Abstract
Background
Curcuma phaeocaulis (CP) is utilized as a Chinese herbal medicine to treat abdominal distension and other related ailments. Curcuma phaeocaulis extracts (CPE) are rich in active components and exhibit anti-tumor properties.
Objectives
Purpose: This work was to assess the impact of CPE on the proliferation, apoptosis, migration, and invasion of gallbladder cancer cells (GCC), aiming to investigate its potential underlying mechanisms of action.
Materials and Methods
CP was utilized as the raw material to prepare CPE volatile oil via drying and heating processes. Total phenolic content analysis was conducted, assessing its scavenging capacity against 2,2-diphenyl-1-picrylhydrazyl (DPPH•) and •OH free radicals, as well as its inhibitory effects on the growth of Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa. Human GCC line GBC-SD was selected, cultured conventionally as a control, and treated with CP volatile oil at concentrations of 25 µg/mL (Low-CPE group), 50 µg/mL (Middle-CPE group), and 100 µg/mL (High-CPE group). Cell proliferation activity was evaluated using MTT assay, apoptosis rate was assessed via flow cytometry, migration, and invasion were measured using the Transwell chamber assay, and Western blot was performed to assess proteins in the mTOR/p70S6K signaling pathway.
Results
The total phenolic content in the volatile oil prepared from warm CPE was approximately 30.9 mg/g, consisting of over 20 active constituents. The warm CP volatile oil exhibited scavenging rates of 85.3% for DPPH• radicals and 91.5% for •OH radicals, with minimum inhibitory concentrations against S. aureus, E. coli, and P. aeruginosa at 4.0 mg/mL, 8.0 mg/mL, and 1.0 mg/mL, respectively. Relative to the Ctrl group, Low-CPE, Middle-CPE, and High-CPE groups demonstrated decreased cell proliferation activity, increased apoptosis rates, reduced cell migration and invasion, decreased phosphorylation levels of mTOR and p70S6K proteins (p < 0.05), exhibiting concentration-dependent characteristics.
Conclusion
The volatile oil derived from warm CPE exhibited robust anti-oxidant and anti-bacterial activities. The warm CP volatile oil effectively inhibited GCC proliferation, migration, and invasion by suppressing the activation of the mTOR/p70S6K signaling, while promoting apoptosis.
Introduction
Gallbladder cancer (GC) is a common malignant epithelial tumor (Schmidt et al., 2019), often presenting insidiously with most patients exhibiting no symptoms in the early stages (Hickman & Contreras, 2019). Surgery stands as the primary modality for treating GC, while adjunct therapies such as chemotherapy and radiotherapy have shown the potential to extend the survival period of some patients or ameliorate symptoms (Javle et al., 2019; Ramachandran et al., 2021; Song et al., 2020). Nevertheless, prognosis varies markedly based on various stages of GC tumors. For patients in advanced stages where surgical resection is not feasible, adjunctive therapeutic approaches like chemotherapy have shown limited clinical significance in improving survival rates, necessitating further research in this domain.
Traditional Chinese medicine (TCM) has achieved notable success in the treatment of cancerous diseases, including GC (Sun et al., 2023). Curcuma phaeocaulis (CP) refers to the dried root and stem of the plant Curcuma wenyujin, a member of the Zingiberaceae family. CP is characterized by its pungent, bitter, and warm properties, and it is attributed to the liver and spleen meridians. Its pharmacological actions involve promoting blood circulation, alleviating pain, resolving stagnation, and treating severe conditions related to blood stasis, qi stagnation, and food accumulation (Lin et al., 2024; Salehi et al., 2020). Modern pharmacological studies have confirmed CP’s anti-tumor, anti-oxidant, and anti-bacterial effects, demonstrating its efficacy in treating respiratory, digestive, and circulatory system-related disorders (Wu et al., 2022). The anti-tumor effect of CP extract (CPE) is notably attributed to enhancing the body’s immune response, inhibiting cancer cell proliferation and migration, as well as inducing apoptosis in cancer cells (Sun et al., 2022; Xu et al., 2019). Feng et al. (2021) established an in vitro co-culture model using colon cancer cells and human umbilical vein endothelial cells, demonstrating that CP volatile oil can inhibit the proliferation of colon cancer cells and endothelial cells, as well as suppress the expression of angiogenesis-related factors in endothelial cells, indicating its ability to hinder colon cancer angiogenesis. Chen et al. (2011) discovered that alcohol-extracted CP can inhibit breast cancer cell proliferation and, in a dose-dependent manner, reduce the mitochondrial membrane potential of the cells. Moreover, apart from these effects, CPE also demonstrates potent anti-bacterial, anti-inflammatory, and anti-viral properties. Nam et al. (2022) found that CP can inhibit the activation of the NOD-like receptor domain-containing protein 3 (NLRP3) inflammasome, thereby exerting an anti-inflammatory therapeutic effect. CPE also exhibits hepatoprotective effects. Gao et al. (2024) utilized network pharmacology to predict CP’s application in treating liver injury, identifying 1,023 genes associated with liver damage, predominantly functioning through the TLR4/MyD88/NF-κB pathway to ameliorate liver injury. These findings collectively indicate the multifaceted pharmacological actions of CP.
