Abstract
Introduction
Objectives
This study aimed to explore the PNS’s potential tumor-inhibitory effects and related mechanisms in LUAD.
Materials and Methods
LUAD cell lines A549 and Lewis were utilized to examine the proliferation, colony formation, and migration of LUAD cells after PNS treatment. WI38 cells were employed to evaluate the toxic impact of PNS on human lung fibroblasts. A Lewis subcutaneous tumor-bearing model was established to assess the combined effects of PNS and carboplatin (CBP) or immune checkpoint blockade with programmed cell death 1 antibody (PD1ab). Lentivirus transfection and quantitative polymerase chain reaction (qPCR) assays were conducted to explore the role of serine protease inhibitor clade E member 1 (SERPINE1) in PNS’s anti-tumor mechanism. Immunohistochemistry was utilized to detect CD8+ T cell infiltration in tumor tissue. Cell counting kit-8 and qPCR assays were applied to determine the specific PNS components exerting anti-tumor effects.
Results
Conclusion
PNS suppressed LUAD progression and enhanced CBP and PD1ab therapeutic effects by downregulating SERPINE1 expression, indicating its potential as a promising agent for LUAD therapy.
Introduction
Lung cancer remains the malignancy with the highest global morbidity and mortality rates, with lung adenocarcinoma (LUAD) as the most common pathological type, comprising approximately 40% of lung cancer (Herbst et al., 2018). Patients with LUAD require individualized therapeutic regimens based on tumor progression. Those with early-stage disease typically undergo surgical treatment (Musika et al., 2021), whereas those with advanced-stage and metastatic LUAD require systemic therapy (Kris et al., 2017). Although treatments such as chemotherapy effectively slow tumor progression and improve their prognosis to some extent, anti-tumor efficiency in patients with LUAD is usually limited by decreased drug sensitivity, resulting in tumor recurrence and poor prognosis (Arbour & Riely, 2019; Zhang et al., 2020). Additionally, reduced sensitivity to drugs often brings unnecessary toxicity and side effects to patients with LUAD. Recently, immune checkpoint blockade (ICB) therapies have been applied to therapeutic regimens of various tumors, with programmed cell death 1 antibody (PD1ab) and programmed death ligand 1 antibody (PD-L1ab) being the most frequently used agents (Dermani et al., 2019; H. Zhang et al., 2021). It has been reported that the overexpression of immune checkpoint molecules, such as programmed death ligand 1 (PD-L1), in tumor cells promotes the formation of an immunosuppressive microenvironment that significantly enhances malignant biological properties. Consequently, ICBs, such as PD1ab, targeting to block the PD-1/PD-L1 can mitigate these knock-on effects and are considered a powerful and commonly used immune therapy strategy. Currently, checkpoint blockade agents, such as PD1ab (pembrolizumab and nivolumab), are approved for clinical cancer therapy, offering improved prognosis and lower toxic side effects than traditional chemotherapeutic regimens (Tang et al., 2022). However, the average PD1ab objective response rate for all cancer types is only 20%–25% (Topalian et al., 2012). In LUAD cases treated with PD1ab, approximately 80% of patients developed resistance and could not acquire satisfying therapeutic benefits (Shukuya & Carbone, 2016). Thus, there is an urgent need to identify novel strategies to prevent LUAD progression and increase sensitivity to therapies such as chemotherapy and immunotherapy.
Due to their rich chemical diversity and pharmacological activities, natural products have significantly contributed to drug discovery for treating various diseases (Atanasov et al., 2021; Mishra, Mishra, Rai, Singh, et al., 2023). For example, the commonly used chemotherapy drug paclitaxel, derived from
This study investigated the activity and mechanisms by which PNS prevented LUAD progression utilizing
Materials and Methods
Referring to a recent report, we bought PNS from Chuxiong Yunzhi Pharmaceutical Co., Ltd. (purity percentage: ≥80%). Moreover, PNS (B21102, purity percentage: ≥90%), GRb1 (B21050, purity percentage: ≥98%), NR1 (B21099, purity percentage: ≥98%), GRg1 (B21057, purity percentage: ≥98%), GRe (B21055, purity percentage: ≥98%), and GRd (B21054, purity percentage: ≥98%) from Shanghai Yuanye Biotechnology Co., Ltd., CBP (T1058) was purchased from Shanghai Topscience Biotechnology Co., Ltd., PD1ab (BE0146) was purchased from Shenzhen Neobioscience Biotechnology Co., Ltd.
