Abstract
Background
Osteoarthritis affects millions of people worldwide. Currently, available pharmacological treatment is limited to topical and oral non-steroidal anti-inflammatory drugs (NSAIDs). Several studies have reported the potential of curcumin in the management of osteoarthritis, owing to its anti-osteoarthritic, anti-inflammatory, and antioxidant effects. However, the poor bioavailability of curcumin limits its safe and effective use in the long-term management of osteoarthritis.
Objectives
The present research study was undertaken to understand the anti-osteoarthritis effect of a newly developed, highly bioavailable curcumin formulation, Maxicuma®, at the preclinical level. The anti-osteoarthritic activity was evaluated using the monosodium iodoacetate (MIA)-induced osteoarthritis model in rats at 10 and 25 mg/kg bw doses.
Materials and Methods
Anti-inflammatory and antioxidant effects were studied in the MIA-induced osteoarthritis model and the lipopolysaccharide (LPS)-induced mouse macrophages. The extent of efficacy was investigated by examining the expression of catabolic and anabolic genes from the joint capsule, along with histopathology examination of the joint.
Results
Maxicuma® administration in osteoarthritic rats reduced the edema of affected knee joints, increased the expression of anabolic gene COL2A1, decreased the expression of catabolic genes MMP-3 and MMP-7, and reduced the glycosaminoglycan contents in the affected knee joints. Chondrocyte scoring through histopathology of the joint tissues has confirmed the anti-osteoarthritic effects of Maxicuma®. Maxicuma® reduced the inflammatory mediators and enhanced the antioxidant defense system in osteoarthritis rats and LPS-induced mouse macrophages.
Conclusion
Thus, Maxicuma® showed an anti-osteoarthritic potential by balancing the synthesis and degradation of joint cartilage.
Introduction
Osteoarthritis (OA) is the most common form of arthritis, affecting millions of people worldwide (Cui et al., 2020; Hunter et al., 2020). The global prevalence of knee osteoarthritis was reported as 16% in individuals with age ≥15 years and 22.9% in individuals with age ≥40 years (Cui et al., 2020). OA occurs when the cartilage that cushions the ends of the bones wears down over time. The risk of osteoarthritis increases with age, making aging a significant factor in its development. Joint injuries, whether from sports, accidents, or repetitive use, can also elevate the risk. Obesity is another crucial risk factor, as excess weight stresses weight-bearing joints. The most common symptoms of OA include joint swelling and pain. From a molecular perspective, higher levels of proinflammatory cytokines, C-reactive protein (CRP) (inflammatory marker), prostaglandin E2 (PGE2), cyclooxygenase-2 (COX-2), nitric oxide (NO), reactive oxygen species (ROS), matrix metalloproteinases (MMPs), cartilage matrix breakdown products (e.g., COMP, glycosaminoglycans) and reduced levels of anabolic growth factors (COL2A1), anti-inflammatory cytokines, and cytoprotective chaperones have been reported to be involved in osteoarthritis (Jovanovic et al., 1998; Mobasheri et al., 2023; Molnar et al., 2021). Substances with anti-inflammatory and antioxidant properties could be potentially helpful in the management of osteoarthritis. Currently, recommended options in the management of osteoarthritis are mostly limited to physical exercise, education, weight loss, and topical non-steroidal anti-inflammatory drugs (NSAIDs) and oral NSAIDs (Nice Guideline [NG226], 2022). Long-term use of oral NSAIDs is associated with complications of the upper gastrointestinal, cardiovascular, and renal systems (Mobasheri et al., 2024).
Curcumin, the active component of turmeric, is clinically effective and safe in the management of osteoarthritis, either alone or in combination with NSAIDs (Zhao et al., 2024). Several clinical studies indicate that curcumin helps reduce pain and improve joint function in individuals with osteoarthritis. Curcumin helps to reduce inflammation, a key factor in the progression of osteoarthritis, by affecting the activity of inflammatory enzymes and cytokines (Deng et al., 2024; Park et al., 2020). Additionally, curcumin’s antioxidant effects help to neutralize free radicals, which can damage joint tissues, and clinically reduce oxidative stress in patients with osteoarthritis (Panahi et al., 2016). Curcumin has also been shown to help protect cartilage by reducing cartilage degradation and promoting its repair (Hamdalla et al., 2022). However, curcumin’s bioavailability is relatively low, and to enhance its effectiveness, formulations often include piperine (found in black pepper) or are developed using advanced delivery systems (Yakubu & Pandey, 2024).
Turmeric extracts available in the market are usually standardized to 95% curcuminoids and are recommended to be used at a daily dose of more than or equal to 1,500 mg unless they are unique high-bioavailable formulations. Maxicuma® is a newly developed proprietary high-bioavailable turmeric standardized to 40% curcuminoids, and its recommended dose ranges from 100 to 250 mg/day. The objective of the current study was to find the anti-osteoarthritic potential of Maxicuma® in rats and to understand its possible mode of action in the management of osteoarthritis, using mouse macrophage cell lines as well as in the animal study.
Materials and Methods
Test Item
Maxicuma® used in the study was prepared from an ethanol extract of turmeric (Curcuma longa). The ethanol extract was prepared using green ethanol obtained from sugarcane molasses and was standardized to contain 95% curcuminoids by high-performance liquid chromatography (HPLC). The powder of the 95% product was subjected to patent-pending LIMAN technology (India Glycols Ltd. & Bhartia, 2024; Mahanta et al., 2023), in which the 95% curcuminoid mixture was encapsulated with excipients to protect curcuminoids from metabolism, and the curcuminoids content of the final formulation (Maxicuma®) was maintained at 40% by HPLC.
