Abstract
Background
Anorexia–cachexia syndrome in head and neck cancer (ACSHNC) seriously affects the patient’s quality of life and prognosis. Curcumin inhibits head and neck cancer and anorexic cachexia syndrome. S100A9 is related to tumors, but its role in ACSHNC requires further study.
Objectives
This study intends to investigate the role of S100A9 in ACSHNC.
Materials and Methods
In this study, subjects were randomly divided into negative control (NC) group, low-, medium-, and high-dose curcumin groups, and cell proliferation was detected; flow cytometry detected cell apoptosis; Western blot detected CyclinB1, P21, DR5, and DR4, cleaved caspase-9, cleaved poly (ADP-ribose) polymerase (PARP), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) protein expression along with analysis of serum albumin and prealbumin levels.
Results
Before treatment, the patient had obvious symptoms such as loss of appetite, weight loss, and fatigue, and levels of lipid mobilization factors and S100A9 in plasma were significantly increased. After curcumin treatment, the patient’s symptoms improved considerably, and the levels of lipid mobilization factors and S100A9 in plasma were reduced. In addition, urinary excretion of lipid mobilization factors was also reduced.
Conclusion
Curcumin can improve the symptoms and prognosis of patients with ACSHNC by regulating lipid mobilization factors and S100A9 levels, indicating that it might be utilized as a new possibility for ACSHNC treatment.
Introduction
Anorexia–cachexia syndrome with head and neck cancer (ACSHNC) is a common cancer complication. The disease is caused by tumor cells releasing tumor necrosis factor (TNF), interleukin (IL)-6, and other inflammatory factors, as well as gastrin, motilin, and so on, and the further metabolites produced directly act on the stomach. The intestines stimulate the feeding center and satiety center of the hypothalamus or medulla oblongata, leading to loss of appetite or anorexia, affecting appetite and digestive function (Mäkitie et al., 2022). In head and neck cancer cells, not only epidermal growth factor receptor (EGFR) gene mutations occur but also abnormal activation of PI3K, Akt, and mammalian target of rapamycin (mTOR) pathways, which leads to abnormal tumor proliferation and enhanced viability. In addition, it also releases a variety of microribonucleic acid (RNAs), which will further affect the normal functions of the neuroendocrine system and gastrointestinal tract, causing anorexia and cachexia syndrome. At present, no new clinical treatment for this syndrome has been found.
In recent years, more and more studies have discovered that Chinese patent medicines are of great value in oncology. Ginseng can regulate the hypothalamic–pituitary–adrenal (HPA) axis, promote body metabolism and energy balance, and promote food digestion and absorption, thereby improving the patient’s nutritional status (Lyu et al., 2021). Astragalus can stimulate the secretion of insulin, promote the metabolism of sugar and protein, and regulate the body’s energy metabolism balance through PI3K/Akt/mTOR signaling (Chen et al., 2022). In addition, curcumin, Ganoderma lucidum, and so on, can inhibit Ras protein, and G. lucidum cannot only negatively regulate CyclinD1 through nuclear factor-kappa B (NF-κB), causing tumor cells to stagnate in the G0/G1 phase but also lead to the loss of CDK4 to a certain extent. According to the survival status, RB dephosphorylates, inactivating E2F to inhibit tumor cell growth, and reducing the consumption of the tumor, thereby alleviating the cachexia symptoms of patients (Grammatikopoulou et al., 2020). These indicate that traditional Chinese medicine (TCM) ingredients have great research potential in cachexia syndrome. Curcumin is a phenolic antioxidant extracted from rhizomes such as turmeric, zedoary, and mustard. It not only has antioxidant, anti-inflammatory, hypolipidemic, and anti-cancer effects but can also reduce plasma viscosity by inhibiting platelet aggregation; in addition, it also has anti-tumor, hypolipidemic, anti-bacterial, and other effects. However, few research reports support the use of curcumin in ACSHNC treatment. It has been reported that curcumin can inhibit SREBP-1, PPARγc, C/EBPα, and inhibit hormone-sensitive lipase (HSL), adipose triglyceride lipase (ATGL), fatty acid-binding protein (FABP), and other factors, thereby reducing the synthesis of cholesterol and fatty acids, thereby affecting lipid mobilization (Wang et al., 2019). In addition, PKA and Ca²⁺ can promote lipolysis and lipid mobilization by phosphorylating enzymes related to lipolysis. Studies believe that curcumin’s regulatory effect on S100A9 is multifaceted in some cases. Curcumin can regulate the expression of genes related to apoptosis, thereby affecting the inflammatory response and immune response of melanoma cells and promoting the apoptosis of cancer cells. This substance can also inhibit NF-κB activation and upregulate CDKI to regulate the expression of the above genes. In addition, curcumin inhibits Bcl-2 and Bax expression and COX-2 and iNOS, thus reducing the inflammatory response to a certain extent. The impact of apoptosis in ACSHNC needs to be further explored (Sancar et al., 2022).
