Abstract
Background
Diabetic nephropathy (DN) is a severe complication in patients with diabetes mellitus, which is mainly characterized by glomerular podocyte injury. Ferroptosis is crucial in the pathogenesis of DN. Moringa oleifera leaf extract (MOLE) has attracted attention due to its multiple pharmacological activities, but it is unclear whether it can delay the progression of DN by inhibiting ferroptosis.
Objectives
To investigate whether MOLE can alleviate podocyte injury and delay the progression of DN by inhibiting ferroptosis.
Materials and Methods
DN model cells were established using rat podocytes induced by high glucose (HG); the cells were processed with MOLE, dapagliflozin (DAPA, positive control), and acyl-CoA synthetase long-chain family member 4 (ACSL4) overexpression plasmids. Cell viability, Fe2+ concentration, reactive oxygen species (ROS), malondialdehyde (MDA), and glutathione (GSH) levels were monitored by Cell Counting Kit-8 (CCK-8) and corresponding kits.
Results
MOLE treatment markedly enhanced HG-induced rat podocyte viability, decreased intracellular Fe2+ concentration, ROS level, MDA concentration, and elevated GSH concentration. ACSL4 overexpression reversed the protective effect of MOLE on podocytes.
Conclusion
MOLE ameliorates rat podocyte injury and delays DN progression by inhibiting ACSL4-mediated ferroptosis. This finding provides a theoretical basis for further developing MOLE-based therapeutic agents for DN treatment.
Introduction
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disease (Wang, Yang, et al., 2023), and some patients suffer from multiple complications due to poorly controlled blood glucose levels (Majety et al., 2023). Nephropathy is a characteristic microvascular complication of T2DM, which can result in increased proinflammatory factors and proteinuria and eventually develop into end-stage renal disease (ESRD) (Gupta et al., 2023). Effective glycemic control is the primary goal in the clinical management of patients with diabetic nephropathy (DN) (Meng et al., 2023). The use of hypoglycemic drugs is the mainstay of DN treatment, but there are limitations to their effectiveness. Relevant statistics show that over the past 10 years, the global morbidity and mortality rate of ESRD has increased by about 32%, of which DN accounts for 35–50% (Gupta et al., 2022). Therefore, drugs for the effective treatment of DN need to be actively explored. Currently, management strategies for DN focus on controlling body weight, blood glucose, and blood pressure, with renin–angiotensin system inhibitors, such as angiotensin-converting enzyme inhibitors (ACE inhibitors) and angiotensin receptor blockers (ARBs), serving as first-line treatments (Heerspink et al., 2020; Shubrook et al., 2022). Dapagliflozin (DAPA), a sodium–glucose cotransporter-2 (SGLT2) inhibitor, is commonly used as a first-line therapeutic agent for DN in clinical practice (Jensen et al., 2024; Vart et al., 2024). However, these treatments have limitations and potential side effects (Raptis & Dimitriadis, 2001). DN has the characteristics of multiple signaling pathways and multiple targets, which undoubtedly increases the difficulty of treatment but also brings opportunities for natural product treatment (Hu et al., 2023). Notably, natural product offers promising therapeutic targets with the potential to improve DN treatment outcomes and have a relatively favorable safety profile with fewer reported adverse effects (Hu et al., 2023). Therefore, identifying effective targets and novel therapeutic approaches for DN remains of great clinical importance.
Moringa oleifera (MO) is a plant of the genus Moringa in the Moringa family (Islam et al., 2021). In 2012, the Ministry of Health approved Moringa oleifera leaves (MOL) as a new food resource (Deepa et al., 2022). MOL contains phenolic acids, flavonoids, tannins, glucosides, and other active substances, which have anti-oxidation, anti-tumor, anti-bacterial, hypoglycemic, lipid-lowering, blood-pressure lowering, and anti-inflammatory effects (Gómez-Martínez et al., 2021; Mushtaq et al., 2021). Numerous studies have proven that MOL can be applied as a medicinal plant for diabetes treatment (Gómez-Martínez et al., 2021; Owens et al., 2020). A recent study revealed that MOL could reduce protein glycosylation in streptozotocin (STZ)-induced diabetic rats and thus protect the kidney (Ahmad et al., 2023). Moringa oleifera leaves extract (MOLE) was also confirmed to have anti-angiogenic and anti-inflammatory effects in STZ-induced DN rats (Thongrung et al., 2022). These studies suggest that MOLE can positively affect the DN process. However, the mechanism by which MOLE alleviates the DN progression is not fully understood.
