Abstract
Background
Excessive levels of oxidative radicals injure organs by directly reacting with macromolecules [proteins, lipids, and deoxyribonucleic acid (DNA)]. Antioxidant supplements with compounds rich in antioxidants, such as essential oils, combat oxidative radicals.
Objectives
This investigation aimed to assess the theory that supplementation with thyme essential oil (TEO) and basil essential oil (BEO) lessen the harmful consequences of hepatic oxidative stress that occur when hydrogen peroxide (H2O2) is added to drinking water.
Materials and Methods
Rats were randomly divided into four groups: normal control (NC) received a basal diet; positive control (PC) was intraperitoneally injected with a single dose of 3% H2O2; and TEO or BEO groups were PC groups received 200 mg/kg of TEO or BEO for 30 days.
Results
BEO demonstrated a higher capacity for radical scavenging than TEO in 2,2-diphenyl-1-picrylhydrazyl (DPPH) and H2O2 assays. Treatment with BEO and TEO significantly improved various liver functions, including total protein, total bilirubin, albumin, and liver enzymes [alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transferase (GGT), and alkaline phosphatase (ALP)] with BEO preferred over TEO. TEO and BEO administration significantly reduced oxidative stress and inflammation by lowering malondialdehyde (MDA) values and enhancing superoxide dismutase (SOD), glutathione peroxidase (GPx), and catalase (CAT) activities, and maintaining normal levels of myeloperoxidase (MPO) and nitrite production in H2O2-treated rats. TEO and BEO also repaired the deformed liver structure to the normal.
Conclusion
These results provide critical insight into how thyme and basil essential oils might protect the liver from H2O2-induced oxidative stress and inflammation.
Introduction
Reactive chemicals, including reactive oxygen species (ROS), reactive nitrogen species (RNS), and reactive sulfur species (RSS), play crucial roles in cellular damage, particularly in hepatocytes, which are central to detoxification in the liver. These reactive species are highly unstable due to unpaired electrons, which make them capable of interacting with cellular components, leading to oxidative and nitrosative stress. ROS, such as superoxide anions (O₂•⁻), hydrogen peroxide (H2O2), and hydroxyl radicals (•OH), are primarily generated during mitochondrial respiration and various metabolic processes (El-Shazly et al., 2024). Excess ROS production can lead to lipid peroxidation, protein oxidation, and deoxyribonucleic acid (DNA) damage, compromising cell membrane integrity and inducing apoptosis or necrosis (Jaeschke &Ramachandran, 2018). RNS, including nitric oxide (NO) and peroxynitrite (ONOO⁻), are reactive nitrogen-containing molecules that can modify proteins through nitrosylation, altering their function. High levels of RNS disrupt mitochondrial function and adenosine triphosphate (ATP) production, leading to energy deficits and cell death in hepatocytes (Tejero et al., 2019). Similarly, RSS, such as hydrogen sulfide (H2S) and sulfite radicals, can interfere with protein and enzyme function through thiol modification. Though RSS can have physiological signaling roles, their excessive accumulation is toxic, further damaging hepatocyte proteins and membranes (Filipovic, 2015). Overall, the imbalance between reactive species and the antioxidant defense mechanisms, such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), promotes oxidative stress, which is a key driver in hepatocellular injury. This oxidative damage is implicated in liver diseases, including hepatitis, fibrosis, and cirrhosis (Cichoż-Lach &Michalak, 2014). By understanding these mechanisms, researchers can better appreciate the role of oxidative stress in liver pathology and the importance of antioxidant systems in hepatocyte protection.