Nevertheless, the anti-GC effects of CPE remain unclear. To address this, warm CP volatile oil was prepared, and its anti-oxidative and anti-bacterial activities were initially analyzed. Subsequently, by using the human gallbladder cancer cells (GCC) line GBC-SD as the research subject, the impact of warm CP volatile oil on cell proliferation, apoptosis, migration, and invasion was investigated, concurrently assessing the status of the mTOR/p70S6K signaling pathway. This work aimed to provide research data for further exploration of the pharmacological actions of CP and to analyze its mechanisms underlying anti-GC effects.
Materials and Methods
Preparation and Analysis of CPE
Preparation of CP Volatile Oil
About 100 g of dried warm CP sample (Huozhentang Pharmaceutical Co., Ltd., Bozhou, China) was coarsely powdered and sieved through a 50-mesh screen. The sample was soaked in distilled water at eight times its mass for 1 h, after which it was boiled, and water vapor was distilled for 8 h. The oil from the oil collector was drawn using a pipette and placed in a beaker, followed by the addition of anhydrous sodium sulfate (Sigma–Aldrich, USA) to remove water. Petroleum ether (Dongguan Xunye Chemical Reagent Co., Ltd., China) was utilized to rinse the pipette and oil collector (Shanghai Yuanhuai Intelligent Technology Co., Ltd., China), and the mixture was left until the petroleum ether evaporated. After multiple extractions, the obtained CP volatile oil sample was stored in a sealed container at 4°C. The preparation process of CPE is shown in Figure 1.
Technical Roadmap.
Gas Chromatography–Mass Spectrometry (GC–MS) Analysis
Ten milligrams of CP essential oil were analyzed using a GC–MS-QP2020 NX gas chromatography–mass spectrometry system (Shimadzu Corporation, Japan) (Simeonova et al., 2022). The analysis employed an HP-5MS capillary column (30 m × 250 µm × 0.25 µm) with helium as the carrier gas at a flow rate of 0.95 mL/min. A sample volume of 1 µL was injected with a split ratio of 80:1. The initial temperature was set at 45°C and maintained for 2 min, followed by a temperature ramp of 4°C/min to a maximum temperature of 250°C, which was held for an additional 2 min. The electron impact source operated at an electron energy of 70 eV, with an ion source temperature of 200°C and a scan range of 20–400 amu.
Total Phenolic Content Detection
The total phenolic content test in CP volatile oil was conducted using the Folin-Ciocalteu methodology (Carmona-Hernandez et al., 2021). About 2.5 mg gallic acid standard (Sigma–Aldrich, USA) was dissolved in 25 mL distilled water to form a 0.1 mg/mL standard solution. Then, 0.0, 0.2, 0.4, 0.6, 0.8, and 1.0 mL standard solution were taken, mixed with 6 mL distilled water, supplemented with 0.5 mL Folin-Ciocalteu reagent (Guangzhou Hewei Pharmaceutical Technology Co. Ltd., China) and 1.5 mL 20% Na2CO3 reagent (Tianjin Jinhui Taiya Chemical Reagents, China). The mixture reacted in the dark at 25°C for 2 h, and absorbance was measured at 760 nm wavelength. Additionally, 1 mL of the CP volatile oil solution was mixed with 6 mL distilled water, supplemented with 0.5 mL Folin-Ciocalteu reagent and 1.5 mL 20% Na2CO3 reagent. The mixture was reacted in the dark at 25°C for 2 h, and absorbance was measured at 760 nm. The total phenolic content in CP volatile oil was calculated.