Cell Culture
Human LUAD cell lines A549, HCC827, H1299, murine LUAD cell lines Lewis, and normal lung fibroblast cells WI-38 were obtained from Procell (Wuhan, China). The cells were cultured in Dulbecco’s modified Eagle medium (DMEM) (Cat#C11995500BT, Gibco, USA)/nutrient mixture F-12 medium containing 1% penicillin–streptomycin and 10% fetal bovine serum (FBS, Cat#10091148, Gibco, USA) in a conventional incubator at 37°C with 5% carbon dioxide (CO2). A549, HCC827, and H1299 cells were cultured in Roswell Park Memorial Institute-1640 medium (Cat#C11875500BT, Gibco, USA), while Lewis cells were maintained in DMEM and WI-38 cells in Eagle’s minimum essential medium. All cultured media were supplemented with 10% FBS and 1% penicillin–streptomycin. All cells were incubated at 37°C in a 5% CO2 humidified incubator.
Cell Viability Assay
The CCK-8 kit (Cat#GW770, Dojindo, Kumamoto, Japan) was used to measure cell viability quantitatively. Briefly, cells were digested by trypsin and uniformly seeded in a 96-well plate at 2 × 103 cells/well density. After 24 h, the medium was replaced with a medium containing specified PNS and carboplatin (CBP) concentrations. From days 1 to 5, the CCK-8 solution was added at 10 µL/well and incubated at 37°C in a 5% CO2 humidified incubator for 2 h. Cell viability was measured at 450 nm with a microplate reader (Tecan Sunrise, Salzburg, Austria). The procedure references the material and method used in recent literature (Lin et al., 2024).
Colony-formation Assay
After trypsin digestion, cells were seeded at a density of 600 cells in a six-well plate. The wells were mixed with 2 mL of complete medium with specified PNS and CBP concentrations. After approximately 10 days, the cells in the wells were fixed utilizing paraformaldehyde for 20 min and stained with Giemsa. All plates were counted for the number of colonies. This experiment references the methodologies used in a recent report (X. Li et al., 2022).
Wound-healing Assay
A density of 1 × 106 cells was first settled in a 6-well plate and allowed for fusion. Scratch wounds were generated using a 10 µL sterile pipette tip. After washing three times with phosphate-buffered saline, a series of images were captured to estimate the level of migration in each group of cells at 0, 12, and 24 h after scratching. The procedure references the material and method used in recent literature (Lin et al., 2024).
Reverse Transcription-Polymerase Chain Reaction (PCR)
After the specified treatment, cells were collected, and the total ribonucleic acid (RNA) was extracted utilizing TRIzol reagent (Cat#15596026CN, Invitrogen, USA). Complementary deoxyribonucleic acid (cDNA) was synthesized using the PrimeScript RT reagent kit (Cat#RR037A, TakaRa, Japan) in preparation for the PCR process. The primers for serine protease inhibitor clade E member 1 (SERPINE1) were sense 5′-ATTTGACGACGAATCGGACCC-3′ and anti-sense 5′-GTTCTTGCGGTCTTTCTGGGA-3′ (human), 5′-GTCACCAA CCGATTCGACCAG-3′, 5′-GGGACTCTTTACGCAACTGTT-3′ (mouse) and the primers for a-actin were sense 5′-CCTAAACAGGTTGATAGGCCAAA-3′ and anti-sense 5′-CTCGCCTTCCACAGAATCCA-3′. A comparative 2–∇∇CT method was used to quantify relative gene expression. This assay references the material and method used in a recent report (Li et al., 2021).
Cell Transfection
The Plasmid PCDNA3.1-mouse-SERPINE1 was obtained from Vigene (Cat#WZ010002, Shandong, China). Using Lipofectamine 2000 (Cat#11668019, Invitrogen, USA), the package tool cells (HEK293) were first transfected with plasmids for 24 h. Then, the medium was abandoned and replaced with a fresh, complete culture medium. After about 2 days, the targeted recombinant lentiviruses were collected and transfected into LUAD cells. A quantitative polymerase chain reaction (qPCR) assay was used to detect knockdown efficiency. The material and method followed this experiment in a recent report (W. Zhang et al., 2021).
Bioinformatic Analysis
Clinical data of patients with LUAD were initially downloaded from the cancer genome atlas (TCGA) database and analyzed using gene expression profiling interactive analysis (GEPIA2, gepia2.cancer-pku.cn). Overall survival (OS) and disease-free survival (DFS) were generated from GEPIA2. The analysis process followed the bioinformatic analysis in recent reports (Mishra, Mishra, Vamanu, et al., 2023a, 2023b).
