Abstract
Background
Anxiety and depression are among the most widespread mental health conditions worldwide, impacting millions and imposing a considerable burden on individuals and healthcare systems.
Objectives
The present research work was dedicated to examining the therapeutic values of nodakenin against anxiolytic and depressive-like conditions in an experimental mice model.
Materials and Methods
The experimental Swiss mice were administered 25 and 50 mg/kg of nodakenin and/or 20 mg/kg of imipramine, a standard anti-depressive drug. Various behavioral examinations, such as forced swimming, tail suspension, open field test, elevated plus-maze, and light and dark exploration tests, were performed to investigate the anxiolytic and anti-depressant properties of the nodakenin. Furthermore, the activity of monoamine oxidase-A (MAO-A) was assessed in the brain tissues of the experimental mice using a test kit.
Results
The present results indicated that nodakenin treatment reduced the immobility period, elevated the swimming duration in the forced swimming test, and reduced the immobility period in the tail immersion test. The nodakenin treatment considerably increased the exploratory behavior of the experimental mice. The results of the elevated plus maze test and light–dark box tests indicated the anti-depressant and anxiolytic properties of nodakenin in experimental mice. Furthermore, the nodakenin treatment also inhibited the MAO-A enzyme activity in the brain tissues of the experimental mice.
Conclusion
The current research demonstrates that the nodakenin treatment exhibits anxiolytic and anti-depressant properties in murine models. The present findings indicate the therapeutic potential of nodakenin as an alternate treatment for comorbid anxiety and depression conditions.
Introduction
Anxiety and depression are among the most common mental health disorders, affecting millions globally. The global burden of disease indicated that anxiety disorders accounted for 26.8 million disability-adjusted life years, underscoring the substantial burden these problems place on individuals and society. Depression is identified by the World Health Organization (WHO) as the foremost contributor to global disability, affecting around 300 million individuals worldwide, which is 4.4% of the global population (WHO, 2017). The high prevalence and disease burden of anxiety and depression further strain healthcare systems, increase rates of absenteeism, and reduce productivity, with the economic cost of lost productivity for these conditions estimated to be $1 trillion per year and forecast to reach $16 trillion by 2030 (Chodavadia et al., 2023). The causes of anxiety and depression are multifaceted, with both genetic and environmental factors playing a role. Addressing the significant burden of anxiety and depression requires a multifaceted approach, including improved access to evidence-based treatments, increased public awareness, and a focus on prevention and early intervention strategies (Lorvellec et al., 2024).
Anxiety and depression disorders share numerous characteristics and often manifest concurrently, highlighting the complex interplay between the underlying biological and environmental causes that participate in their progression. A growing body of research has implicated the serotonergic system and its interaction with the prefrontal cortex as a pivotal mechanism in the development of anxiety and depression. Specifically, dysregulation in the serotonin-prefrontal cortical circuitry has been connected with the emergence of distinct depression and anxiety phenotypes, highlighting the crucial role of this neural network in the manifestation of these disorders (Liu et al., 2024). Moreover, the role of environmental factors, such as stress and early-life adversity, in shaping the biological predisposition to depression has been widely recognized (Hong et al., 2022). Anxiety disorders, on the other hand, have been linked to a complex interplay between genetic, neurobiological, and psychological causes. The prefrontal cortex and its interactions with the amygdala and other limbic structures have been identified as a crucial neural circuit implicated in the regulation of anxiety responses. The underlying mechanisms of pathophysiology in anxiety and depression are multifaceted and involve the complex interplay of various neurobiological, genetic, and environmental factors. These disorders are not a result of a single defect but rather a dynamic and intricate process that requires a comprehensive understanding of the various systems and processes involved (Bie et al., 2024).
