Abstract
Background
Diabetic cataract, an ocular complication arising from diabetes, can lead to damage to ocular tissues, particularly affecting the lens and causing symptoms such as blurred vision.
Objectives
This work aimed to demonstrate the therapeutic effects of Dendrobium extract (DE) on pathogenic lens alterations in diabetic cataract patients and an ocular complication arising from diabetes, and to comprehensively assess its in vitro and clinical research outcomes.
Materials and Methods
Initially, a diabetic cataract cell model was established using high glucose (HG)-induced human lens epithelial cells (HLEB3). The cells were categorized into three groups: the untreated control (UC) group, the HG treatment group, and the HG+DE treatment group. Apoptosis status, reactive oxygen species (ROS), superoxide dismutase (SOD), as well as Bcl-2, Bax, and caspase-3 within the cells, were comparatively analyzed to evaluate the impact of DE on diabetic lens epithelial cells. Subsequently, a cohort of 60 diabetic cataract patients were recruited and randomly allocated into Group C (control group, 30 cases), which received conventional ophthalmic medications and blood glucose control, and Group E (experimental group, 30 cases), which received the DE intervention in addition to the Group C regimen. A comparative assessment of visual acuity, intraocular pressure (IOP), and cataract grading before and after treatment was conducted between the two groups to evaluate the therapeutic efficacy of DE on patient lenses.
Results
In the in vitro study, the HG+DE group exhibited a drastically inferior apoptotic rate compared to the HG group (.05). The production of ROS in the HG+DE group was drastically inferior, and the SOD levels were markedly superior to those in the HG group (p < .05). The HG group and HG+DE group showed a greater decrease in Bcl-2 protein levels and a greater increase in Bax and caspase-3 protein levels versus the UC group (p < .05). Moreover, the HG+DE group displayed notably elevated Bcl-2 protein levels and greatly reduced Bax and caspase-3 protein levels versus the HG group (p < .05). The in vivo study demonstrated that Group E patients exhibited markedly improved visual acuity recovery versus Group C patients (p < .05), along with more stable IOP. The cataract grading of Group E patients was remarkably superior to that of Group C patients (p < .05).
Conclusion
DE may contribute to ameliorating ocular lesions in diabetic cataract patients, mitigating lens damage, and could potentially emerge as a promising therapeutic option.
Introduction
Diabetic cataract, an ocular complication arising from diabetes, is characterized by prolonged hyperglycemia-induced damage to ocular tissues, particularly the lens, resulting in diminished lens transparency and the emergence of blurred and hazy visual perceptions (Greenberg & Bamba, 2021; Xia, 2022). The progression of diabetic cataracts typically occurs gradually, and as the condition advances, patients may progressively experience symptoms such as blurred vision, photophobia, and diminished color perception (Mrugacz et al., 2023). Timely intervention can avert severe visual impairment, thereby preserving the quality of life for affected individuals (Mhaibes & Ali, 2024).
Dendrobium nobile Lindl. (Dendrobium), a member of the Orchidaceae family is extensively utilized in traditional Chinese medicine (Guo et al., 2020). Dendrobium extract (DE), a natural botanical extract derived from Dendrobium, encompasses a diverse array of bioactive constituents, including polysaccharides, flavonoids, alkaloids, and organic acids (Wang, 2021). Owing to its rich profile of bioactive components, DE has garnered significant attention. Research has elucidated a multifaceted pharmacological spectrum of DE, encompassing antioxidant, anti-inflammatory, immunomodulatory, anti-cancer, and neuroprotective effects (Xu et al., 2023; Zhan et al., 2023; Zhang et al., 2020). These bioactive constituents contribute to the maintenance of human health, bolstering immunity, disease resistance, and conferring beneficial effects on the cardiovascular, digestive, and nervous systems (Chen et al., 2023; Gao et al., 2023; Zhang et al., 2022). Within the realm of traditional Chinese medicine, Dendrobium is traditionally employed for its purported effects in clearing heat, detoxification, nourishing Yin, and moistening the lungs, as well as promoting gastric health and generating body fluids (Feng et al., 2022; Qi et al., 2022). Furthermore, DE finds wide-ranging applications in dietary supplements and cosmetic products due to its moisturizing, antioxidant, and anti-aging properties for the skin (Nie et al., 2020). Notably, Dendrobium polysaccharides within the extract exhibit diverse biological activities, encompassing antioxidant, anti-inflammatory, and immunomodulatory effects, rendering them a potential natural substance for the treatment of cataracts (Fan et al., 2023). Research indicates that Dendrobium polysaccharides can mitigate lens oxidative stress and inflammatory responses, impeding the progression of lens opacification. As a result, they hold promise for playing a pivotal role in the prevention and treatment of cataracts (Xu et al., 2022). Additionally, Dendrobium polysaccharides contribute to maintaining ocular tissue health, fostering lens metabolism and repair, thereby potentially enhancing vision and reducing the incidence of other ocular complications (Ling et al., 2022; Yang et al., 2022).