In Vivo Efficacy Study
The C57BL/6 mice (female, 18–20 g, 5 weeks) were purchased from the Medical Laboratory Animal Center of Taizhou University. After feeding for 1 week with routine feed and sterile water, a density of 0.5–1 × 106 Lewis cells was subcutaneously injected into the flanks of C57BL/6 mice. When the volume of the tumors reached approximately 50–100 mm3, the mice were divided into groups for specified treatment, including PNS (160 mg/kg, oral administration, daily), CBP 5 mg/kg, and PD1ab (intraperitoneal injection, 100 µg/per mouse). The calculated formula for the tumor volume (TV) was as follows: TV = 0.5 ×
Statistical Analysis
The experimental data are presented as the mean ± standard deviation and statistically analyzed using GraphPad Prism software (version 9.0). The statistical significance of the differences was analyzed using Student’s and unpaired
Results
PNS Selectively Impaired the Malignant Biological Ability of LUAD Cells
To investigate the anti-tumor effect of PNS, CCK-8 detection was first used to examine the proliferation of human LUAD cell A549 and murine LUAD cell Lewis after treatment. After PNS treatment with concentrations of 25, 50, 100, 250, and 500 µg/mL, the viability of A549 cells was impaired and displayed dose-dependent (Figure 1A,
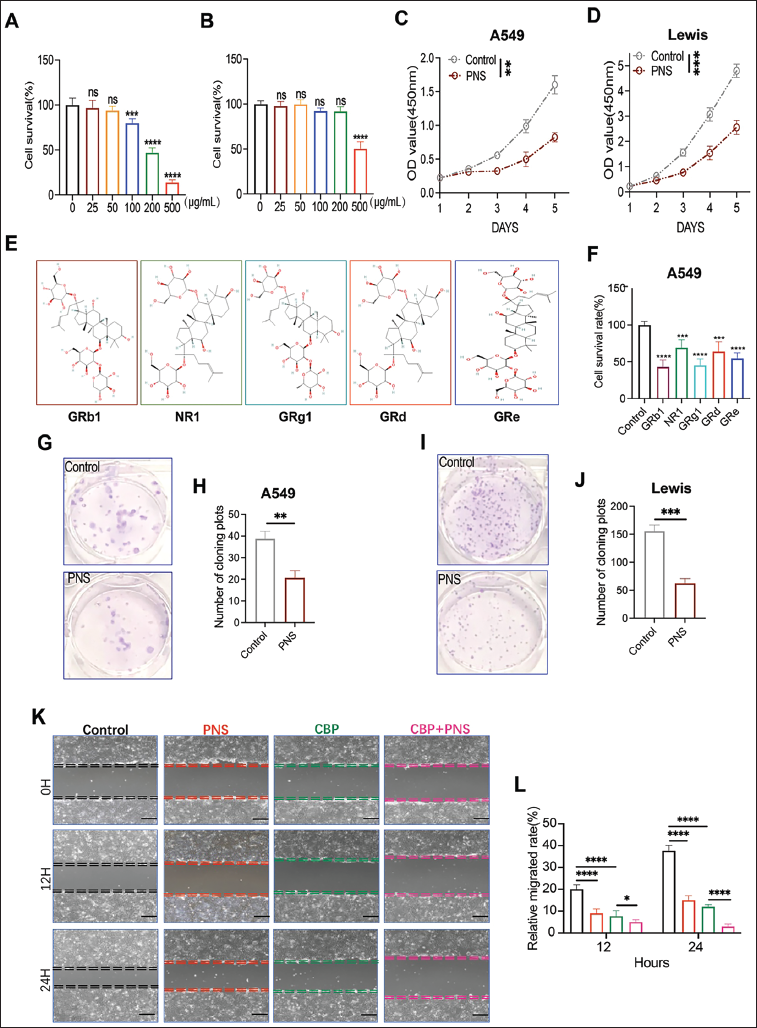
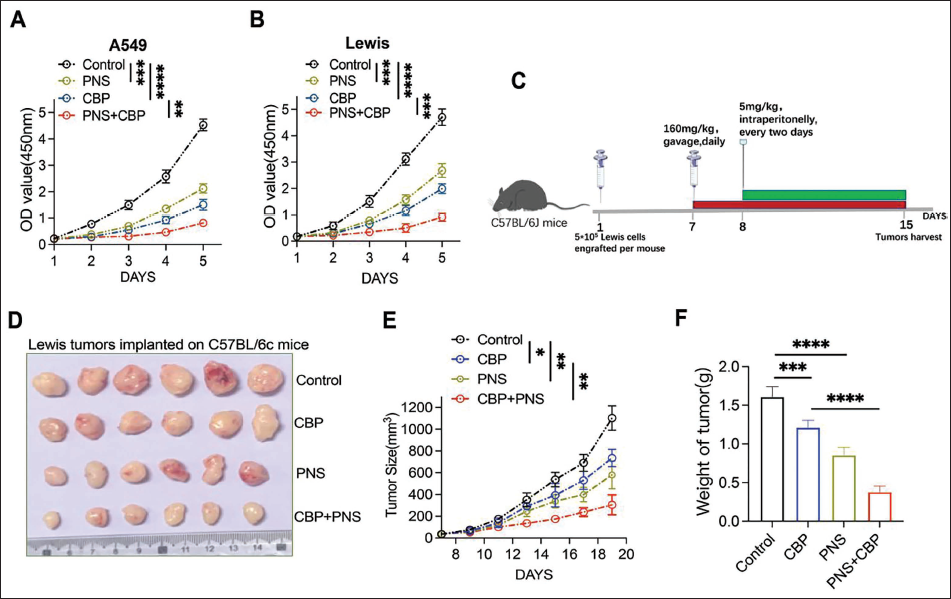
PNS Enhanced CBP’s Therapeutic Effect and Inhibited LUAD Growth In Vitro and In Vivo
First, CCK-8 detection was performed to detect the proliferation capacity after PNS and CBP treatment. As displayed in Figure 2A and B, treatment with PNS or CBP alone significantly inhibited cell growth compared to the control group (
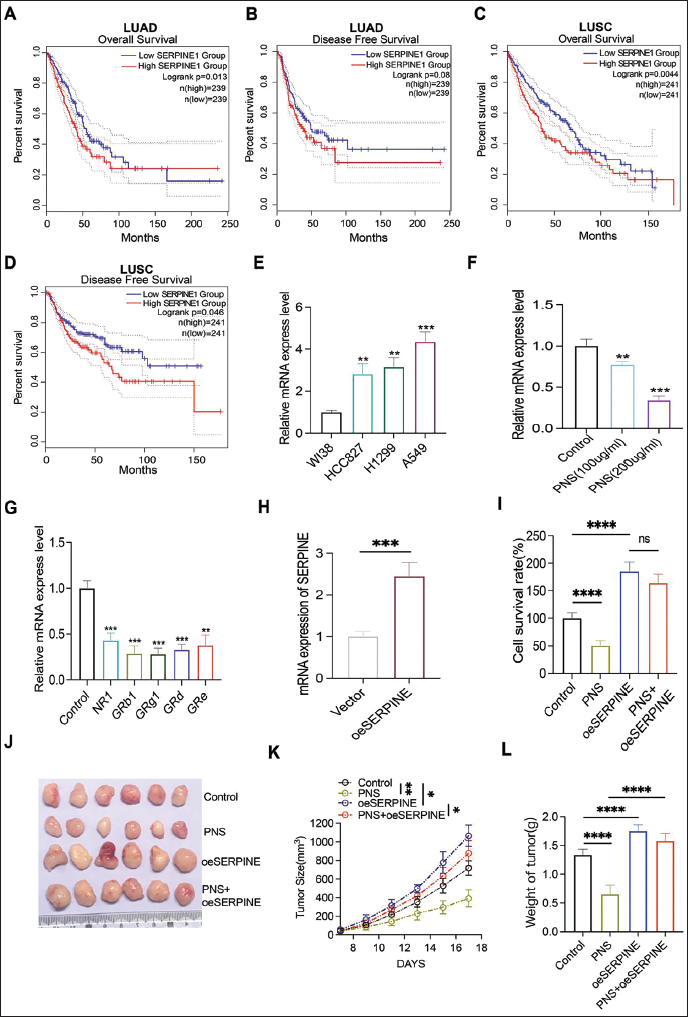
PNS Suppressed LUAD Progression by Downregulating SERPINE1
The RNA-seq data from a recently published study demonstrated that the expression levels of many vital genes, such as SERPINE1 (also known as PAI-1), were significantly altered after PNS treatment in breast cancer (Xia et al., 2023). The SERPINE1 was reported to be involved in various malignant processes, such as cell migration and immune evasion. It exerted a significant influence on many types of cancer, including cervical squamous cell carcinoma (Wei et al., 2023), bladder cancer (Furuya et al., 2022), and ovarian cancer (Furuya et al., 2022). However, its role in LUAD remains largely unknown. Consequently, we hypothesized that SERPINE1 is crucial in the mechanism