Abstract
Background
Sophocarpine, a quinoline alkaloid derived from Sophora alopecuroides L., is known for its anti-inflammatory effects. These effects are achieved by modulating both nuclear factor-κB (NF-κB) pathway and node-like receptors pyrin domain containing 3 (NLRP3) inflammasome. However, whether sophocarpine could alleviate colonic inflammation requires further exploration.
Objectives
The present work focused on investigating the potential of sophocarpine for treating colonic epithelial barrier dysfunction in ulcerative colitis.
Materials and Methods
An inflammatory model was initially developed using human colorectal adenocarcinoma cells (Caco-2) induced by lipopolysaccharide (LPS) and tumor necrosis factor-α (TNF-α), which was used for investigating how sophocarpine affected inflammation-mediated colonic epithelial barrier function and monitoring levels of inflammatory responses. Besides, the colonic epithelial barrier integrity was evaluated through measuring transepithelial/transendothelial electrical resistance (TEER). Lastly, we conducted Western blotting for assessing zonula occludens-1 (ZO-1), claudin-1, p65, p-p65, IκBα, p-IκBα, MLC, p-MLC, and myosin light chain kinase (MLCK) protein levels. Besides, the beneficial effect of sophocarpine was also evaluated in a mouse mode of ulcerative colitis.
Results
Upon LPS stimulation, Caco-2 cells exhibited enhanced TNF-α and interleukin (IL)-1β levels, along with reduced IL-4 and IL-10 levels, decreased TEER values, increased fluorescein isothiocyanate (FITC)-dextran concentrations, and suppressed ZO-1 and claudin-1 protein levels. In contrast, sophocarpine treatment at varying concentrations attenuated the detrimental effects of LPS induction. Notably, sophocarpine could reverse TNF-α-induced colonic epithelial barrier dysfunction through significantly decreasing p65, IκBα, and MLC protein phosphorylation levels within LPS-treated Caco-2 cells. Sophocarpine administration also mitigated inflammatory damages within the ulcerative colitis mouse model.
Conclusion
According to these findings, sophocarpine prevents inflammatory damage in the colonic epithelial barrier by targeting the NF-κB/MLCK/MLC signaling pathway, indicating a novel intervention strategy to ameliorate ulcerative colitis.
Keywords
Introduction
Ulcerative colitis is an inflammatory bowel disease (IBD) associated with severe damage to colonic mucosa. This pathological condition presents with a variety of symptoms, such as, but not limited to, abdominal discomfort, frequent bowel movements, and rectal bleeding (Neurath & Leppkes, 2019; Sklyarov et al., 2011). The development of this condition is connected to a range of factors, including genetic predisposition, compromised colonic epithelial barrier, and exposure to certain environmental triggers, among others (Anbazhagan et al., 2018; Priyamvada et al., 2015). Over the past years, the number of cases of colitis in China has been on the rise, constituting approximately 20%–30% of colitis patients requiring surgery, imposing significant financial strains on affected families (Shao et al., 2019; Subasinghe et al., 2011). Current treatment options for colitis involve the use of steroids, aminosalicylates, immunosuppressants, and biologics (such as vaccines and growth factors) (Wan et al., 2014). However, these medications are often linked to severe adverse effects, such as fever, infections, diarrhea, and high rates of disease recurrence (Zou et al., 2020). Consequently, there is a crucial need to formulate novel and effective therapeutic alternatives for colitis. Physiologically active phytochemicals are convenient and efficient replacements for existing medications (Surguchov et al., 2021).
Sophocarpine is an alkaloid derived from Sophora alopecuroides L., which possesses various beneficial biological and pharmacological properties (Li et al., 2020). Sophocarpine was previously suggested to possess diverse activities, like anti-inflammation, anti-cancer, neuroprotection, and immunomodulation (Surguchov et al., 2021). For example, Lu and his team reported that sophocarpine provided protection against acute lung injury induced by lipopolysaccharide (LPS) (Lu et al., 2019). Additionally, Li and his colleagues demonstrated that sophocarpine alleviated lupus nephritis in the mouse model by suppressing node-like receptors pyrin domain containing 3 (NLRP3) inflammasome and nuclear factor-κB (NF-κB) activation (Li et al., 2018). Nevertheless, whether sophocarpine has a beneficial effect on colitis and the underlying mechanisms needs further clarification.