The management of anxiety and depression remains a significant challenge in the area of mental health. While a range of treatment options, including pharmacotherapy, psychotherapy, and somatic therapies, have been developed, their efficacy is limited, and many patients continue to experience treatment-resistant symptoms. The search for more effective and well-tolerated treatments has led researchers to study the efficacy of plant-derived bioactive compounds in the treatment of these disorders (Gutiérrez-Rodelo et al., 2023). Standard treatment modalities for anxiety and depression frequently include anti-depressant pharmacotherapy. While these treatments exhibit certain efficiency, they frequently entail various side effects, such as sexual dysfunction, weight gain, and gastrointestinal issues. Moreover, a significant proportion of patients, estimated at 20%–30%, do not respond adequately to these treatments and are considered to have treatment-resistant depression (Mrozek et al., 2023). Recently, there has been increasing attention on the use of plant-derived bioactive compounds as therapeutic agents for depression and anxiety disorders. These natural compounds, derived from medicinal plants, have been utilized in conventional medicine systems for centuries and are increasingly being investigated for their potential to modulate neurotransmitter systems and mitigate the underlying pathophysiological mechanisms of these disorders (Kenda et al., 2024). Nodakenin is a well-known bioactive furanocoumarin glycoside compound, which is extensively found in the Angelica gigas, Angelica biserrata, Rhodiola sachalinensis, and Peucedanum decursivum plants. Numerous previous studies already reported the various pharmacological properties of nodakenin, such as hepatoprotective (Lim et al., 2021), anti-obesity (Jin et al., 2021), anti-dermatitis (Park et al., 2014), anti-tumor (Kim, 2023), nephroprotective (Liao et al., 2021), and anti-osteoporotic (Liu et al., 2023) effects. However, there are not many previous reports to claim the anti-anxiolytic and anti-depressive effects of nodakenin. Therefore, the present research work was dedicated to examining the therapeutic values of nodakenin against anxiolytic and depressive-like conditions in an experimental mice model.
Materials and Methods
Chemicals
Nodakenin, imipramine and other chemicals utilized in this work were purchased commercially in Sigma–Aldrich, USA. The assay kit was procured from Elabscience, USA.
Experimental Mice
In the institutional animal facility, the 7–8 weeks-old male Swiss mice weighing around 25–30 g were obtained and maintained under controlled laboratory settings at a temperature of 23°C–25°C and 12-h light/dark series. They were given full access to standard rodent food (protein: 14 g; fat: 4 g; fiber: 5 g; and carbohydrate: 42 g), pellets (Bio-Serv, USA) and drinking water. The protocols for animal studies were approved, and experiments were performed in strict adherence to the guidelines established by the institutional animal ethics committee (no: 2024-15/023). The experimental mice were acclimated for 7 days in the laboratory prior to the initiation of experiments.
Treatment Groups
The acclimatized mice were distributed into four experimental groups (n = 6). Group I comprised mice designated as the control group. The mice from groups II and III were administered 25 and 50 mg/kg of nodakenin via oral route. Group IV mice were treated with 20 mg/kg of imipramine, a standard anti-depressive drug (Figure 1).
Timeline of Analysis of Anxiolytic and Anti-depressant-like Activities of Nodakenin.
Forced Swimming Test
The procedure was performed on the experimental mice following the protocol established previously (Campus et al., 2015). The device consists of a translucent glass cylinder filled with water. Before 24 h to the test, each mouse was individually confined in the cylinder for approximately 15 min. Initially, mice positioned in the cylinder exhibit heightened activity, swimming energetically in circles, striving to ascend the walls, or diving downward. After 2 min, performance starts to diminish, primarily associated with immobility or prolonged floating duration. Prior to their return to the home confines, mice were extracted from the cylinder and permitted to desiccate in a heated enclosure. After 24 h, they are reintroduced into a cylinder, and the duration of immobility is assessed during the 5-min trial. An animal is considered stationary when it remains inactively buoyant in the water, with its snout positioned above the surface and its body slightly bent yet erect.
Tail Suspension Test
It is extensively utilized to evaluate the efficacy of anti-depressive drug candidates. This assay was conducted as per the earlier literature (Steru et al., 1985). The experimental mice were separately affixed with adhesive tape 60 cm above the ground for 6 min, positioned roughly 1 cm from the tip of the tail. The immobility period was recorded when the mouse remained inactively suspended and exhibited total stillness.
Open Field Test
The assay was conducted in accordance with the previous method (Covington et al., 2009). The experimental mice were separately positioned in an apparatus (100 × 100 × 40 cm) with 25 equal places (5 × 5 cm) for 5 min under subdued lighting situations. The quantities of square crossing and raising exhibited by each animal during the testing time were recorded. Prior to each test, the device was sanitized properly.