Studies have reported that lipid mobilization factors can inhibit the activity of JAK/STAT and mTOR, thereby downregulating this pathway and reducing the malignancy of tumor cells. Curcumin can not only mitigate PTEN phosphorylation and inhibit the expression of miR-21, thereby promoting PI3K/AKT signaling activation, but also inhibit activation of Ras/MAPK signaling through regulation of Ras, ultimately inhibiting PI3K/AKT signaling (Ding et al., 2022). Studies have pointed out that the inhibitory effect of curcumin on S100A9 is related to the MAPK pathway. On the one hand, it can inhibit the growth of gastric cancer cells to a certain extent by regulating miR-34a, S100A9, NF-κB, and MAPK pathways. On the other hand, it can inhibit the growth of gastric cancer cells to a certain extent. On the other hand, curcumin also inhibits miRs to reduce the expression of S100A9 further and inhibit TNF-α and IL-1β levels, thereby reducing the inflammatory response and inhibiting cancer cells (Schmitt et al., 2021). For cancer disease syndromes represented by ACSHNC, studying the mechanism of curcumin regulating lipid mobilization factors through S100A9 may discover new drug treatment targets and provide a reference for the treatment. It may provide a reference for the treatment of other cancers and promote the development of cancer-related research (Grabner et al., 2021). Zhao Chengzhi and others pointed out that curcumin has great potential for head and neck squamous cell carcinoma (HNSCC) treatment. It can exert anti-cancer effects through cell cycle arrest and promotion of apoptosis (Mäkitie et al., 2022). Some scholars have found that for HNSCC, curcumin synergizes with DTX by down-regulating MDSCs, M2 macrophages, and upregulating CD8+ T cells. At the same time, it reduces the secretion of CXCL1 and interferon in tumor tissues. The secretion of interferon (IFN)-γ and TNF-α is increased, which in turn shows a more significant tumor inhibitory effect (Talbert & Guttridge, 2022). At the same time, Kürten et al. (2021) also found that curcumin not only reduced the expression of IC ligands in HNSCC, thereby regulating epithelial-mesenchymal transition-mediated tumor invasion but also effectively restored CD8+ cytotoxic T cell’s ability to lyse cancer cells to further increase T cell proliferation, TIL and effector cytokine levels (Kürten et al., 2021). In addition, curcumin can effectively inhibit the Treg attraction effect of PIC, while reversing the EMT process back to MET, reducing Treg attraction chemokine CCL22, significantly inhibiting Treg migration, and has an immunomodulatory effect in HNSCC. It can be seen that curcumin has a positive inhibitory effect on HNSCC (Johnson et al., 2020). Compared with placebo, curcumin can significantly improve muscle mass and reduce the rate of body fat loss in patients undergoing cancer treatment. At the same time, adding 1,600 mg of curcumin has a marginal benefit in cancer cachexia treatment (Brabletz et al., 2021). It was also pointed out that the improvement effect of curcumin on anorexia–cachexia syndrome was better than that of placebo to a certain extent (Zhang et al., 2021).