This work was to demonstrate the therapeutic effects of DE on lens alterations in diabetic cataract patients, comprehensively evaluating its efficacy through both in vitro and clinical investigations. In the in vitro experiments, a diabetic cataract cell model was established through high glucose (HG) induction of lens epithelial cells, followed by intervention with DE to elucidate its impact on the diabetic cataract cell model. Subsequently, diabetic cataract patients were recruited for an in vivo study to examine the therapeutic effects of DE on patient lenses.
Materials and Methods
Cell Culture
Human lens epithelial cells (HLEB3) obtained from Shanghai Guandao Biotechnology Co., Ltd., China, were utilized. The purity of the cell line, confirmed through immunofluorescent detection of beaded filament structural protein 2 (BFSP2), exceeded 90%. Moreover, the cells were certified as devoid of contaminants such as human immunodeficiency virus-1 (HIV-1), hepatitis B virus (HBV), hepatitis C virus (HCV), mycoplasma, bacteria, yeast, and fungi, rendering them suitable for experimental purposes. Thawed frozen cells were promptly placed in a 37°C water bath and gently agitated to ensure complete thawing. Subsequently, the cells were centrifuged at 1,000 rpm for 5 min at 25°C using phosphate-buffered saline (PBS) (Thermo Fisher Scientific, USA), followed by three consecutive washes with PBS. Subsequently, 1 mL of culture medium with 10% fetal bovine serum (FBS) (Ausbian, Australia) was gently pipetted and transferred into a 25 cm2 culture flask, supplemented with 4 mL of culture medium. The mixture was gently agitated again and then cultured at 37°C in a 5% CO2 incubator (Kenton Instrument Co., Ltd., China). Approximately 24 h later, upon observing cell adhesion and growth along the flask walls, the culture medium was replaced. For experimental purposes, cells were subcultured normally when reaching a density of 70%–80%.
Construction and Grouping of Lens Epithelial Cell Model of Diabetes
HLEB3 cells in the logarithmic growth phase (LGF) were enzymatically digested with pancreatin (Sichuan Deebio Pharmaceutical, China) and subsequently dissociated into a single-cell suspension through gentle pipetting. Cell suspension density was determined using a hemocytometer (Sigma–Aldrich, USA), and cells were subsequently diluted to the desired density of 4 × 105 cells/9 mL. The experimental design encompassed three groups: the untreated control (UC) group, the HG treatment group, and the HG+DE group, each consisting of six replicates. Cells were seeded into 96-well cell culture plates (Sigma–Aldrich, USA) at 4,000 cells/well, then cultured for 6–12 h. Upon cell adhesion, the following interventions were applied: the UC group received no treatment, the HG group was exposed to minimum essential medium (MEM) culture (Sigma–Aldrich, USA) medium supplemented with glucose at a concentration of 30 mmol/L (150 µL), and the HG+DE group received MEM culture medium containing glucose at 30 mmol/L (150 µL) along with 40 mmol/L DE (Fufeng Sinuote Biotechnology Co., Ltd., China). The 40 mmol/L concentration of DE used in the cell experiments was determined based on the optimal concentration identified from preliminary experimental results. Subsequently, cells were incubated at 37°C with 5% CO2 for 24 h.