Motivated by these previous observations, we surmise that the anti-inflammatory effect of sophocarpine could mitigate inflammation and preserve colonic epithelial barrier integrity. This work established an inflammation cell model utilizing LPS-stimulated human colorectal adenocarcinoma cells (Caco-2). Subsequently, we treated Caco-2 cells with tumor necrosis factor-α (TNF-α) to evaluate how sophocarpine affected colonic epithelial barrier integrity after inflammatory stimulation. To determine the protective roles of sophocarpine, we co-treated inflammatory-induced cells with sophocarpine at diverse doses. Then, inflammatory markers, including TNF-α, interleukin (IL)-4, IL-10, and IL-1β, were quantified using quantitative reverse transcriptase-polymerase chain reaction (RT-qPCR) and enzyme-linked immunosorbent assay (ELISA) techniques. Epithelial barrier integrity in cell monolayer was analyzed through transepithelial/transendothelial electrical resistance (TEER) measurement as well as fluorescein isothiocyanate (FITC)-dextran labeling methods. Moreover, we conducted Western blotting for assessing zonula occludens-1 (ZO-1), claudin-1, p65, p-p65, IκBα, p-IκBα, MLC, p-MLC, and myosin light chain kinase (MLCK) protein levels. We also evaluated the beneficial effect of sophocarpine in a mouse mode of ulcerative colitis. Our findings demonstrated the protective effect of sophocarpine against inflammatory damage in the colonic epithelial barrier through targeting the NF-κB/MLCK/MLC signaling pathway, indicating a novel intervention strategy to ameliorate ulcerative colitis.
Materials and Methods
Cell Culture and Treatment
Caco-2 cells were provided by Wuhan Prosperity Life Science and Technology Co., Ltd., in Wuhan, China, and cultivated within the Caco-2 cell-specific medium provided by the same company and incubated in a humidified incubator at 37°C with 5% CO2, with medium change at 2-day intervals for ensuring proper cell development and maintenance. For inducing inflammatory damages to the Caco-2 cell monolayer culture, 1 mg/L LPS (L2630, Sigma–Aldrich, St. Louis, MO, USA) or 10 ng/mL recombinant human TNF-α (300-01A, PeproTech Rocky Hill, NJ, USA) was added to treat cells for 6 h to induce acute inflammatory response within the cells. For intervention, sophocarpine (HY-N0103, MedChemExpress, Shanghai, China) was applied at 50–800 µg/mL for 48 h after inflammatory stimulation.
Cell Counting Kit-8 (CCK-8) Cell Viability Assay
CCK-8 (Dojindo Molecular Technologies, Inc., Rockville, MD, USA) was utilized to assess how sophocarpine affected Caco-2 cell viability in line with specific instructions. In brief, we inoculated Caco-2 cells (5 × 103/well) into 96-well plates for overnight incubation to allow adherence. On the following day, various concentrations of sophocarpine (50, 100, 200, 400, and 800 µg/mL) supplemented with dimethyl sulfoxide (DMSO, Sigma–Aldrich, St. Louis, MO, USA) were added to treat the cells. Untreated cells were used in the control group, and DMSO-treated cells (vehicle control) were included for assessing potential toxicity induced by the solvent. We eliminated the culture medium after treatment for 48 h and later introduced fresh medium (100 µL) containing 10 µL of CCK-8 reagent into every well. Following 2 h of incubation in dark under 37°C, the microplate reader (SpectraMax M5, Molecular Devices, San Jose, CA, USA) was adopted for measuring absorbance values at 450 nm.
RT-qPCR Analysis
Total ribonucleic acid (RNA) sample was isolated from cultured Caco-2 cells (1 × 106 cells) under different experimental conditions utilizing the RNA-easy Isolation Reagent Kit (Novozymes Inc., Nanjing, China) following specific protocols. Subsequently, total RNA (1 µg) was prepared into complementary deoxyribonucleic acid (cDNA) with RevertAid reverse transcriptase (Thermo Fisher Scientific, Waltham, USA) through reverse transcription. Following this, a qRT-PCR analysis was conducted employing a 2× Universal SYBR Green Fast qPCR Mix (Abtek Co. Ltd., Wuhan, China) on the Roche LightCyler 480 system (Roche, Basel Switzerland). The following cycling conditions were adopted for qPCR: 2–5-min denaturation under 95°C; 10–30-s denaturation under 95°C, 30 s to 1 min annealing under 55°C–65°C, and 30 s to 1 min extension under 72°C for 40 cycles, with 10–30-s data acquisition under 60°C–80°C and 5–10 min optional final extension under 72°C and melting curve analysis. We adopted 2−∇∇Ct approach for calculating gene expression, with glyceraldehyde-3-phosphate dehydrogenase (GAPDH) being a reference. Primer sequences utilized are stated below:
TNF-α: Forward—CCTCTCTCTAATCAGCCCTCTG Reverse—GAGGACCTGGGAGTAGATGAG IL-1β: Forward—ATGATGGCTTATTACAGTGGCAA Reverse—GTCGGAGATTCGTAGCTGGA GAPDH: Forward—TGAACGGGAAGCTCACTGG Reverse—GCTTCACCACCTTCTTGATGTC IL-4: Forward—CCAACTGCTTCCCCCTCTG Reverse—TCTGTTACGGTCAACTCGGTG IL-10: Forward—GACTTTAAGGGTTACCTGGGGTTG Reverse—TCACATGCGCCTTGATGTCTG
ELISA
We determined the levels of different inflammatory and anti-inflammatory agents, like TNF-α, IL-1β, IL-4, and IL-10, in different cell groups using corresponding ELISA kits from Sigma in St. Louis, USA. We then gathered logarithmic-phase Caco-2 cells and planted them into six-well plates for each treatment set at 1 × 106/well. After inflammatory stimulation and sophocarpine treatment, we obtained culture supernatant and spun it under 4°C, 1,000 × g for a 10-min duration. After spinning, 100 µL of liquid sample was explored through ELISA following the instructions of the manufacturer. For the tissue samples, 10 mg of colonic tissues were lysed using the tissue lysis buffer included in the kit. After centrifugation at 4°C, 10,000 × g for 10 min, 100 µL of sample was used for ELISA quantification.
