Abstract
Background
Diabetes mellitus, a persistent metabolic disorder defined by elevated blood glucose, poses a global public health challenge. One of the most debilitating complications of this disease is the progression of chronic, non-healing wounds, particularly in the lower extremities.
Objectives
The present work was dedicated to studying the beneficial effects of albiflorin to enhance wound healing in streptozotocin (STZ)-induced diabetic rats.
Materials and Methods
In the current investigation, rats received an injection of STZ and were subjected to excision wound development. The rats were treated with albiflorin for 14 days. After the treatments were completed, the glucose and insulin levels, wound diameter, wound closure rates, and wound healing biomarker levels were estimated in the experimental rats. The concentrations of inflammation and oxidative stress biomarkers were evaluated using commercial kits. The wound tissues were subjected to a histopathological study to investigate histological alterations. The anti-bacterial effects of the albiflorin were assessed against Staphylococcus aureus and Pseudomonas aeruginosa.
Results
The albiflorin treatment significantly reduced glucose, elevated insulin levels, and increased the wound closure rate in diabetic rats. Furthermore, the albiflorin treatment diminished the inflammatory marker levels and reduced oxidative stress via upregulating the antioxidant mechanisms. The albiflorin treatment also efficiently regulated the wound healing biomarker levels in diabetic rats and showed better anti-bacterial activity. Moreover, histological analyses have demonstrated that albiflorin treatment can improve the wound healing process in diabetic rats.
Conclusion
The findings indicate that albiflorin may significantly increase wound healing in diabetic rats. Consequently, the current data suggest that albiflorin may offer a useful therapeutic alternative for diabetic wound management.
Introduction
Diabetes mellitus is a chronic metabolic disorder defined by elevated blood glucose due to the body’s incapacity to make or efficiently utilize the insulin hormone. This condition can result in various debilitating problems, including diabetic foot ulcers, which are a pivotal consequence of the disease (Gong et al., 2023). The worldwide incidence of diabetes is increasing, with around 463 million individuals affected in 2019, a number predicted to escalate to 700.2 million by 2045. Diabetic foot ulcers, a prevalent consequence of diabetes, impact around 26.1 million individuals annually, with lifetime cases of foot ulcers among diabetics predicted to range from 19% to 34% (McDermott et al., 2023). Diabetic foot ulcers have a multifactorial origin, resulting from a mix of variables including peripheral neuropathy, which causes loss of sensation in the feet, and peripheral artery disease, which hinders blood flow and healing. Additionally, chronic inflammation associated with diabetes can disrupt the normal tissue healing process, further exacerbating the advancement of these ulcers (Hicks et al., 2020). The burden of diabetic foot ulcers extends beyond the individual patient, as these complications can lead to increased morbidity, mortality, and significant healthcare expenditures. Diabetic foot ulcers account for over 90% of lower-limb amputations in adults with diabetes, with a global economic burden estimated at billions of dollars each year (Maldonado-Valer et al., 2023).
The pathophysiology of diabetic wounds is a complex process influenced by various intrinsic and extrinsic causes that hinder wound healing (Raja et al., 2023). Hyperglycemia, a defining characteristic of diabetes, has been known as a primary cause of the onset of diabetic wounds. Chronic hyperglycemia creates advanced glycation end-products, which can stimulate the production of inflammatory mediators and disrupt collagen synthesis, essential elements of the wound healing process. Additionally, hyperglycemia-induced oxidative stress can negatively impact cellular functions, hindering the body’s ability to heal wounds effectively (Ran et al., 2024). An earlier study aimed to explore predictive risk factors for diabetic foot ulcers in China. A total of 968 patients with diabetes were selected as research participants. After a 5-year follow-up, the incidence of diabetic foot was 25.83%. Independent predictors of diabetic foot included body mass index, abnormal pinprick sensation, history of fungal foot infection, abnormal 128-Hz tuning fork test, and HbA1c ≥ 8%. The study highlights clinically relevant indicators that may help prevent diabetic foot ulcers and guide timely interventions. The findings can help clinicians identify high-risk diabetic feet and guide timely interventions (Su et al., 2024).