Detecting Cell Apoptosis
HLEB3 cells in LGF were seeded at 2.5 × 105 cells/well in 24-well plates (Sigma–Aldrich, USA) containing glass coverslips. After 24 h cultivation, the original medium was discarded, and cells were subjected to respective experimental treatments. Following an additional 48 h of incubation, the original medium was replaced, and coverslips were retrieved. Cells were fixed with a mixture of methanol [TCI (Shanghai) Chemical Industry Development Co., Ltd., China]/glacial acetic acid (Purex Chemical Co., Ltd., China) for 5 min, and subsequently stained with Hoechst 33258 (Sigma–Aldrich, USA) by gently resuspending cells, followed by incubation at 25°C in the absence of light for 1 h. Following cell washing with PBS, cells were treated with a quenching fluorescence working solution (Beyotime Biotechnology Co., Ltd., China) before mounting onto slides. Cell staining was visualized under a fluorescence microscope (Leica, Germany), where viable cell nuclei appeared blue, whereas apoptotic cell nuclei appeared white. Five random fields were selected for apoptotic cell counting, and the apoptotic cell percentage was calculated.
Detection of Indicators Related to Cellular Oxidative Damage
Fluorescence microscopy observation was implemented. The in situ generation of intracellular reactive oxygen species (ROS) in HLEB3 cells in LGF was qualitatively observed using the ROS fluorescent probe H2DCFDA, also known as dichloro-dihydro-fluorescein diacetate (DCFH-DA) (AbMole, USA), under a fluorescence microscope. DCFH-DA was diluted in a 1:1,000 ratio using low-glucose Dulbecco’s modified Eagle medium (DMEM) culture medium (Thermo Fisher Scientific, USA). The diluted DCFH-DA was gently pipetted and mixed, and the original culture medium was aspirated from the 6-well culture plates (Sigma–Aldrich, USA). Cells were then washed twice with serum-free DMEM to remove residual serum. Subsequently, 2 mL of the diluted DCFH-DA solution was applied and incubated in a cell culture incubator for 20 min, with gentle agitation every 5 min to ensure optimal probe-cell interaction. Following this, cells were rinsed three times with serum-free low glucose DMEM under subdued light conditions to thoroughly remove extracellular DCFH-DA. Finally, 2 mL of serum-free DMEM was applied, and cell fluorescence was visualized under a fluorescence microscope.
An enzymatic assay of activity was performed. HLEB3 cells in LGF were subjected to digestion using 0.25 g/L pancreatin. According to the instructions provided with the superoxide dismutase (SOD) assay kit (Assay Genie, Ireland), cell disruption was achieved using an ultrasonic disruptor (Thermo Fisher Scientific, USA) set at 200 A current, with each ultrasound burst lasting 5 s followed by a 5-s interval, repeated 10 times, to disrupt the cells. The resulting cell lysate was subsequently centrifuged, and the supernatant was aspirated for determination of SOD activity employing an enzyme-linked immunosorbent assay (ELISA) reader, following the manufacturer’s instructions. This process was performed in triplicate, and the results of the three replicates were averaged.
Detection of Bcl-2, Bax, and Caspase-3
Western blotting was conducted to assess Bcl-2, Bax, and caspase-3 protein levels in various cell groups. Cell culture media was aspirated, and cells were lysed with cell lysis buffer (Sartorius, Germany) to release proteins. The resulting cell lysates were collected, and protein concentrations were determined. Protein samples were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis to separate proteins by size. Subsequently, proteins were transferred onto a polyacrylamide gel membrane. The rabbit monoclonal antibodies Bcl-2 (1:2,000), Bax (1:5,000), caspase-3 (1:5,000), and mouse monoclonal antibody β-actin (1:10,000) were added and incubated overnight at 4°C. Subsequently, a horseradish peroxidase-conjugated goat anti-human IgG antibody (1:10,000) was applied, and the samples were incubated at room temperature for 2 h. Chemiluminescent or fluorescent imaging systems (Shanghai Licai Biotechnology Co., Ltd., China) were employed to capture and record images of the target proteins, and software was used for protein band analysis.