TEER Testing
TEER testing, an important measure of the intact tight junction of the epithelial layer, was conducted by utilizing a Millicell ERS-2 voltage-resistance meter from Millipore (Massachusetts, USA). Initially, we inoculated Caco-2 cells into the 6-well plate till forming one tight cell monolayer. We then selected cell samples with TEER values greater than 400 Ω-cm2 for various experiments on the Transwell chamber basolateral surface. The experimental conditions included a negative control group (untreated), LPS exposure (Caco-2 cells treated with 1 µg/mL LPS), sophocarpine treatment (800 µg/mL), and combined LPS + sophocarpine treatment. Notably, the cells were first exposed to LPS for a duration of 6 h, followed by sophocarpine treatment. Furthermore, the impact of TNF-α on colonic epithelial barrier function was investigated in different treatment groups, including a negative control group (untreated), TNF-α treatment group (Caco-2 cells treated with 10 ng/mL TNF-α), sophocarpine treatment (800 µg/mL), and TNF-α + sophocarpine treatment group. It is important to highlight that TNF-α treatment lasted for 8 h in the TNF-α group. In the TNF-α + sophocarpine treatment group, sophocarpine was administered 48 h after TNF-α treatment. TEER was assessed using the formula: Resistance (TEER) (Ω-cm2) = (RC − RE) × membrane area (cm2), where RC and RE denote resistance values (Ω) of the Caco-2 cells and control separately.
Detection of FITC-dextran 4kd Leakage
We inoculated Caco-2 cells into the 6-well plate till forming one tight cell monolayer and utilized FITC-dextran 4kd (Sigma–Aldrich, St. Louis, MO, USA) for assessing epithelial permeability across Caco-2 monolayers. After monolayer washing by PBS, Hank’s balanced salt solution (600 µL) was added to basolateral chambers. Additionally, 1 mg/mL FITC-dextran contained within HBSS (100 µL) was loaded onto the parietal chambers for 2 h under 37°C. Subsequently, HBSS (20 µL) was obtained in the basolateral chamber, followed by dilution using 10-fold HBSS before measuring the fluorescence signal at Ex/Em of 480/520 nm using a fluorescence plate reader.
Western Blotting
Initially, proteins were gathered by lysing cells using the radioimmunoprecipitation assay (RIPA) buffer (Xavier, Wuhan, China) supplemented with 0.01% phenylmethyl sulfonyl fluoride (PMSF, Xavier) in line with specific protocols. Further, protein content was analyzed with the bicinchoninic acid (BCA kit, 23250, Thermo Fisher Scientific, Waltham, USA). Further, proteins were subjected to the Bio-Rad Mini Trans-blot system (Bio-Rad, 88518, Hercules, USA), followed by transfer of the blots to the poly(vinylidene difluoride) (PVDF) membrane as well as primary and secondary antibody incubation for 24 and 2 h, respectively [1:1,000, Cell Signaling Technology (CST, Danvers, USA and Abcam, Cambridge, UK), including ZO-1 (#13663), claudin-1 (#13255), GAPDH (#5174), p-p65 (#3033), p65 (#8242), p-IκBα (#2859), IκBα (#4814), p-MLC (#3675), MLC (#3672) (CST); and MLCK (ab232949, Abcam), anti-mouse (#7076) and anti-rabbit (#7074) IgG horseradish peroxidase (HRP)-conjugated antibodies (1:3,000, CST)]. At last, we applied enhanced chemiluminescence (ECL, Bio-Rad) substrate for chemiluminescence development. Image J (NIH, USA) was adopted for quantitative result analysis.