Vascular complications, both macro and microvascular, are also significant contributors to the reduced wound healing observed in individuals with diabetes. Thickening of the basement membrane of capillaries and arterioles, a common feature in diabetes, can result in impaired tissue perfusion and diminished oxygen and nutrient delivery to the wound site, ultimately slowing the healing mechanism (Bonnet & Sultan, 2022). Furthermore, diabetic neuropathy can lead to decreased sensation in the affected limbs, making the individual more susceptible to repeated trauma or mechanical stress. This, in turn, can perpetuate the cycle of wound formation and delayed healing. Chronic inflammation, a hallmark of the diabetic wound, is another key factor in impaired healing. Sustained release of inflammatory cytokines and proteases and a suboptimal immune response can further exacerbate the wound (Akkus & Sert, 2022).
A previous study on diabetic rats found that alpha-linolenic acid (AL) improved cognitive function, reduced Aβ plaque density, and improved oral glucose tolerance, suggesting AL may alleviate STZ-induced cognitive impairment by regulating oxidative stress and inflammation (Ma et al., 2021). Albiflorin, a small molecule from Paeoniae radix, has been found to alleviate obesity, improve insulin-stimulated glucose disposal, reverse liver triglyceride and cholesterol levels, and increase energy expenditure and physical activity (Zhou et al., 2019). The earlier study investigates the therapeutic effects of albiflorin on spinal cord injury-induced rats, finding that it can alleviate motor dysfunction and neuron cell loss by attenuating inflammatory responses (Fang et al., 2023). Fracture healing is linked to osteogenic differentiation and mineralization, with natural product-based chemical studies revealing albiflorin’s role in promoting osteoblastogenesis, RUNX2 expression, osteogenic gene upregulation, and bone healing, thereby enhancing fracture healing (Kim et al., 2021).
Current treatment options for diabetic wounds include a range of approaches, such as wound debridement, offloading, moisture-retentive dressings, and advanced therapies like biological skin substitutes and growth factor-based treatments. However, these interventions often have limitations, such as the need for specialized care, high costs, and variable efficacy (Ramirez-Acuña et al., 2019). In this context, the potential of plant bioactive compounds has garnered increasing attention as a complementary or alternative approach to managing diabetic wounds. Plant-derived compounds have demonstrated several therapeutic values, which can be beneficial in managing diabetic wounds (Accipe et al., 2023). Several studies have reported the efficacy of plant-based compounds in enhancing wound healing and diminishing the problems in diabetic animal models and clinical settings. These compounds have been shown to modulate key pathways that participate in wound healing, like angiogenesis, collagen deposition, and regulating inflammatory mediators (Herman & Herman, 2023). Albiflorin is a well-known and major bioactive monoterpene glycoside compound, which is found in the Radix paeoniae and Paeonia lactiflora plants. Numerous previous studies have already highlighted the various pharmacological properties of the albiflorin, including anti-depressant (Wang et al., 2016), anti-asthma (Cai et al., 2019), anti-osteolytic (Wang et al., 2024), anti-obesity (Jeong et al., 2017), antioxidant and anti-inflammatory (Fang et al., 2023), anti-enteritis (Zhang et al., 2022), and anti-neuropathy (Zhou et al., 2016) properties. Apart from these activities, there is no evidence of the beneficial effects of albiflorin on diabetic complications. Therefore, this work was dedicated to studying the beneficial values of albiflorin to promote wound healing in streptozotocin (STZ)-induced diabetic rats.
Materials and Methods
Animals
The Sprague Dawley rats, aged 8–10 weeks and weighing 200–250 g, were employed in this work. They were sustained in sterile enclosures under laboratory settings, with a 21°C ± 1°C temperature, 60%–70% humidity, and a 12-h light and dark series. Animals were granted free access to diet and water throughout the study. The standard diet used in this investigation was a commercially available rodent chow obtained from Trophic Animal Feed High-tech Co., China. The diet composition included 20% protein, 5% fat, 55% carbohydrate, 5% fiber, vitamins, and minerals, ensuring adequate nutrition for the experimental animals. All the protocols for animal experimentations were followed by the specifications and regulations given by the institutional ethical committee (SYXK[冀]2023-009).