of the PNS anti-tumor effect. First, using the TCGA database, we found that higher expression of SERPINE1 was associated with poor prognoses in LUAD, including OS and DFS (Figure 3A and B,
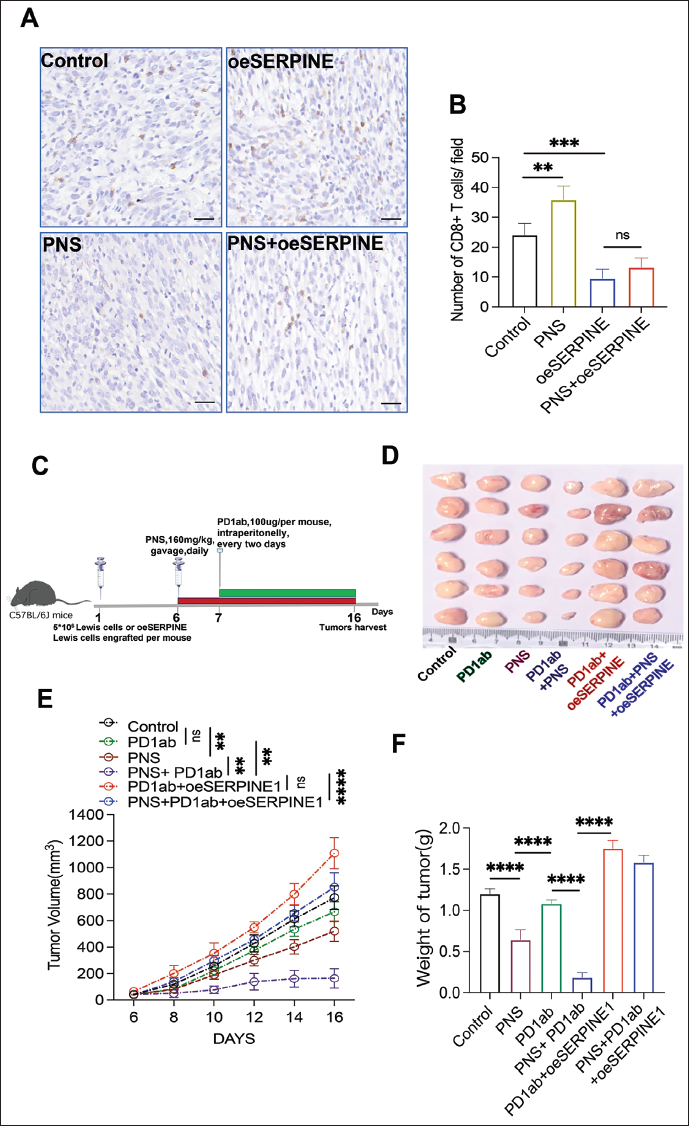
PNS Enhanced PD1ab Therapeutic Effect Via SERPINE1 on LUAD Tumors
Considering that SERPINE1 was reported to be strongly associated with immune cell infiltration and based on the aforementioned results, which indicated that PNS treatment significantly altered SERPINE1 expression, we examined the vital immune cell, CD8+ T cell infiltration, after PNS treatment. The results suggested that CD8+ T cells increased after PNS treatment compared with the control group (
Discussion
This study revealed that by downregulating SERPINE1, PNS selectively impaired the proliferative, colony formation, and migratory abilities of LUAD cells. Furthermore, PNS enhanced the therapeutic effect of the common chemotherapy drugs CBP and ICBs PD1ab. To our knowledge, this is the first study to explore the activity and underlying mechanism of PNS in tumor cells and the tumor immune environment. Overall, the results of this research suggested that PNS may be a potential therapeutic or preventive agent for treating LUAD.