Animal Experiment
We raised the 8–10-week-old male C57BL/6 mice (weight, 20–25 g) under controlled environmental conditions (22°C ± 2°C, 50% ± 0% relative humidity, and 12-h light/dark cycle). They could take standard rodent chow and water ad libitum. Every experiment was approved by the Institutional Animal Care and Use Committee (IACUC) of our hospital (2024-017). The animals were randomized into three groups (n = 6 each): Sham group: Mice were given standard drinking water and performed sham procedures without colitis induction; Model group: Mice were given standard drinking water and performed colitis induction; sophocarpine intervention group: Mice received sophocarpine (100 mg/kg/day) by oral gavage and were subjected to colitis induction.
Ulcerative colitis was induced in the model and sophocarpine intervention groups using the dextran sulfate sodium (DSS) method. Mice in these groups received 3% DSS (molecular weight 36,000–50,000 Da, Sigma) dissolved with drinking water for a 7-day period and subsequently another 7 days of standard drinking water. Mice in the sham group were given standard drinking water during this study. Sophocarpine (MedChemExpress, Shanghai, China) was blended into 0.5% carboxymethyl cellulose (CMC) solution and orally administered for a 14-day duration, starting from the colitis induction day. On day 14, the animals were euthanized by CO2 asphyxiation to collect colonic tissues for histological examination and molecular analysis.
Hematoxylin and Eosin (H&E) Staining
After fixation within 10% neutral-buffered formalin, colonic tissue samples were dehydrated into graded ethanol solutions before paraffin embedding. The 5-µM tissue sections were prepared using the microtome and later added onto glass slides. Following xylene deparaffinage and gradient ethanol rehydration, we performed H&E staining using a commercial kit (Hematoxylin and Eosin Stain Kit, Catalog No. HT110116, Sigma–Aldrich, St. Louis, MO, USA) in line with specific protocols. To be specific, the sections were subjected to 5 min of Mayer’s hematoxylin solution staining, washing with tap water, and differentiation within 1% acid-alcohol solution. After rinsing in tap water, 0.2% ammonia water was added to blue the sections before 2 min of counterstaining using eosin Y solution. Finally, gradient ethanol dehydration, xylene transparentizing, and mounting of sections with a permanent mounting medium were completed for histological evaluation under a light microscope.
Statistical Analysis
All the results were represented by mean ± standard deviation (SD) from three separate assays. We analyzed data between diverse groups through Student’s t-test or one-way ANOVA followed by Tukey’s post hoc test, with p < .05 standing for statistical significance.
Results
Sophocarpine Reduces LPS-induced Inflammation in the Caco-2 Cells
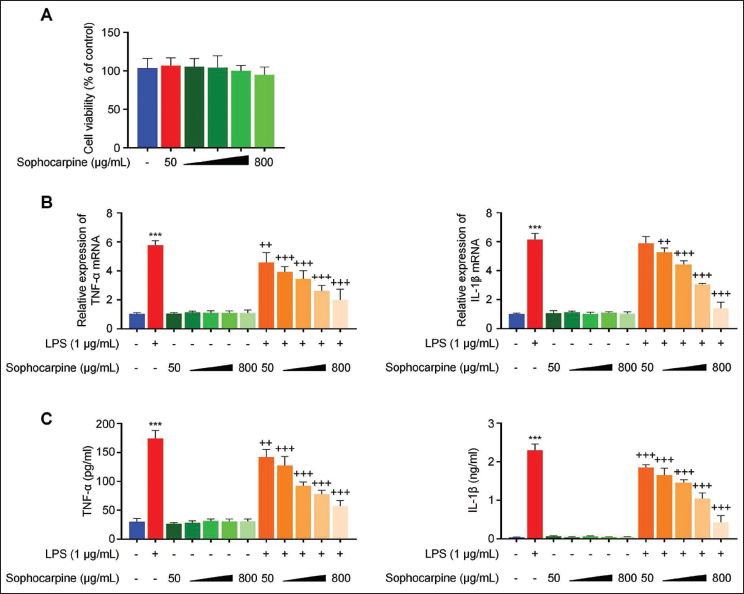
Initially, sophocarpine’s activity of anti-inflammation was assessed through measuring various inflammatory factor levels in cells exposed to LPS-induced inflammation. We initially incubated Caco-2 cells with varying doses of sophocarpine (50–800 µg/mL) for a 48-h duration. CCK-8 assay showed no significant cytotoxicity (Figure 1A). RT-qPCR results indicated a significant increase in TNF-α and IL-1β expression of LPS treatment versus control groups (p < .001). Conversely, cells treated with sophocarpine at varying concentrations showed a gradual decrease in TNF-α and IL-1β expression versus the LPS group (p < .01, Figure 1B). Nevertheless, treatment with sophocarpine alone did not significantly impact TNF-α or IL-1β expression. Consistently, ELISA analysis revealed that the LPS treatment group had significantly increased TNF-α and IL-1β protein levels relative to the control group (p < .001). Sophocarpine treatment decreased TNF-α and IL-1β expression dose-dependently (p < .01, Figure 1C), indicating that sophocarpine treatment effectively reduced TNF-α and IL-1β production triggered by LPS.