Antioxidant Activity Test
According to the instruction of 2,2-diphenyl-1-picrylhydrazyl (DPPH) (Sigma–Aldrich, USA), a total of 2 mL of CP volatile oil methanol solution at various concentrations was mixed with 1 mL of 0.2 mM DPPH• methanol solution, thoroughly blended, and left in the dark for 30 min. The V-5800 UV–vis spectrophotometer (Shanghai Yuanxi Instrument Co., Ltd., China) was employed to detect the optical density at 517 nm. The methanol solution of CP volatile oil without DPPH• served as a blank control, while vitamin C was utilized as a positive control. DPPH• free radical scavenging rate was calculated.
A total of 0.2 mL of a mixture containing 3 mM FeSO4-ethylenediaminetetraacetic acid, 0.5 mL of 5 mM deoxyribose (Chengdu Biyang Phytochemicals Ltd., China), 1.5 mL of CP volatile oil methanol solution (Tianjin Concord Technology Co., Ltd., China) at various concentrations, 0.6 mL of 20 mM phosphate buffer (pH = 7.4), and 0.2 mL of 3 mM H2O2 solution were thoroughly mixed and subjected to a water bath at 37 °C for 1 h. Subsequently, 1 mL of 2.8% (w/v) trichloroacetic acid solution and 1 mL of 1% thiobarbituric acid solution were applied, and the reaction was maintained in a boiling water bath for 15 min. After cooling, absorbance was measured at 532 nm. Methanol solution of CP volatile oil without the aforementioned reagents served as a blank control, while vitamin C was utilized as a positive control. The •OH free radical scavenging rate was calculated.
Anti-bacterial Activity Determination
CP volatile oil was dissolved in 1% Tween-80 Mueller-Hinton Broth (MHB) culture medium (Qingdao Jisskang Biotechnology Co., Ltd., China) to create a dilution series of 0.125, 0.25, 0.5, 1.0, 2.0, 4.0, 8.0, 16.0, 32.0, and 64.0 mg/mL concentrations. ATCC43300 Staphylococcus aureus, ATCC25922 Escherichia coli, and ATCC9027 Pseudomonas aeruginosa (China General Microbiological Culture Collection Center, China) were inoculated on fresh Mueller-Hinton Broth culture medium and cultured at a constant temperature of 37°C for 24 h in LRH-150 biochemical incubator (Nanbei Instrument Co., Ltd., China). After bacterial activation, the bacteria were inoculated into sterile physiological saline using an inoculation loop to prepare a bacterial suspension with a McFarland turbidity of 0.5, containing bacterial counts of 105–106 CFU/mL. Subsequently, 200 µL of the diluted CP volatile oil solution was applied to a 96-well plate, then added with 50 µL of the bacterial suspension, thoroughly mixed, and then cultured at a constant temperature of 37°C for 24 h. The bacteria growth status was observed, and the lowest concentration of CP volatile oil that showed a clear culture medium, indicating inhibition of bacterial growth, was determined as the minimum inhibitory concentration (MIC).
Effects of CPE on the Biological Behavior of GCC
GCC Culture and Grouping
Human GCC line GBC-SD (Shanghai Yaji Biological Technology Co., Ltd., China) were cultured in Dulbecco’s Modified Eagle Medium (Gibco, USA) and 10% fetal bovine serum (Gibco, USA) at 37°C with 5% CO2 until reaching approximately 90% confluency in VS-80Q CO2 constant temperature incubator (Wuxi Voxin Instrument Co., Ltd., China). Cells (all cells used in this research were in the logarithmic growth phase) were seeded at 1 × 104 cells/well in a 96-well plate and then subjected to grouped cultivation. The Ctrl group was cultured in a normal medium. GBC-SD cells were rolled into Low-CPE, Middle-CPE, and High-CPE groups. Ctrl group cells were cultured in a normal medium. The Low-CPE, Middle-CPE, and High-CPE groups were cultured in a medium containing 25, 50, and 100 µg/mL CP volatile oil, respectively. Each group was repeated three times.