Real-time quantitative polymerase chain reaction (PCR) technology was employed to quantify Bcl-2, Bax, and caspase-3 messenger ribonucleic acid (mRNA) levels in various cell groups. Total ribonucleic acid (RNA) was extracted from various cell groups using an RNA extraction kit (Nanjing ATG Biotechnology Co., Ltd., China).
Research Subjects
A total of 60 diabetic cataract patients undergoing ophthalmic treatment in the hospital from March 2021 to September 2022 were recruited as the study subjects. This work received approval from the hospital’s ethics committee, and informed consent was obtained from the participants.
Inclusion Criteria
Participants were required to meet the following criteria: (a) age ≥ 18 years with a confirmed history of diabetes; (b) diagnosed with cataract and not meeting surgical indications (corrected visual acuity < 0.3); (c) duration of both diabetes and cataract diagnosis exceeding 6 months; (d) willingness to voluntarily participate in the study and provide informed consent.
Exclusion Criteria
Participants were excluded if they were with (a) the presence of severe concurrent ocular disorders, such as macular degeneration or glaucoma; (b) the concurrent presence of severe systemic diseases, including cardiovascular or renal conditions; (c) excessive use of medications or alcohol abuse; (d) severe cognitive impairment or intellectual disabilities; (e) pregnant or lactating females.
Grouping and Treatment
Sixty patients were randomly and equally assigned into Group C (control group) and Group E (experimental group).
Group C received the following therapies: (a) Ocular treatment: patients received Pirenoxine sodium eye drops (Hubei Yuanda Tiantianming Pharmaceutical Co., Ltd., China) at 4 to 6-h intervals, with 1–2 drops instilled into the conjunctival sac. During the initial 1–2 days, the dosage could be increased to every 2 h. Zhenshiming eye drops (Jiangxi Zhenshiming Pharmaceutical Co., Ltd., China) were administered 3–5 times per day, at 1–2 drops per time, with potential dosage adjustments as needed. (b) Blood glucose control: metformin hydrochloride (Shanghai Squibb Pharmaceutical Co., Ltd., China) was administered once daily, with one tablet (500 mg) taken during dinner. Dosage was adjusted based on blood glucose and urinary sugar levels, with a maximum daily dose not exceeding 4 tablets. Acarbose tablets (Bayer Healthcare Co., Ltd., China) were taken at 50 mg per dose (1 tablet), three times daily, and gradually increased to 0.1 g per dose (2 tablets) taken three times daily or adjusted as per medical advice. Ginkgo leaf capsules (Wuhu Luye Pharmaceutical Co., Ltd., China) were taken at 1–2 capsules per dose, three times daily, or adjusted as per medical advice. The treatment duration was 2 months. The 2 g per dose, twice daily dosage of Dendrobium dry powder used in the clinical trials was based on the recommended dosage provided in the product’s official instructions.
Group E received the following therapies: in addition to the treatment received by Group C, patients in Group E were administered Dendrobium dry powder (Liuankang Shuntang Dendrobium Co., Ltd., China) treatment was accepted orally, twice daily, at 2 g per dose. The treatment duration was 2 months, consistent with Group C.
Efficiency Evaluation
Visual acuity assessment was conducted at baseline, and at 2-, 4-, 6-, and 8-weeks post-treatment. The patient’s best corrected visual acuity (BCVA) was recorded using a standard Snellen visual acuity chart. The BCVA was noted based on the smallest line of letters that the patient could identify. The measured BCVA was then converted to Logarithm of the Minimum Angle of Resolution (LogMAR) units using logarithmic calculations.
Intraocular pressure (IOP) evaluation was performed at baseline and at 2-, 4-, 6-, and 8-weeks post-treatment. Non-contact tonometry was employed to measure and record the patient’s IOP.