Diabetes Induction and Wound Model
All the rats underwent overnight fasting before the STZ treatment. STZ was delivered at a dosage of 50 mg/kg intraperitoneally. Following 2 days of STZ treatment, glucose levels were assessed using a glucometer. Animals exhibiting glucose concentrations over 25 mmol/L were classified as diabetic and utilized for subsequent research. The variations in insulin levels were assessed after the study using kits. The rear fur of normal and diabetic rats was shaved, and the rodents were anesthetized using isoflurane. A full-thickness circular wound with a 2.5 cm diameter and depth of 0.2 cm was developed on the dorsal skin of the rat using a round hole punch and surgical scissors.
Experimental Groups
Rats were categorized into the following four groups (n = 6). Group I had non-diabetic rats, while Groups II–IV comprised diabetic animals. Group I: rats received distilled water and functioned as normal wound control; Group II: rats were treated with distilled water and acted as diabetic wound control; Group III: rats treated orally with albiflorin (10 mg/kg); and Group IV: rats treated orally with albiflorin (20 mg/kg). The wound closure rate was monitored on the 1st, 7th, and 14th days following the injury. Day 0 was designated as the day of wound formation.
Wound Contraction Rate (WCR) Analysis
The WCR was determined using the following equation, which considers the starting wound size of 2.5 cm as 100%. The duration of epithelialization was determined by monitoring the moment when the necrotic tissue entirely obscured the wound, devoid of any indications of an unhealed surface. WCR (%) = wound area (day 0) – wound area (observation day)/wound area (day 0) × 100.
Analysis of Collagen and Total Protein Levels
The wound tissues were removed and studied for collagen and protein contents. The concentrations of collagen and total protein were quantified using an assay kit per the manufacturer’s guidelines (Abcam, USA).
Analysis of Inflammatory and Oxidative Stress Marker Levels
The harvested wound skin tissues were homogenized in ice-cold saline and centrifuged for 10 min. Using commercial test kits, the resulting supernatant was employed to assess oxidative stress markers and antioxidant levels. The antioxidants superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), glutathione-S-transferase (GST) along with malondialdehyde (MDA), were assessed using the kits. The tests were conducted in triplicate using the manufacturer’s specified methods (MyBioSource, USA). The tumor necrosis factor-alpha (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6) concentrations in the serum were assessed using kits. The tests were conducted with three replicates following the assay kit manufacturer’s specifications (Abcam, USA).
Histopathological Analysis
The wound skin tissue was removed for histological studies on the concluding day of the experiment. Skin tissues were subsequently cut into 7 µm diameter and stained with eosin and hematoxylin following fixation in formalin (10%) and embedding in paraffin. Microscopic slides were qualitatively assessed for histopathological alterations using the microscope.
As previously outlined, the histological assessment of skin excision wounds was conducted in a blinded mode (Uckun et al., 2022).
Anti-bacterial Activity
The well diffusion technique was utilized to study the anti-bacterial characteristics of the albiflorin. The diverse pathogens, including Staphylococcus aureus and Pseudomonas aeruginosa, were cultured on agar plates. Subsequently, 6 mm wells were created on the agar plate surface. The wells were filled with 10, 20, and 30 µM/mL of albiflorin, respectively, and incubated for 24 h. The standard anti-biotic amoxicillin, at a dosage of 30 µg/well, was utilized as a standard control. The inhibitory zones were evaluated, and the results were documented.
Statistical Analysis
The findings were portrayed as a mean ± standard deviation (SD) from three replicates. A one-way analysis of variance (ANOVA) and Tukey’s post hoc test were employed to assess the significance, with p < .05 denoting statistical significance. Statistical analysis was performed using GraphPad Prism V 20 software.
Results
Effect of Albiflorin on Glucose and Insulin Levels in Experimental Rats
The impact of albiflorin on blood glucose and insulin concentrations was assessed in rats, with results presented in Figure 1. The diabetic rats exhibited a drastic elevation in glucose, accompanied by a reduction in insulin concentration compared to the control. However, the albiflorin at a dosage of 50 and 100 mg/kg concentration remarkably diminished the glucose and increased insulin concentrations in the diabetic rats.