One major risk factor for human health is thought to be oxidative stress due to the serious diseases it causes, such as liver cirrhosis, kidney dysfunction, cancer, and atherosclerosis (Abass et al., 2024; Diab et al., 2024). H2O2 is commonly used in the study of oxidative damage and increased oxidative stress in experimental animals (Abozid, 2013). The liver is important for the detoxification and elimination of drugs. Therefore, xenobiotics such as alcohol and a variety of drugs can cause hepatic damage. Liver damage is a common condition that primarily results from oxidative stress and involves the progression of steatosis to liver cancer (El-Magd et al., 2022; Elmetwalli et al., 2023). One common biomarker for the state of peroxidation of lipids is malondialdehyde (MDA) (Attia et al., 2022). The hepatic antioxidant defense system is made up of SOD, CAT, and GPx. Its functions include reducing oxidative cell damage and regulating redox homeostasis (Mohamed et al., 2021). Intracellular lipids are shielded from peroxidation by the enzyme GPx, while tissues are protected against oxidation by CAT and SOD. Increased MDA concentrations and decreased GPx, CAT, and SOD activities were utilized to determine the severity of hepatocyte damage (El-Demerdash et al., 2021; El-Magd et al., 2019). In animal experiments, antioxidant-rich diets were linked to improved redox states and some tissue functions that were oxidatively damaged (Abdel-Aleem et al., 2019; Abu Gazia &El-Magd, 2018; Abu Khudir et al., 2019).
Many researchers have emphasized the significance and efficiency of essential oils (EO) as a natural antioxidant that can be obtained from food (Abu El-Hamd et al., 2021; Ademiluyi et al., 2016; Elsied et al., 2023). A wide and varied class of plant secondary metabolites, including amines, sulfides, phenolic compounds, and terpenoids, combine to produce EO (Avetisyan et al., 2017). Thymus linearis Benth., a medicinal plant used traditionally, contains valuable bioactive compounds (Thakur et al., 2023). Thyme and basil are common spices or daily drinks in the Middle East (Avetisyan et al., 2017). Thyme essential oil (TEO) and basil essential oil (BEO) are characterized by containing a large amount of thymol and
Numerous in vivo and in vitro studies have been conducted on the antioxidant effects of TEO and BEO or plant extracts containing these oils (Avetisyan et al., 2017; Diniz do Nascimento et al., 2020; El-Boshy et al., 2019; Emami et al., 2010; Miguel, 2010; Ramadan et al., 2022), nevertheless, as far as we know, there have been no studies to evaluate the role of these EO in protecting the liver from H2O2-induced oxidative stress. Therefore, the purpose of this study was to investigate the antioxidant activity of TEO and BEO, and, more importantly, to assess their potential role in preventing H2O2-induced liver damage in rats.
Materials and Methods
Plant Oils
Ocimum basilicum (Basil), Thymus vulgaris (thyme), and EO were obtained from Fifa Herbalist Shop, Jeddah, Saudi Arabia.
Gas Chromatography–Mass Selective (GC–MS) Assessment of TEO and BEO
The GC–MS examination was executed utilizing an Agilent 5975 gas chromatography–mass selective detector (GC–MSD) system with (60 m × 0.25 mm, 0.25 µm film thicknesses) Innowax free-standing capillary (FSC) column. Helium (He) (0.8 mL/min) was utilized as a carrier gas. The oven was set to reach 220°C at a rate of 4°C/min after being at 60°C for 10 min. After being held steady at 220°C for 10 min, the temperature was raised to 240°C at a rate of 1°C per min. With a mass range of m/z 35–450, mass spectra were recorded at 70 eV. TEO and BEO components were identified by comparing their retention periods with reference samples or their relative retention indices to a series of n-alkanes. The components of the EO were defined through computer reference with commercial libraries (Wiley GC/MS, Adams, and Mass Finder 2.1) and internal libraries, “Baser Library of Essential Oil Constituents,” which was compiled by real components and known oil components, furthermore, mass spectrometry (MS) literature data (Adams, 2001).
2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Scavenging Assay
The DPPH radical scavenging capacity (RSC) was assessed utilizing a methodology made by Karamać et al. (2005). RSC is calculated as follows: RSC (%) = (Ablank − Asample)/Ablank × 100, whereas Ablank is the absorbance of DPPH in which the sample has been substituted with methanol, and Asample is DPPH absorbance combined with EO.