Materials and Methods
Instruments and Reagents
Curcumin (purity > 98º; batch number: 110823-201706; molecular structure diagram 2) Curcumin is a phenolic antioxidant extracted from rhizomes such as turmeric, zedoary, and mustard. It not only has antioxidant, anti-inflammatory, hypolipidemic, and anti-cancer effects but can also reduce plasma viscosity by inhibiting platelet aggregation.
About 80 patients with ACSHNC who were treated in our hospital from May 2022 to August 2023 were enrolled in this study, aged (52.32 ± 4.42) years old, and were divided into negative control (NC) groups, as well as low-curcumin, medium-curcumin, and high-curcumin dose groups. In addition, 20 normal healthy people during the same period were selected as the normal group, with an age of 49.36 ± 3.89. Patients in the NC group took 160 mg of medroxyprogesterone acetate tablets orally every day. Curcumin low-, medium-, and high-dose groups took 800, 1,000, and 1,200 mg of curcumin orally every day based on the NC1 group.
The following materials were used in the study: biological microscope (Japanese OLYMPUS Company); bovine human albumin (BSA) (Wuhan ASPEN Company); cell culture flask (American Corning Company); and Petri dish (American Corning Company). This study was approved by the ethics committee of our hospital, and all patients provided informed consent.
Inclusion criteria: patients diagnosed with head and neck cancer; those experiencing symptoms such as anorexia, weight loss, muscle wasting, fat loss, fatigue, anemia, and cachexia; and those willing to participate in research or treatment plans.
Exclusion criteria: the presence of other serious diseases, such as heart disease, liver disease, and kidney disease; inability to tolerate the side effects related to the study or treatment plan; currently receipt of other anti-tumor treatments that may affect the results of the study or treatment plan; or the presence of mental illness or cognition impairment and inability to cooperate with research or treatment plans.
Cell Culture
Human head and neck cancer cell HN4 were cultured in RPMI 1640 culture medium, cultured in an incubator with 100% saturated humidity, 5% CO2 volume fraction, and 37°C. The cells grow adherently in a single layer. After the adherent cells reach 80%–90% confluence, they are digested with 0.25% trypsin, and the pellet is resuspended in a culture medium containing 10% fetal bovine serum, 1 × 106/L. Density inoculation, passage at a ratio of 1:2 for later use. Divided into NC group, curcumin group, ABR-215757 group, agonist group, and curcumin + agonist group.
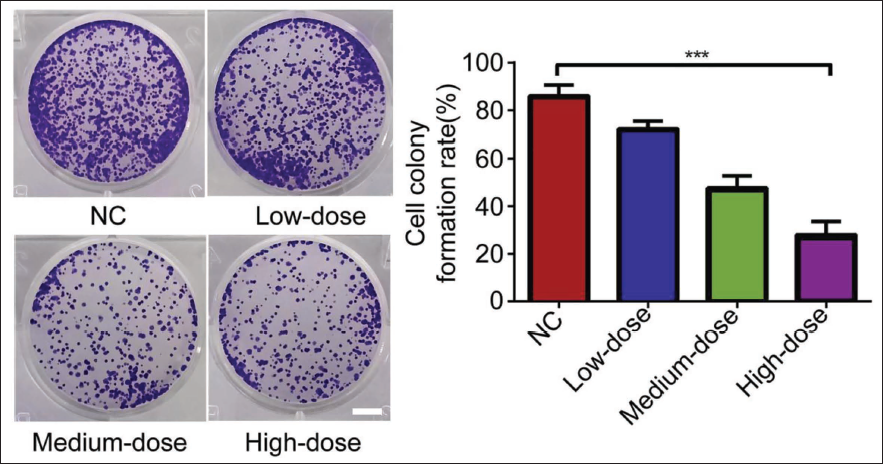
Clone Formation Assay to Detect Cell Proliferation
The NC group, curcumin group, ABR-215757 group, agonist group, and curcumin + agonist group were respectively inoculated into 96-well or 24-well cell culture plates, and each well contained one or more groups of cells to achieve the ultimate goal of cell colony formation followed by detection of cell proliferation.