Elevated Plus-maze Test
This behavioral investigation was conducted to assess the efficacy of nodakenin in alleviating anxiety. This test was predicated on the instinctual response of rodents to evade perilous environments (open arm) and to favor secure settings (closed arm) (Walf et al., 2007). The “+” shaped device consisted of four arms (110 × 10 cm), two of which were open and two closed, positioned 50 cm above the floor. The experimental mice were situated at the maze’s center, oriented towards the open arms. Parameters, including the durations of open and closed arms and the frequency of open-arm entries, were recorded. Elevated entrances into open arms and prolonged duration spent inside were interpreted as anxiety-free conditions in mice.
Light–Dark Box Test
The light and dark exploration assay was carried out to identify the anxiolytic effects of drug samples on the experimental animals (Haefely et al., 1990). The light–dark model apparatus comprised a wooden box positioned above. There are two distinct sections: one little section lined with black plywood on its top. A well-lit chamber, white painted and illuminated by a white light source, was positioned above the open box. The two connected areas were linked by a small open doorway at the center of the divider on the floor level; following 60 min of oral treatments (nodakenin and/or imipramine), the mouse was separately positioned in the center of the lightbox and monitored for 5 min.
Analysis of Monoamine Oxidase-A (MAO-A) Activity
At the end of the behavioral tests, the mice were fasted overnight and sacrificed. Subsequently, the entire brain tissue was promptly extracted and homogenized with iced saline to assess the MAO-A activity. The measurement of MAO-A activity in mouse brains was conducted using the commercial test kit (#E-BC-K008-M). The assay was conducted with three replicates in accordance with the manufacturer’s handbook (Elabscience, USA).
Statistical Analysis
The outcomes are studied with GraphPad software (version 9.5.0; GraphPad Software, Inc., USA), and the results were given as a Mean ± SD of three tests. The data are examined with a one-way analysis of variance (ANOVA) and Duncan’s multiple range test (DMRT) to assess the significance. A significance level of p < .005 is employed to ascertain statistical significance.
Results
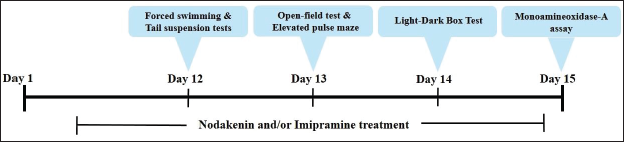
Effect of Nodakenin on the Immobility and Swimming Time of the Experimental Mice
The duration of immobility and swimming activity of the experimental mice was studied using the forced swim test (Figure 2). The present results indicated a significant (p < .005) decrease in the immobility period and an elevation in the swimming duration of the mice with nodakenin (25 and 50 mg/kg) treatment, which is in contrast to the control mice. Furthermore, these findings were also supported by the results of standard drug imipramine-treated mice, which also reduced the immobility period and elevated the swimming period of the experimental mice.
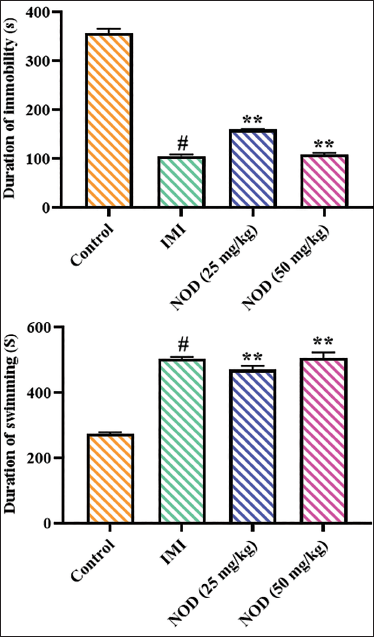
Effect of Nodakenin on Tail-immersion Test in the Experimental Mice
The anti-depressive effects of nodakenin on the experimental mice were tested using a forced-immersion test, with the results illustrated in Figure 3. The immobility duration of the experimental mice was significantly (p < .005) reduced in response to the 25 and 50 mg/kg of nodakenin treatment in comparison to the control group. Interestingly, the standard drug imipramine treatment also notably reduced the immobility period of the experimental mice, which further supports the anti-depressive effects of nodakenin.
