Abstract
Background
Periodontitis is a persistent inflammatory condition that mainly affects the supportive tissues of the teeth, comprising the gingiva, periodontal ligament, and alveolar bone.
Objectives
The present study focused on addressing fisetin’s therapeutic activities against the experimental periodontitis model in rats.
Materials and Methods
Experimental periodontitis was initiated in rats by ligation procedure. The fisetin treatment was given with three different dosages for 10 days, and finally, rats were sacrificed on the 11th day, and subsequently, samples were collected for additional assays. The extent of periodontal damage was assessed by quantifying alveolar bone loss (ABL). The inflammatory and osteoblast cell quantities were enumerated on stained tissue slides. The concentrations of oxidative stress biomarkers, bone mineral contents, inflammatory cytokines, glucose, and glycated hemoglobin (HbA) levels in the experimental rats were assessed using commercial kits. The bone morphogenetic protein-2 (BMP-2), matrix metalloproteinase-8 (MMP-8), tissue inhibitor of metalloproteinase-1 (TIMP-1), receptor activator of nuclear factor kappa-Β ligand (RANKL), and osteoprotegerin (OPG) concentrations were assessed using commercially procured kits.
Results
The results of this study demonstrated that fisetin treatment successfully reduced ABL, osteoclast, and inflammatory cell counts and increased osteoblast cell numbers in the rats with periodontitis. The levels of glucose and HbA1c were reduced, and bone mineral levels were elevated by fisetin in periodontitis-induced rats. The contents of oxidative stress biomarkers, inflammatory cytokines, inducible nitric oxide synthase, and MMP-8 were effectively reduced in the fisetin-treated rats with periodontitis. Furthermore, the fisetin treatment successfully ameliorated the pathological changes induced by periodontitis in rats by regulating BMP-2/TIMP-1 and OPG/RANKL expressions.
Conclusion
These results highlight that fisetin treatment successfully reduced ABL and promoted periodontal tissue regeneration in rats with experimental periodontitis. Therefore, it can be a talented salutary candidate to treat periodontal diseases in the future.
Introduction
Periodontitis is a persistent inflammatory disease that primarily affects the supportive tissues of the teeth, comprising the gingiva, periodontal ligament, and alveolar bone. Periodontitis is characterized by the progressive destruction of the periodontal tissues, ultimately resulting in tooth loss (Takedachi et al., 2022). The global prevalence of periodontitis is quite high, with estimates suggesting that nearly 50% of the population is affected by this condition. Moreover, the severe form of periodontitis is reported to affect around 11% of the global population, accounting for approximately 734 million people worldwide. Factors such as age, socio-economic status, and geographic location have been found to influence the prevalence of periodontitis (Dionigi et al., 2022). The pathophysiology of periodontitis is complex and encompasses a combination of bacterial, host, and environmental factors. The disease process begins with bacterial plaque, which initiates inflammation in the gingival tissues. The uncontrolled inflammatory response can destroy the periodontal ligament and alveolar bone (Sczepanik et al., 2020). The chronic inflammatory state connected with periodontitis can result in the release of inflammatory mediators, potentially participating in the development of these systemic conditions (Veljovic et al., 2022).
The current treatment approaches for periodontitis, which primarily involve mechanical debridement and the utilization of non-steroidal anti-inflammatory drugs (NSAIDs), have shown limited long-term success. The side effects associated with chronic NSAID use have further emphasized the need for alternative therapies that can effectively manage the disease while minimizing adverse effects. While current treatments aim to control the infection and inflammation, they often fail to provide long-term solutions, highlighting the need for innovative approaches (Perez-Pacheco et al., 2021). In this context, plant bioactive compounds have appeared as a hopeful alternative, offering practical therapeutic benefits to treat periodontitis. These natural compounds, obtained from plant sources, have shown antimicrobial, anti-inflammatory, and anti-adhesive activities, making them a compelling option for addressing the multifaceted challenges associated with this disease (Bhattarai et al., 2019). Incorporating plant bioactive compounds into the management of periodontitis holds significant promise in addressing the shortcomings of current treatment approaches. By targeting multiple pathological pathways, these natural compounds offer a holistic approach to managing this complex disease, potentially leading to improved clinical results and quality of life for patients (Wei et al., 2021).