Sophocarpine Elevates the Expression of Anti-inflammatory Factors
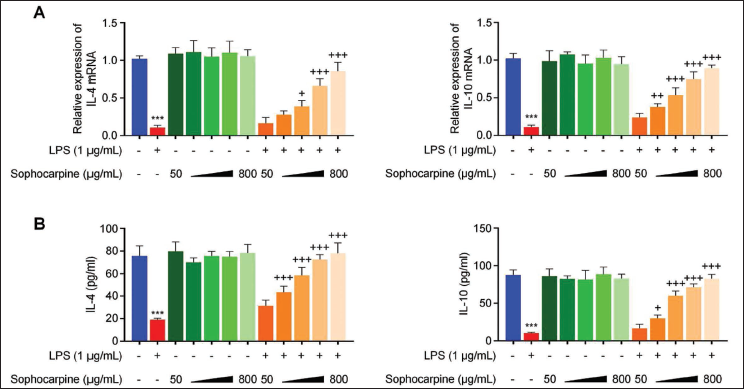
Moreover, we confirmed sophocarpine’s anti-inflammatory activity by analyzing the expression of anti-inflammatory factors. RT-qPCR analysis showed a marked reduction in IL-4 and IL-10 expression in cells treated with LPS versus the control group (p < .001). Sophocarpine administration apparently increased IL-4 and IL-10 expression within cells at various concentrations versus the LPS group (p < .05, Figure 2A). Nonetheless, sophocarpine administration alone did not have a significant effect on IL-4 or IL-10 expression. Furthermore, ELISA assay results demonstrated the apparently reduced IL-4 and IL-10 secretion within the cell culture supernatant of the LPS treatment versus control groups (p < .001). IL-4 and IL-10 secretion levels were elevated by increasing doses of sophocarpine after LPS induction (p < .05, Figure 2B). Consequently, sophocarpine upregulates anti-inflammatory factors, specifically IL-4 and IL-10, highlighting its anti-inflammatory properties.
Sophocarpine Improves LPS-induced Colonic Epithelial Barrier Dysfunction
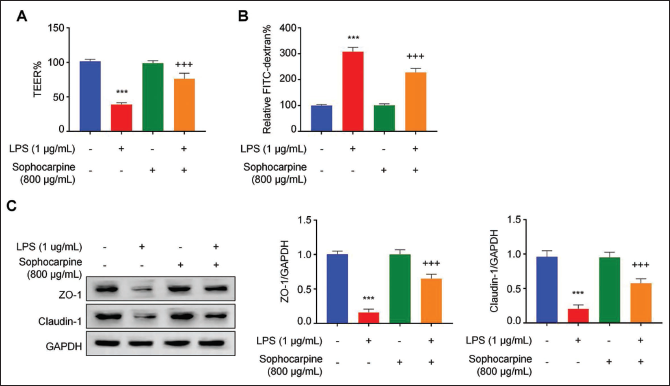
Next, we attempted to examine whether sophocarpine offers protection against colonic epithelial barrier dysfunction after LPS induction. TEER measurement was conducted to assess monolayer integrity after LPS stimulation and sophocarpine treatment. The LPS treatment group showed markedly decreased TEER values versus the control group (p < .001). Sophocarpine alone did not apparently affect TEER values. Nonetheless, sophocarpine administration remarkably elevated TEER values after LPS stimulation (p < .001, Figure 3A). Additionally, in the FITC-dextran leakage assay, the amount of FITC-dextran leaked from the Caco-2 monolayer in the LPS group markedly increased relative to the control group, while sophocarpine administration partially rescued this effect (p < .001, Figure 3B). Moreover, Western blotting assay suggested significantly decreased ZO-1 and claudin-1 protein levels (the proteins mediating tight junctions in the cell epithelial layer) in LPS versus the control groups (p < .001). Moreover, sophocarpine treatment alone did not significantly affect ZO-1 and claudin-1 protein levels, while sophocarpine treatment increased their expression levels relative to the LPS treatment group (p < .001, Figure 3C). Therefore, sophocarpine prevents LPS-mediated epithelial barrier dysfunction of Caco-2 cells.