Flow Cytometry to Detect Cell Apoptosis in Each Group
RNA is digested, and RNA inhibitors are used. Propidium iodide (PI) is used to combine RNA and deoxyribonucleic acid (DNA). Flow cytometry is used to detect the fluorescence intensity of PI combined with DNA to understand the DNA content and analyze various phases of the cell cycle: S phase, In G2/M and G1/G0 phase, take at least 105 cells from each group 24 h after transfection. After washing with PBS, resuspend the cells in binding buffer and label cells with 500 μL of labeling solution. Apoptotic cells are analyzed and detected through the loss instrument, and the status of living cells, individual apoptotic cells, and necrotic cells in each group can be accurately distinguished to a certain extent.
Western Blot
Cells were extracted and digested in lysis buffer, and their total protein content was measured. Separate by 10% SDS-PAGE, and then place it at 4°C with primary antibody CyclinB1 (ab32053, 1:1,000); P21 (ab109520, 1:1,000); DR5 (ab199357, 1:1,000); DR4 (ab312848, 1:1,000); cleaved caspase-9 (ab65608, 1:1,000); cleaved PARP (ab32064, 1:1,000) and maintained overnight. Wash and incubate the membrane with HRP-labeled secondary antibody for 1 h, followed by detection with GAPDH as the internal reference.
Clinical Symptom Score
Referring to the 2002 edition of the “Guiding Principles for Research on New Traditional Chinese Medicines” issued by the former Ministry of Health, the five symptoms of appetite, bloating, fatigue, insomnia, and palpitations were divided into three levels: none, mild, moderate, and severe according to severity, and were recorded as 0, 1, 2, and 3 points, respectively. Add the scores of each symptom to get the total symptom score. If the symptom score after treatment decreases by ≥70% compared with the score before treatment, it is considered effective; if the score decreases by ≥30%, it is considered stable; if the score decreases by <30% or even increases, it is considered ineffective.
Observe the Patient’s Physical Strength, Food Intake, and Weight Changes
The Karnofsky score was used to record changes in the physical strength of patients in each group.
Observe the Occurrence of Adverse Events in Patients
Adverse event incidence rate = number of adverse event cases/total number of cases × 100%.
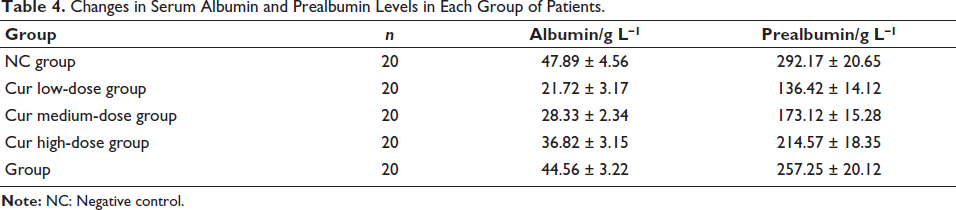
Detect Serum Albumin and Prealbumin Levels
About 5 mL of fasting blood was collected from each group of patients in the morning, centrifuged at 3,500 rpm for 5 min, and detected by Abbott 2000 fully automatic chemiluminescence instrument.
Statistical Analysis
The data obtained in each of the above experiments were displayed as mean ± SD and analyzed using SPSS21.0 and GraphPad Prism software. If no special requirements exist, p < .05 is used as the test standard.