H2O2 Scavenging Assay
The scavenging activity of H2O2 was calculated in accordance with Ruch et al. (1989). The scavenging activity percentage was calculated as follows: Scavenging of H2O2 (%) = (1 – [Asample/Acontrol]) × 100, Acontrol and Asample are the absorbance of control and sample, respectively.
Oxygen Radical Absorbance Capacity (ORAC) Assay
The ORAC assay was conducted to detect the antioxidant potential of TEO and BEO in accordance with Bentayeb et al. (2014). To summarize, 100 µL of antioxidant solution was combined with 800 µL of fluorescein solution. To initiate the radical reaction, 600 µL of the 2,2′-azobis(2-methylpropionamidine) dihydrochloride (AAPH) solution was subsequently added. Using excitation and emission wavelengths of 540 and 565 nm, respectively, a 474 Scanning Fluorescence Detector (Waters, Milford, MA, USA) was used to observe the fluorescence. The antioxidant capacity of the two EO was presented as g Trolox/g essential oil (gT/gEO).
NO Scavenging Activity Assay
The Griess reagent method was used to study the NO scavenging capacity of TEO and BEO as previously detailed (Fandakli et al., 2021). The various concentrations of the two oils were incubated at 25°C for 150 min in 1 mL sodium nitroprusside in pH 7.4 phosphate-buffered saline. Then, half a milliliter of Griess reagent, which consisted of 1% sulphanilamide, 0.1% naphthyl ethylenediamine dihydrochloride, and 2.5% phosphoric acid, was added. To determine NO scavenging capability, the optical density (OD) (measured at 540 nm) values of the control and reaction mixture were quantified to compute the NO% scavenging capability of the extracts and gallic acid. Sodium nitroprusside was used as a NO donor in an in vitro study of compounds with a regulatory impact on NO concentration. The Griess reagent can be used to detect nitrite ions, which are formed when NO is transformed under aerobic conditions.
Experimental Design
Thirty-two male Albino Wistar rats (weighing between 140 and 180 g) were used for this investigation. Rats, obtained from the experimental animal unit of the Faculty of Pharmacy, King Abdulaziz University, Jeddah, Saudi Arabia, were housed in cages with wood shavings for bedding. They were randomly allocated into the following four groups (8 rats/group). Normal control (NC) without any treatments. Positive control (PC) received a single intraperitoneal (IP) injection of 3 mL/kg b.w of 5% v/v of H2O2 (Okagu et al., 2020). The TEO-treated group was injected with H2O2 as in the PC group and given 200 mg/kg b.wt TEO. The BEO-treated group was injected with H2O2 as in the PC group 200 mg/kg b.wt BEO. BEO and TEO were administered via stomach tube every day for 30 days. The doses of BEO and TEO were selected as 1/10 LD50 of these oils as evidenced by our pilot study. The animal study was reviewed and approved by the IRRB-ER/EC committee and was performed following the guidelines of the Implementing Regulations of the Law of Ethics of Research on Living Creatures in the Kingdom of Saudi Arabia and other applicable national and international regulations. The Research Ethics Committee Reference Number IRRB-ER/EC-20102024.
Blood and Liver Samples
Following a 30-day treatment period, blood samples were collected from the orbital sinus veins using heparinized capillary tubes. These samples were then transferred into 1.5 mL Eppendorf tubes. Heparin was present in the tubes as an anti-coagulant. The samples were centrifuged at 2,000 g for 15 min to separate the plasma. Plasma samples were kept in a deep freezer at −20°C until use. The animals underwent anesthesia, an overnight food fast, and cervical decapitation as a means of sacrifice. The removed liver samples were split into two portions: the first was homogenized in a 50 mM Tris–KCl buffer at pH 7.4 with 1.15% KCl, centrifuged at 12,000 g for 15 min at 4°C (for biochemical tests), and the second was fixed in 10% formalin (for histopathology).
Determination of Liver Function Parameters
The method outlined by Young (1997) was utilized to estimate aspartate aminotransferase (AST) and alanine aminotransferase (ALT) activities. The activity of gamma-glutamyl transferase (GGT) was measured in accordance with Shaw et al. (1983). Alkaline phosphatase (ALP) activity was assessed in accordance with Statland (1979). Total bilirubin was measured according to Perry et al. (1983). The albumin level was assessed according to Henry (1964), and total protein was calculated using the methodology of Schultze and Heremans (1966).