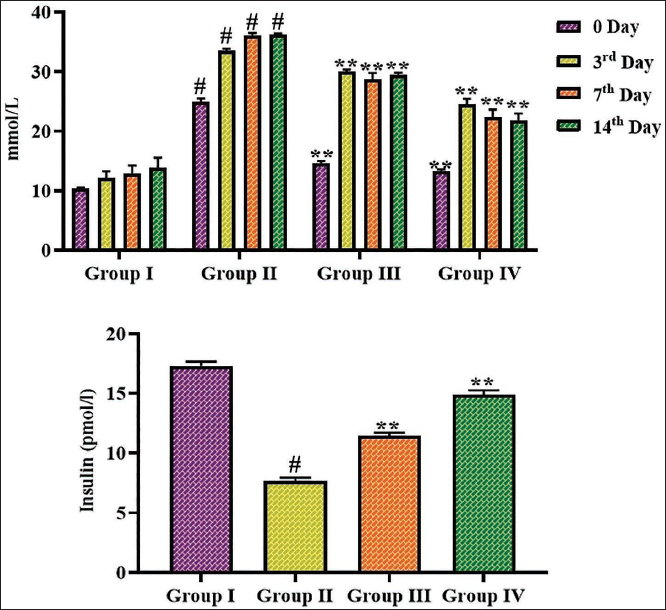
Effect of Albiflorin on the Wound Diameter and WCR in Experimental Rats
Figure 2 illustrates the results of albiflorin on wound diameter and WCR in the rats. The diabetic rats illustrated a significantly increased wound diameter accompanied by reduced WCR relative to the control group. The albiflorin treatment at 50 and 100 mg/kg concentrations demonstrated a significant reduction in wound diameter and subsequent increase in WCR in the diabetic rats. These findings evidenced that albiflorin effectively increased wound healing in diabetic rats.
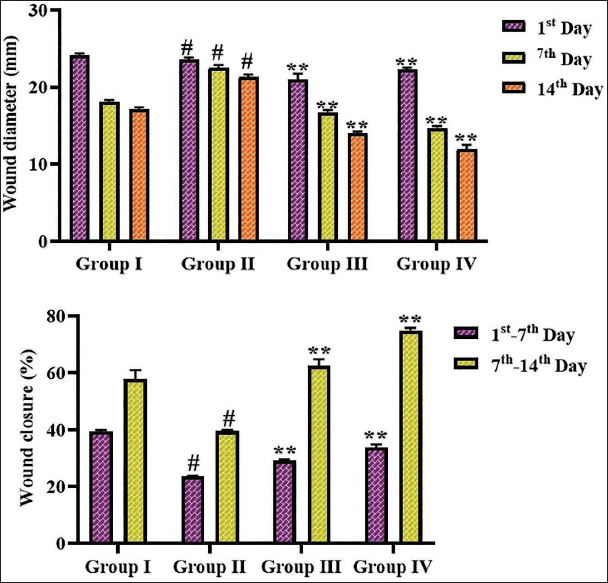
Effect of Albiflorin on Wound Healing Markers Level in the Experimental Rats
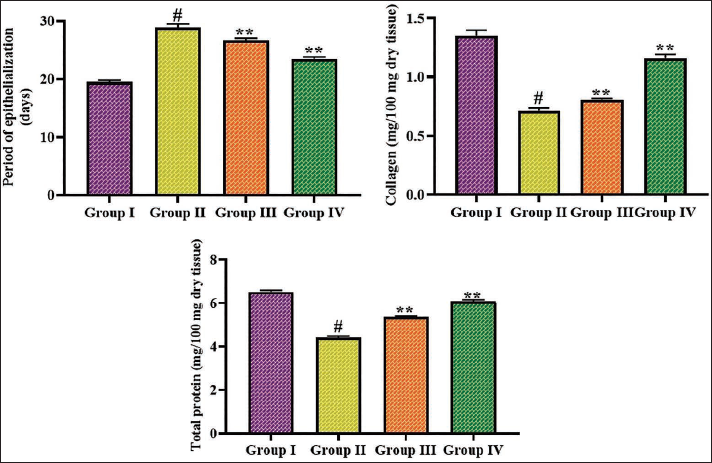
The wound-healing effect of albiflorin in diabetic rats was studied by evaluating the concentrations of wound healing biomarkers, including duration of epithelialization, total protein, and collagen contents in the experimental rats, as shown in Figure 3. The rats with diabetes demonstrated significant elevation in epithelialization and reduction in total protein and collagen contents compared to the control. Remarkably, the 50 and 100 mg/kg of albiflorin noticeably diminished the epithelialization and elevated the total protein and collagen contents in the diabetic rats, evidencing enhanced wound healing in diabetic rats in response to albiflorin treatment.