Fisetin is a naturally occurring bioactive flavonoid compound that has been shown to hold various bioactive properties. It is prevalent in numerous fruits and vegetables, including cucumber, persimmon, strawberry, and apple (Yamaura et al., 2022). Fisetin exhibits several biological actions, including amelioration of pulmonary disease (Hussain et al., 2019), nephroprotective (Sahu et al., 2014), attenuation of metabolic dysfunction (Shi et al., 2018), anti-inflammatory (Molagoda et al., 2021), cardioprotective (Li et al., 2022), antioxidant (Khan et al., 2013), anti-cancer (Kumar et al., 2023), and neuroprotective (Hassan et al., 2022) properties. Apart from these biological properties, fisetin’s salutary roles against periodontitis have not been scientifically assessed yet. Hence, the present study focused on addressing fisetin’s therapeutic activities against the experimental periodontal disease model in rats.
Materials and Methods
Chemicals
The primary chemicals and reagents, including fisetin, anesthetics xylazine, ketamine, and other chemicals employed in this work, were obtained commercially from Sigma-Aldrich, USA. The assay kits for assessing biochemical markers were obtained from Elabscience, CusaBio, and LSBio, USA, respectively.
Experimental Rats
The current investigation employed healthy male Wistar rats, aged 6–8 weeks, with an approximate weight of 150 ± 40 g. The rats were housed in a regulated laboratory setting for 7 days. The animals were caged in sterile polypropylene enclosures. Rigorous hygiene standards were maintained throughout the research period, and cages and bedding were routinely substituted. Standard temperature and relative humidity were consistently maintained, and a 12-h light/dark sequence was implemented. The protocols for the animal assays were verified and sanctioned by the institutional animal ethics committee.
Induction of Experimental Periodontitis
Periodontitis was initiated by a ligation procedure. Sterilized nylon thread sutures were positioned around the cervix of the maxillary left second molar tooth. A well-experienced physician conducted all experimental procedures. Anesthesia was administered using ketamine (10%) and xylazine (2%). The sutures were securely ligated and subsequently verified post-application. The contralateral side without ligature functioned as the control group (no periodontitis induction and treatments).
Treatment Groups
The rats were assigned into four groups, each comprising six rats (n = 6). Group I consisted of normal control rats only administered a standard pellet diet and excluded from the ligature procedure. Rats in Group II were induced with periodontitis by ligation procedure. Groups III–V rats underwent periodontitis induction using a ligation procedure and were treated with 5, 10, and 15 mg/kg of fisetin, respectively, via oral gavage route 1 h before induction and continued daily for 10 days. Upon completion of the treatment regimen, rats were anesthetized on the 11th day, sacrificed, and samples were collected for additional assays.
Analysis of Alveolar Bone Loss (ABL)
The maxillae were removed from the experimental rats and preserved in 10% formalin. Subsequently, the maxillary halves were defleshed and stained with 1% methylene blue to distinguish bone from teeth. The maxillae were subsequently fixed in wax, and images were taken. The photos were processed using imaging software (ImageJ 1:48), and ABL was quantified.
Histopathological Analyses
The removed maxillae underwent demineralization in a 7% nitric oxide (NO) solution, were dehydrated, and fixed in paraffin. The samples were stained using Mallory’s trichrome stain. Sections of 6 µm in thickness, corresponding to the region between the first and second molars, were examined using microscopy. Each sample was assessed using three slides. The average of these measurements was documented. Osteoblasts and inflammatory cells were quantified in stained slides. Osteoclast cells were enumerated following the visualization of tartrate-resistant acid phosphatase (TRAP)-positive cells by enzyme histochemistry. Cuboidal osteoblasts located on the alveolar bone surface next to the periodontal ligament were identified as active osteoblasts and enumerated. Inflammatory cells, including neutrophils, lymphocytes, eosinophils, and macrophages, were similarly enumerated. All histology procedures were conducted by a seasoned researcher who was blind to the study groups.
Analysis of Bone Minerals and TRAP Levels
The concentrations of calcium (Ca), phosphorus (P), creatinine (Cr), and TRAP in the serum of experimental rats were assessed utilizing kits as per the manufacturer’s protocols (Elabscience, USA).
Analysis of Glycated Hemoglobin A1c (HbA1c) and Glucose Levels
The concentrations of HbA1c and glucose in the serum of experimental rats were investigated using commercially procured kits per the manufacturer’s protocols (CusaBio, USA).