3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) Assay for Detecting Cell Proliferation
GBC-SD cells in the logarithmic growth phase at 4 × 105 cells/well in a 6-well plate were subjected to grouping. According to the instruction of the MTT cell proliferation assay kit (Sigma–Aldrich, USA), after conventional culture for 24, 48, 72, and 96 h, 20 µL of 0.5% MTT reagent was applied to each well, followed by an additional 4-h incubation. The original solution was then removed. Cells were rinsed twice with phosphate-buffered saline, after which 150 µL of dimethyl sulfoxide (Hubei Xingfa Chemicals Group Co., Ltd., China) was applied to each well and shaken for 10 min. The absorbance of each well was measured using a SpectraMax iD3 multimode microplate reader (Zhejiang Meigu Biological Technology Co. Ltd., China) at 490 nm. This process was repeated thrice for each group, the average was calculated, and the cell proliferation activity was determined.
Detection of Cell Apoptosis by Flow Cytometry (FCM)
GBC-SD cells (1 × 104 cells/well in a 96-well plate) were subjected to grouping. As per the instruction for Annexin V-FITC/PI cell apoptosis assay kit (Solarbio, USA), after conventional culture for 48 h, 5 µL of Annexin V-FITC reagent and 5 µL of PI reagent were applied to each well, mixed thoroughly, and incubated in the dark for 15 min. Standard Bio Tools flow cytometer (Shanghai Standard Bio Tools Instrument Technology Co., Ltd., China) was immediately performed to assess the apoptosis rate of cells in each group. This process was repeated thrice, and the mean values were calculated.
Transwell Assay for Detecting Cell Migration and Invasion
Log-phase GBC-SD cells were collected, rinsed twice with a serum-free culture medium, and resuspended. The diluted Matrigel matrix gel (Sigma–Aldrich, USA) was applied to the Transwell upper chamber for invasion assays, while the non-coated insert was utilized for migration assays; the remaining steps were identical. Each lower chamber of the Transwell was filled with 600 µL of drug-containing or drug-free culture medium, while 200 µL of cell suspension was applied to the upper chamber. The cells were conventionally cultured for 24 h. After the medium was removed from the upper chamber, the cells were fixed with methanol for 15 min and stained with crystal violet staining solution (Beijing Solarbio Technology Co. Ltd., China) for 30 min. A sterile cotton swab was utilized to remove non-migrated cells, and the migrated cells were visualized and counted under an IX73 inverted microscope (Olympus, Japan).
Western Blotting
GBC-SD cells in the logarithmic growth phase at 4 × 105 cells/well in a 6-well plate were subsequently subjected to grouping. After conventional culturing for 48 h, cells from each group were collected and lysed using radioimmunoprecipitation assay lysis buffer (Beijing Solarbio Technology Co., Ltd., China). Protein concentration was measured using a bicinchoninic acid protein quantification assay kit (Shanghai Beyotime Biotechnology Co., Ltd., China). Following denaturation at high temperatures, proteins were separated via SDS-PAGE gel electrophoresis. Post-transfer, membranes were blocked and incubated overnight at 4°C with the respective primary anti-bodies against mTOR (1:2,000), p-mTOR (1:2,000), p70S6K (1:2,000), p-p70S6K (1:2,000), and GAPDH (1:2,000) (Shanghai Aibo Antibody Trade Co., Ltd., China). After washing thrice, horseradish peroxidase-conjugated secondary IgG anti-bodies (1:5,000) (Shanghai Aibo Antibody Trade Co., Ltd., China) were applied and left to incubate at 25°C for 1 h. After another three washes, the electrochemiluminescence solution (Shanghai Beyotime Biotechnology Co., Ltd., China) was initiated using an electrochemiluminescence solution. In addition, the WD-9403C gel imaging system (Beijing Liuyi, China) was adopted. The ImageJ was utilized for quantitative analysis of target band relative grayscale values with GAPDH as the reference.
Statistical Analysis
Using SPSS 22.0 (IBM, USA), all data were normally distributed and denoted as mean ± standard deviation. The t-test was employed for between-group comparisons, while the one-way analysis of variance for multiple-group comparisons. p < 0.05 was statistically considerable.