Cataract grading assessment was carried out at baseline and at the end of the treatment period. Patients’ anterior segments were observed under a slit-lamp microscope to determine the cataract grade. The grading criteria were as follows: no cataract (transparent lens without cataract signs); mild cataract (slight lens opacity, minimal impact on disc clarity); moderate cataract (increased lens opacity, slight impact on disc clarity); and severe cataract (distinct lens opacity, significant impact on disc clarity, notable decrease in visual acuity).
Statistical Methods
All experimental data were subjected to statistical analysis using statistical package for the social sciences (SPSS) 19.0. Data were denoted as Mean ± Standard Deviation. Differences between two independent samples were compared using Student’s t-test, while differences among multiple groups were assessed using one-way analysis of variance. p < .05 indicated statistically significant differences.
Results
Cell Apoptosis Detection Results
The detection results revealed that after 24 and 48 h of incubation, the apoptotic rates of the UC group cells were 1.44% ± 0.52% and 1.84% ± 0.63%, respectively. In the HG group, the apoptotic rates were 8.33% ± 2.07% and 11.36% ± 3.34%, respectively, while in the HG+DE group, the apoptotic rates were 3.29% ± 1.05% and 5.14% ± 1.93%, respectively. In comparison, both HG and HG+DE groups exhibited a great increase in apoptotic rates versus the UC group (p < .05), with the HG group showing substantially superior apoptotic rates to the HG+DE group (p < .05) (Figure 1).
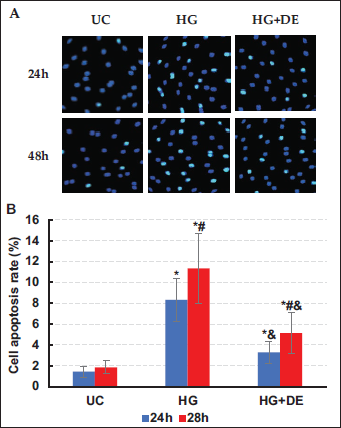
Evaluation Results of Cell Oxidative Damage Indicators
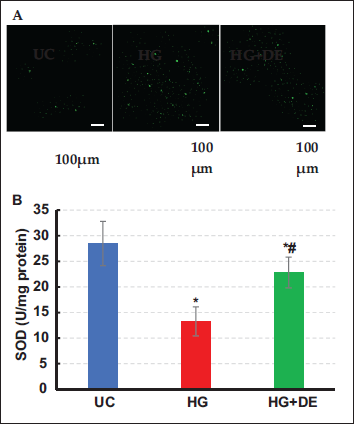
Under fluorescence microscopy, it was observed that even under normal conditions, cells produce a small amount of ROS. However, comparatively, both the HG group and the HG+DE group exhibited substantially superior levels of ROS production to the UC group. Additionally, the ROS production in the HG group was notably higher than in the HG+DE group (Figure 2A). The SOD activities of cells in the UC group, HG group, and HG+DE group were measured as 28.47 ± 4.33 U/mg protein, 13.26 ± 2.85 U/mg protein, and 22.84 ± 3.01 U/mg protein, respectively. Among the three groups, the SOD activity in the HG group was drastically inferior to the other two groups (p < .05), with the UC group exhibiting the highest SOD activity (Figure 2B).
Comparison of Oxidative Damage Indicators. (A) Reactive Oxygen Species (ROS) Microscope Observation Results (×400); (B) Superoxide Dismutase (SOD) Activity Detection Results.
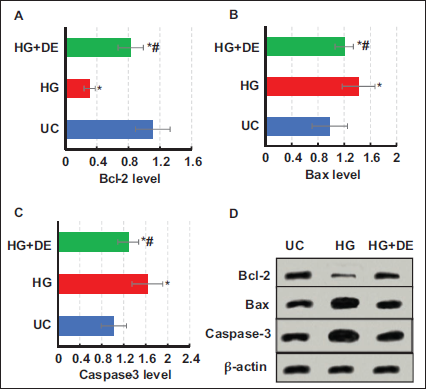
Cell Bcl-2, Bax, and Caspase-3 Level Detection Results
The analysis indicated that, versus the UC group, Bcl-2 protein levels were greatly reduced, while those of Bax and caspase-3 proteins were markedly increased in both HG and HG+DE groups (p < .05). Moreover, in Figure 3, the HG+DE group exhibited substantially superior Bcl-2 protein levels to the HG group, along with drastically inferior levels of Bax and caspase-3 proteins (p < .05).