Additionally, PNS has been reported to treat myocardial ischemia, cerebral infarction, atherosclerosis, and several other metabolic diseases (Wu et al., 2023). Recently, many studies have reported that PNS exhibits anti-tumor activity in various cancers. Xia et al. (2023) demonstrated that PNS normalizes tumor blood vessels by inhibiting EphA2 gene expression, thereby modulating the tumor microenvironment in breast cancer. Yao et al. (2021) highlighted that PNS promotes cell death and chemosensitivity in pancreatic cancer through the apoptosis and autophagy pathways. Cancer research on PNS focuses on its anti-tumorigenic action and the direct inhibition of cancer proliferation and angiogenesis. In this study, we also verified that PNS impaired the proliferative, colony formation, and migratory abilities of LUAD cells. A549 and Lewis were the main cell lines used in the present work and were reported to have multiple mutations in Kirsten rat sarcoma viral oncogene homolog (KRAS), epidermal growth factor receptor (EGFR), and other lung cancer-related genes (Bagnyukova et al., 2024; Xia et al., 2024), which gives a hint that the efficacy of PNS in treating LUAD may be limited by genetic variations. Generally, the results of this study corroborated the anti-tumorigenic activity, suggesting that its tumor-inhibitory effects may be less toxic and extend to various types of cancer.
The inhibition of SERPINE1 expression has been proven to prevent tumor progression in some types of malignancies. Accordingly, previous studies have highlighted that the inhibition of SERPINE1 may be a promising novel strategy for cancer therapy. In this study, we found that by downregulating SERPINE1, PNS selectively impaired the proliferative, colony-forming, and migratory abilities of LUAD cells
Targeted immune checkpoint drugs, such as PD-L1 antibodies and PD-1, have become promising therapeutic strategies due to their long-lasting effects and low toxicity. Despite this, some patients demonstrate a low response to ICBs, and only a small percentage of patients with advanced LUAD benefit from this form of treatment (Lahiri et al., 2023; Rotow & Bivona, 2017). As a result, strategies should be developed to enhance the therapeutic efficacy of ICBs, such as PD1ab, and to extend the benefits of immunotherapy to patients with low-response LUAD. Based on the previous result, PNS significantly altered SERPINE1 expression level, and a recent literature report that SERPINE1 exhibited close association with infiltrated immune cells (Li et al., 2023). We explored and verified that the inhibition of SERPINE1 promotes CD8+ T cell infiltration in LUAD tumors. The infiltration number of CD8+ T cells is the key to the anti-tumor activity of ICBs, such as PD1ab (Mariathasan et al., 2018). Consequently, our findings suggested that PNS enhanced the PD1ab therapeutic effect by downregulating SERPINE1 on LUAD tumors.
Conclusion
Our findings suggest that extracts from traditional Chinese medicine PNS enhance the common chemotherapy drug CBP and ICBs PD1ab inhibition effects on LUAD by downregulating SERPINE1. This may provide a preliminary theoretical basis for using LUAD to treat PNS. However, some limitations remain, such as understanding how PNS affects SERPINE1 and what the downstream of SERPINE1 is in the PNS anti-tumor action. Further experiments should be conducted to provide more direct and detailed evidence, which we will explore in more depth in the future.
Footnotes
Abbreviations
CCK-8: Cell counting kit-8; CBP: Carboplatin; cDNA: Complementary deoxyribonucleic acid; CO2: Carbon dioxide; DFS: Disease-free survival; DMEM: Dulbecco’s modified Eagle medium; DNA: Deoxyribonucleic acid; EGFR: Epidermal growth factor receptor; FBS: Fetal bovine serum; GRb1: Ginsenoside Rb1; GRd: Ginsenoside Rd; GRe: Ginsenoside Re; GRg1: Ginsenoside Rg1; ICB: Immune checkpoint blockade; IHC: Immunohistochemistry; KRAS: Kirsten rat sarcoma viral oncogene homolog; LUSC: Lung squamous cell carcinoma; LUAD: Lung adenocarcinoma; NR1: Notoginsenoside R1; OS: Overall survival; PAI-1: Plasminogen activator inhibitor-1 (alternative name for SERPINE1); PD1ab: Programmed cell death 1 antibody; PD-L1: Programmed death ligand 1; PD-L1ab: Programmed death ligand 1 antibody; PNS:
Acknowledgments
The authors are thankful to Professor Huirong Lin for providing technical support in the animal study.
Author Contributions
Yuhuai Wang, Ping Wang, Jiaqi Wang, Yeze Yang, and Fagen Huang are the major contributor in writing the manuscript. All authors read and approved the final manuscript.
Data Availability
The original contributions presented in the study are included in the article; further inquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Wenling Social Development Science and Technology Project (No.2023S00023).
Informed Consent
Not applicable.
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