Cell Culture and Test Solution
The mouse macrophage cell line (RAW 264.7) was purchased from the National Center for Cell Science, Pune, India and cultured in Dulbecco’s modified Eagle medium-HG/MEM (DMEM-HG/MEM) (Gibco, USA) media supplemented with 10% inactivated fetal bovine serum (FBS) (Gibco, USA), penicillin (100 IU/mL), streptomycin (100 µg/mL), and amphotericin B (5 µg/mL) (HiMedia, India) in a humidified atmosphere of 5% CO2 at 37°C until confluent. A test solution of 10 mg/mL concentration was prepared by using 10 mg of Maxicuma® in DMEM-HG supplemented with 2% inactivated FBS. Serial twofold dilutions were used for cytotoxic studies.
Cytotoxicity Study of Maxicuma® in Mouse Macrophage Cell Line
The stock cultures of the mouse macrophage cell line (RAW 264.7) were grown in 25 cm2 culture flasks, and cytotoxicity studies were carried out in a 96-well microtiter plate. Cell viability was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay (Denizot & Lang, 1986). The monolayer cell culture was trypsinized, and the cell count was adjusted to 1.0 × 105 cells/mL using DMEM-HG, which contained 10% FBS. To each well of the 96-well microtiter plate, 0.1 mL of the diluted cell suspension (approximately 10,000 cells) was added. After 24 h, when a partial monolayer had formed, the supernatant was flicked off, the monolayer was washed once with medium, and 100 µL of different test concentrations (0.4–100 µg/mL) of the test substance was added to the partial monolayer in microtiter plates. The plate was incubated at 37°C for 3 days in a 5% CO2 atmosphere, and microscopic observations were carried out at every 24-h interval. After 72 h, the test solution in the wells was discarded, and 50 µL of MTT (HiMedia, India) in Dulbecco’s phosphate-buffered saline (DBPS) (HiMedia, India) was added to each well (Hassani et al., 2022).
The plates were gently shaken and incubated for 3 h at 37°C in a 5% CO2 atmosphere. The supernatant was removed, and 100 µL of propanol was added to solubilize formazan. Absorbance was measured using a microplate reader (Biotek, USA) at a wavelength of 540 nm. The percentage growth inhibition was calculated using the standard formula:
Percentage cytotoxicity = Control absorbance – Test absorbance/control absorbance × 100
Percentage cell viability = 100 – Percentage cytotoxicity.
Antioxidant Study in LPS-stimulated Mouse Macrophage Cell Line
The mouse macrophage cell line (RAW 264.7) was plated at a density of 1 × 105 cells/well in 96-well plates and preincubated with different concentrations (0.4, 0.6, 0.8, 1, 2, and 4 µg/mL) of Maxicuma® at 37°C in a 5% CO2 incubator for 2 h. After 2 h, LPS at 1 µg/mL was added to the cells and further incubated for 16 h (Kacem et al., 2015).
The medium was removed after 16 h, and the cells were washed twice with the medium. One milliliter of ice-cold tris-buffered saline (TBS) with pH 7.4 was added, and cells were scraped. Cell suspensions were sonicated three times for 5 s each time on ice. The sonicated cells were centrifuged at 10,000 × g for 10 min at 4°C. The obtained supernatants were then used for antioxidant enzyme activities. Kits for superoxide dismutase (SOD) (0612248), glutathione peroxidase (GPx) (0589927), catalase (CAT) (0602429), and glutathione (GSH) (0616926) were procured from Cayman, USA. Quantification of SOD, CAT, GSH, and GPx levels in the supernatant was performed as per the procedure mentioned in the respective kits. Samples were tested in triplicates.
Anti-inflammatory Study in Lipopolysaccharide (LPS)-stimulated Mouse Macrophage Cell Line
The mouse macrophage cell line (RAW 264.7) was plated in a 96-well plate (1 × 105 cells/well) and incubated for 24 h at 37°C in a humidified atmosphere of 5% CO2. The culture medium was discharged, the cells were then supplemented with fresh medium, and 100 µL of Maxicuma® at different concentrations (0.4, 0.6, 0.8, 1, 2, and 4 µg/mL) was added. After 1 h, LPS (1 µg/mL) was added into each well, and the plate was incubated for 24 h at 37°C in a humidified atmosphere of 5% CO2. The medium was removed, and the cells were washed twice with the medium and centrifuged at 2,000 × g for 10 min to get the supernatant (Kang et al., 2021; Phumsuay et al., 2020).
Enzyme-linked immunosorbent assay (ELISA) kits for tumor necrosis factor-alpha (TNF-α), interleukin (IL)-10, IL-1β, and IL-13 were obtained from Invitrogen, USA, and COX-2 from Elabscience, USA. Quantification of IL-10 (88-50629-22), IL-13 (BMS6015), TNF-α (88-7340-22), IL-1-β (371088-005), and COX-2 (GY01R8VJ7276) in the supernatant was performed as per the procedure mentioned in the respective ELISA kits. Samples were tested in triplicates.
NO Inhibition Assay in LPS-stimulated Mouse Macrophage Cell Line
The NO inhibition assay was carried out as per Kang et al. (2021). Briefly, the mouse macrophage cell line (RAW 264.7), plated at 1 × 105 cells/well in 24-well plates, was used for this study. Cells were pretreated with various concentrations of Maxicuma® for 1 h, then treated with LPS (1 µg/mL) and incubated for 24 h. After 24 h, 100 µL of cell culture medium was mixed with 100 µL of Griess reagent and incubated at room temperature for 10 min. Absorbance was taken at 540 nm. Fresh culture medium was used as a blank in every experiment. The NO production accumulated in the cell culture was measured as nitrite using Griess reagent. The quantity of nitrite was calculated using a sodium nitrite standard curve.