Therapy Efficiency Evaluation
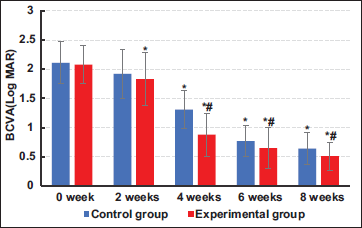
Vision Test Results
The results of visual acuity assessment are presented in Figure 4. The BCVA (LogMAR) values of both patient groups gradually decreased over time, with those in Group E showing a drastic reduction versus Group C starting from the 4th week of treatment (p < .05).
Eye Pressure Test Results
The results of IOP measurements at various stages are depicted in Figure 5. After 2 weeks of treatment, both patient groups exhibited a considerable increase in IOP (p < .05). Following the 2-week treatment period, patients in Group E demonstrated a relatively stable IOP, while those in Group C exhibited more pronounced fluctuations, with considerable differences versus the previous assessment (p < .05).
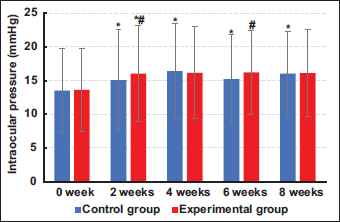
Cataract Grading Results
No considerable difference existed in the pre-treatment cataract grading between the two patient groups (p > .05). At the end of the treatment period, the improvement in cataract status was greatly better in Group E versus Group C (p < .05) (Table 1).
Comparison of Cataract Grading Among Patients.
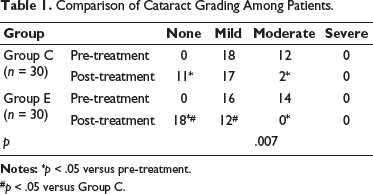
#p < .05 versus Group C.
Discussion
Diabetic cataracts, a common complication of diabetes, can lead to damage to ocular tissues, particularly affecting the lens and causing symptoms such as blurred vision. Apoptosis, oxidative stress, and inflammatory levels of lens epithelial cells are closely associated with the development of diabetic cataracts. Under prolonged hyperglycemic conditions, lens epithelial cells are prone to apoptosis, while oxidative stress and inflammatory reactions exacerbate, thereby accelerating the progression of diabetic cataracts. DE, rich in polysaccharides, flavonoids, and other active ingredients, exhibits various pharmacological effects, including antioxidation and anti-inflammation, contributing to the improvement of the lens and maintenance of ocular tissue health. This study initially investigated the impact of DE on apoptosis and oxidative stress in HLEB3 through in vitro experiments.