Analysis of Oxidative Stress-related Marker Levels
The levels of malondialdehyde (MDA), myeloperoxidase (MPO), glutathione (GSH), and nitrite/nitrate (NOX) in the gingival tissues of the rats were investigated using kits as per the manufacturer’s protocols (CusaBio, USA). The concentrations of inducible nitric oxide synthase (iNOS), matrix metalloproteinase-8 (MMP-8), and total alkaline phosphatase (TAP) in the rats’ gingival tissues were studied using kits per the manufacturer’s protocols (Elabscience, USA).
Analysis of Inflammatory Marker Levels in the Serum
The serum concentrations of interleukin (IL)-1β, IL-10, and tumor necrosis factor-alpha (TNF-α) in both control and experimental rats were assessed using commercially available assay kits. All tests were done in triplicate as per the manufacturer’s procedures (LSBio, USA).
Analysis of Bone-related Biomarker Levels
The concentrations of bone-related biochemical marker levels such as bone morphogenetic protein-2 (BMP-2), tissue inhibitor of metalloproteinase-1 (TIMP-1), osteoprotegerin (OPG), and receptor activator of nuclear factor kappa-Β ligand (RANKL) in the gingival tissues of the rats were assessed using kits as per the manufacturer’s protocols (Elabscience, USA).
Statistical Analysis
The findings are illustrated as Mean ± SD of three replicates, which were studied using GraphPad Prism. The one-way analysis of variance (ANOVA) and Tukey’s post hoc assay were utilized for group comparisons, with p < .05 as significant.
Results
Effect of Fisetin on the ABL in Experimental Rats
The findings revealed that rats who underwent periodontitis induction had a significant increase in ABL levels when compared to rats without periodontitis induction (Figure 1). The oral treatment of fisetin at diverse concentrations (5, 10, and 15 mg/kg) substantially reduced the ABL level in the rats with periodontitis. The present outcomes proved that the fisetin treatment effectively inhibited bone loss in the periodontitis-induced rats (Figure 1).
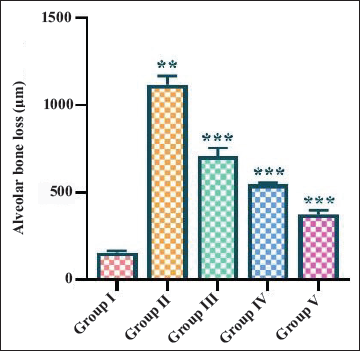
Effect of Fisetin on Osteoblast, Osteoclast, and Inflammatory Cell Counts in the Experimental Rats
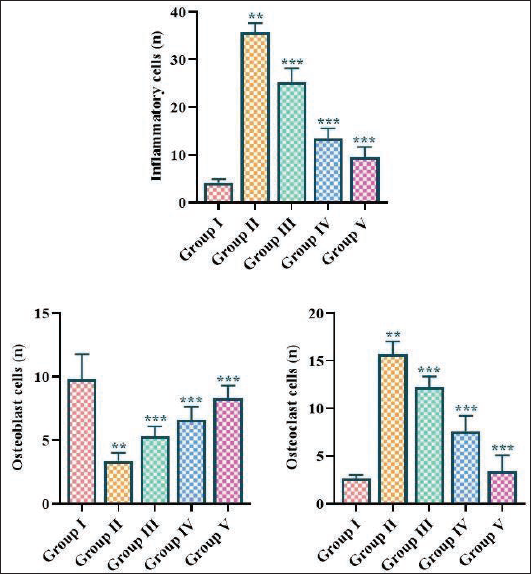
The results demonstrated that rats with periodontitis induction experienced a significant increase in osteoclast and inflammatory cells while drastically reducing the osteoblast cell counts compared with rats without periodontitis induction (Figure 2). Remarkably, the treatment with fisetin at 5, 10, and 15 mg/kg concentrations, respectively, successfully reduced the levels of osteoclast and inflammatory cell counts while elevating the osteoblast cell counts in the periodontitis-induced rats.