Assessment of Antioxidant and Oxidative Stress Biomarkers
The content of MDA was determined according to the method of Ohkawa et al. (1979). The activities of CAT, GPx, and SOD were determined as previously described (Aebi, 1984; Nishikimi et al., 1972; Paglia &Valentine, 1967).
Assessment of Inflammatory Biomarkers in the Liver
The method detailed by Granell et al. (2003) was used to determine the myeloperoxidase (MPO) activity, while an established protocol by Green et al. (1982) was used to measure the concentration of nitrate.
Histological Examination
The 10% formalin-fixed specimens of the liver were washed, transferred to 70% ethanol, dehydrated in rising ethanol, cleared in xylene, embedded in paraffin wax, cut into 5 µm thick sections, and stained with Harries Hematoxylin and Eosin.
Statistical Analysis
Analysis of variance (ANOVA) was utilized in the statistical analysis, and Duncan’s multiple range (post hoc) test was employed to compare the treatment means. CoStat version 6.311 (Copyright 1998–2005, CoHort software) was used for all analyses. Data were presented as mean ± standard error of the mean (SEM). p values less than 0.05 were considered significant.
Results
Volatiles Composition of TEO and BEO
Results of GC–MS revealed that TEO and BEO were rich in phenolic compounds. p-Cymene (33.3%) and thymol (27.88%) were the largest compounds in TEO. On the other hand, β-linalool (44.53%) was the main component in BEO, followed by eugenol (29.34%), 2-piperitone (14.34%), and eugenol acetate (4.34%) (Table 1, Figures 1 and 2).
Gas Chromatography–Mass Selective (GC–MS) Results for Thyme Essential Oil (TEO) and Basil Essential Oil (BEO).
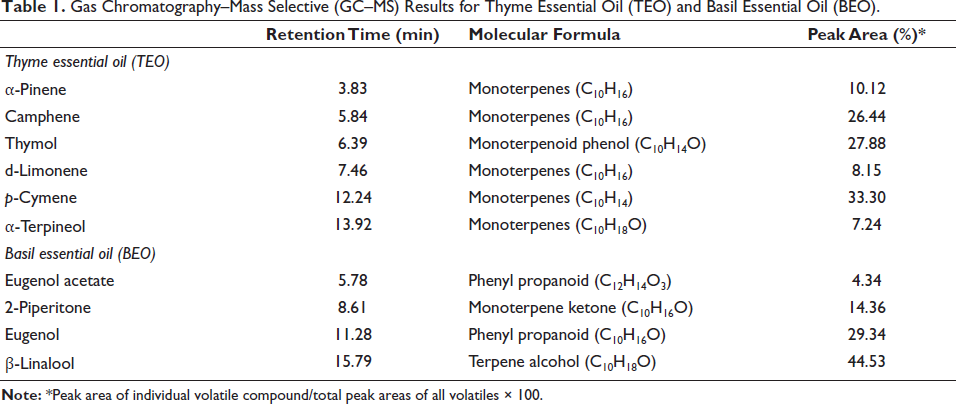
Gas Chromatography–Mass Selective (GC–MS) Results for Thyme Essential Oil (TEO).
