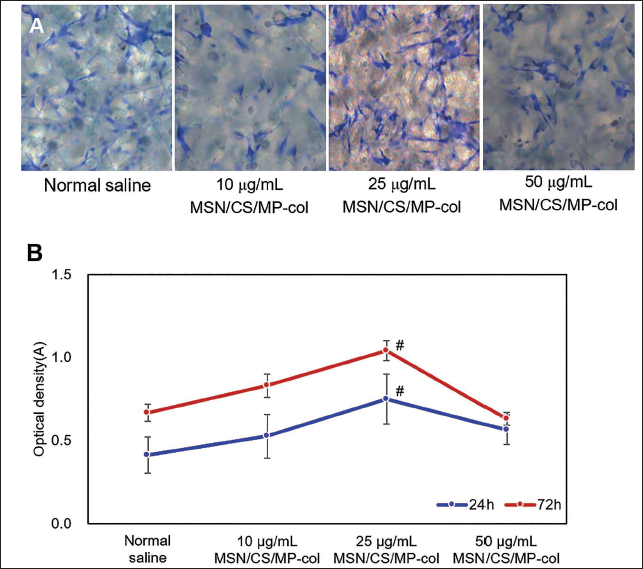
HE staining results of SH-SY5Y cells cultured with physiological saline and 10, 25, and 50 µg/mL MSN/CS/MP-col are illustrated. In the visual fields, the majority of SH-SY5Y cells remained in a normal state. The cells using physiological saline grew in a spindle shape without any obvious abnormalities (Figure 3A). However, the SH-SY5Y with 10 and 50 µg/mL MSN/CS/MP-col did not show a visible promotion of cell growth (Figure 3B–3C). In contrast, the SH-SY5Y with 25 µg/mL MSN/CS/MP-col not only grew in a spindle shape but also extended along the axons, showing a more active growth state (Figure 3D). The results of the MTT assay were similar to those of the H&E staining, indicating that the proliferation and activity of SH-SY5Y cells increased, and then decreased with time and concentration of MSN/CS/MP-col, reaching the highest activity at 25 µg/mL (Figure 3E).
Behavioral Results
As time and the number of experiments increased, the escape latency of all rats suggested a decreasing trend. The escape latency of the KG was visibly higher than that of the CG (
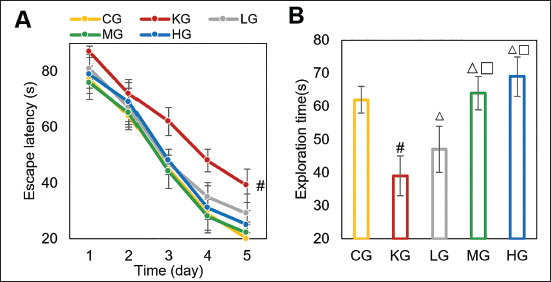
In Figure 4B, the exploration time of the KG was visibly lower than the CG. The LG, MG, and HG extended their exploration time in the same area, and the exploration duration was visibly higher than the KG, with the MG and HG showing a visibly higher degree of recovery in memory function than the LG (
Contrast of Protein Expression
As against the CG, p-Akt/Akt, PSD-95, and Bcl-2 in the KG were markedly decreased; as against the KG, those were markedly increased in the LG, MG, and HG (
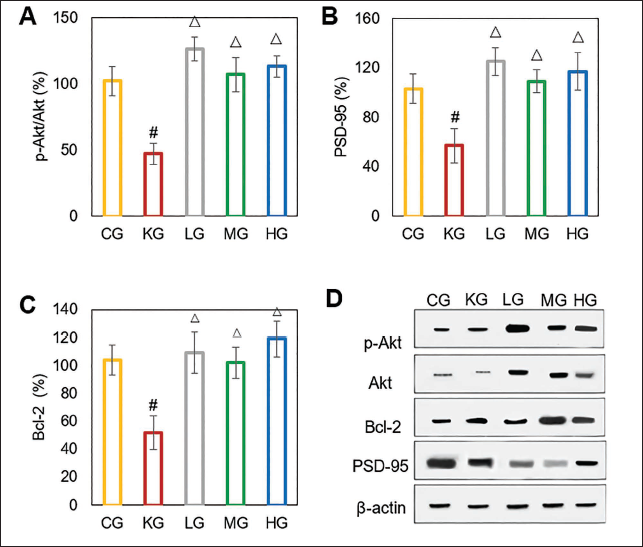
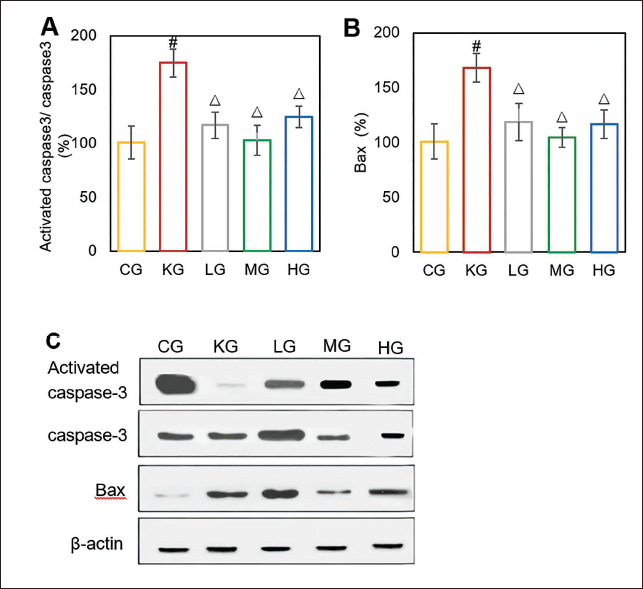
As against the CG, activated Cas-3/Cas-3 and Bax in the KG were markedly reduced; as against the KG, those were markedly raised in the LG, MG, and HG (
Neuronal Apoptosis Detection
Apoptotic cells appeared black and were generally shrunken. From the H&E staining images, there were only a few apoptotic cells in the CG, with most cells showing a normal morphology (Figure 7A). In contrast, the KG suggested many black apoptotic cells with few normal cells (Figure 7B). In the LG, MG, and HG, as the concentration increased, the number of apoptotic cells first decreased and then increased (Figure 7C–7E), with the MG showing a reduction in apoptotic cells comparable to that of the CG.