Monosodium Iodoacetate (MIA)-induced OA in Rats
Animals
The current animal study, MIA-induced osteoarthritis in rats, was approved by the Institutional Animals Ethics Committee (IAEC) with the approval number PAL/IAEC/2022/01/01/44 on January 27, 2024.
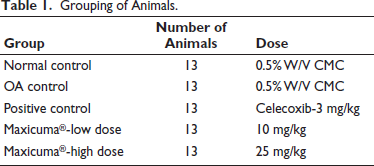
Sixty-five male in-house-bred Sprague–Dawley (SD) rats, aged 6–8 weeks (170–270 g), were used in this study. All the animals were housed inside polypropylene rat cages in the animal house, maintained at a temperature of 22°C ± 3°C, humidity of 30%–70%, good air circulation, and a cycle of 12 h in light and 12 h in the dark period. Each animal was identified using a cage card and an animal identification number. All the animals were given free access to commercially available feed (Krishna Valley Agrotech, Pune, India) and RO water. Animals (n = 65) were divided into five groups, namely, normal control (G1), osteoarthritic control (G2), positive control (G3), Maxicuma® low dose (G4), and Maxicuma® high dose (G5), with 13 animals per group.
Evaluation in MIA-induced OA
Test and reference samples for oral administration were prepared freshly every day before dosing as a suspension in a 0.5% carboxymethyl cellulose (CMC) solution. The experiment was initiated after an acclimatization period of 5 days. Animals in the G1 were administered 25 µL of normal saline (0.9% NaCl) intra-articularly into the right knee of the rats under isoflurane anesthesia, and no treatment was given. Animals in the groups G2–G5 were administered 25 µL of saline (0.9% NaCl) containing 0.4 mg of MIA intra-articularly into the right knee of the rats under isoflurane anesthesia. After 5 days of MIA administration, the G3, G4, and G5 groups were given respective treatment for 28 days. The G4 and G5 group animals were administered with Maxicuma® at a once-daily oral dose of 10 mg/kg bw and 25 mg/kg bw, respectively. The dose of Maxicuma® used in this animal study was based on the dose of an adult human (weighing 60 kg), calculated using the body surface area—conversion factor of 6.17. Celecoxib was administered orally at a once-daily oral dose of 3 mg/kg bw to the G3 group animals (Jeong et al., 2017), as mentioned in Table 1.
Grouping of Animals.
All animals were observed for general clinical signs once daily during the acclimatization and dosing periods. Body weights were recorded on the day of MIA administration and on days 1 and 28 of the dosing period. The hind limb knee joint perimeter was measured on day 1 (5 days after MIA administration) and days 7, 14, 21, and 28 by the classical thread method. In brief, white thread was surrounded at the hind limb joint and marked with a marker pen. The thread was then laid over a graduated scale, and the perimeter was recorded. The increase in joint perimeter was calculated by subtracting the left hind limb joint perimeter from the right hind limb joint perimeter. On day 28, blood samples were drawn from the retro-orbital plexus from six animals of each group using a non-heparinized glass capillary tube and centrifuged at 4°C with 4,000 rpm for 10 min for serum separation. The separated serum was stored at –80°C until use. The stored serum samples were analysed for SOD and CAT using commercially available kits (catalog no. KTB1030 and KTB1040 for SOD and CAT, respectively, from Abbkine Scientific Co., Ltd., Georgia, USA). On day 28 of the dosing period, all the animals were sacrificed humanely by asphyxia under carbon dioxide inhalation. The hind limb knee joint was collected from all the animals and processed for histopathological analysis, as well as gene and protein expression analysis. The joint tissue homogenates were analysed for glycosaminoglycans content using a commercially available ELISA kit (catalog no. ELK8486, ELK Biotechnology Co., Ltd., Denver, USA).
Gene Expression Analysis
The right knee joint tissues of four animals from each group were processed for gene expression analysis using reverse transcription-polymerase chain reaction (RT-PCR) for GAPDH, COL2A1, MMP-3, and MMP-7 genes. In brief, the joint tissues were homogenized, ribonucleic acid (RNA) was isolated using Trizol, complementary deoxyribonucleic acid (cDNA) was synthesized using Takara Prime Script cDNA synthesis kit, and PCR amplification was performed using SYBR Green Supermix. Denaturation occurred at 95°C for 10 min, separating the DNA strands. Next, primer extension and annealing took place at 95°C for 10 s, allowing DNA synthesis. Finally, the melting temperature step occurred at 60°C for 60 s, ensuring specific primer binding. These steps were repeated for 35 cycles to amplify and quantify gene expression levels (Qiagen Rotor-Gene Q). The primers used are shown in Table 2 (Jeong et al., 2017).
Primers Used in the Gene Expression Analysis.

Analysis of Markers of Inflammation
Joint tissues from the right knee of five animals from each group was homogenized and processed for COMP (E-EL-R1059, Elabsciences Biotechnology Inc., Texas, USA), COX-2 (KTE100863, Abbkine Scientific Co., Ltd., Georgia, USA), IL-10 (KTE101029, Abbkine Scientific Co., Ltd., Georgia, USA), CRP (KTE101074, Abbkine Scientific Co., Ltd., Georgia, USA), PGE2 (E0504Ra, BT Lab, Zhejiang, China), and TNF-α (KTE9007, Abbkine Scientific Co., Ltd., Georgia, USA) proteins using commercially available ELISA kits.