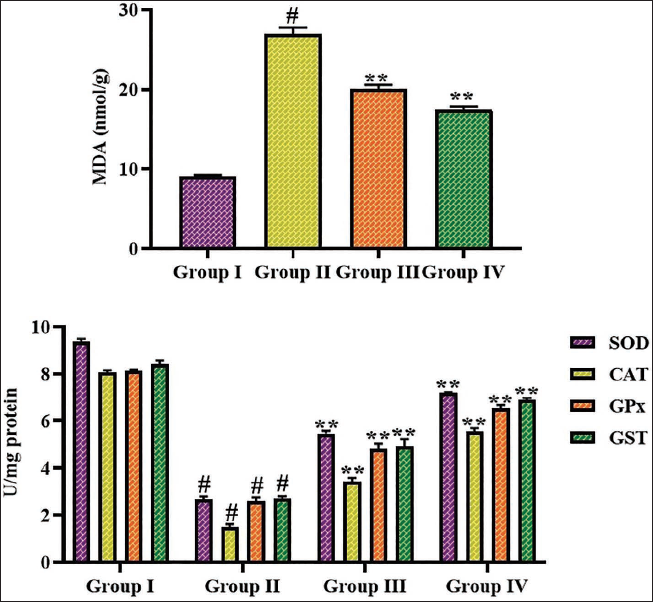
Effect of Albiflorin on Oxidative Stress in Experimental Rats
The oxidative stress marker and antioxidants were quantified in the wound tissues of control and treated rats. The concentrations of the antioxidants, including SOD, CAT, GPx, and GST were reduced drastically and MDA content was elevated in the wound tissues of the diabetic rats. Nonetheless, treating the albiflorin at 50 and 100 mg/kg concentrations successfully elevated the antioxidants and diminished the MDA concentration in the wound tissues of the diabetic rats (Figure 4). These outcomes demonstrated the antioxidant properties of the albiflorin.
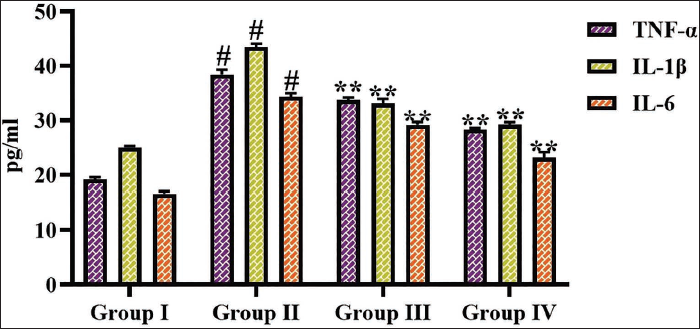
Effect of Albiflorin on Inflammatory Cytokines in Experimental Rats
The rats with diabetes displayed a drastic increase in the TNF-α, IL-1β, and IL-6 concentrations in their serum compared with the control. Interestingly, the diabetic rats administered with 50 and 100 mg/kg of albiflorin demonstrated a notable diminution in these inflammatory cytokines in their serum. The current results indicated the anti-inflammatory activity of the albiflorin (Figure 5).