Iron is a trace element that maintains normal physiological processes in the body (Zhang, Lu, et al., 2022). Iron overload is a vital cause of ferroptosis (Dutt et al., 2022). Ferroptosis is a novel mode of non-apoptotic cell death (Chu et al., 2023). The primary mechanism is to catalyze lipid peroxidation of polyunsaturated fatty acids (PUFA) highly expressed on the cell membrane in Fe2+ or ester oxygenase (Jiang et al., 2021). In DN, intracellular reactive oxygen species (ROS) and Fe2+ concentrations are increased, which can eliminate PUFA through lipid peroxidation, leading to ferroptosis (Li, Dai, et al., 2023; Mengstie et al., 2023). However, whether MOLE can improve DN by inhibiting ferroptosis has yet to be elucidated.
In the present study, we further investigated MOLE’s protective effect and mechanism on rat podocyte injury by establishing a DN cell model to provide a theoretical basis for the relevant clinical treatment of DN.
Materials and Methods
Preparation of MOLE (Whole Extract)
MO powder (200 g; Taike Biotechnology Co. Ltd., Shanxi, China) and ultrapure water (2 L) were mixed and boiled for 5 min, then cooled naturally to room temperature. The sample was filtered using a 2 µm membrane to remove undissolved impurities. After vacuum freeze–drying, the brown powder was obtained and stored frozen. The lyophilized powder was dissolved in a culture medium for 1 mg/mL solution. After filtration with a 0.22 µm filter membrane, the samples were dispensed into centrifuge tubes and stored at –20℃.
Cell Culture
Rat podocytes (BNCC338697, Beina Biology, Beijing, China) were maintained in low-glucose Dulbecco’s modified Eagle medium (DMEM) (Gibco; Cat. no. 10567014) with 10% fetal bovine serum (FBS, Sigma) at 37°C with 5% CO2.
Construction of DN Model Cells
DN model cells were established with rat podocytes stimulated with 30 mmol/L glucose for 48 h; this group was the high glucose (HG) group (Jiang et al., 2022; Su et al., 2022). The rat podocytes were treated with 44.4 mmol/L mannitol for 48 h (MG group), which was applied to increase the osmotic pressure. Normal cultured cells (NG group) were only cultured with low-glucose DMEM.
Cell Treatment and Transfection
Rat podocytes were first treated with 0, 25, 50, 100, 200, and 300 µg/mL MOLE. The cells in the HG group were treated with 100 µg/mL MOLE, 200 µg/mL MOLE, or 2 µM DAPA (Yuanye Bio-Technology). DAPA was applied as a positive control (Lv et al., 2023) and was given 30 min before glucose. ACSL4 overexpression plasmids (OE-ACSL4) and the negative control (NC) were purchased from GenePharma (Shanghai, China). The treated rat podocytes were transfected with 50 ng ACSL4 plasmids with Lipofectamine 3000 (Invitrogen). Transfection efficiency was evaluated by quantitative real-time polymerase chain reaction (qRT-PCR) for ACSL4 expression. To minimize off-target effects, we included a negative control group (NC; transfected with empty vector) to account for any non-specific effects induced by the transfection procedure itself.
Cell Counting Kit-8 (CCK-8)
The cell suspension (200 µL, 5 × 103 cells/well) was homogeneously added to 96-well plates for incubation. After 48 h, the cells were supplemented with 10 µL CCK-8 (Dojindo, Japan). After 2 h of incubation, cell viability was calculated by measuring the absorbance value at 450 nm using an enzyme marker.
qRT-PCR
Total ribonucleic acid (RNA) was extracted from the treated rat podocytes using the TRIzol method. After quantification, reverse transcription and qPCR were conducted using the PrimeScript RT Reagent Kit (Takara) and the TaqMan Master Mix kit (Thermo Fisher Scientific). The following primers were employed: ACSL4: 5′-TCCTTTTTGCGAGCTTTCCGA-3′ and 5′-GCCATAGCGTTTTTAAATGTCCT-3′; β-actin: 5′-GTCGTACCAGGCATTGTGTAGG-3′ and 5′-GCAATGCCTGGGTACATGGTGG-3′. The ACSL4 level was calculated in line with the 2–△△Ct.