Sophocarpine Enhances TNF-α-induced Colonic Epithelial Barrier Dysfunction
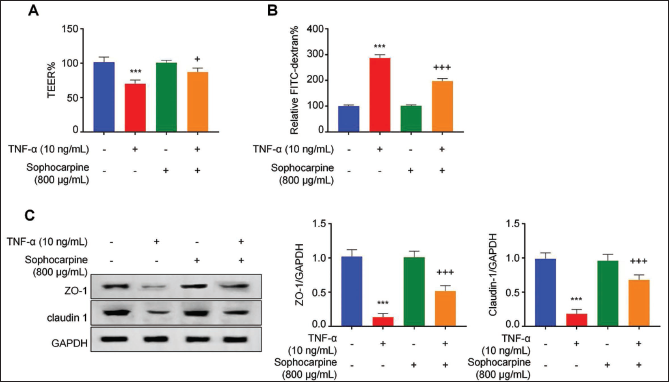
We further examined sophocarpine’s protection on TNF-α-mediated colonic epithelial barrier dysfunction. Similar to LPS induction, the TEER values in the TNF-α treatment group decreased compared with the control treatment group of cells (p < .001), and sophocarpine intervention significantly increased the TEER values after TNF-α treatment (p < .05, Figure 4A). FITC-dextran leakage experiment revealed the similar protection of sophocarpine from TNF-α-mediated colonic epithelial layer leakage (p < .001, Figure 4B). Additionally, as confirmed by WB analysis, ZO-1, and claudin-1 protein levels apparently lowered within cells in the TNF-α treatment group, and sophocarpine treatment partially restored their expression (p < .001, Figure 4C). These findings indicate that sophocarpine could also alleviate the TNF-α-mediated colonic epithelial barrier dysfunction.
Sophocarpine Negatively Regulates NF-κB/MLCK/MLC Pathway
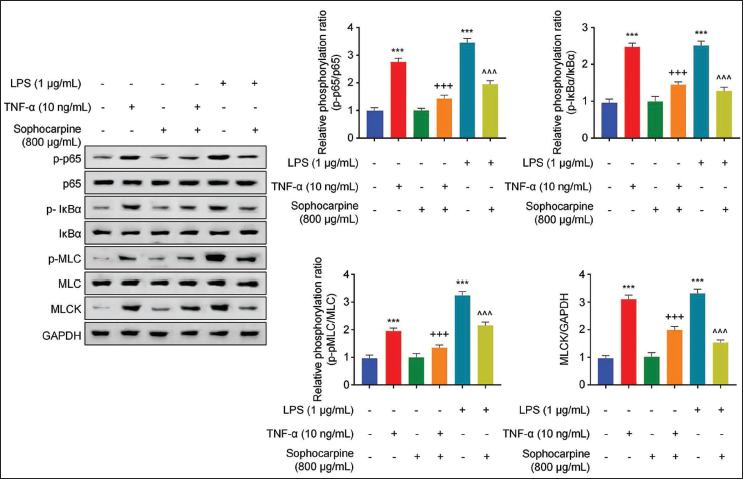
The mechanism of sophocarpine in alleviating inflammation was demonstrated through NF-κB/MLCK/MLC pathway. The relevant p65, IκBα, and MLC proteins (p-p65, p-IκBα, p-MLC) phosphorylation levels were assessed using WB analysis. LPS and TNF-α remarkably elevated the protein phosphorylation compared to the control group. Sophocarpine administration showed no significant effect. Interestingly, p-p65, p-IκBα, and p-MLC proteins had significantly reduced levels in cells treated with sophocarpine after either LPS or TNF-α stimulation (p < .001, see Figure 5). Consequently, sophocarpine could suppress NF-κB/MLCK/MLC pathway activation after inflammatory stimulation.
Sophocarpine Mitigates DSS-induced Ulcerative Colitis Through Modulating the NF-κB/MLCK/MLC Signaling Pathway
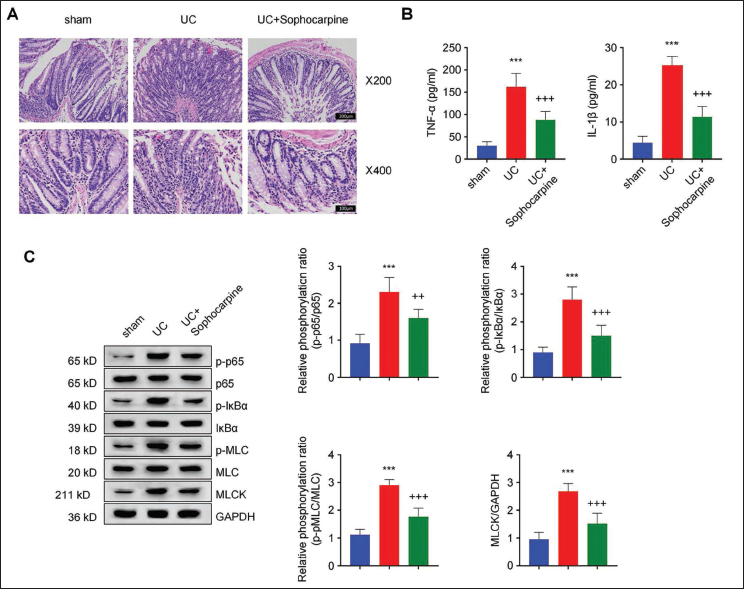
We next established an ulcerative colitis mouse model by gavaging DSS-containing drinking water, and the intervention group was administered with sophocarpine for 14 days. DSS induction induces notate damage to the tissue integrity of the colonic mucosa, manifested by the disruption of the colonic epithelial layer (Figure 6A). This effect was alleviated by sophocarpine treatment. Sophocarpine administration also remarkably decreased inflammatory factor (TNF-α, IL-1β) levels in the DSS-mediated model group (p < .001, Figure 6B). Furthermore, Western blot analyses demonstrated the suppression of phosphorylation of p65, IκBα, and MLC proteins in DSS-induced colon tissues after sophocarpine treatment (Figure 6C), indicating that sophocarpine improves DSS-induced ulcerative colitis by dampening NF-κB/MLCK/MLC pathway.