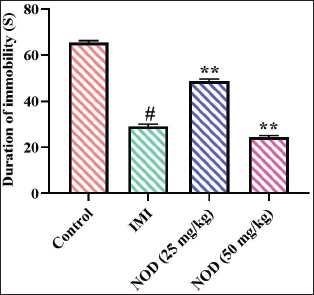
Effect of Nodakenin on Exploratory Behavior of the Experimental Mice
The result of nodakenin treatment on the exploratory behavior of the experimental mice was assessed using an open-field test, and the findings are depicted in Figure 4. The slight elevation in the number of crossings and the significant (p < .005) increase in the crossing numbers in the center area was observed in the mice with 25 and 50 mg/kg of nodakenin treatment, which is in comparison to the control. Similar results were also observed in the mice with imipramine treatment, which supports the activity of nodakenin in increasing the exploratory behaviors of the experimental mice.
Effect of Nodakenin on the Elevated Plus Maze Assay in the Experimental Mice
The anxiolytic properties of the nodakenin in the experimental mice were assessed using elevated plus maze assay, and outcomes are presented in Figure 5. A significant (p < .005) elevation in time spent in the open arm, number of head dips, and a subsequent diminution in the time spent in the closed arm were found in the nodakenin (25 and 50 mg/kg)-treated experimental mice related to the control group. Notably, the standard drug imipramine treatment also produced similar results in the experimental mice, which further supported the activity of nodakenin treatment.
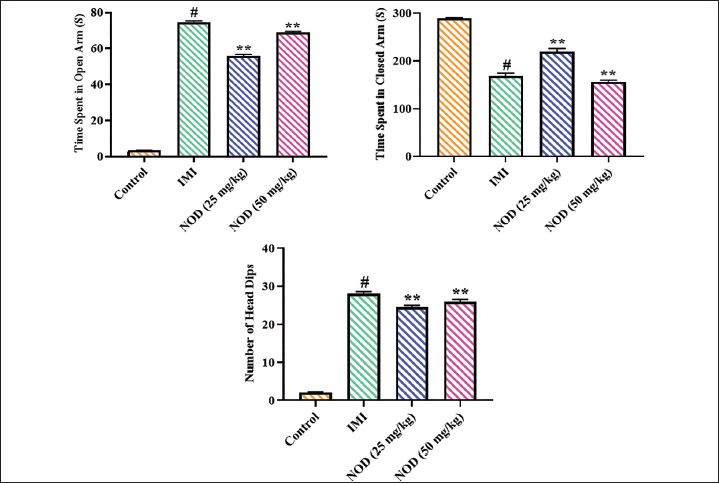
Effect of Nodakenin on the Light–Dark Box Test in Experimental Mice
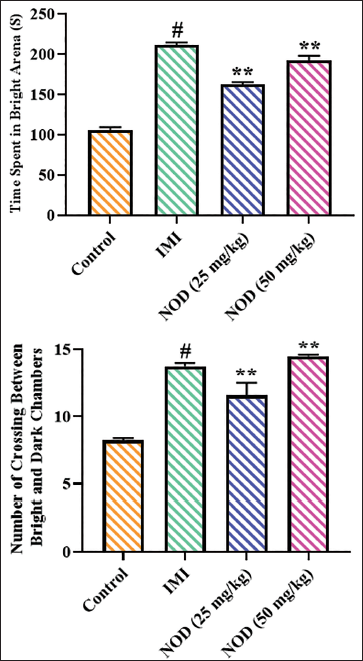
Figure 6 illustrates the findings of the light–dark box assay, which is conducted on the experimental mice to assess the anxiolytic properties of the nodakenin. The present results indicated that mice with 25 and 50 mg/kg of nodakenin treatment demonstrated significant (p < .005) elevation in the time spent in bright areas and total crossing numbers between bright and dark chambers, which is in contrast to the control mice. Similarly, the standard drug imipramine treatment also demonstrated similar results, which supports the anxiolytic properties of nodakenin.