Results
Curcumin can Effectively Inhibit the Development of ACSHNC and has a Certain Relationship with S100A9
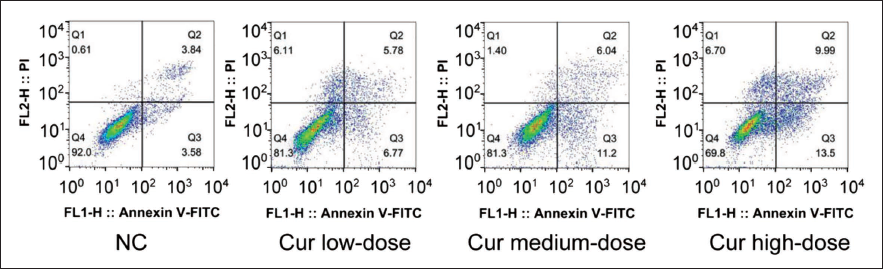
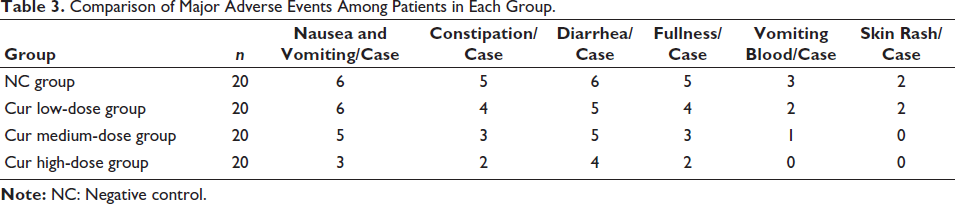
It can be seen that curcumin can effectively inhibit the development of ACSHNC to a certain extent (Figure 1). The mitochondrial collapse has been improved; the higher the dose of curcumin, the more the apoptotic ability of head and neck cancer cells was significantly enhanced, and the apoptosis rate was greatly increased (Figure 2); and then, by comparing the patient’s physical strength, food intake, and weight, it can be found that the effective rate of the Cur high-dose group was not less than 95% (Table 1). Before and after the TCM symptom score treatment, after comparing the differences among the patients, it was found that the conditions of patients with anorexia were significantly improved under the suppression of high-dose curcumin (p < .05) (Table 2). In the case of adverse effects caused by the use of curcumin, judging from the situation, the proportion of Cur high-dose group is lower than that of the other two groups (Table 3).
Changes in Physical Strength, Food Intake, and Weight of Patients in Each Group.
Comparison of Differences in Patients’ Traditional Chinese Medicine (TCM) Symptom Scores Before and After Treatment.
Comparison of Major Adverse Events Among Patients in Each Group.
When the patient’s serum albumin and prealbumin levels were abnormally high, it was found that Cur’s high-dose group effectively inhibited the development of the patient’s disease and restored the serum protein to normal levels (Figure 3, Table 4).
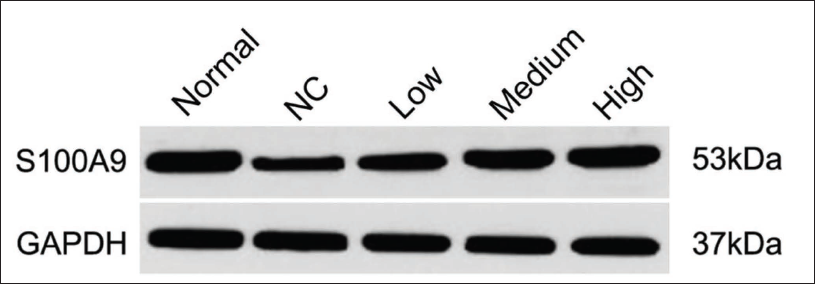
Changes in Serum Albumin and Prealbumin Levels in Each Group of Patients.