Results
Identification of Components in CP Volatile Oil
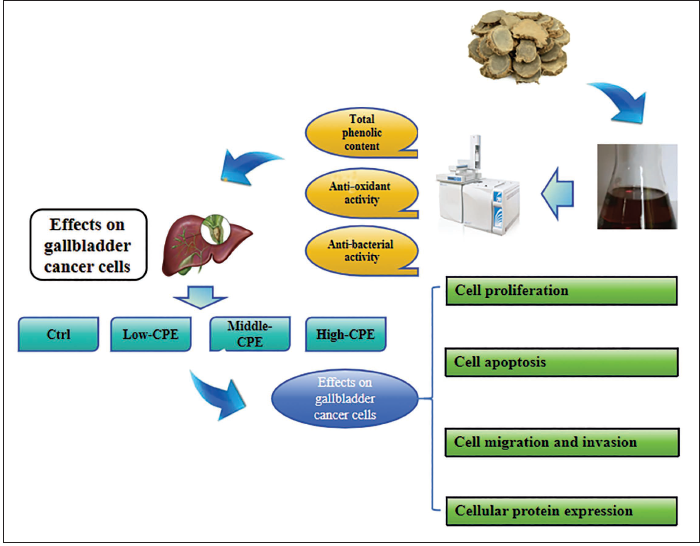
The analysis of CP volatile oil derived from CP was conducted using GC–MS technology, and a total ion chromatogram was generated (Figure 2). Analysis revealed the presence of 25 components in the CP volatile oil. The major constituents identified were curdione (33.5%), curzerene (23.4%), germacrone (16.2%), β-elemene (7.8%), neocurdione (3.7%), and atractylone (2.8%). The determination of total phenolic content in the warm CP volatile oil using the gallic acid standard curve indicated a content of approximately 30.9 mg/g. Notably, warm CP also contains a rich array of phenolic compounds.
Anti-oxidant Activity Analysis of CP Volatile Oil
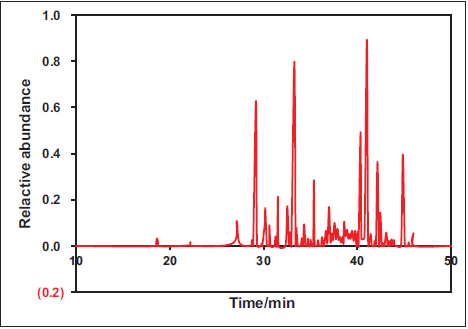
The alterations in scavenging rates of DPPH• and •OH radicals at various concentrations of CP volatile oil were analyzed, and concentration-radical scavenging rate curves were plotted (Figure 3). With Vc as the positive control, it was observed that the scavenging rates of both DPPH• and •OH radicals gradually increased with the escalating concentration of CP volatile oil. At a concentration of 5 mg/mL, the scavenging rate against DPPH• radicals gradually stabilized, while at 1 mg/mL, the scavenging rate against •OH radicals reached a steady state. The efficacy of CP volatile oil in clearing DPPH• and •OH radicals was found to be highly similar to that of Vc.
Anti-bacterial Activity Analysis of CP Volatile Oil
The inhibitory effect of various concentrations of warm CP volatile oil on the growth of bacterial strains was analyzed (Table 1). The MICs of warm CP volatile oil against S. aureus, E. coli, and P. aeruginosa were found to be 4.0 mg/mL, 8.0 mg/mL, and 1.0 mg/mL, respectively.
MICs of Warm CP Volatile Oil against Pathogens.
Effect of CP Volatile Oil on GCC Proliferation
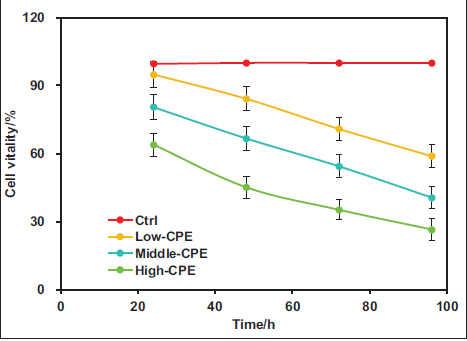
First, the impact of warm CPE on the proliferation activity of GC-related GBC-SD cells was assessed using the MTT assay, and the cell proliferation activity curve was plotted, as depicted in Figure 4. The proliferation activity of cells in the Ctrl group remained around 100%. Nevertheless, in Low-CPE, Middle-CPE, and High-CPE groups, cell proliferation activity decreased with increasing concentrations. The proliferation activity markedly decreased in the Low-CPE, Middle-CPE, and High-CPE groups versus the Ctrl group (p < 0.05). Moreover, the proliferation activity was notably lower in the Middle-CPE and High-CPE groups versus the Low-CPE group (p < 0.05). Additionally, relative to the Middle-CPE group, the High-CPE group exhibited markedly reduced cell proliferation activity (p < 0.05).