Western Blot
Total protein from the treated rat podocytes was extracted using radioimmunoprecipitation assay (RIPA) (Beyotime, Beijing, China). After quantification using a Pierce BCA Protein Assay Kit (Thermo Scientific, USA), the protein (40 µg) was denatured by heating, separated, and transferred to the polyvinylidene fluoride (PVDF) membrane (Millipore). After sealing, the membrane was soaked in anti-ACSL4 (1:1,000, Abcam, ab227256) and anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (1:1,000, Abcam, ab8245) overnight at 4℃ and processed with a secondary antibody (1:2,000, Abcam, ab288151). Then, the proteins were visualized after treatment with a chemiluminescence kit (Millipore, MA, USA).
Iron Assay
Based on the manufacturer’s protocols, the Fe2+ content was confirmed using a Fe2+ colorimetric kit (Applygen Technologies, Beijing, China, E1042).
ROS Detection
Rat podocytes from each group were treated with 20 µM 2′,7′-dichlorofluorescin diacetate (DCFH-DA) (Sigma) for 30 min, and the fluorescence intensity was quantified by fluorescence spectrophotometer at 485 nm (excitation) and 530 nm (emission).
Malondialdehyde (MDA) and Glutathione (GSH) Detection
The levels of MDA and GSH were examined using the MDA assay kit (Jiancheng Bioengineering, Nanjing, China, A003-1-2) and the GSH assay kit (Jiancheng, China, A006-1-1) in line with the protocol descriptions.
Statistical Analysis
The measurement data, repeated three times independently, were expressed as mean ± standard deviation (SD). Statistical analysis was conducted using SPSS22.0 (SPSS Inc., Chicago, IL, USA) with one-way analysis of variance (ANOVA). p < .05 denoted the statistical significance.
Results
MOLE Enhances the Viability of Rat Podocytes Induced by HG
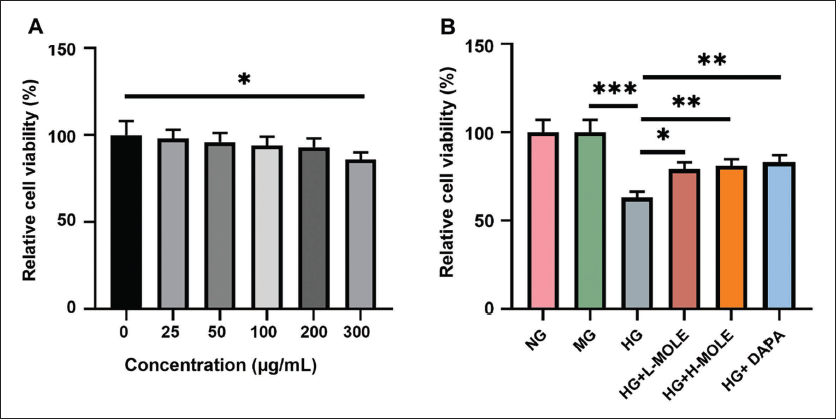
To verify the effect of MOLE on DN, we first treated rat podocytes with different concentrations of MOLE. CCK-8 results denoted that 0–200 µg/mL MOLE did not affect the viability of rat podocytes, and 300 µg/mL MOLE significantly reduced rat podocyte viability (Figure 1A). Therefore, 100 and 200 µg/mL MOLE were selected as low and high concentrations for subsequent experiments in this study. Then, the podocyte injury model was established using 30 mmol/L glucose. CCK-8 data revealed that MG did not affect the viability of rat podocytes, while HG significantly decreased their viability; the inhibitory effect of HG on rat podocyte viability could also be partially reversed by MOLE or DAPA (Figure 1B).