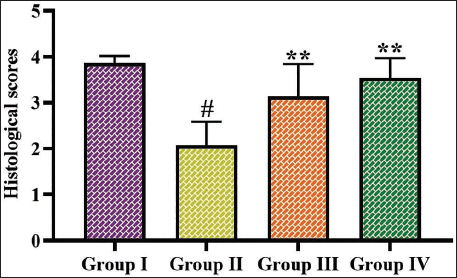
Effect of Albiflorin on Wound Tissue Histopathology
Figure 6 demonstrates the findings of the histopathological analysis conducted on the wound tissues. The diabetic rats exhibited significant alterations, including fibrosis, inflammatory cell infiltration, and epithelial deterioration. The wound tissues of the diabetic rats treated with albiflorin exhibited organized collagen fiber formation, a thickened epidermal layer, and fewer inflammatory signs. The findings of the histological analysis evidenced increased wound healing in diabetic rats due to the treatment with albiflorin.
Anti-bacterial Activity of the Albiflorin
The anti-bacterial efficacy of albiflorin against P. aeruginosa and S. aureus was studied utilizing the well diffusion technique (Table 1). The administration of varying doses of albiflorin has shown a substantial impact in suppressing the growth of all examined strains. The treatment with albiflorin successfully suppressed the growth of P. aeruginosa and S. aureus. The measured inhibition zones around the wells treated with albiflorin indicate the suppression of pathogen growth.
Anti-bacterial Activity of the Albiflorin. The Treatment of Albiflorin at Varying Concentrations has Shown a Substantial Reduction in the Growth of Pseudomonas aeruginosa and Staphylococcus aureus in Dose Dependence.
Discussion
Diabetes is a significant concern in the realm of chronic wound management, with diabetic foot ulcers affecting 15% of individuals with diabetes and often leading to amputations. Diabetic wounds pose a significant challenge in healthcare, as the underlying metabolic imbalances and impaired immune function in diabetic individuals often lead to prolonged wound healing and a high risk of complications (Edmonds et al., 2021). The pathogenesis of diabetic wounds is complex, implicating factors like persistent hyperglycemia, disrupted barrier function, infection, oxidative stress, neuropathy, and suboptimal inflammatory response (Lee et al., 2024). Proper glycemic control has been demonstrated to significantly impact wound healing rates in diabetic patients. Diabetes can impair wound healing by forming inflammatory molecules and interfering with collagen synthesis (Lane et al., 2020). Furthermore, hyperglycemia contributes to the deregulation of angiogenesis, chronic inflammation, and persistent bacterial colonization in diabetic wounds. Diabetic wounds often exhibit a prolonged and suboptimal healing process, which can lead to the development of chronic, hard-to-treat ulcers (Sidhu & Harbuzova, 2024). Consequently, analyzing glucose and insulin levels is crucial in understanding and managing the complex pathology of diabetic wounds. In this work, the diabetic rats exhibited a significant elevation in glucose accompanied by a diminution in insulin concentrations. The albiflorin treatment significantly reduced the glucose and insulin concentrations in the diabetic rats. These findings demonstrate that albiflorin can reduce hyperglycemic conditions and aid in wound healing in diabetic conditions.
Diabetic wounds present a considerable issue in healthcare owing to their tendency for protracted healing and heightened risk of difficulties. Examining the dynamics of epithelialization, total collagen, and protein levels in diabetic wounds is essential for understanding the underlying mechanisms and developing targeted therapeutic interventions (Jiang et al., 2024). The role of matrix metalloproteinases is of particular interest in this regard. Excessive collagenolytic effect in diabetic wounds is attributed to diminished tissue inhibitor of metalloproteinase 1. This disproportion between matrix metalloproteinases and their inhibitors can lead to the degradation of extracellular matrices, impeding normal wound healing (Zheng et al., 2023). Furthermore, hyperglycemia-induced oxidative stress has been shown to interfere with collagen synthesis and disrupt the delicate balance of inflammatory markers, implicating the perpetuation of the chronic wound state (Li et al., 2021). Improving our understanding of the temporal and spatial dynamics of epithelialization, total collagen, and protein levels in diabetic wounds can offer useful insights into the underlying pathophysiology. This data can guide the advancement of novel diagnostic tools and targeted therapeutic strategies to overcome the challenges associated with diabetic wound management. By proactively identifying and addressing the imbalances in these key wound healing parameters, clinicians can able to inhibit the advancement of normal wounds into chronic, hard-to-treat conditions, ultimately reducing the significant morbidity associated with this health problem (Yan et al., 2024). In this work, the wound-healing effect of the albiflorin in diabetic rats was assessed by studying the concentrations of wound-healing biomarkers. The rats with diabetes demonstrated significant elevation in epithelialization duration and reduction in total protein and collagen contents in comparison to the control. The treatment of albiflorin significantly reduced the epithelialization period and elevated the total protein and collagen contents in the diabetic rats, which evidences the improved wound healing mechanism in diabetic rats due to the albiflorin treatment.