Effect of Fisetin on the Bone Mineral Levels in the Experimental Rats
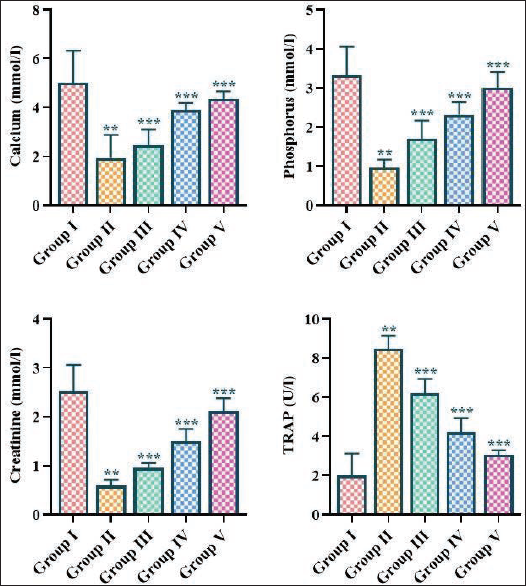
The current results demonstrated that the concentrations of bone minerals, including Ca, P, and Cr, were significantly diminished while the TRAP level was considerably elevated in the serum of periodontitis-induced rats (Figure 3). Contrastingly, the oral administration of fisetin at 5, 10, and 15 mg/kg concentrations, respectively, led to a remarkable upsurge in the levels of Ca, P, and Cr while diminishing the TRAP concentration in the periodontitis-induced rats (Figure 3).
Effect of Fisetin on HbA1c and Glucose Levels in the Experimental Rats
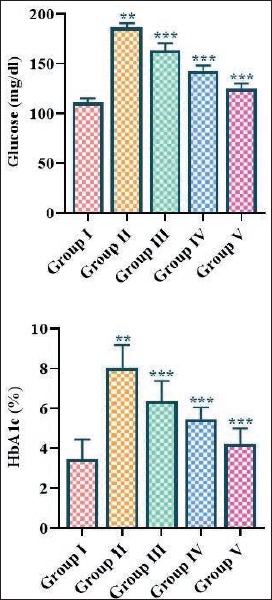
Figure 4 depicts the results of the fisetin treatment on the levels of HbA1c and glucose in the experimental rats. The rats with periodontitis induction demonstrated an increase in HbA1c and glucose concentrations compared to control. The treatment of fisetin at 5, 10, and 15 mg/kg concentrations, respectively, effectively reduced both HbA1c and glucose concentrations in the periodontitis-induced rats.
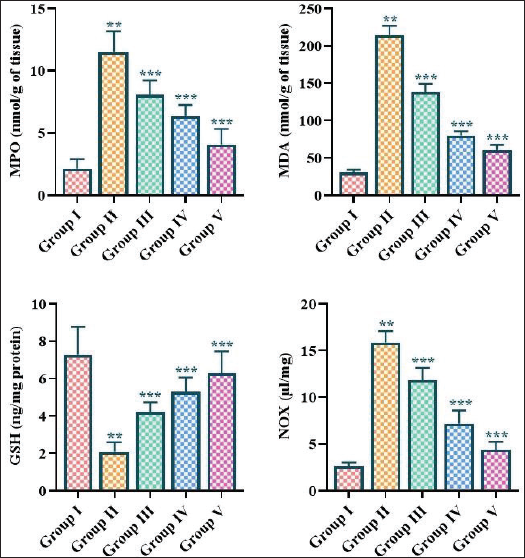
Effect of Fisetin on the Oxidative Stress-related Biomarker Levels in the Experimental Rats
The influence of fisetin on oxidative stress was evaluated by measuring the concentrations of MPO, MDA, NOX, and GSH in the experimental rats. The current outcomes demonstrated a reduction in GSH levels, accompanied by an elevation in MDA, MPO, and NOX concentrations in the periodontitis-induced rats in comparison to control rats. Interestingly, the various dosages (5, 10, and 15 mg/kg) of the fisetin treatment significantly enhanced the antioxidant GSH while substantially reducing MDA, MPO, and NOX levels in rats with periodontitis (Figure 5).