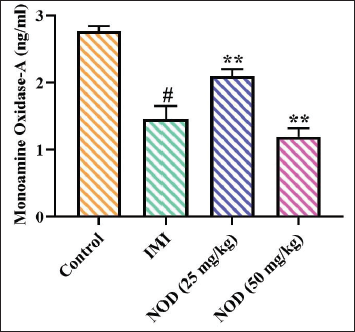
Effect of Nodakenin on the MAO-A Activity Brain Tissues of Experimental Mice
The MAO-A activity in the brains of both control and treatment mice was analyzed using assay kits, with the results displayed in Figure 7. A significant (p < .005) reduction in the MAO-A enzyme activity was observed in the brain tissues of the mice with 25 and 50 mg/kg of nodakenin treatment. Similarly, the standard drug imipramine treatment also reduced the MAO-A concentration in the brain tissues of the mice.
Discussion
Depression and anxiety are among the most common mental health conditions worldwide, impacting millions and imposing a considerable burden on individuals and healthcare systems. It is estimated that approximately 300 million individuals worldwide experience serious depressive illnesses, representing 4.4% of the global population. The same portion of individuals are likewise impacted by anxiety disorders, frequently accompanied by concurrent depression. These mental health conditions are ranked by the WHO as the single largest cause of global disability (for depression) and the sixth largest contributor (for anxiety) (GBD 2019 Mental Disorders Collaborators, 2022). The etiology of anxiety and depressive conditions is multifactorial, encompassing a complex interaction of genetic, environmental, and psychological elements. Genetic predisposition, traumatic experiences, chronic stress, and specific medical problems have been recognized as potential factors in the onset of various disorders (Tian et al., 2022). The influence of anxiety and depression on individual health and well-being is substantial. These disorders can lead to physical changes in the body, such as disruptions in sleep patterns, appetite, and energy levels, which in turn can exacerbate the psychological symptoms. Moreover, individuals with anxiety and depression are at a higher risk of developing other chronic health problems, like cardiovascular disease, diabetes and chronic pain, further compounding the burden on their overall health. Addressing this public health challenge requires a multifaceted approach that encompasses early detection, access to quality mental health services, and the integration of mental health into primary care and community-based settings (Hopwood, 2023).
The forced swimming test has emerged as a widely employed paradigm for assessing the anxiolytic and anti-depressant efficacy of sample drug agents in rodent models. This test capitalizes on the observation that when rodents are placed in an inescapable water-filled cylinder, they eventually exhibit a characteristic immobile posture, interpreted as a state of behavioral depression. The forced swimming study is a dependable and sensitive tool that can detect the effects of several anti-depressant and anxiolytic drugs, which makes it a useful tool for the preclinical evaluation of new therapeutic compounds (Yankelevitch-Yahav et al., 2015). The forced swimming test has been instrumental in elucidating the underlying mechanisms of anxiety and depression (Becker et al., 2023). This test has been instrumental in advancing our understanding of the underlying mechanisms of depression and anxiety. It continues to be a valuable model for the preclinical screening of new therapeutic compounds (Nadeau et al., 2022). In this work, the findings of the forced swimming test have demonstrated the diminished immobility period and an elevation in the swimming time of the nodakenin-treated mice, which is also supported by the results of standard drug imipramine-treated mice. These findings proved that nodakenin treatment had mitigated the depression and anxiety states in the experimental mice.
The tail suspension assay is a broadly utilized behavioral paradigm for assessing the anti-depressant and anxiolytic-like activities of pharmacological compounds in experimental mouse models. This test exploits the natural tendency of mice to become immobile when suspended by the tail, a behavior that is sensitive to the administration of anti-depressant drugs. Behavioral studies on anxiety and depression in animal models have contributed to the elucidation of anti-depressant targets and the identification of dysregulated systems in anxious or depressed states (Cryan et al., 2005). The tail suspension assay, in particular, provides a useful tool for assessing novel compounds for their potential anti-depressant and anxiolytic-like properties in mice. This test has several advantages over others utilized to examine anxiety and depression-like behaviors in mice. It is a relatively simple and inexpensive procedure that can be performed with minimal training and equipment, making it a practical choice for screening large numbers of compounds (Bourin et al., 2005). The test encompasses suspending a mouse by its tail and measuring the time it spends immobile, which is considered a measure of behavioral despair. Decreased immobility time is taken as an anti-depressant-like condition, while increased immobility is associated with a depressive-like phenotype (Andreasen et al., 2009). In this study, the anti-depressive properties of nodakenin were assessed on the experimental mice using the forced-immersion test. The results demonstrated the reduced immobility period of the nodakenin-treated mice, which highlights the anti-depressive effects of the nodakenin.