Discussion
Colitis has emerged as a serious medical problem affecting the health of people worldwide. Considering its severity, developing effective treatment to achieve optimal disease control is urgently needed. In this context, various novel active pharmaceutical ingredients, including drugs and biologics (vaccines and antibodies), have been developed and tested clinically (Chudy-Onwugaje et al., 2019). However, the moderate efficacy of the drugs and their associated side effects limit the treatment outcome (Hisamatsu et al., 2019; Martinez et al., 2017). Sophocarpine, the naturally derived compound which possesses various pharmacological activities, is well-known for its anti-tumor and anti-inflammatory effects (Jiang et al., 2018; Weng et al., 2022). This study demonstrated that sophocarpine, a natural quinoline alkaloid, can resist inflammation and protect colonic epithelial barrier integrity within cellular and animal models of ulcerative colitis. Sophocarpine suppressed pro-inflammatory factors and upregulated anti-inflammatory factors. It alleviated epithelial barrier disruption by increasing tight junction protein levels and improving transepithelial resistance. Mechanistically, sophocarpine inhibited NF-κB/MLCK/MLC pathway for mitigating inflammation while preserving barrier function. In the colitis mouse model, sophocarpine mitigated mucosal damage and inflammatory responses. These findings highlight the therapeutic potential of sophocarpine for ulcerative colitis by targeting NF-κB/MLCK/MLC pathway to ameliorate colonic inflammation and restore epithelial barrier integrity, presenting a novel intervention strategy for IBDs.
According to previous studies, sophocarpine possesses a diverse range of biological properties. For example, Yang et al. (2020) found that sophocarpine effectively reduced injury-induced carotid intima-media hyperplasia in a rat model by inhibiting inflammatory signaling. Similarly, another study by Zou et al. (2019) demonstrated that sophocarpine could inhibit NF-κB-mediated inflammation both in vitro and in vivo, thereby alleviating diabetic cardiomyopathy. In our current investigation, we utilized RT-qPCR and ELISA analyses to examine how LPS-mediated inflammation affected Caco-2 cells. We observed the significantly upregulated pro-inflammatory factors (TNF-α, IL-1β) whereas downregulated anti-inflammatory factors (IL-4, IL-10) upon LPS stimulation. Interestingly, sophocarpine was able to reverse these effects through reducing pro-inflammatory cytokines while stimulating anti-inflammatory cytokines in the LPS-induced inflammatory cell model. Notably, sophocarpine had no apparent influence on untreated control cells, indicating no direct toxic effect. Therefore, sophocarpine can mitigate LPS-mediated inflammation. This study also verified sophocarpine’s benefits for DSS-mediated colitis model. Our findings are consistent with previous studies in which sophocarpine was shown to decrease the expression levels of LPS-induced inflammatory factors in carotid artery restenosis and cardiomyopathy (Fu et al., 2023; Yang et al., 2021).
The selective permeability of colonic epithelial barrier serves to selectively transport substances across the colonic lumen through transcellular or paracellular pathways. The selective transport mechanisms depend on the sealing of intercellular spaces by the structural and functional engagement of claudin-1 and ZO-1 proteins to form tight junctions (Di Tommaso et al., 2021). According to our findings, both LPS and TNF-α induced colonic epithelial barrier dysfunction in human colon cancer epithelial Caco-2 cells, which was evidenced by the decreased TEER values, the increased FITC-dextran leakage, as well as the downregulated levels of ZO-1 and claudin-1 proteins. By contrast, treatment with sophocarpine improved colonic epithelial barrier integrity after LPS and TNF-α treatment. Published studies have shown that TNF-α may impair tight junction function in epithelial and vascular endothelial cell lines, leading to NF-κB and MLCK activation, actin filament contraction, and epithelial permeability increase (Zareie et al., 1998). Notably, tight junctions regulate bidirectional signaling between intra- and extracellular environments, with the MLCK/MLC pathway being one of the crucial pathways that impact transportation through tight junctions. Previous studies have suggested that the pathogenesis underlying IBD is linked to MLCK activation and MLC hyperphosphorylation. Conversely, the administration of an MLCK inhibitor (ML-7) can stabilize intercellular tight junctions, thus significantly preserving the epithelial barrier function (Herring et al., 2006; Hong et al., 2009). Therefore, our study revealed that sophocarpine treatment led to suppression of NF-κB/MLCK/MLC signaling pathway within LPS- and TNF-α-induced cells, indicating that sophocarpine protects epithelial barrier through regulating tight junction integrity.