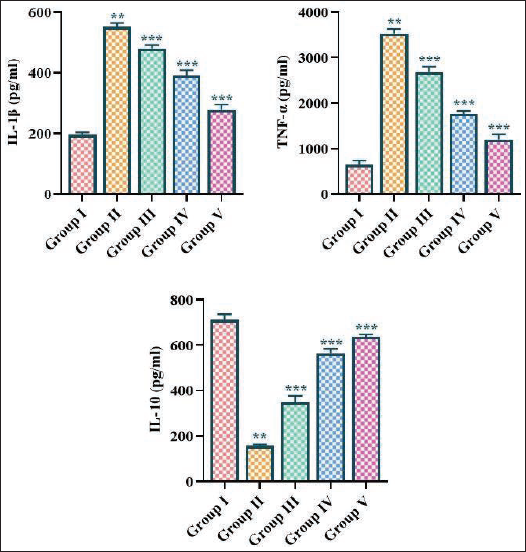
Effect of Fisetin on the Inflammatory Cytokine Levels in Experimental Rats
The results demonstrated a considerable elevation in the concentrations of IL-1β and TNF-α with a reduction in the IL-10 concentrations in the periodontitis-induced rats. Remarkably, the treatment of diverse concentrations of fisetin (5, 10, and 15 mg/kg, respectively) considerably diminished the concentrations of IL-1β and TNF-α and boosted the IL-10 concentrations in the periodontitis-induced rats (Figure 6).
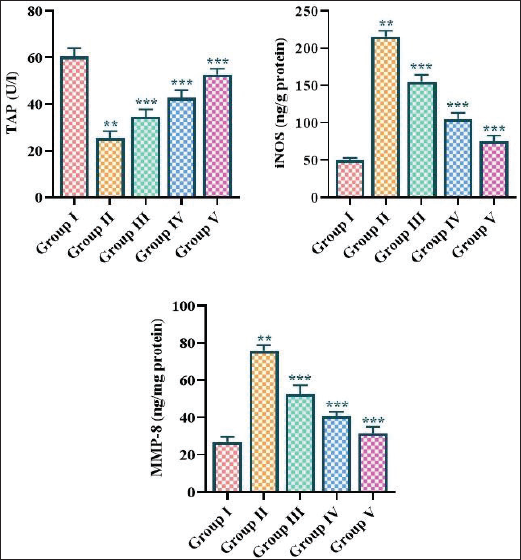
Effect of Fisetin on the TAP, iNOS, and MMP-8 Levels in the Experimental Rats
The current results indicated that periodontal disease induction significantly increased the iNOS and MMP-8 concentrations while diminishing the TAP levels when compared to normal rats. Nonetheless, the treatment of fisetin at a dosage of 5, 10, and 15 mg/kg, respectively, markedly reduced iNOS and MMP-8 concentrations while considerably elevating TAP levels in comparison to rats with periodontitis (Figure 7).
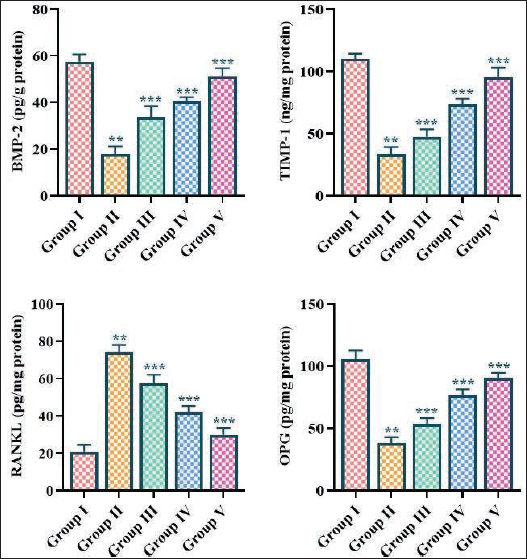
Effect of Fisetin on the BMP-2, TIMP-1, RANKL, and OPG Levels in the Experimental Rats
The present results revealed a considerable elevation in the RANKL level and a reduction in BMP-2, TIMP-1, and OPG concentrations in the rats with periodontal disease induction (Figure 8). Conversely, the oral treatment of fisetin at a dosage of 5, 10, and 15 mg/kg, respectively, resulted in a remarkable decrease in the RANKL level and an upsurge in the BMP-2, TIMP-1, and OPG concentrations in the rats with periodontal disease (Figure 8).