The open-field test is a widely utilized paradigm in preclinical research to investigate anxiety-like behaviors and general locomotor activity in rodents. This test provides valuable insights into the effects of various pharmacological compounds, including those with potential anxiolytic (anxiety-reducing) and anti-depressant properties (Mehrhoff et al., 2023). The open field test is a simple yet effective tool for evaluating the emotional and behavioral reactions of mice to a novel environment. The test measures parameters such as time spent in the central versus peripheral zones, the number of entries into the center and the overall distance traveled, which collectively reflect the subject’s level of anxiety and exploratory drive (Zhou et al., 2022). The open-field test is specifically beneficial for screening and evaluating the efficacy of potential anxiolytic and anti-depressant drug candidates. Compounds with anxiolytic effects have been shown to increase time spent in the central area and reduce thigmotaxis (the tendency to stay close to the walls), indicating reduced anxiety-like behavior. Conversely, anxiogenic agents have been found to decrease the time spent in the central zone and increase peripheral exploration, reflecting an increase in anxiety-like behavior. This test also provides valuable information about the effects of these compounds on general locomotor activity, which is an important factor to consider when interpreting the anxiolytic or anti-depressant-like effects (Snyder et al., 2021). In this work, the influence of nodakenin on the exploratory behavior of the experimental mice was assessed using an open-field test. The present results showed a slight elevation in the total number of crossings and a remarkable increase in the crossing numbers in the center area of the mice with nodakenin treatment, which proves that nodakenin increases the exploratory behaviors of the mice.
The elevated plus maze test has developed as a crucial model in the study of anxiolytic and anti-depressant properties of various pharmacological compounds using experimental mouse models. This test leverages rodents’ inherent aversion to open areas, a recognized predictor of anxiety-like behavior (Figueiredo Cerqueira et al., 2023). The behavioral measures recorded in this test include time spent in open arms, number of entries into the open arms, and other ethological parameters such as rearing, head dips, and defecation, all of which are indicative of the animal’s emotional state. The face validity of this test is well-established, as the avoidance of open arms reflects the rodent’s fear of exposed and elevated spaces, which is analogous to the human experience of anxiety (Gaspar et al., 2023). The elevated plus maze test has been extensively utilized to evaluate the anti-depressant and anxiolytic properties of a wide range of drugs in experimental mice models. This approach allows researchers to gain valuable insights into the potential therapeutic properties of novel pharmacological compounds, which can then be further explored in more complex animal models and ultimately translated into clinical trials. By providing a standardized and reliable platform for the assessment of emotional behaviors in rodents, the elevated plus maze test has become an indispensable tool in the field of psychopharmacology and neuroscience research (Bruijnzeel et al., 2022). In this study, the anxiolytic activity of the nodakenin in the experimental mice was assessed using an elevated plus maze test. A considerable elevation in time spent in the open arm, the number of head dips, and a subsequent diminution in the time spent in the closed arm were found in the nodakenin-treated experimental mice related to the control. These outcomes support the anxiolytic properties of the nodakenin.