Oxidative stress, characterized by a disproportion between reactive oxygen species (ROS) production and the body’s antioxidant defenses, has been recognized as a significant component in the pathophysiology of diabetic wound healing. Analyzing oxidative stress markers and antioxidants in individuals with diabetes is crucial for understanding the underlying mechanisms and developing effective treatment strategies (Deng et al., 2021). Diabetic wounds demonstrate a multifaceted pathophysiology resulting from sustained hyperglycemia and related consequences, including barrier disruption, infection, elevated oxidative stress, and inflammation. Oxidative stress has been shown to compromise insulin action and promote the development of diabetic complications, including impaired wound healing (Zhao et al., 2024). Moreover, the thickening of the basement membrane of capillaries and arterioles in diabetic persons can result in compromised wound healing and advanced chronic ulcers. The examination of oxidative stress indicators and antioxidants in diabetic persons can yield significant insights into the pathophysiological mechanisms that contribute to poor wound healing (He et al., 2023). Evaluating the levels of these markers can help identify the extent of oxidative stress, which may participate in the prolonged wound healing noted in diabetes. By understanding the role of oxidative stress and its associated markers in individuals with diabetes, researchers can develop more targeted therapies to improve wound healing outcomes and decrease the burden of this debilitating complication (Jiang et al., 2023). Here, the oxidative stress marker and antioxidant levels were quantified in the wound tissues. The concentrations of the antioxidants were markedly reduced, and MDA content was elevated in the wound tissues of the diabetic rats. Nonetheless, the albiflorin successfully increased the antioxidants and reduced the MDA concentration in wound tissues of diabetic rats, demonstrating the antioxidant properties of the albiflorin.
The mechanism of diabetic wound healing is complicated, involving a combination of neuropathy, vascular dysfunction, and dysregulated immunity. A key aspect of this process is the dysregulation of the inflammatory response, defined by persistent inflammation and the overproduction of inflammatory cytokines (Gao et al., 2024). Hyperglycemia, a hallmark of diabetes, has been shown to induce the chronic inflammatory state observed in diabetic wounds. This sustained inflammation can result in further tissue injury, late wound closure, and an elevated risk of infection. Chronic infection and inflammation are key factors contributing to the chronicity of diabetic wounds. Pathologically extensive inflammation disrupts the normal healing cascade, perpetuating the rogue inflammatory response and preventing resolution (Worsley et al., 2023). Inflammatory cytokines, including IL-1β, IL-6, and TNF-α, play a central role in the progression of diabetic wound conditions. These inflammatory mediators can contribute to the persistence of inflammation, impair angiogenesis, and inhibit the growth of key cell types that participate in wound healing, like fibroblasts and keratinocytes (Deng et al., 2023). The elevated levels of these inflammatory mediators in diabetic wounds contribute to a prolonged and ineffective inflammatory phase, which can inhibit the subsequent stages of wound healing and tissue remodeling (Dasari et al., 2021). Despite recent advances in wound care strategies, including debridement, dressings, and the utilization of anti-biotics, managing chronic diabetic wounds remains a significant challenge. Novel interventions targeting the modulation of inflammation and inflammatory cytokines may hold promise in driving diabetic wounds toward resolution and improved healing outcomes (Kupczyk et al., 2021). In this work, the results proved that the rats with diabetes displayed drastic elevation in the TNF-α, IL-1β, and IL-6 concentrations in their serum when compared with the control. Captivatingly, the treatment of albiflorin in diabetic rats resulted in a notable diminution in these inflammatory cytokines in their serum, highlighting the anti-inflammatory properties of the albiflorin.