Discussion
Periodontitis, a chronic inflammatory condition, is characterized by the progressive destruction of the periodontal tissues, including the alveolar bone. Analyzing the changes in alveolar bone levels is crucial to understanding the severity and progression of this disease (Carrizales-Sepulveda et al., 2018). In experimental periodontitis models, rodents, particularly rats, are commonly used to mimic the human condition. Inducing periodontitis in rats allows researchers to investigate the pathological changes in the alveolar bone and develop effective treatment strategies. The systematic evaluation of ABL in these models offers valuable insights into the underlying mechanisms of periodontitis and its influence on oral health (Iova et al., 2021). The development of oral implantology has increased the interest in preserving both bone and soft tissues after tooth extraction (Bagavad Gita et al., 2019). Therefore, understanding the mechanisms of ABL is essential for developing potential strategies to treat this debilitating disease, ultimately improving the overall oral health of individuals. The present findings clearly proved that the rats with experimental periodontitis induction showed a drastic ABL level. However, the fisetin treatment at diverse concentrations effectively reduced the ABL in the periodontitis-induced rats.
Inflammatory cells, such as neutrophils, lymphocytes, and macrophages, play a crucial role in the development and progression of periodontitis. These cells are responsible for the immune response to the microbial biofilm that accumulates on the tooth surfaces (Leira et al., 2019). Lipopolysaccharide (LPS), a bacterial membrane protein, stimulates these inflammatory cells to release more IL-1β, IL-6, and TNF-α. These markers can trigger the accumulation of MMPs, which degrade the connective tissues of the gingiva and periodontal ligament, and prostaglandins, which mediate alveolar bone destruction (Justo et al., 2022). Osteoclasts, which are specialized cells responsible for bone resorption, are also pivotal in the onset of periodontitis. The inflammatory cytokines released by the inflammatory cells can trigger the differentiation and activation of osteoclasts, leading to the breakdown of alveolar bone. This imbalance between bone resorption and bone formation, with osteoclasts dominating over osteoblasts, leads to the net loss of alveolar bone, a hallmark of periodontal disease (Xu et al., 2021). Conversely, osteoblasts, which are responsible for bone formation, play an essential role in the maintenance of alveolar bone homeostasis. The dysregulation of osteoblast function, either due to the direct effects of inflammatory mediators or the imbalance between osteoclasts and osteoblasts, can contribute to the progressive destruction of alveolar bone observed in periodontitis (Robinson et al., 2020). The persistent inflammatory response, coupled with the imbalance between bone formation and resorption, leads to the characteristic tissue destruction and ABL seen in this disease (Kawai et al., 2006). Similarly, the present findings clearly proved that rats with periodontitis induction demonstrated increased inflammatory and osteoclast cells while reduced osteoblast cells. Interestingly, the fisetin treatment successfully diminished the inflammatory and osteoclast cells and increased the osteoblast cells, which proves that it decreased inflammation and regulated bone resorption and bone formation homeostasis.
The pathophysiology of periodontitis encompasses a complex interplay between various biochemical and physiological factors, including the levels of specific macromolecules and minerals. One such crucial factor is the role of Ca, P, Cr, and TRAP in the progression of periodontitis (Naghsh et al., 2017). Ca and P are pivotal minerals for regulating the structural integrity of the teeth and alveolar bone, which provide the foundation for the periodontal tissues. Cr, a byproduct of muscle metabolism, is considered a marker of kidney function, and its elevated levels may indicate underlying health issues that can contribute to periodontal disease. TRAP, on the other hand, is an enzyme associated with osteoclast activity, which is crucial for bone resorption and remodeling processes (Rodrigues et al., 2014). The dysregulation of these biochemical factors can have an influence on the pathophysiology of periodontitis. The delicate balance and interplay between Ca, P, Cr, and TRAP play an essential role in the pathogenesis of periodontitis. Understanding the underlying mechanisms and the interactions of these biochemical factors can facilitate the development of potential therapies for this debilitating oral condition (Dembowska et al., 2022). The present findings showed a drastic diminution in the levels of Ca, P, and Cr and a subsequent increase in the TRAP levels in the rats with periodontitis. Interestingly, the treatment with the fisetin successfully increased the Ca, P, and Cr and reduced the TRAP levels in the periodontitis-induced rats.