Histopathology
The right knee joint tissues of four animals from each group were stored in 10% neutral buffered formalin. The stored tissues were processed, embedded, and cut cross-sectionally for the joint cartilage at an approximate thickness of 3–5 µm and stained with hematoxylin and eosin (H&E) staining and toluidine staining. In brief, histological changes were assessed to confirm the efficacy of Maxicuma® and celecoxib on the cartilage tissue of MIA-induced OA rats. Following the sacrifice after 28 days, each knee joint was resected, fixed in 10% formalin for 24 h at 4°C, and decalcified with 5% hydrochloric acid for 4 days at 4°C. After decalcification, specimens were dehydrated with graded acetone and embedded in paraffin. Finally, sections (thickness, 2–3 µm) were stained with 0.2% hematoxylin and 1% eosin for 5 min and 3 min, respectively. For toluidine blue staining, sections were stained with 0.04% toluidine blue for 5–10 min, then rinsed in tap water and dried in warm air. A blind observer then observed sections according to the modified Mankin scoring system. The slides were observed in 4×, 10×, and 40× objective lenses using Leica DM500 microscope (images of 10× were provided). All stained slides were histologically evaluated and graded on a scale of 0–13.
Statistical Analysis
The raw data was processed using GraphPad Prism, Version 9.5.1 (GraphPad Software, Inc, CA, USA). Data was subjected to analysis of variance (ANOVA) followed by Tukey’s multiple comparison test for in vivo data and Dunnett’s test for in vitro data. Values were expressed as Mean ± Standard Deviation (SD).
Results
Cytotoxicity of Maxicuma® in Mouse Macrophage Cell Line
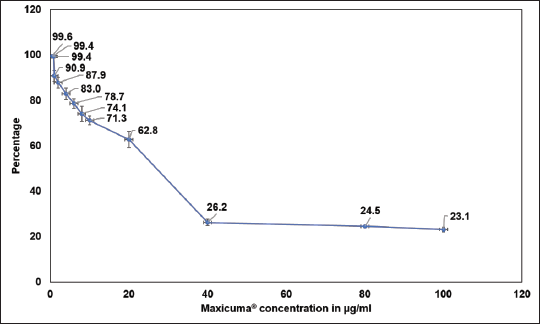
The percentage cell viability of the mouse macrophage cells after treatment with various concentrations (0.4–100 µg/mL) of Maxicuma® is presented in Figure 1. Cell viability after incubation with Maxicuma® remained at >80% up to a concentration of 4 µg/mL in the cell viability assay using MTT. Therefore, the 4 µg/mL concentration was selected as the maximum concentration of Maxicuma® to be tested in the mouse macrophage cell line (RAW 264.7) to study its antioxidant and anti-inflammatory effects.
Cytotoxicity of Maxicuma® in Mouse Macrophage Cells.
Antioxidant Effect of Maxicuma® in LPS-induced Mouse Macrophages
Results of the antioxidant study of Maxicuma® in the LPS-induced mouse macrophage cell line (RAW 264.7) were expressed as Mean ± SD of triplicates and are shown in Figure 2. The SOD activity of mouse macrophage cells (219.75 ± 4.09 pg/mL) decreased significantly (p < .001) to 54.17 ± 4.56 pg/mL in the presence of LPS. Maxicuma® significantly (p < .001) prevented the LPS-induced reduction in SOD activity at 0.8, 1.0, 2.0, and 4.0 µg/mL concentrations, and the preventive effect was concentration-dependent up to the maximum test concentration of 4.0 µg/mL (Figure 2a). The CAT activity of mouse macrophage cells (207.33 ± 4.23 pg/mL) decreased significantly (p < .001) to 45.28 ± 0.99 pg/mL in the presence of LPS. Maxicuma® significantly (p < .001) prevented the LPS-induced reduction in CAT at 0.6, 0.8, 1.0, 2.0, and 4.0 µg/mL concentrations, and the preventive effect was concentration-dependent up to the maximum test concentration of 4.0 µg/mL (Figure 2b). The GPx activity of mouse macrophage cells (383.67 ± 6.46 pg/mL) decreased significantly (p < .001) to 84.19 ± 7.06 pg/mL in the presence of LPS. Maxicuma® significantly (p < .001) prevented the LPS-induced reduction in GPx activity at 0.6, 0.8, 1.0, 2.0, and 4.0 µg/mL concentrations, and the preventive effect was concentration-dependent up to the maximum test concentration of 4.0 µg/mL (Figure 2c). The GSH level of mouse macrophage cells (383.27 ± 4.27 pg/mL) decreased significantly (p < .001) to 66.63 ± 5.11 pg/mL in the presence of LPS. Maxicuma® significantly (p < .001) prevented the LPS-induced reduction in the GSH activity at 0.6, 0.8, 1.0, 2.0, and 4.0 µg/mL concentrations, and the preventive effect was concentration-dependent up to the maximum test concentration of 4.0 µg/mL (Figure 2d).