MOLE Suppresses Ferroptosis of HG-induced Rat Podocytes
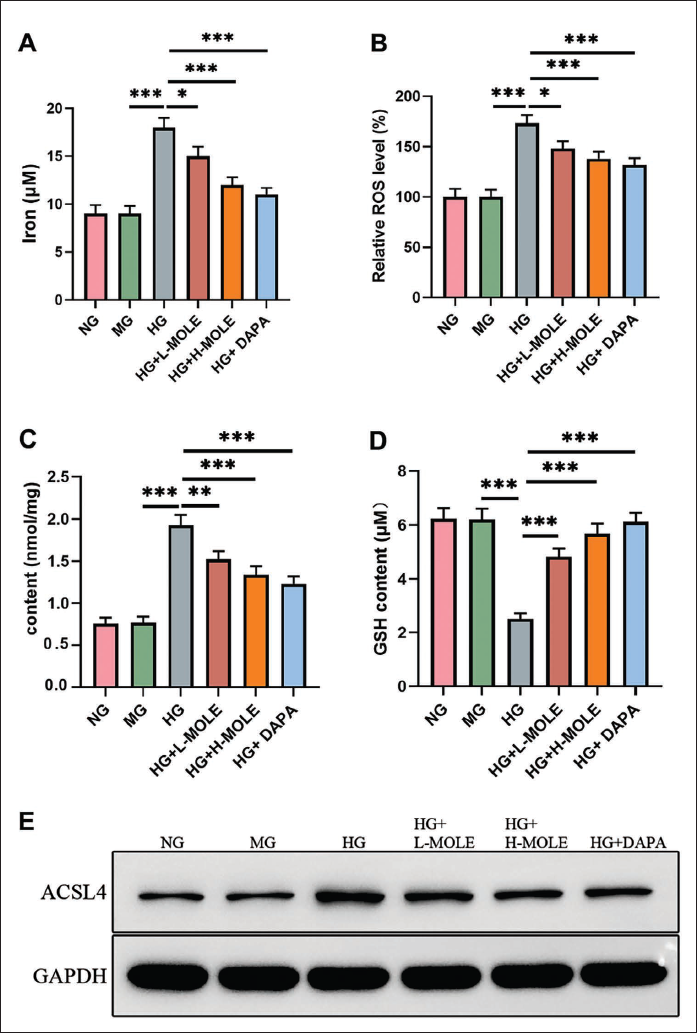
Ferroptosis is associated with DN and lipid-ROS accumulation (Li, Dai, et al., 2023). After HG treatment, Fe2+ content was markedly elevated in rat podocytes. MOLE or DAPA treatment reduced HG-induced Fe2+ content in rat podocytes; a higher concentration of MOLE had a more substantial reduction effect on Fe2+ content than a lower concentration (Figure 2A). HG treatment increased ROS level and MDA content and decreased GSH content; the increase in ROS and MDA levels and the decrease in GSH level mediated by HG treatment could also be reversed by MOLE or DAPA in rat podocytes, with exceptionally high concentration of MOLE and DAPA (Figure 2B–D). Besides, we discovered that HG stimulation upregulated ACSL4 expression in rat podocytes, which could also be reduced by MOLE or DAPA in rat podocytes (Figure 2E). Altogether, MOLE inhibits ferroptosis of HG-stimulated rat podocytes.
ACSL4 Overexpression Reverses the Increase in Cell Viability and Ferroptosis Inhibition Mediated by MOLE in HG-treated Rat Podocytes
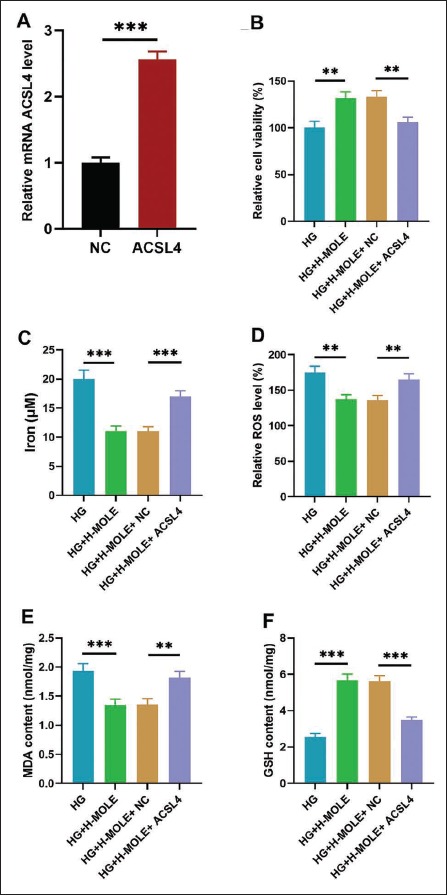
To further verify whether the inhibition of MOLE on ferroptosis can be realized by regulating ACSL4, we first overexpressed ACSL4 in rat podocytes by transfection. qRT-PCR data denoted that after OE-ACSL4 transfection, ACSL4 expression was dramatically increased in rat podocytes, showing successful overexpression of ACSL4 (Figure 3A). Then, CCK-8 results disclosed that the increase of cell viability mediated by MOLE treatment could be attenuated by ACSL4 overexpression in HG-induced rat podocytes (Figure 3B). The data also uncovered that the reduction of Fe2+ content mediated by MOLE was, in turn, reversed by ACSL4 overexpression in HG-mediated rat podocytes (Figure 3C). Besides, ACSL4 overexpression also attenuated the decrease in ROS and MDA levels and the increase in GSH level mediated by MOLE in HG-treated rat podocytes (Figure 3D–F).