Apoptosis analysis revealed a significant increase in the apoptotic rate of cells in the HG group compared to the HG+DE group (p < .05), indicating that under HG conditions, lens epithelial cells undergo more apoptosis, signifying an adverse effect of HG on cells. Consistent with our findings, a study by Bai et al. (2022) also demonstrated a significant increase in lens cell apoptosis in a diabetic cataract rat model induced by HG, corroborating our results. Moreover, the apoptotic rate of cells in the HG group was significantly higher than that in the HG+DE group (p < .05), suggesting a potential protective effect of DE, which may mitigate the damage to lens epithelial cells induced by HG environments, thereby reducing the occurrence of cell apoptosis. Building upon these observations, the study further investigated changes in the levels of Bcl-2, Bax, and caspase-3. Bcl-2, an anti-apoptotic protein, primarily acts by regulating mitochondrial membrane permeability to counter cell apoptosis. A decrease in Bcl-2 levels indicates a downregulation in anti-apoptotic capability (Arulananda et al., 2021). This study revealed that compared to Group C, the levels of Bcl-2 protein were significantly reduced in cells of both the HG and HG+DE groups, with the HG+DE group exhibiting significantly higher Bcl-2 protein levels than the HG group (p < .05). This suggests that in an HG environment, the addition of DE can partially counteract the inhibitory effect on Bcl-2 levels, thereby aiding in maintaining the cells’ anti-apoptotic capability. Bax is a pro-apoptotic protein that primarily functions by increasing mitochondrial membrane permeability, leading to cell apoptosis (Spitz & Gavathiotis, 2022). The study found that compared to Group C, cells in both the HG and HG+DE groups showed a significant increase in Bax protein levels (p < .05), indicating that HG environments may enhance Bax expression, thereby increasing the likelihood of cell apoptosis. Moreover, the Bax protein levels in the HG+DE group were lower than those in the HG group (p < .05), suggesting that the addition of DE could mitigate the stimulation of Bax expression induced by HG environments, thereby reducing the risk of cell apoptosis. Caspase-3 is a key executor enzyme in the process of cell apoptosis, and its activation is a crucial step in cell apoptosis (Beroske et al., 2021). The study found that compared to Group C, both the HG and HG+DE groups showed a significant increase in caspase-3 protein levels (p < .05), indicating that HG environments triggered the activation of caspase-3, thereby promoting cell apoptosis. Furthermore, the caspase-3 protein levels in the HG+DE group were lower than those in the HG group (p < .05), suggesting that the addition of DE could inhibit the activation of caspase-3 induced by HG environments, thereby reducing the occurrence of cell apoptosis. Taken together, DE may modulate the expression and activity of Bcl-2, Bax, and caspase-3, thereby influencing cell apoptosis and inhibiting cell apoptosis induced by HG environments, ultimately exerting effects against the progression of diabetic cataracts.
Oxidative damage reveals that, under normal conditions, there exists a small amount of ROS within cells. ROS, a class of highly reactive oxidative molecules, is generated through cellular redox reactions involving oxygen and participates as signaling molecules in regulating many important biological processes within cells, such as cell proliferation, cell signaling, and apoptosis (Tripathi et al., 2022). Moderate ROS production is a part of normal cellular metabolism and is also an essential mechanism used by the immune system to combat pathogens; hence, a small amount of ROS can be observed in normal cells. ROS plays a crucial role within cells, maintaining internal balance and regulating the cell cycle. However, when ROS production exceeds the cell’s clearance capacity, it may lead to oxidative stress, disrupting the cellular environment, causing damage to cell membranes, proteins, deoxyribonucleic acid (DNA), and so on, and even triggering cell apoptosis, thus causing damage to cells and tissues (Ariestanti et al., 2024; Jing et al., 2022). This study found that cells in both the HG and HG+DE groups produced significantly more ROS compared to Group C, indicating that an HG environment may induce increased oxidative stress within cells. However, the HG group exhibited significantly higher ROS levels than the HG+DE group, confirming the potential inhibitory effect of DE on ROS production. SOD is an important antioxidant enzyme that helps maintain cellular redox balance and protects cells from the harmful effects of ROS (Gulcin, 2020). The study revealed that SOD activity in cells of the HG group was significantly lower than that in the other two groups, indicating that an HG environment suppressed SOD activity, thereby reducing the cells’ resistance to oxidative stress. In comparison to the HG group, cells in the HG+DE group showed increased SOD activity, suggesting that DE may enhance SOD activity to counteract oxidative stress induced by HG environments. Previous studies showed similar findings, indicating that Dendrobium can enhance SOD activity and alleviate oxidative stress, consistent with the results of our study (Han et al., 2021; Horng et al., 2021; Hsu et al., 2022; Li et al., 2022; Long et al., 2023).