The pathophysiology of periodontitis is multifaceted and several key oxidative stress markers have been implicated in the disease process, including MDA, MPO, GSH, and NOX. MDA, a byproduct of lipid peroxidation, is a well-established oxidative stress marker. Increased MDA concentrations have been consistently noted in patients with periodontitis, reflecting increased oxidative damage to cellular membranes and lipids (Almerich-Silla et al., 2015). MPO, an enzyme released by activated neutrophils, catalyzes the production of potent oxidants, contributing to the perpetuation of the inflammatory response. GSH, a critical antioxidant, is often depleted in periodontal disease, compromising the body’s defense against oxidative insults. Additionally, NOX levels, indicative of NO production, have been found to be altered in periodontitis, suggesting a potential role in the onset of the disease (Kose et al., 2016). The interplay between these oxidative stress markers and the underlying mechanisms in periodontitis is not yet fully elucidated. The precise mechanisms by which oxidative stress participates in the initiation and regulation of periodontitis remain to be further investigated (Arana et al., 2017). Emerging evidence suggests that the dysregulation of these oxidative stress markers may not only contribute to the local periodontal destruction but also influence the progression of systemic diseases through the dissemination of bacterial components and inflammatory mediators into the circulatory system (Chen et al., 2019). As a result, the assessment of oxidative stress markers provides valuable insights into the pathophysiology of periodontitis. The present findings proved that periodontitis induction resulted in increased levels of MDA, MPO, and NOX and a reduction in GSH levels in rats. Whereas, the fisetin treatment successfully decreased the MDA, MPO, and NOX levels and increased the GSH level in the rats with periodontitis, which proves its antioxidant properties.
iNOS is an enzyme that catalyzes the production of NO, a potent vasodilator and signaling molecule that can also exert pro-inflammatory and cytotoxic effects. Elevated levels of iNOS have been observed in periodontal tissues affected by inflammation, and it has been suggested that iNOS-mediated NO production may participate in the pathogenesis of periodontitis (Cataldi et al., 2022). MMP-8 is a zinc-dependent endopeptidase that plays an essential role in the degradation of the extracellular matrix, including the collagen fibers that provide structural support to the periodontal tissues. Increased MMP-8 levels have been connected with the progression of periodontitis (Luchian et al., 2022). TAP is an enzyme involved in the mineralization of bone and the regulation of bone metabolism. Decreased levels of TAP have been observed in individuals with periodontitis, suggesting that impaired bone metabolism and homeostasis may contribute to the onset of this disease (Nagasaki et al., 2021). The dysregulation of iNOS, MMP-8, and TAP levels plays an essential role in the pathophysiology of periodontitis. Elevated iNOS and MMP-8 levels can promote inflammation and tissue destruction, while decreased TAP levels may impair bone homeostasis and contribute to ABL (Plemmenos & Piperi, 2022). In a similar manner, the present results also showed a drastic increase in iNOS and MMP-8 while the TAP levels were reduced in the rats with periodontitis. Remarkably, the fisetin treatment successfully reduced the iNOS and MMP-8 levels and increased the TAP content in the periodontitis-induced rats.
The initiation and development of periodontitis are primarily driven by the inflammatory response to the presence of a subgingival periodontal biofilm, which contains a diverse array of pathogenic bacteria (Ramadan et al., 2020). These bacteria release various virulence factors, including LPS, which can stimulate the host’s immune cells to generate increased IL-1β and TNF-α. These cytokines can then trigger a cascade of events, leading to the destruction of the periodontium (Hosokawa et al., 2016). Interestingly, IL-10 has been found to play a protective role in the development of periodontitis. IL-10 can inhibit the production of pro-inflammatory cytokines and downregulate the inflammatory response, potentially preventing or slowing the progression of the disease (Preshaw, 2018). Furthermore, the dysregulation of the balance between pro- and anti-inflammatory cytokines may facilitate periodontitis. Persistent inflammation can lead to the continuous production of MMPs and prostaglandins, which can further destroy the periodontal tissues. The inflammatory mediators released during the course of periodontal disease may have far-reaching effects on the body, contributing to the progression of these systemic conditions (Relvas et al., 2024). In this work, the outcomes revealed increased IL-1β and TNF-α concentrations and a reduction in IL-10 contents in the periodontitis-induced rats. Interestingly, the fisetin treatment effectively diminished the IL-1β and TNF-α concentrations and boosted the IL-10 concentration in the rats with periodontitis.