Effect of CP Volatile Oil on GCC Apoptosis
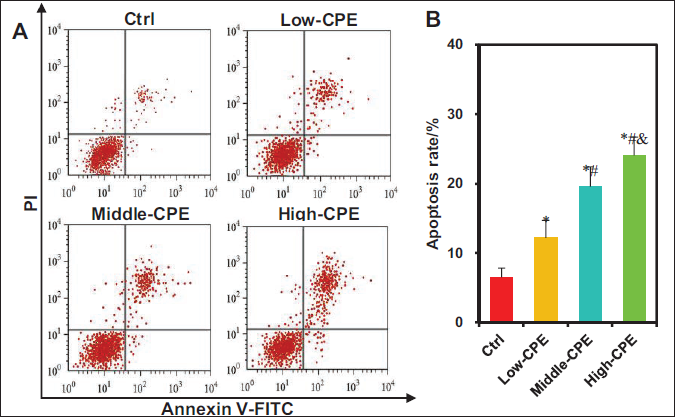
The impact of warm CP volatile oil on apoptosis in GBC-SD cells was evaluated using FCM (Figure 5). The apoptosis rates markedly increased in the Low-CPE, Middle-CPE, and High-CPE groups versus the Ctrl group (p < 0.05). Moreover, the Middle-CPE and High-CPE groups exhibited markedly elevated apoptosis rates relative to the Low-CPE group (p < 0.05). Similarly, the High-CPE group demonstrated a substantial rise in apoptosis rates versus the Middle-CPE group (p < 0.05).
Effect of CP Volatile Oil on GCC Migration and Invasion
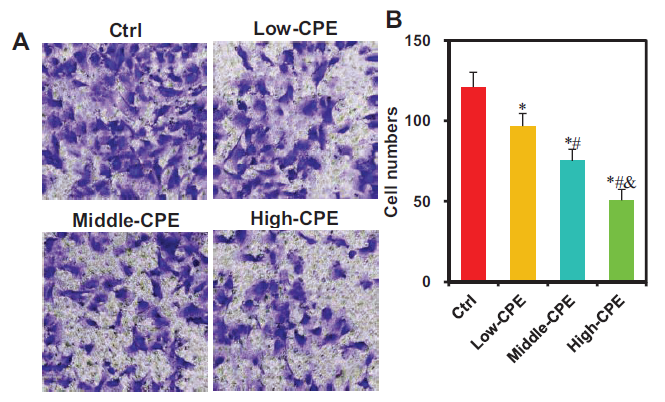
The impact of warm CP volatile oil on the migration of GBC-SD cells was assessed using the Transwell chamber (Figure 6). Relative to the Ctrl group, the number of migrating cells markedly decreased in Low-CPE, Middle-CPE, and High-CPE groups (p < 0.05). Additionally, relative to the Low-CPE group, Middle-CPE and High-CPE groups exhibited a considerable decrease in migrating cells (p < 0.05). Furthermore, the High-CPE group displayed a considerable reduction in migrating cells versus the Middle-CPE group (p < 0.05).
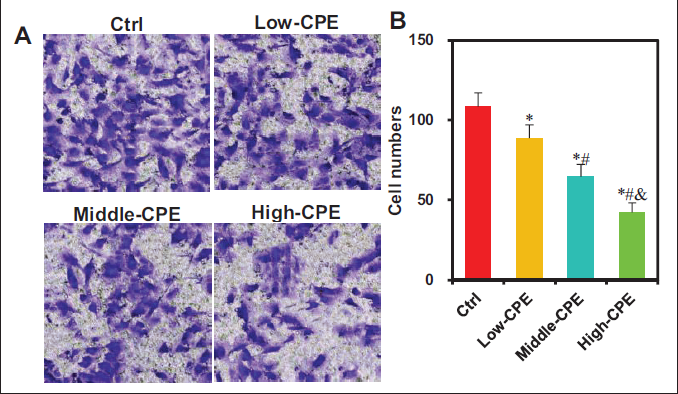
The effect of warm CP volatile oil on the invasion of GBC-SD cells was examined using the Transwell chamber assay (Figure 7). The numbers of invasive cells were markedly reduced in the Low-CPE, Middle-CPE, and High-CPE groups relative to the Ctrl group (p < 0.05). The Middle-CPE and High-CPE groups exhibited a drastic decrease in the number of invasive cells versus the Low-CPE group (p < 0.05). Moreover, the High-CPE group showed a notable reduction in the number of invasive cells versus the Middle-CPE group (p < 0.05).