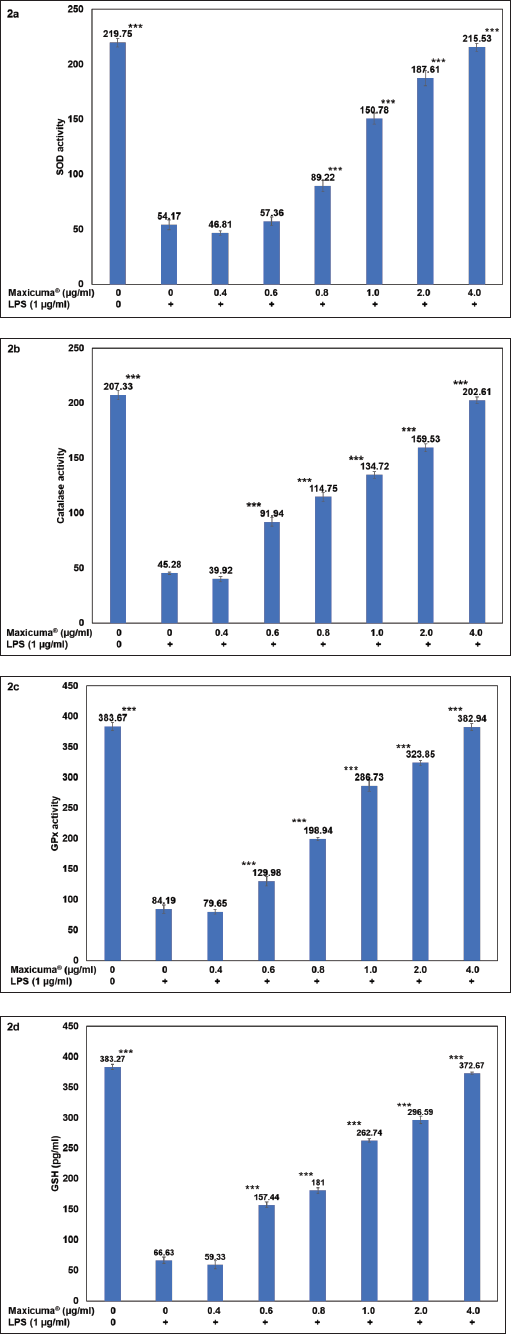
The mouse macrophage cells (RAW 264.7) were treated with Maxicuma® at different concentrations (0.4, 0.6, 0.8, 1, 2, and 4 µg/mL) and incubated at 37°C in a 5% CO2 incubator for 2 h. LPS at 1 µg/mL was added after 2 h to the cells and further incubated for 16 h. After 16 h, the cell culture supernatant was analysed for (a) SOD activity, (b) CAT, (c) GPx, and (d) GSH. Each value represents Mean ± SD with n = 3. ***p < .001—significant compared to LPS-induced mouse macrophage cells.
Maxicuma® was tested at concentrations of 0.4, 0.6, 0.8, 1, 2, 4, 6, 8, 10, 20, 40, 80, and 100 µg/mL in the mouse macrophage cell line (RAW 264.7).
Anti-inflammatory Effect of Maxicuma® in LPS-induced Mouse Macrophages
Results of the anti-inflammatory study of Maxicuma® in the LPS-induced mouse macrophage cell line (RAW 264.7) were expressed as the Mean ± SD of triplicates and are represented in Figure 3.
The mouse macrophage cells (RAW 264.7) were treated with Maxicuma® at different concentrations (0.4, 0.6, 0.8, 1, 2, and 4 µg/mL) and incubated at 37°C in a 5% CO2 incubator for 1 h. After incubation, LPS at 1 µg/mL was added to the treated cells and further incubated for 24 h. After 24 h, the cell culture supernatant was analysed for (a) IL-10, (b) IL-13, (c) TNF-α, (d) IL-1β, (e) COX-2, and (f) NO. Each value represents mean ± SD with n = 3. ***p < .001—significant compared to LPS-induced mouse macrophage cells.
The levels of IL-10 (246.85 ± 0.69 pg/mL) and IL-13 (214.44 ± 2.4 pg/mL) of the mouse macrophage cells decreased significantly (p < .001) to 31.92 ± 1.58 pg/mL and 32.68 ± 2.70 pg/mL, respectively, in the presence of LPS. Maxicuma® significantly (p < .001) prevented the LPS-induced reduction in the levels of IL-10 (at 0.6, 0.8, 1.0, 2.0, and 4.0 µg/mL) and IL-13 (at 0.4, 0.6, 0.8, 1.0, 2.0, and 4.0 µg/mL) in a concentration-dependent manner (Figure 3a and b). The levels of TNF-α (107.94 ± 1.3 pg/mL), IL-1β (143.01 ± 3.3 pg/mL), and COX-2 (115.86 ± 2.0 pg/mL) of the mouse macrophage cells increased significantly (p < .001) to 430.78 ± 1.7 pg/mL, 365.05 ± 3.5, and 253.03 ± 1.3 pg/mL, respectively, in the presence of LPS. Maxicuma® significantly (p < .01) prevented the LPS-induced increase in the levels of TNF-α, IL-1β, and COX-2 at all the tested concentrations (0.4–4.0 µg/mL) in a concentration-dependent manner (Figure 3c–e).
Effect of Maxicuma® on NO in LPS-induced Mouse Macrophages
A significantly (p < .001) increased concentration of NO was found in the LPS-induced mouse macrophages, as assessed in terms of the nitrite level. Pretreatment of the mouse macrophages with Maxicuma® at concentrations from 0.4 to 4 µg/mL significantly (p < .001) prevented the increase in the concentration of nitrite compared with the levels in the LPS-induced mouse macrophages, demonstrating the NO-inhibiting effect of Maxicuma® (Figure 3f).
Effect on Maxicuma® in MIA-induced OA
Maxicuma® was evaluated for its anti-osteoarthritic properties by MIA-induced osteoarthritis in rats. The effect of Maxicuma® on edema, content of glycosaminoglycans, inflammatory markers, antioxidants, and gene expression was assessed. Histopathology was also performed to visualize the impact of Maxicuma® on knee joints. No mortality or clinical signs of toxicity were observed in any of the animals during the entire study period. No statistically significant difference in body weight was seen in the treated groups compared with the control group animals.