Emerging evidence suggests that specific molecular mediators, such as BMP-2 and TIMP-1, play crucial roles in the pathogenesis of this disease. BMP-2 is a member of the TGF-β superfamily and participates in various biological activities, including bone formation and remodeling. In the context of periodontitis, BMP-2 has been shown to exhibit both protective and detrimental effects. On one hand, BMP-2 can promote the regeneration of periodontal tissues by stimulating the differentiation of osteoblasts and the deposition of mineralized extracellular matrix (Wang et al., 2023). However, dysregulation of BMP-2 signaling can also participate in the onset of periodontitis. TIMP-1 is an important regulator of MMPs, which are enzymes responsible for degrading the extracellular matrix in the periodontal tissues. TIMP-1 has been found to be downregulated in the gingival tissues of periodontitis patients, leading to an imbalance between MMPs and their inhibitors. This imbalance can lead to the excessive breakdown of the connective tissue and alveolar bone, ultimately contributing to the progression of the disease (de Brouwer et al., 2022). The role of BMP-2 and TIMP-1 in the pathophysiology of periodontitis highlights the need for a better understanding of the complex molecular mechanisms that participate in this disease. Elucidating the intricate interplay between these and other key mediators could result in the development of potential therapies for the management of periodontitis (Sirisereephap et al., 2022). The present findings demonstrated a reduction in both BMP-2 and TIMP-1 levels in the rats with periodontitis. However, the fisetin treatment successfully increased both BMP-2 and TIMP-1 levels in the periodontitis-induced rats.
Two key regulators of osteoclastogenesis and bone metabolism are OPG and RANKL, which play the role of a major role in the pathophysiology of periodontitis. OPG is a soluble decoy receptor that binds to RANKL, preventing it from contacting with its receptor activator of nuclear factor kappa-Β (RANK) on osteoclast precursors and inhibiting osteoclast differentiation and activation. In contrast, RANKL is a pro-osteoclastogenic cytokine that promotes osteoclast formation and function (Asif et al., 2022). In periodontal disease, an imbalance between OPG and RANKL levels can lead to excessive osteoclast activity and ABL. It has been reported that patients with periodontitis have higher RANKL levels and diminished OPG levels in gingival tissues and serum compared to healthy controls. This shift in the OPG/RANKL ratio can tip the balance toward increased osteoclast production and bone resorption, participating in the onset of periodontitis (Abdullameer & Abdulkareem, 2023). The modulation of the OPG/RANKL axis may represent a hopeful target for treating periodontitis. For instance, pharmacological agents that can increase OPG levels or inhibit RANKL signaling may be effective in reducing ABL and promoting periodontal tissue regeneration (López Roldán et al., 2020). Similarly, the present findings also evidenced that fisetin successfully increased OPG levels and subsequently reduced the RANKL levels in the rats with periodontitis. These findings prove the positive effects of fisetin in reducing ABL and promoting periodontal tissue regeneration in rats.
Conclusion
Fisetin, a potent natural compound with anti-inflammatory properties, ameliorated the pathological changes induced by periodontitis in rats by regulating BMP-2/TIMP-1 and OPG/RANKL expressions while reducing inflammation, osteoclast cell numbers, MMP-8 activity, decreasing oxidative stress markers, and inflammatory biomarkers. These results suggest that fisetin effectively reduced ABL and promoted periodontal tissue regeneration in periodontitis-induced rats. However, further studies are still required in the future to comprehend the therapeutic roles of fisetin against periodontal disease fully.
Footnotes
Abbreviations
ABL: Alveolar bone loss; BMP-2: Bone morphogenetic protein-2; Ca: Calcium; Cr: Creatinine; GSH: Glutathione; HbA1c: Glycated hemoglobin A1c; IL: Interleukin; iNOS: Inducible nitric oxide synthase; LPS: Lipopolysaccharide; MDA: Malondialdehyde; MMP: Matrix metalloproteinase; MPO: Myeloperoxidase; NOX: Nitrite/Nitrate; NSAID: Non-steroidal anti-inflammatory drug; OPG: Osteoprotegerin; P: Phosphorus; RANK: Receptor activator of nuclear factor kappa-Β; RANKL: Receptor activator of nuclear factor kappa-Β ligand; TAP: Total alkaline phosphatase; TIMP-1: Tissue inhibitor of metalloproteinase-1; TNF-α: Tumor necrosis factor-alpha; TRAP: Tartrate-resistant acid phosphatase.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This work has been approved by the institutional animal ethical committee at the Stomatological Hospital of Fujian Medical University, Fuzhou, China.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Fujian Provincial Department of Education Young and Middle-aged Backbone Teachers Project JAT210113 and Fujian Medical University Sailing Project 2021QH1136.
Informed Consent
Not applicable.