Discussion
DN is one of the microvascular complications of diabetes (Thipsawat, 2021). The functional and structural integrity of podocytes is critical for the glomerular filtration barrier (GFB) (Barutta et al., 2022). Podocyte injury is a central mechanism in the development of DN (Li, Zhang, et al., 2023). In our study, we further proved that MOLE could enhance the viability of HG-induced rat podocytes. Thus, our data further confirm that MOLE has a protective effect on DN, which is generally consistent with previous studies. Moreover, DAPA was applied as a positive control. DAPA is a new type of hypoglycemic drug that can effectively protect the kidneys by controlling the absorption of glucose by the kidneys and enhancing the excretion of glucose in the urine to exert therapeutic effects (Wheeler et al., 2021). Our study also revealed that MOLE enhanced HG-induced rat podocyte viability comparable to DAPA.
Renal exposure to HG and oxidative stress in diabetic patients can promote the progression of DN (Xu et al., 2023). The pathology of DN is characterized by aggregation of the extracellular matrix, thickening of the basement membrane, and the development of diffuse glomerulosclerosis and interstitial fibrosis (Jin et al., 2023). Under DN conditions, renal intrinsic cells can undergo ferroptosis, exacerbating renal injury (Feng et al., 2021). Ferroptosis is associated with iron-dependent oxidative stress and lipid peroxidation (Li et al., 2021; Wang et al., 2020). Earlier studies revealed that excessive iron accumulation in the kidney could accelerate renal injury in diabetic rats (Li et al., 2021; Wang, Liu, et al., 2023). Under the HG environment, renal cells showed prominent ferroptosis characteristics, including the increase of membrane density, the reduction of mitochondrial volume, and the reduction or even disappearance of mitochondrial cristae (Ye et al., 2023). Ferroptosis may promote DN progression by activating the hypoxia-inducible factor/heme oxygenase 1 pathway, damaging the renal tubules (Feng et al., 2021). All of the above studies testified that ferroptosis plays a crucial role in DN’s progression. Currently, treatments for DN are not limited to glucose reduction, lipid reduction, reduction of urinary protein, and so on. Some drugs can slow down the progression of DN by inhibiting ferroptosis. Therefore, an in-depth study of the molecular mechanism of ferroptosis and DN may provide a new direction for treating DN. Whether MOLE regulates ferroptosis in the DN process has yet to be reported.