Albiflorin has shown promising results in wound healing at low doses (10 mg/kg) and high doses (20 mg/kg). Low doses enhance re-epithelialization and reduce inflammation, accelerating recovery. High doses may enhance antioxidant activity and mitigate oxidative stress, as observed in diabetic wound models. Albiflorin’s therapeutic potential is enhanced by modulating inflammatory responses and oxidative stress (Fang et al., 2023). Recent studies have demonstrated that albiflorin facilitates wound closure, enhances fibroblast proliferation, reduces pro-inflammatory cytokine levels, and modulates vascular endothelial growth factor (VEGF) expression, aiding wound healing (Zulkefli et al, 2023). Combining these findings helps better understand the dose-dependent effects of albiflorin.
Diabetic wounds are a significant healthcare challenge, with high rates of infection and delayed healing. S. aureus and P. aeruginosa are two well-known bacteria implicated in these infections, posing a significant threat to effective wound management (Norton et al., 2024). S. aureus is a pyogenic bacterium that produces the β-lactamase enzyme, which can render many anti-biotics ineffective. This bacterium is a prevalent cause of skin and soft tissue infections, including those connected with diabetic wounds. Notably, the presence of S. aureus can extend the inflammatory condition of wound healing, delaying the overall recovery process (Pajon et al., 2023). Similarly, P. aeruginosa is another opportunistic pathogen that frequently colonizes chronic wounds, including those in diabetic patients. P. aeruginosa is particularly adept at forming biofilms, which can shield the bacteria from the host’s immunity and anti-microbial therapies, further hindering wound healing (Garousi et al., 2023).
Bacterial infections, particularly with S. aureus and P. aeruginosa, are a significant factor that exacerbates delayed wound healing in diabetic patients. These pathogens contribute to chronic wound infections, further impairing the healing process. The anti-bacterial activity of albiflorin was evaluated as an exploratory measure to determine whether its effects on wound healing could be partially attributed to its ability to mitigate bacterial load. While our study primarily focuses on wound healing and oxidative stress parameters, understanding the anti-microbial potential of albiflorin could be crucial in future therapeutic applications for diabetic wound management. Biofilm-forming bacteria have been identified as a significant challenge in chronic wound care. These biofilms can absorb nutrients from the extracellular matrix, providing a stable environment for the bacteria to thrive and propagate. The presence of biofilms is associated with delayed healing and reduced clinical effectiveness of topical treatments (Genito et al., 2023). In this work, the anti-bacterial property of albiflorin was studied against P. aeruginosa and S. aureus.
Limitations of the present findings, such as the study on diabetic rats, may not fully replicate the complexity of diabetic wound healing in humans. Further validation through human clinical trials is needed to confirm the efficacy and safety of albiflorin in diabetic patients. The study may have focused on short-term wound healing effects, and further research is needed to understand the molecular pathways involved fully. The present results proved that the treatment of varying concentrations of albiflorin has shown a substantial impact in suppressing the growth of these pathogens. These findings prove that albiflorin can inhibit infection through its anti-microbial properties and speed up wound healing in diabetic rats.
Conclusion
The present results indicate that albiflorin may significantly increase wound healing in diabetic rats. The albiflorin treatment significantly reduced glucose and insulin levels while increasing WCR in diabetic rats. Moreover, the treatment with albiflorin significantly regulated the wound healing marker levels, reduced inflammatory response, and oxidative stress via upregulating the antioxidant mechanisms. Additionally, histological analyses demonstrated that albiflorin can speed up wound healing in diabetic rats. Consequently, the present data highlight that albiflorin can be a useful therapeutic alternative for diabetic wound treatment.
Footnotes
Abbreviations
DR: Diabetic rats; GPx: Glutathione peroxidase; IL-6: Interleukin-6; IL-1β: Interleukin-1 beta; MDA: Malondialdehyde; NF-κB: Nuclear factor-kappa B; ROS: Reactive oxygen species; SOD: Superoxide dismutase; TNF-α: Tumor necrosis factor-alpha.
Acknowledgments
The authors would like to thank the Affiliated Hospital of Hebei University, Baoding 071000, China, for the support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study is approved by the Ethical Committee of Shanxi Bethune Hospital (No. SYXK(冀) 2023-009), following all guidelines, regulations, and legal and ethical standards as required for animals.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