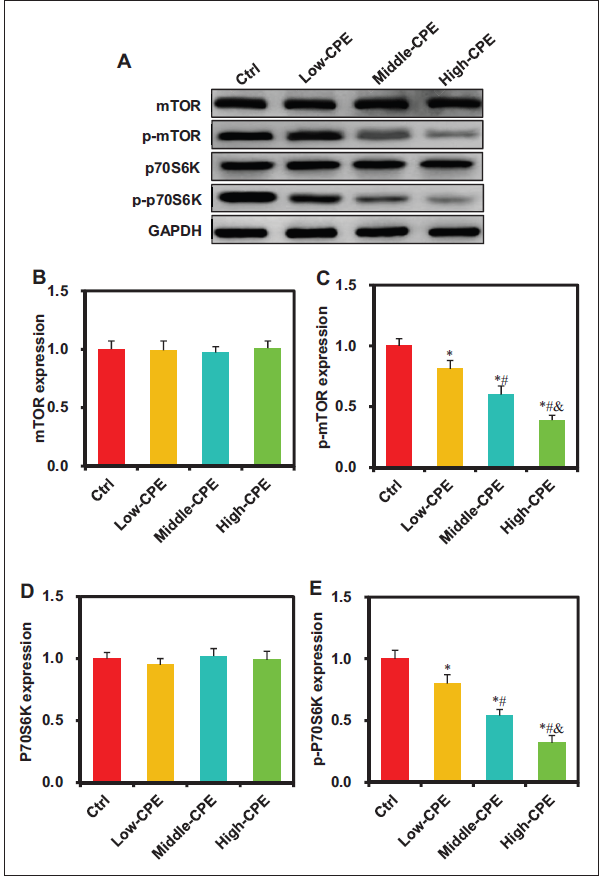
The Effect of CP Volatile Oil on the mTOR/p70S6K Signaling in GCC
The impact of CP volatile oil on target protein levels in mTOR/p70S6K signaling of GC GBC-SD cells was assessed using Western blot analysis (Figure 8). No marked differences were observed in mTOR and p70S6K protein levels among Ctrl, Low-CPE, Middle-CPE, and High-CPE groups (p > 0.05). The p-mTOR and p-p70S6K levels in Low-CPE, Middle-CPE, and High-CPE groups exhibited considerable decreases versus the Ctrl group (p < 0.05). In comparison with the Low-CPE group, Middle-CPE and High-CPE groups demonstrated markedly reduced p-mTOR and p-p70S6K expression (p < 0.05). Moreover, when compared with the Middle-CPE group, the High-CPE group exhibited markedly decreased p-mTOR and p-p70S6K expression (p < 0.05).
Discussion
CP is a commonly used TCM, and its main bioactive components have various pharmacological effects. In this study, CPE was prepared, and the analysis revealed that curdione, curzerene, and germacrone were among the highest in content, with a total phenol content as high as 30.9 mg/g. Zhao et al. (2023) demonstrated that turmeric crude extract contains seven major active phytochemical components, namely, curcumol, β-elemene, furanodiene, furanodienone, germacrone, curdione, and curcumin. Turmeric has pharmacological properties such as inhibiting the proliferation, migration, and invasion of breast cancer cells and reversing resistance to chemotherapy drugs (Zhao et al., 2023). Plant phenolic compounds in TCM materials exhibit a wide range of pharmacological activities, such as enhancing immunity, promoting blood circulation, reducing blood pressure, and anti-cancer effects (Coc et al., 2022; Freitas et al., 2022; Santos-Buelga et al., 2019; Schieber & Wüst, 2020). The similarity of active components in the extracts of CP and turmeric, along with the rich phenolic compounds, suggests that CP may also have strong anti-tumor activity. Furthermore, this study analyzed the scavenging effects of CPE on DPPH• and •OH free radicals to evaluate their anti-oxidant activity, and the results showed a similar scavenging effect to Vitamin C. Zhao et al. (2010) also found that CP contains abundant anti-oxidant sesquiterpene compounds, which can effectively eliminate DPPH• free radicals. Repeated occurrences of oxidative stress in the body can lead to internal and external disruptions, possibly causing immune system dysfunction and accelerating the growth and spread of cancer cells (Singh et al., 2019). Therefore, it is hypothesized that CPE may play a role in inhibiting cancer progression by suppressing cellular oxidative stress.