Anti-edema Effect on Knee Joints
A statistically significant increase in the knee joint perimeter was observed in the animals of groups G2–G5 after 5 days of MIA administration compared with the control group (G1) animals, confirming the induction of osteoarthritis. Maxicuma® at the dose of 25 mg/day showed a significant reduction in the edema induced by MIA, as assessed from the reduction in the perimeter on day 21 (p < .05) and on day 28 (p < .001), compared with the MIA control group (G2). Results of the knee joint perimeter are shown in Figure 4.
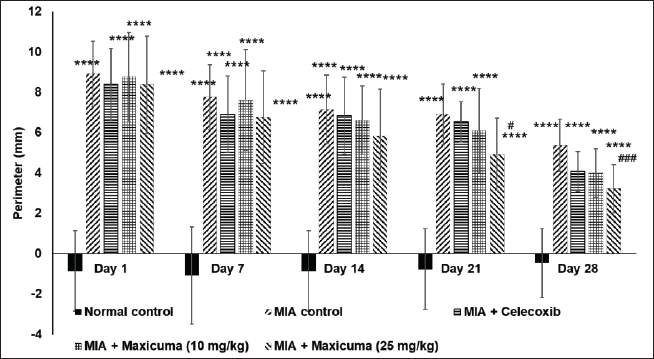
Effect on Glycosaminoglycans
The glycosaminoglycans level in the knee joint tissue was significantly (p < .0001) increased in the osteoarthritic control (G2) group compared to the normal control group animals (G1). Maxicuma® administration at a dose of 10 and 25 mg/kg bw for 28 days in the MIA-induced osteoarthritic rats significantly reduced the joint tissue degradation as indicated by the reduced levels of GAG (Table 3).
Effect of Maxicuma® on Antioxidant Status and Glycosaminoglycans in the Monosodium Iodoacetate (MIA)-induced Osteoarthritic Rats.
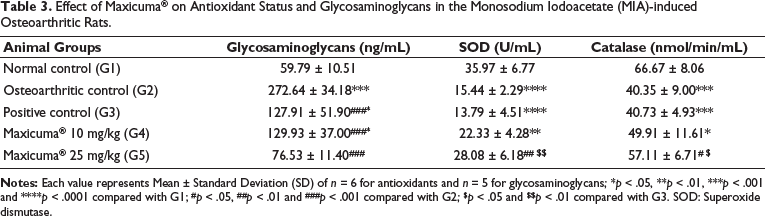
Effect on Antioxidant Status
The serum levels of SOD activity (p < .0001) and CAT (p < .001) were significantly lower in the osteoarthritic rats compared to those of the normal control group (Table 3). Rats administered with Maxicuma® at a 25 mg/kg bw dose for 28 days had significantly higher SOD activity (p < .01) and CAT (p < .05) levels compared to the osteoarthritic rats, indicating the antioxidant potential of Maxicuma®. The antioxidant effect of Maxicuma® at a 25 mg/kg bw dose for 28 days was also significantly higher compared with celecoxib (G3) concerning the SOD activity (p < .01) and CAT (p < .05) levels.
Effect on Inflammatory Mediators
The markers of inflammation, namely, COMP (p < .001), COX-2 (p < .001), CRP (p < .0001), PGE2 (p < .001), and TNF-α (p < .001), increased significantly. The anti-inflammatory marker IL-10 decreased significantly (p < .0001) in the MIA-induced osteoarthritic rats compared with the control group animals (Table 4). Maxicuma® (at doses of 10 mg/kg bw and 25 mg/kg bw) and celecoxib treatment significantly reversed the levels of COX-2, CRP, IL-10, PGE2, and TNF-α in the osteoarthritic rats compared with the MIA-induced osteoarthritic rats. Additionally, Maxicuma® treatment at a 25 mg/kg bw dose significantly (p < .01) reduced the level of COMP in the osteoarthritic rats compared with the MIA-induced osteoarthritic rats. Maxicuma® at the 25 mg/kg bw dose showed significantly better anti-inflammatory action in positively modulating the levels of IL-10 and TNF-α compared with the Maxicuma® at the 10 mg/kg bw dose.
Anti-inflammatory Action of Maxicuma® in the Monosodium Iodoacetate (MIA)-induced Osteoarthritic Rats.

Effect on Gene Expression
The results of the gene expression analysis are presented in Table 5. The genes MMP-3 and MMP-7 were significantly upregulated, and the gene COL2A1 was significantly downregulated in the osteoarthritic rats. Maxicuma® at both doses significantly reversed the gene expressions seen in the osteoarthritic control rats, indicating the anti-arthritic potential of Maxicuma®. Celecoxib, the positive used in this study, significantly reversed the gene expressions seen in the osteoarthritic control rats.
Results of the Gene Expression Analysis.

Histopathology
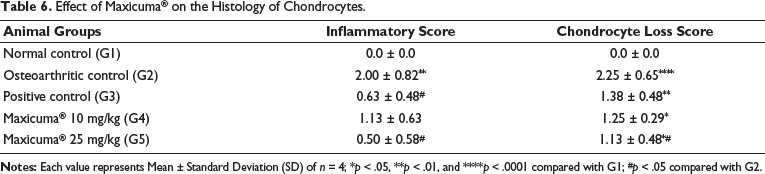
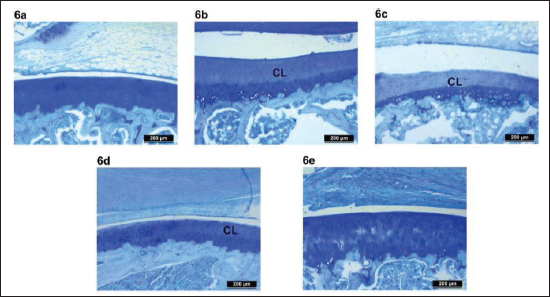
Microscopic observations of the joint cartilage tissue stained with H&E displayed inflammatory stain after H&E staining and chondrocyte losses after toluidine staining. Statistically significant higher inflammatory scores and chondrocyte loss scores were seen in the osteoarthritic rats. Maxicuma® at a 25 mg/kg bw dose significantly reduced both the inflammatory score and chondrocyte loss score compared with the osteoarthritic control rats. Celecoxib showed a significant reduction only in the inflammatory score compared with the osteoarthritic control rats (Table 6).