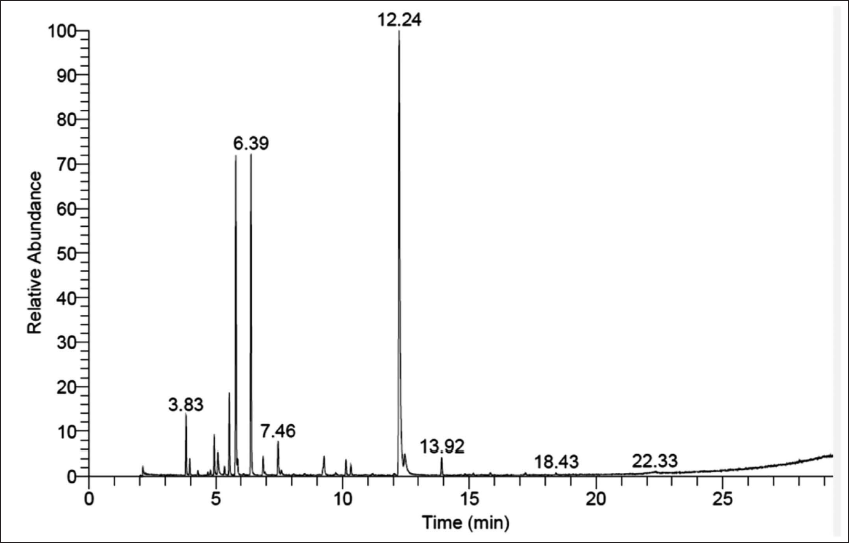
Gas Chromatography–Mass Selective (GC–MS) Results for Basil Essential Oil (BEO).
In Vitro Antioxidant Activities for TEO and BEO
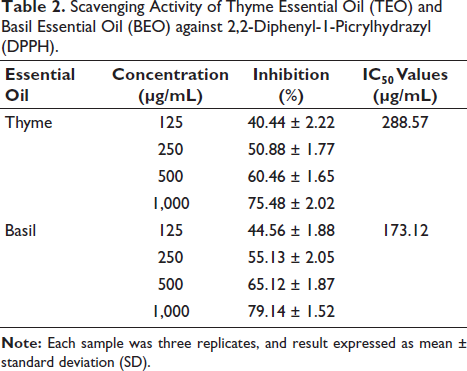
The scavenging activities of TEO and BEO against DPPH are presented in Table 2. Comparing the IC50 values of TEO and BEO, it clearly showed that BEO was more efficient as an antioxidant, as it had a much lower IC50 value (173.12 µg/mL) than TEO (288.75 µg/mL). The scavenging activities of TEO and BEO against H2O2 are shown in Table 3. The findings showed through comparison with IC50 values that BEO (IC50 = 77.48 µg/mL) was more effective versus TEO (123.46 µg/mL). To further confirm the antioxidant potential of TEO and BEO, we performed the ORAC assay, and the results showed significantly (p ˂ 0.05) higher antioxidant capacity for BEO (1.35 ± 0.05 gT/gEO) than TEO (0.92 ± 0.03 gT/gEO). Regulating the NO concentration is necessary to prevent the cell from being negatively affected by an excess of NO. NO scavenging activity assay was also used to measure the antioxidant activity of the two oils, and the results exhibited significantly higher NO scavenging activity for BEO (190.85 ± 7.33 µg/mL) than TEO (237.09 ± 8.51 µg/mL).
Scavenging Activity of Thyme Essential Oil (TEO) and Basil Essential Oil (BEO) against 2,2-Diphenyl-1-Picrylhydrazyl (DPPH).
Scavenging Activity of Thyme Essential Oil (TEO) and Basil Essential Oil (BEO) against Hydrogen Peroxide (H2O2).
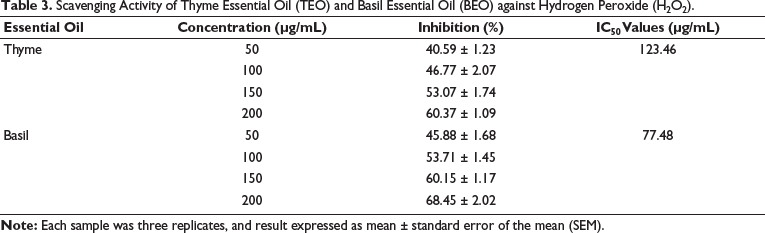
TEO and BEO Improve Liver Functions Deteriorated by H2O2
The results, which are shown in Table 4 and Figure 3, demonstrated that H2O2 (PC) induced highly significant (p ˂ 0.05) increases in the activities of liver enzymes (AST, ALT, ALP, and GGT) and plasma levels of the total bilirubin in comparison to the NC. On the other hand, treatment with H2O2 caused the levels of albumin and total protein to significantly (p ˂ 0.05) decrease when compared to the NC (p ˂ 0.05). As opposed to the PC group, the administration of TEO and BEO significantly reduced the activities of ALT (103.47 and 85.72 U/L, respectively), AST (99.17 and 81.12 U/L, respectively), ALP (111.9 and 94 U/L, respectively), and GGT (105.77 and 90.05 U/L, respectively), as well as total bilirubin (2.69 and 2.17 mg/dL, respectively). However, treatment with TEO and BEO significantly increased albumin (2.47 and 2.85 g/dL, respectively) and total protein (4.23 and 4.69 g/dL, respectively) levels compared to the PC group. The group treated with BEO significantly outperformed its counterparts treated with TEO in all measures of liver function.