GC is a highly common malignant tumor of the digestive system. Due to its insidious onset, most patients are diagnosed in the middle to late stages of the disease, with a 5-year survival rate of only around 5% (Sung et al., 2022). Radical surgery is an effective method for treating GC, but for some patients who cannot undergo resection, the prognosis is less than ideal even after receiving chemotherapy or radiotherapy (Krell & Wei, 2019). CP, a TCM, contains various effective active ingredients and exhibits excellent anti-tumor activity. CPE contains multiple active components, with β-elemene belonging to sesquiterpene compounds. It participates in regulating tumor angiogenesis and metastasis has immunomodulatory effects, enhances cancer cell sensitivity to radiotherapy and chemotherapy, and improves the multidrug resistance of cancer cells (Zhai et al., 2019). This work found that warm CPE can significantly inhibit the proliferation, migration, and invasion of GCC, inducing cell apoptosis. Li et al. (2014) discovered that CP can dose-dependently inhibit tumor growth, increase the expression of pro-apoptotic proteins, and inhibit the expression of anti-apoptotic proteins, thereby suppressing tumor cell proliferation and promoting apoptosis. Hermansyah et al. (2023) found that CP can inhibit the proliferation of triple-negative breast cancer cells and reduce cell invasion and migration. This indicates that CPE can effectively inhibit the invasion of GCC, showing concentration-dependent characteristics. In summary, the diverse bioactive compounds in CPE have anti-cancer effects, and they also demonstrate a strong anti-GC effect.
mTOR belongs to the class of protein kinases, and it is closely associated with the occurrence and development of various cancer cells. mTOR primarily regulates cell growth and proliferation through two signaling pathways: the PI3K/Akt/mTOR pathway and the Akt/TSC1-2/mTOR/p70S6K pathway (Kenerson et al., 2007). Studies have confirmed that the mTOR/p70S6K signaling pathway, when activated by upstream growth factor receptors and insulin receptors, can inhibit apoptosis, accelerate the cell cycle, promote tumor angiogenesis, and facilitate the invasion and metastasis of tumor cells (Heinonen et al., 2008; Jiang et al., 2018). The mTOR signaling pathway plays a crucial role in the malignant transformation of normal cells into cancer cells. Chen et al. (2021) found that the PI3K/Akt/mTOR signaling pathway is abnormally activated in GCC, and inhibiting this pathway activation can suppress cancer cell growth and induce apoptosis. Han et al. (2018) discovered that the mTOR/p70S6K signaling pathway is abnormally activated in cancer cells, and inhibiting this pathway activation can suppress cancer cell growth and induce mitochondria-mediated autophagy-related apoptosis. In this work, it was observed that the expression levels of p-mTOR and p-P70S6K proteins in GCC significantly decreased after treatment with warm CPE. This indicates that warm CPE can inhibit the activation of the mTOR/p70S6K signaling pathway, thereby exerting an inhibitory effect on the proliferation, migration, and invasion of GCC, and inducing apoptosis.
The findings of this study contribute to the understanding of the pathogenesis of gallbladder cancer, elucidate the anti-cancer mechanisms of CPE, and provide a reference for the development of novel therapeutic agents for gallbladder cancer. However, this study also has certain limitations; specifically, it only analyzed the anti-cancer effects of CP essential oil at the in vitro cellular level. Future research should involve the establishment of gallbladder cancer xenograft animal models to further investigate its effects on tumor behavior and angiogenesis.
Conclusion
The essential oil of warm CP contains various bioactive components and exhibits strong anti-oxidant and anti-bacterial properties. This essential oil can inhibit the proliferation, migration, and invasion of human GBC-SD, induce apoptosis, and demonstrate a concentration-dependent effect. Furthermore, the essential oil regulates the malignant biological characteristics of GCC by suppressing the abnormal activation of the mTOR/p70S6K signaling pathway.
Footnotes
Abbreviations
CP: Curcuma phaeocaulis; CPE: Curcuma phaeocaulis extracts; DPPH: 2,2-Diphenyl-1-picrylhydrazyl; GCC: Gallbladder cancer cells; MIC: Minimum inhibitory concentration; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; NLRP3: NOD-like receptor domain-containing protein 3; TCM: Traditional Chinese medicine.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
None.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 82270695) and the Key Research and Development Program Project of Shaanxi Province (No. 2023-YBSF-371).