Ferroptosis is associated with ROS production, MDA accumulation, and decreased GSH levels (Yang et al., 2022). An increase in ferric ions promotes lipid ROS production, causing lipid peroxidation and cell death (Endale et al., 2023). MDA is one of the products of lipid peroxidation. During ferroptosis, the level of MDA rises (Chen et al., 2021). GSH is a vital intracellular anti-oxidant that scavenges lipid peroxidation and inhibits ferroptosis (Ursini & Maiorino, 2020). Our results first proved that MOLE could decrease ROS and MDA levels and increase GSH content in HG-treated rat podocytes, suggesting that MOLE can suppress ferroptosis in DN. The GFB consists of podocytes, the glomerular basement membrane, and endothelial cells, with podocytes playing a critical role in maintaining filtration integrity. In DN, the dysfunction of podocytes leads to the breakdown of the GFB, resulting in proteinuria and kidney damage. In our study, treatment with MOLE led to significant improvements in podocyte viability and reduced ferroptosis markers (ROS, MDA, and Fe²⁺), which likely contributed to the structural restoration of the GFB. These findings highlight the potential of MOLE as a therapeutic agent for preserving GFB integrity in DN by targeting ferroptosis-related pathways. Besides, the findings of this study are consistent with previous reports demonstrating the protective effects of MOLE against oxidative stress and inflammation in kidney models. For instance, previous studies have shown that Moringa oleifera ethanolic extract (MOEE) ameliorates renal oxidative damage and inflammation in rat models exposed to cobalt chloride (CoCl2) and in diabetic rats (Abdel-Daim et al., 2020). These studies have reported improvements in anti-oxidant levels (SOD, CAT, and GSH) and reductions in oxidative stress markers (MDA and H2O2), similar to the effects of MOLE observed in our study. Furthermore, the anti-inflammatory effects of MOLE, including the suppression of proinflammatory cytokines such as interleukin (IL)-1β, IL-6, and nuclear factor-kappa B (NF-κB) activation, align with our findings showing reductions in oxidative stress and inflammatory markers in podocytes treated with HG. These consistent results across different renal models highlight the broad therapeutic potential of MO for managing DN and other kidney-related complications. Besides, ACSL4 is a key enzyme that catalyzes the activation of long-chain fatty acids and plays an essential role in lipid metabolic pathways (Zhou et al., 2023). ACSL4 drives intracellular lipid peroxidation and is a key determinant of ferroptosis sensitivity (Zhang, Hu, et al., 2022). A study showed that ACSL4 deficiency could attenuate acute kidney injury mediated by ferroptosis (Wang et al., 2022). The results of our study significantly advance the understanding of the mechanistic role of ACSL4 in ferroptosis and its contribution to the progression of DN. Our data demonstrate that ACSL4 is upregulated in rat podocytes under HG conditions, promoting the accumulation of lipid peroxides and contributing to ferroptosis. This finding establishes ACSL4 as a key player in ferroptosis in the context of DN. Furthermore, we show that MOLE can downregulate ACSL4 expression and mitigate the markers of ferroptosis, providing therapeutic evidence that targeting ACSL4 could protect podocytes from HG-induced injury. The ability of MOLE to inhibit ACSL4-mediated ferroptosis suggests that ACSL4 could be a promising therapeutic target for preventing or slowing the progression of DN, highlighting its mechanistic role in podocyte injury and kidney damage in diabetic conditions. While the inhibition of ACSL4 is central to MOLE’s protective effects, we propose that MOLE also exerts broader effects on other ferroptosis-related pathways, such as anti-oxidant defenses and lipid metabolism. Besides, the broader implications of MOLE’s modulation of ACSL4 extend to other diseases where ferroptosis plays a key role. Ferroptosis is implicated in a range of conditions, including neurodegenerative diseases (e.g., Alzheimer’s and Parkinson’s), ischemia–reperfusion injury, and cancer. In these diseases, ACSL4 regulates lipid peroxidation, a central mechanism of ferroptosis. By modulating ACSL4, MOLE could offer a novel therapeutic approach to reduce ferroptosis-induced damage in these conditions. Future research should explore MOLE’s effects on ACSL4 regulation and ferroptosis in models of neurodegeneration, ischemia–reperfusion injury, and cancer to better understand its potential for treating these diseases. In this study, MOLE demonstrated significant protective effects in reducing ferroptosis markers in HG-induced podocytes. However, a direct comparison with other well-known ferroptosis inhibitors, such as ferrostatin-1 (Fer-1) or liproxstatin-1, was not included in the experimental design. These inhibitors are commonly used to suppress ferroptosis by preventing lipid peroxidation or stabilizing key ferroptosis-related proteins, such as GPX4. Future studies should aim to compare MOLE’s efficacy in reducing ferroptosis with these standard inhibitors to evaluate its relative potency. These comparisons will be crucial for understanding the therapeutic potential of MOLE in comparison to other ferroptosis-targeting interventions. Besides, we have considered two recent studies in the context of our findings on the protective effects of MOLE against DN. The study on Pleurotus fossulatus as a natural product demonstrated its protective effects against kidney and liver damage in diabetic rats, reversing histopathological changes and preventing DN (Dubey et al., 2023). These results parallel our observations of MOLE’s ability to reduce oxidative stress and modulate ferroptosis in podocytes, suggesting that MOLE could similarly offer reno-protective benefits. Another study reviewed the role of inflammatory cytokines, such as tumor necrosis factor alpha (TNF-α) and IL-6, in insulin resistance and obesity, which are major contributing factors to DN (Singh & Rai, 2019). The findings emphasize the importance of targeting inflammation and oxidative stress pathways, both of which are modulated by MOLE. As our study has shown, MOLE’s anti-oxidant and anti-inflammatory properties may mitigate the harmful effects of elevated inflammatory cytokines, further supporting its potential as a therapeutic agent in managing not only DN but also insulin resistance and obesity.