The current study demonstrated that sophocarpine protects against inflammation-mediated colonic epithelial barrier dysfunction in ulcerative colitis, targeting the NF-κB/MLCK/MLC pathway. Nonetheless, limitations such as using simplified in vitro models, lack of intracellular protein targets, and unexplored mechanisms warrant further investigations. Future research should focus on more physiologically relevant models, dose optimization, exploration of additional molecular targets (including intracellular protein targets through proteomics approaches), long-term efficacy and safety evaluations, and potential combinatorial therapies. Addressing these aspects could help translate sophocarpine into clinical practice as a novel treatment intervention against ulcerative colitis and other IBDs.
Conclusion
As shown in Figure 7, our study showed that sophocarpine reduced inflammation in LPS-stimulated Caco-2 cells. Sophocarpine treatment also restored the compromised epithelial barrier function, which was verified through the improved transepithelial electrical resistance, reduced FITC-dextran leakage, and upregulated ZO-1 and claudin-1. From the mechanistic perspective, sophocarpine targeted NF-κB/MLCK/MLC pathway to inhibit inflammatory signaling. In vivo studies further confirmed sophocarpine’s protective effects against colitis in mice. Consequently, sophocarpine is a potential treatment against ulcerative colitis by preserving colonic epithelial integrity and modulating inflammatory responses. Future directions include investigating sophocarpine’s intracellular protein targets through proteomics, optimizing dosage, and evaluating long-term safety for potential clinical translation as a novel treatment for IBDs.
A Schematic Drawing Showing the Regulatory Mechanism in this Study.
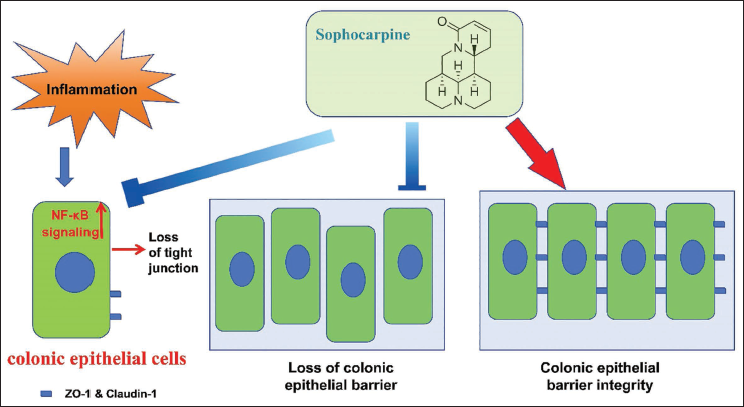
Limitations
Despite the success in alleviating inflammation in vitro, it is required to validate the mechanism of sophocarpine’s action in vivo in treating colitis. The differential regulation mechanism of the sophocarpine’s action is an extremely complex process involving various factors. Therefore, in the next work, we will focus on the other mechanism of sophocarpine’s action.
Footnotes
Abbreviations
CMC: Carboxymethyl cellulose; DSS: Dextran sulfate sodium; FITC: Fluorescein isothiocyanate; IACUC: Institutional Animal Care and Use Committee; IBD: Inflammatory bowel disease; LPS: Lipopolysaccharide; MLCK: Myosin light chain kinase; RIPA: Radioimmunoprecipitation assay; SD: Standard deviation; TEER: Transendothelial electrical resistance.
Authors’ Contributions
Y. M. Zhang and Z. G. Zuo mainly participated in the literature search, study design, writing, and critical revision and prepared Figures 1–3. T. Z. Liu mainly participated in data collection, data analysis, and data interpretation and prepared Figures 4 and ![]() . All authors read and approved the final manuscript.
. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
All experimental procedures were approved by the Institutional Animal Care and Use Committee of our hospital.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Qinhuangdao science and technology research and development program (202301A250), Central Guidance Local Science and Technology Development Fund Project (246Z7736G) and Hebei Provincial Administration of Traditional Chinese Medicine TCM Research Program (2024150).