The light–dark box test is a widely employed experimental paradigm for investigating the anxiolytic and anti-depressant properties of various pharmaceutical compounds using rodent models. This assay exploits the natural propensity of mice and rats to avoid brightly lit open spaces, which are perceived as aversive, and instead seek refuge in darker, enclosed compartments. The light–dark box test offers a simple and efficient means of evaluating the emotional and behavioral responses of experimental subjects to anxiogenic or anxiolytic stimuli, offering useful insights into the therapeutic applications of candidate drugs (Miller et al., 2011). By integrating the data obtained from behavioral tests, researchers can gain a deeper understanding of the interplay between anxiety, depression and other emotional and behavioral domains (Campos-Cardoso et al., 2023). The open field test measures locomotor activity and exploratory behavior, while the elevated plus maze assesses anxiety-like behaviors in a different environmental context. Combining the light–dark box test with these other paradigms allows for a more nuanced evaluation of the diverse emotional and behavioral responses elicited by pharmacological interventions (Ramos et al., 2008). The utility of the light–dark box assay in identifying potential anxiolytic and anti-depressant agents has been highlighted. The light–dark box test offers several advantages over other behavioral paradigms, including its simplicity, low cost and ease of implementation. Additionally, the test is less stressful for the animals compared to some other anxiety-related assays, as it capitalizes on their natural tendency to seek out darker, more enclosed spaces (Shemesh & Chen, 2023). The present study utilizes a light–dark box assay to assess the anxiolytic properties of the nodakenin. The present results indicated that the nodakenin-treated mice demonstrated substantial elevation in the time spent in bright areas and a total number of crossings between bright and dark chambers, which is in contrast to the control mice. These findings are supported by the standard drug imipramine treatment, which further suggests the anxiolytic properties of the nodakenin.
Monoaminergic neurotransmitters are essential for the regulation of cognitive, emotional, neuroendocrine and motor activities in the brain. Neurotransmitter systems have been thoroughly examined concerning psychiatric diseases, as successful psychotropic medications predominantly focus on these systems. Recent data indicates that disturbances in these regulatory systems may contribute to the pathogenesis of several mental disorders, such as anxiety and depression (Shao & Zhu, 2020). One key player in the regulation of monoamine neurotransmitters is the enzyme MAO-A, which is responsible for the degradation of the neurotransmitters. Impaired monoaminergic function, particularly related to serotonin and noradrenaline, has been consistently found in depression conditions (Jiang et al., 2022). MAO-A is an enzyme that plays a crucial role in the degradation of important neurotransmitters, like serotonin, norepinephrine, and dopamine, which are implicated in the regulation of mood, anxiety, and depression. By studying the effects of test compounds on MAO-A activity, researchers can gain insights into their mode of action and their therapeutic applications to treat anxiety and depression (Fiedorowicz & Swartz, 2004). The altered monoaminergic function has also been linked to the pathophysiology of anxiety disorders. Disruptions in the delicate balance of these neurotransmitter systems can lead to the emergence of anxious behaviors and the onset of anxiety-related disorders. The analysis of MAO-A activity has various applications in the field of neuropsychopharmacology. It can be used to screen for new drug candidates with anxiolytic and anti-depressant properties, to comprehend the underlying mode of actions of existing anti-depressant and anxiolytic drugs, and to investigate the role of MAO-A in the onset of anxiety and depressive conditions. The analysis of MAO-A activity has become increasingly important in studying the anxiolytic and anti-depressant properties of various drug candidates in experimental mouse models (Hamon & Blier, 2013). The current findings revealed a significant reduction in the MAO-A enzyme activity in the brain tissues of the mice with nodakenin treatment. Hence, it was suggested that nodakenin can mitigate the degradation of neurotransmitters by inhibiting the MAO-A enzyme activity.
Conclusion
The current research demonstrates that the nodakenin treatment exhibits anxiolytic and anti-depressant-like properties in murine models. The present findings indicate the therapeutic potential of nodakenin as an alternate treatment for comorbid anxiety and depression conditions. The present study has some limitations, including the use of only murine models and a limited range of behavioral tests. Further in-depth molecular studies are needed to fully elucidate the mechanisms underlying nodakenin’s anxiolytic and anti-depressant-like effects. The current research does not provide a comprehensive understanding of the neurochemical changes induced by nodakenin, which is essential for its potential therapeutic application. Future studies should prioritize investigating the molecular mechanisms of nodakenin to unlock its full therapeutic potential for treating comorbid anxiety and depression.
Footnotes
Abbreviations
DMRT: Duncan’s multiple range test; MAO-A: Monoamine oxidase-A; WHO: World Health Organization.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This work was approved by the Institutional Ethical Committee at Gansu Provincial People’s Hospital, Lanzhou 730000, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