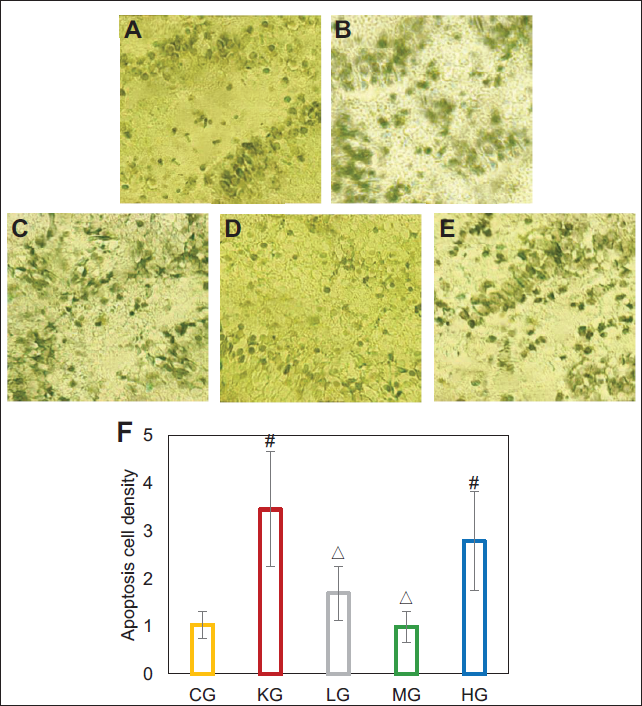
The ImageJ software analysis results were consistent with the H&E staining. The apoptotic cell density in the KG was 3.453, markedly higher than the CG (
Discussion
This study explored the effects of MSN/CS/MP-col on the proliferation of SH-SY5Y, learning and memory abilities in the MWM, and apoptosis of hippocampal neurons. It assessed its reparative role in ketamine-induced neural injury through the expression of proteins. The study analyzed the drug release and sustained-release properties of the MSN/CS/MP-col, indicating that the drug-loaded microspheres effectively delayed drug release, further confirming the excellent sustained-release properties of the prepared microspheres.
This study innovatively employed a multi-level
However, this study has certain limitations. First, although the effects of MSN/CS/MP-col were explored through cellular and animal experiments, the long-term effects and potential side effects were not further investigated, which are crucial for clinical translation. Second, the
In summary, although this study provides compelling evidence of the potential neuroprotective effects of MSN/CS/MP-col, further research is necessary to address the aforementioned limitations, particularly regarding long-term effects, dose dependency, and its application in human neural injury.
Conclusion
This study demonstrates that MSN/CS/MP-col has visible neuroprotective and functional recovery effects in neural injury caused by ketamine overdose. MSN/CS/MP-col effectively promotes the damaged neurons’ survival and functional recovery. In addition, under low- and medium-concentration treatment conditions, MSN/CS/MP-col markedly reduced the level of cell apoptosis, showing potential application value in reducing neuronal injury caused by excessive anesthesia. These findings offer empirical support for the possible application of MSN/CS/MP-col to treat neural injury caused by excessive anesthesia.
Footnotes
Abbreviations
Cas-3: Caspase-3; CG: Control group; CS: Chitosan solution; DMEM: Dulbecco’s modified Eagle medium; FBS: Fetal bovine serum; HG: High concentration MSN/CS/MP-col group; KG: Ketamine group; LG: Low concentration MSN/CS/MP-col group; MG: Medium concentration MSN/CS/MP-col group; MP: Methylprednisolone; MP-col: Methylprednisolone-collagen; MSN: Mesoporous silica nanoparticles; MWM: Morris water maze; PBS: Phosphate-buffered saline; PVDF: Polyvinylidene fluoride.
Authors Contribution
Conception and study design: Shuangju Liao; data acquisition and analysis: Shuangju Liao, Huiqiong Huang, and Yuan Tang; manuscript draft, editing, and revision: Shuangju Liao and Huiqiong Huang. All authors approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This animal study protocol was approved by the Ethics Committee of West China Fourth Hospital of Sichuan University, in compliance with Chinese national guidelines for the care and use of animals.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