Based on in vitro studies, further in vivo experiments were conducted. Visual acuity testing revealed that patients in Group E exhibited a faster improvement in vision after treatment, with their BCVA (LogMAR) values significantly lower than those in Group C. BCVA is a commonly used clinical indicator to assess visual acuity clarity, represented in LogMAR, where smaller values denote better vision (Holbach et al., 2020). In cases of lens opacities, due to inherent lens issues, the refractive function of the optical system is affected, leading to decreased visual acuity. Consequently, lens opacities typically result in increased BCVA (LogMAR) values, indicating poorer vision (Moshfeghi et al., 2020). Combining the findings of this study, it can be concluded that DE contributes to improving visual acuity in patients with lens opacities. Results from IOP measurements at various stages showed minimal fluctuations in IOP among patients in Group E during treatment. IOP serves as a crucial indicator of intraocular fluid balance, and its maintenance within normal ranges generally supports the stability of ocular structures and normal visual function (Alkozi et al., 2020). In conditions such as cataracts and other lens opacities, the structure and transparency of the lens itself may be compromised, potentially impeding intraocular fluid circulation and causing fluctuations in IOP (Schuknecht et al., 2022). As an herbal medicine, DE has been reported to possess anti-inflammatory and antioxidant properties, which may help alleviate ocular inflammation and oxidative stress, thereby maintaining intraocular fluid balance and reducing fluctuations in IOP (Panuthai et al., 2023). Thus, DE appears to contribute to maintaining intraocular fluid balance. Furthermore, cataract grading plays a significant role in ophthalmic clinical practice, serving as a common tool to classify the degree and severity of lens opacity in cataract patients (Mirzaie et al., 2022). The study analyzed patient improvement based on cataract grading. Results indicated that post-treatment, patients in Group E showed significantly better improvement in cataract grading compared to those in Group C. This may be attributed to the antioxidative and anti-inflammatory effects of DE, which help protect lens cells and improve lens structure and function. However, it is important to note that individual variations exist in the effectiveness of DE in treating cataracts, and therapeutic effects may require a longer time to manifest. In summary, DE may serve as a potential therapeutic option, aiding in improving visual acuity and ocular health in patients with lens opacities.
However, this study has certain limitations. The sample size included is relatively small, the patient population is homogeneous, and the study duration is relatively short. Moreover, the specific components and pharmacological effects of DE were not extensively explored, and a comprehensive understanding of its therapeutic mechanism is lacking. Given the limitations of the current study, future research should involve larger-scale, long-term, multicenter randomized controlled trials to validate the safety and efficacy of DE in the treatment of diabetic cataracts. Additionally, further investigation is needed to identify the active components of DE, as well as to explore its effects on lens cells and the associated signaling pathways.
Conclusion
In in vitro experiments, DE has been shown to protect lens cells by inhibiting apoptosis and alleviating oxidative stress. Clinical studies demonstrated that diabetic cataract patients receiving DE intervention exhibited superior visual recovery and more stable IOP after treatment. Overall, DE shows potential therapeutic effects in diabetic cataract patients and can be considered a promising traditional Chinese medicine for treating diabetic cataracts. With advancements in scientific research and technology, we hope to gain a deeper understanding of the underlying mechanisms of DE and provide more effective and safer treatment options for cataract patients in the future.
Footnotes
Abbreviations
BFSP2: Beaded filament structural protein 2; DE: Dendrobium extract; DMEM: Dulbecco’s modified Eagle medium; ELISA: Enzyme-linked immunosorbent assay; FBS: Fetal bovine serum; HBV: Hepatitis B virus; HG: High glucose; HIV-1: Human immunodeficiency virus-1; HLEB3: Human lens epithelial cells; IOP: Intraocular pressure; LGF: Logarithmic growth phase; MEM: Minimum essential medium; PBS: Phosphate-buffered saline; ROS: Reactive oxygen species; SOD: Superoxide dismutase; UC: Untreated control.
Authors Contribution
Baoping Duan and Ping Liu: Conception and study design; Hongyan Ge and Meng Sun: Data acquisition.
Sheng Su and Shiqiang Zhao: Data analysis.
Baoping Duan and Ping Liu: Manuscript draft, editing, and revision.
All authors wrote and approved the final manuscript.
Availability of Data and Material
All data generated during this study are included in this published article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
All experimental procedures in this study follow the guidelines of the Research Ethics Committee of The First Affiliated Hospital of Harbin Medical University.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