Inhibiting S100A9 can Inhibit Lipid Mobilization Factors and Thereby Inhibit ACSHNC
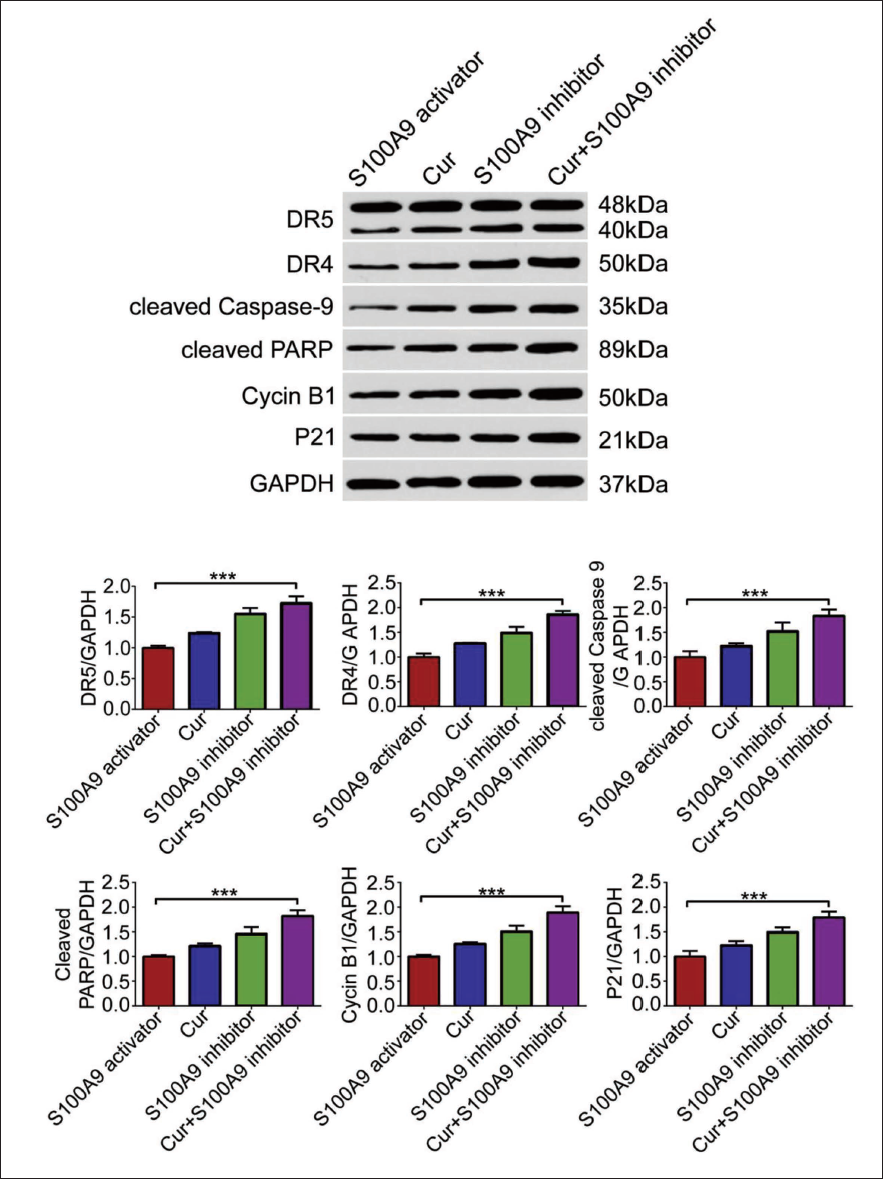
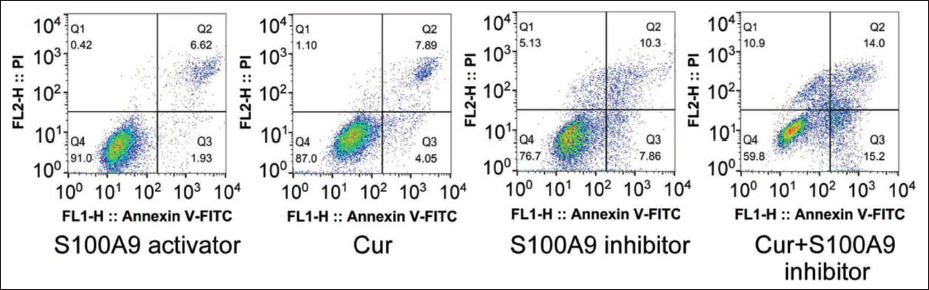
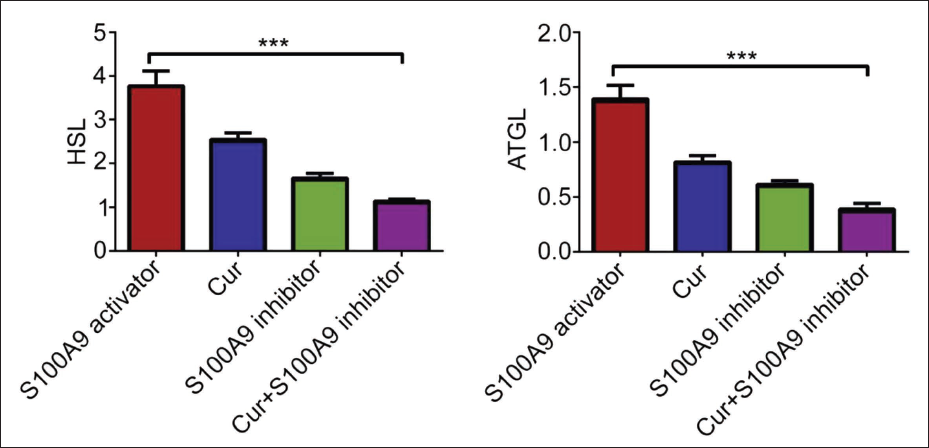
Based on the above research, this topic will further explore the mechanism of curcumin in the apoptosis of ACSHNC cells and use S100A9 agonists and S100A9 inhibitors to intervene under the condition of lipid mobilization factor intervention. The results showed that, to a certain extent, S100A9 can inhibit S100A9. To inhibit lipid mobilization factors and thereby inhibit head and neck cancer (Figure 4A), in comparison, it can be clearly seen that the apoptosis of head and neck cancer cells in the curcumin A + 100A9 inhibitor group was significantly higher than other groups (Figure 4B–F). From the above, the curcumin + S100A9 inhibitor group showed a good inhibitory effect (Figure 5) and a low expression of HSL and ATGL levels (Figure 6). Therefore, the above experimental results demonstrate that the inhibitory effect on head and neck cancer can be promoted by inhibiting S100A9 and thereby inhibiting lipid mobilization factors.
CyclinB1, P21, DR5, DR4, Cleaved Caspase-9, Cleaved Poly (ADP-ribose) Polymerase (PARP), and Glyceraldehyde-3-phosphate Dehydrogenase (GAPDH) Protein Expression in Each Group. ***p < .001.
Apoptosis of Head and Neck Cancer Cells in Each Group.
Discussion
We explored whether curcumin can improve the ACSHNC that accompanies the disease. By setting low, medium, and high doses of curcumin, we found that as the intervention dose of curcumin increased, the apoptosis rate of head and neck cancer cells gradually increased, and cell proliferation also gradually slowed down (Distefano & Goodpaster, 2018). At the same time, the patient’s physical strength, food intake, and weight gradually increased, and the anorexia, fatigue, abdominal distension, insomnia, and heart palpitations also decreased accordingly, and the number of adverse events decreased (Finniss, 2018). Compared with patients who did not take curcumin, the albumin and prealbumin levels of patients who took low, medium, and high doses of curcumin increased steadily with the increase in dose (Sun et al., 2021). It can be seen that curcumin has a positive effect in improving ACSHNC. In addition, during this experiment, we also found that the level of S100A9 in patients with ACSHNC was significantly reduced, but with the intervention of curcumin, its level was increased. Among them, high-dose curcumin intervention had the most significant effect. It is suggested that the ameliorative effect of curcumin on ACSHNC is potentially related to S100A9.