Given the complexity of ferroptosis and the potential for MOLE to modulate multiple pathways, future research will be required to dissect the precise molecular mechanisms through which MOLE its effects. MOLE contains a variety of bioactive compounds, including flavonoids, phenolic acids, and triterpenoids, which may contribute to its anti-oxidant and anti-inflammatory effects. Future studies should focus on isolating individual components of MOLE and testing their effects on ferroptosis-related markers to determine which compounds are responsible for the observed bioactivity. We propose that network pharmacology and molecular docking analysis could be employed to identify the active components of MOLE and their potential targets and pathways in DN. These approaches will provide a more comprehensive understanding of the molecular network involved and help identify additional therapeutic targets for managing ferroptosis in DN. Besides, while this study focused on the protective effects of MOLE in podocytes, exploring the molecular interactions between MOLE flavonoids and enzymes involved in carbohydrate and lipid metabolism is an important avenue for future research. Flavonoids, such as quercetin and kaempferol, are known to interact with key enzymes like α-amylase, α-glucosidase, and enzymes in lipid metabolism such as acetyl-CoA carboxylase and fatty acid synthase. Future studies using molecular docking and network pharmacology could be employed to identify the specific enzymes targeted by MOLE flavonoids. In vitro assays could then be performed to validate these interactions and further elucidate the metabolic regulatory effects of MOLE flavonoids in DN. While this study provides valuable insights into the potential therapeutic effects of MOLE in vitro, we recognize that in vivo studies are crucial to validate these findings in a more complex and physiologically relevant context. In future studies, we plan to investigate the therapeutic potential of MOLE in animal models of DN, such as the STZ-induced diabetic rat or db/db mouse models. These in vivo experiments would allow for the evaluation of MOLE’s effects on kidney function, histopathological changes, and markers of ferroptosis in the kidney. Moreover, exploring the molecular mechanisms of MOLE’s action in vivo, particularly its impact on ACSL4 expression and oxidative stress pathways in kidney tissues, would provide a more comprehensive understanding of its therapeutic potential. These additional in vivo studies will be crucial to confirm the efficacy of MOLE and assess its potential as a therapeutic agent for DN. Besides, we recognize the importance of validating these findings in primary human podocytes to confirm their translational relevance. Human podocytes more closely reflect the clinical pathology of DN, and testing MOLE in these cells will provide a better understanding of its potential therapeutic benefits in human diabetic kidney disease. Additionally, we plan to expand our research to other relevant cell models, such as kidney tubular cells or mesangial cells, to explore the broader applicability of MOLE’s effects across different kidney cell types.