Effect of Maxicuma® on the Histology of Chondrocytes.
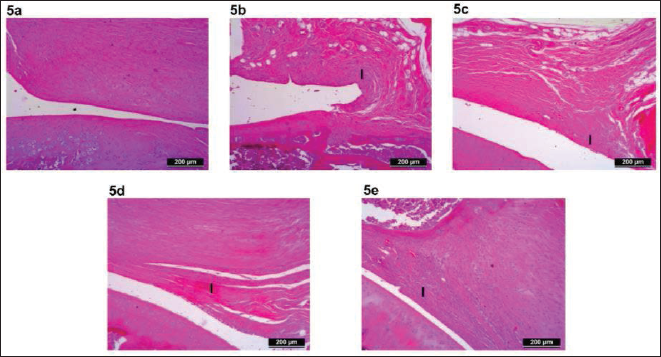
Representative images of the H&E-stained joint tissue cartilage showing inflammation are presented in Figure 5, and the toluidine-stained joint tissue cartilage showing chondrocytes are presented in Figure 6.
Discussion
In the current study, osteoarthritis was induced by intra-articular administration of MIA into the right hind limb of rats. MIA is commonly used to induce osteoarthritis in experimental models (Takahashi et al., 2018; Yoh et al., 2022). MIA works by inhibiting the activity of glyceraldehyde-3-phosphate dehydrogenase, a key enzyme in the glycolytic pathway. This inhibition leads to chondrocyte death, which in turn results in cartilage lesions and functional joint impairment. These changes closely resemble the conditions seen in human OA (Xu et al., 2020). The induction of osteoarthritis in this study was confirmed by the significant increase in the right knee joint perimeter. Administration of Maxicuma® orally at 10 and 25 mg/kg rat body weight for 28 days showed anti-osteoarthritic effect, as observed from the significant reduction in the knee joint perimeter of the affected knee joints compared with the MIA-induced osteoarthritic rats. Similar effects were reported for a high bioavailable curcumin formulation in MIA-induced osteoarthritic rats, but at doses of 100 and 200 mg/kg bw of rats for 4 weeks, which is about 10 times higher dose than that we studied here for Maxicuma® (Yabas et al., 2021). Another bioavailable curcumin formulation (Theracurmin®), containing 30% curcumin at a dose of 2,600 mg/kg bw, was reported to have reduced the knee joint thickness that was elevated by MIA-induced osteoarthritis in rats. A 95% curcuminoids extract has also been reported to have reduced joint swelling at a dose of 200 mg/kg bw for 30 days in MIA-induced osteoarthritic rats (Alwan et al., 2022). A curcumin-loaded nanoparticle at a dose of 200 mg/kg bw was reported to have reduced knee swelling but through intra-articular injections in MIA-induced osteoarthritic rats (Hamdalla et al., 2022). Results of the histopathology studies showed that Maxicuma® at a 25 mg/kg bw dose significantly reduced both the inflammatory score and the chondrocyte loss score. Similar results were reported for a bioavailable curcumin formulation (Theracurmin®) at doses of 1,300 and 2,600 mg/kg bw in MIA-induced osteoarthritic rats (Park et al., 2020).
In osteoarthritis, the proinflammatory cytokines, IL-1β and TNF-α, were found elevated, and they stimulate the production of MMPs, PGE2, COX-2, and NO, leading to joint inflammation, osteophytes formation, and cartilage degradation (Molnar et al., 2021). MMPs, including MMP-3 and MMP-7, are enzymes involved in the degradation of all components of the extracellular matrix, including collagen. High levels of these enzymes were found in osteoarthritis. PGE2, COX-2, and NO mediate inflammation of the joints in osteoarthritis (Han et al., 2018; Mehana et al., 2019). Anti-inflammatory cytokines, namely, IL-10 and IL-13, protect chondrocytes by their multimode action, including anti-apoptotic and anti-inflammatory, by stimulating type II collagen and aggrecan synthesis, and by the inhibition of MMP synthesis. IL-10 and IL-13 basically works against IL-1β and TNF-α. A balance between these proinflammatory and anti-inflammatory cytokines is essential in maintaining healthy cartilage (Jovanovic et al., 1998; Molnar et al., 2021). CRP is a marker of systemic inflammation, and increased levels of CRP have been associated with local joint inflammation in osteoarthritis (Pearle et al., 2004). COMP, an extracellular matrix glycoprotein, is responsible for the assembly and structure of the cartilage matrix. Elevated level of COMP in serum is considered a biomarker in patients with knee osteoarthritis (Udomsinprasert et al., 2024). An increase in glycosaminoglycan content is considered an indicator of cartilage loss/damage (Kulkarni et al., 2016).