As a calcium ion-binding protein, S100A9 is highly expressed in cancer and can participate in cancer development by affecting cancer cells’ biological processes (Falcone et al., 2022). Researchers have found that S100A9 is highly expressed in breast cancer. Related studies have shown that the expression level of S100A9 gradually increases with the degree of cervical intraepithelial neoplasia, while there is no S100A9 expression in normal epithelial tissue (Manolis et al., 2022). The effect of S100A9 on cervical cancer cells is induced through Wnt/β-catenin signaling (Pop et al., 2022). Falcaro et al. (2021) successfully detected an increase in the expression of S100A9 by inducing M1 and M2 macrophages. On the contrary, inhibiting S1000A9 can inhibit the polarization of M2 macrophages, thereby inhibiting the progression of liver cancer. For nasopharyngeal carcinoma, S100A9 also has a significant inhibitory effect, and it is believed that this effect is achieved by inhibiting PI3K/Akt signaling. In addition, S100A8 and S100A9 expression is significantly increased in the tumors of patients with pancreatic cancer and cachexia. S100A8, S100A9, and S100A8/A9 can induce C2C12 myotube atrophy and are the main pathogenic factors causing pancreatic cancer and cachexia (Zhang & Wang, 2020). Zhang et al. (2020) also proved this result when studying the relationship between pancreatic cancer-related diabetes and cachexia. To assess S100A9’s role in ACSHNC, we cultured human head and neck cancer cells HN4 and transfected them into groups. We found that under the intervention of S100A9 inhibitor, CyclinB1, P21, DR5, DR4, cleaved caspase-9. The expression of cleaved PARP and GAPDH proteins increased, the apoptosis of head and neck cancer cells increased, and the levels of lipid mobilization factors HSL and ATGL decreased. This effect was more obvious under the combined intervention of curcumin and S100A9 inhibitor. The intervention results of the S100A9 agonist were the opposite. This shows that inhibiting S100A9 can inhibit the expression of lipid mobilization factors and play a role in improving head and neck cancer. It can also be concluded that curcumin can inhibit S100A9, thereby reducing the levels of lipid-mobilizing factors and then exerting an effect on the anorexia–cachexia syndrome of head and neck cancer. Improve this result. This is because, in cancer, lipid mobilization factors not only promote lipolysis and utilization of tumor cells but also provide energy for tumor growth and spread (Lee et al., 2019). It can also affect the signal transduction and cell cycle progression of tumor cells, thus affecting tumors (Bossi et al., 2021). Meanwhile, some studies have shown that the content of lipid mobilization factors increases significantly in the blood of patients with tumor cachexia. In cancer, HSL and ATGL can catalyze the decomposition of triglycerides into diglycerides and fatty acids (Dunne & Roeland, 2022), inhibit acetyl CoA carboxylase and expression of fatty acid synthase and other enzymes, thereby reducing fat synthesis (Grabner et al., 2021). Another study found that inhibiting the expression of lipid mobilization factors can inhibit lipolysis and utilization of tumor cells, thereby affecting the signal transduction and cell cycle progression of tumor cells (Knudsen et al., 2019). The study also found that inhibiting the expression of lipid mobilization factors can enhance the killing effect of radiotherapy and chemotherapy on tumor cells and improve the therapeutic effect (Tain et al., 2021).
Conclusion
In summary, curcumin can inhibit S100A9, reduce HSL and ATGL levels, and thereby improve ACSHNC. This study helps to deeply reveal the mechanism of curcumin in ACSHNC treatment, provides theoretical support for the clinical application of curcumin, and provides new possibilities for the treatment of ACSHNC. At the same time, this study also has certain limitations, such as the small sample size, which needs to be further expanded to verify the research results. In addition, the therapeutic effect of curcumin on ACSHNC needs to be further verified in clinical trials.
Footnotes
Abbreviations
ACSHNC: Anorexia–cachexia syndrome with head and neck cancer; GAPDH: Glyceraldehyde-3-phosphate dehydrogenase; mTOR: Mammalian target of rapamycin; PARP: Poly (ADP-ribose) polymerase.
Acknowledgments
The authors acknowledge the financial support of the Hebei Provincial Administration of Traditional Chinese Medicine Project for this work.
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The ethics committee of the Affiliated Hospital of Hebei University of Engineering approved this study. Verbal informed consent was obtained from all subjects before the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hebei Provincial Administration of Traditional Chinese Medicine Project (2020226).