While this study demonstrates the short-term protective effects of MOLE in reducing oxidative stress and ferroptosis in HG-induced podocytes, the long-term effects of MOLE need to be further investigated. The reductions in ROS and MDA levels and increases in GSH observed at the 48-h time point indicate initial protection, but long-term studies are required to assess whether these effects are sustained over time. Future research should include in vivo animal models of DN to evaluate the chronic effects of MOLE on oxidative stress, kidney function, and renal histopathology. This would provide a more comprehensive understanding of MOLE’s potential for long-term therapeutic benefit in managing DN and other chronic diseases. While this study focused on the effects of MOLE in vitro, the interaction of MOLE with other therapeutic agents for DN is an important consideration for its clinical potential. MOLE contains various bioactive compounds with anti-oxidant, anti-inflammatory, and lipid-lowering properties, which could interact synergistically with established therapies such as ACE inhibitors, ARBs, SGLT2 inhibitors, and other renoprotective agents. For example, the anti-oxidative and anti-inflammatory effects of MOLE may complement the actions of drugs like SGLT2 inhibitors, potentially enhancing their therapeutic efficacy in DN. However, this study did not explore the potential synergistic or antagonistic effects of MOLE when used in combination with other drugs. Future studies will need to investigate these interactions in more detail, including assessing the combined effects of MOLE with drugs like DAPA and evaluating the potential for synergistic benefits or any antagonistic effects. These studies are crucial to determine whether MOLE can be used safely in conjunction with other therapies or if it could enhance the efficacy of existing treatments for DN. Although the high-glucose-treated rat podocyte model used in this study demonstrates reliable reproducibility and robustness in simulating key pathological changes of DN, it does have certain limitations. First, the model does not fully recapitulate the complexity of human DN, particularly with regard to comorbid conditions such as hypertension. Furthermore, the in vitro nature of the model does not account for the systemic interactions with other cell types and factors that are present in an in vivo environment. Finally, as the study employed a short-term, high-glucose treatment, it cannot fully assess the long-term progression of DN and its effects on kidney function. Future studies may address these limitations by incorporating in vivo models or extending the experimental duration to capture the chronic progression of the disease. Given the promising results observed in this study, the next steps in translating MOLE to potential therapeutic applications for DN include several key stages. First, in vivo validation in DN animal models is necessary to confirm the effectiveness of MOLE in reducing kidney damage, improving renal function, and inhibiting ferroptosis. Mechanistic studies to elucidate the pathways through which MOLE exerts its effects will be crucial for understanding its therapeutic potential. In parallel, formulation development and safety assessments should be conducted to optimize MOLE’s delivery and long-term safety. Finally, preclinical testing followed by clinical trials will be necessary to evaluate MOLE’s safety, efficacy, and therapeutic potential in human DN patients.
Conclusion
MOLE has a protective effect on podocytes in DN rats, and its mechanism may be related to the regulation of ACSL4 protein. Therefore, this study might provide a new direction for DN therapy.
Footnotes
Abbreviations
ACE: Angiotensin-converting enzyme; ACSL4: Acyl-CoA synthetase long-chain family member 4; ANOVA: Analysis of variance; ARBs: Angiotensin receptor blockers; BCA: Bicinchoninic acid; CCK-8: Cell Counting Kit-8; CO2: Carbon dioxide; DAPA: Dapagliflozin; DCFH-DA: 2′,7′-Dichlorofluorescin diacetate; DMEM: Dulbecco’s modified Eagle medium; DN: Diabetic nephropathy; ESRD: End-stage renal disease; FBS: Fetal bovine serum; Fe2+: Ferrous iron; GAPDH: Glyceraldehyde 3-phosphate dehydrogenase; GPX4: Glutathione peroxidase 4; GSH: Glutathione; HG: High glucose; IL-6: Interleukin-6; MDA: Malondialdehyde; MG: Mannitol group; MOL: Moringa oleifera leaves; MOLE: Moringa oleifera leaf extract; NG: Normal glucose; PVDF: Polyvinylidene fluoride; qRT-PCR: Quantitative real-time polymerase chain reaction; RIPA: Radioimmunoprecipitation assay; RNA: Ribonucleic acid; ROS: Reactive oxygen species; SD: Standard deviation; SGLT2: Sodium–glucose cotransporter-2; SPSS: Statistical Package for Social Sciences; STZ: Streptozotocin; T2DM: Type 2 diabetes mellitus; TNF-α: Tumor necrosis factor alpha.
Author Contribution
Conceptualization: ML, HZ.
Data curation: ML, YP, JL, HZ.
Formal analysis: ML, YS, JL, HZ.
Funding acquisition: HZ.
Investigation: ML, HZ.
Methodology: ML, JL, HZ.
Project administration: ML, HZ.
Resources: YP, JL, HZ.
Software: YP, JL, HZ.
Supervision: JL, HZ.
Validation: YP, JL, HZ.
Visualization: YS, YP, HZ.
Writing—Original draft: ML, YS, YP.
Writing—Review & editing: JL, HZ.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
No animal and human participants were involved in this study, and therefore, ethical approval from animal research ethics committee, human research ethics committee and informed consent were not necessary.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