Maxicuma® exhibited anti-inflammatory effect in the osteoarthritic rats by significantly decreasing the inflammatory mediators, namely, COX-2, COMP, CRP, PGE2, TNF-α, and glycosaminoglycan levels, and by increasing IL-10 in the affected knee joint tissues. These results were in line with a high-bioavailable formulation of curcumin (Next Generation Ultrasol Curcumin) that has been reported to have reduced the levels of TNF-α, COMP, and CRP and increased the level of IL-10 in the synovial tissue of rats with MIA-induced osteoarthritis but at doses of 100 and 200 mg/kg bw for 4 weeks (Yabas et al., 2021). A curcumin-loaded nanoparticle was reported to have reduced the serum levels of TNF-α and inhibited the downregulation of IL-10 in MIA-induced osteoarthritic rats at a dose of 200 mg/kg bw through intra-articular injections (Hamdalla et al., 2022). The anti-inflammatory effect of Maxicuma® was also observed in the LPS-induced mouse macrophages, as evident from the decreased levels of proinflammatory cytokines, namely, IL-1β and TNF-α, levels of COX-2, NO, and increased levels of anti-inflammatory cytokines, namely, IL-10 and IL-13. These effects were supported by the significantly lesser inflammatory score and chondrocyte loss score in the histopathology of the Maxicuma administered osteoarthritic rats. A curcumin-loaded nanoparticle was reported to have reduced the mRNA expression of inducible NO synthase in MIA-induced osteoarthritic rats at a dose of 200 mg/kg bw but through intra-articular injections (Hamdalla et al., 2022). Curcumin and its liposomal formulation were found to have inhibited NO production at 10 and 20 µg/mL concentrations, IL-1β and TNF-α at 10 µg/mL concentration in LPS-induced macrophages (RAW 264.7) (Basnet et al., 2012). Whereas, in the current study, Maxicuma® showed these effects at concentrations from 0.4 to 4 µg/mL, indicating the higher potency of Maxicuma® compared to regular curcumin and its liposomal formulation.
COL2A1 (collagen, type II, alpha 1) is an anabolic gene that encodes for type II collagen. Expression of the gene COL2A1 was found to decrease with an increase in the severity of osteoarthritis (Zhong et al., 2016). Increased expression of the anabolic gene (COL2A1) and decreased expression of the catabolic genes, namely, MMP-3 and MMP-7 in osteoarthritic rats was found upon Maxicuma® administration, and this can be attributed to the anti-osteoarthritic effect of Maxicuma®. Downregulation of the protein expression of MMP-3 and COX-2 has been reported for a curcumin formulation as well, at a dose of 100 and 200 mg/kg bw for 4 weeks in MIA-induced osteoarthritic rats (Yabas et al., 2021).
SOD, CAT, GSH, and GPx are part of the cellular antioxidant defense system that protects against oxidative stress. The antioxidant system is compromised in patients with osteoarthritis. As a result, oxidative stress is high in osteoarthritis, which is considered a major cause of chronic inflammation (Ansari et al., 2020). Maxicuma® exhibited antioxidant effect, as evident from the increased SOD activity and CAT levels in osteoarthritic rats. Similar results on the activities of SOD, CAT, and GPx were also reported by Yabas et al. (2021) for a curcumin formulation at doses of 100 and 200 mg/kg bw for 4 weeks in osteoarthritic rats. A 95% curcuminoids extract was reported to have enhanced the activities of GPx and SOD, but not CAT, at a dose of 200 mg/kg bw for 30 days in MIA-induced osteoarthritic rats (Alwan et al., 2022). A curcumin-loaded nanoparticle was reported to have reduced the activity of SOD and GSH in the serum in MIA-induced osteoarthritic rats at a dose of 200 mg/kg bw but through intra-articular injections (Mobasheri et al., 2024). Additionally, the antioxidant effect of Maxicuma® was confirmed in the LPS-induced mouse macrophage cells, where Maxicuma® enhanced the SOD activity, CAT, GSH, and GPx levels. Curcumin and its liposomal formulation were found to have enhanced SOD activity in LPS-induced macrophages (RAW 264.7) at a 10 µg/mL concentration (Jovanovic et al., 1998), whereas, in the current study, Maxicuma® enhanced SOD activity from 0.6 to 4 µg/mL concentrations.
Although Maxicuma® exhibited antioxidant, anti-inflammatory, and anti-osteoarthritic activity at both the tested doses, the 25 mg/kg bw dose of Maxicuma® was found to be better than the dose of 10 mg/kg bw in osteoarthritic rats. Notably, the balancing effect of Maxicuma® on cartilage synthesis and degradation was observed directly in the affected knee joint tissue samples, which is considered more valuable than testing in serum samples. Considering the reported studies on various curcumin formulations that have been reviewed here, we could say that Maxicuma®, having anti-osteoarthritic, anti-inflammatory, and antioxidant activities at a comparatively low dose of 25 mg/kg in a way communicates its true bioavailability and its potential in the management of osteoarthritis.
Conclusion
The anti-osteoarthritic effect of Maxicuma® was observed from the reduction of knee joint perimeter, increased expression of anabolic gene COL2A1, reduced expression of catabolic genes MMP-3 and MMP-7, and reduced glycosaminoglycan content in the MIA-induced osteoarthritic rats. The anti-inflammatory and antioxidant effects were evident from the results of the evaluations in the MIA-induced osteoarthritic rats as well as from the results of the LPS-induced mouse macrophage cells. In conclusion, Maxicuma® can be considered a potential candidate in the management of osteoarthritis on account of its anti-osteoarthritic, anti-inflammatory, and antioxidant effects. Further studies can be planned to evaluate the anti-osteoarthritic potential of Maxicuma® in clinical studies.
Footnotes
Data Availability Statement
All the data were presented in the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study is approved by the Institutional Animals Ethics Committee (IAEC) with the approval number PAL/IAEC/2022/01/01/44 on January 27, 2024.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was financially supported by Everspring Co., Ltd., #1113 Techcenter, SKn Technopark, 124 Sagimakgol-ro, Korea.
Informed Consent
Not applicable.