Plasma Liver Enzymes Activities in Rats Treated with Thyme Essential Oil (TEO) and Basil Essential Oil (BEO).
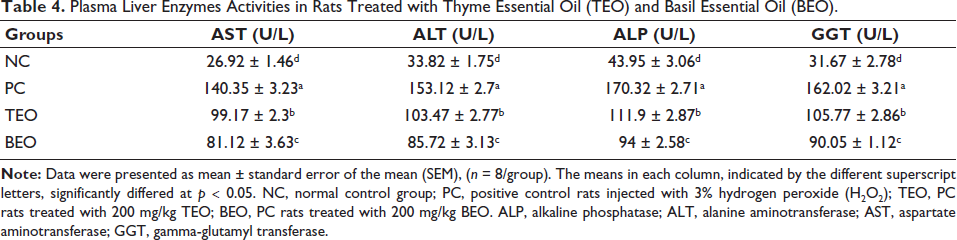
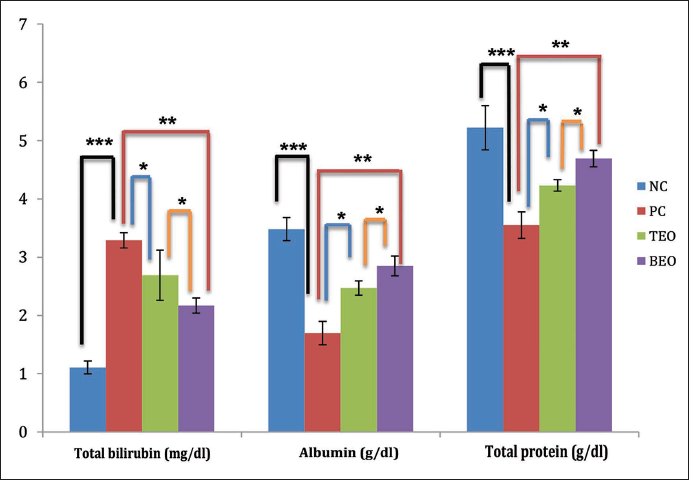
TEO and BEO Relieve H2O2-induced Oxidative Stress
Figure 4 illustrates the effect of TEO and BEO on the activities of SOD, CAT, GPx, and MDA levels in the plasma of all groups. Significantly higher MDA (p < 0.05) was seen in the PC group in plasma (76.85 ± 3.8 nmol mL−1) versus the NC group (25.6 ± 3.71 nmol mL−1). Significant (p < 0.05) reductions were observed in activities of SOD, CAT, and GPx in PC values (12.14 ± 1.22, 5.12 ± 1.09, and 6.36 ± 0.94 U/L, respectively) than in the NC (26.02 ± 1.92, 12.89 ± 2.27, and 15.5 ± 1.5 U/L, respectively). However, treatment with TEO or BEO significantly increased the activity levels of the GPx (8.35 and 10.92 U/L, respectively), SOD (17.8 and 20.85 U/L, respectively) and CAT (7.83 and 10.42 U/L, respectively) enzymes, while these groups (TEO and BEO) showed a significant decline in MDA level (55.68 and 44.74 nmol/mL, respectively) versus the PC group. Again, BEO significantly improved the antioxidant parameters more than TEO.
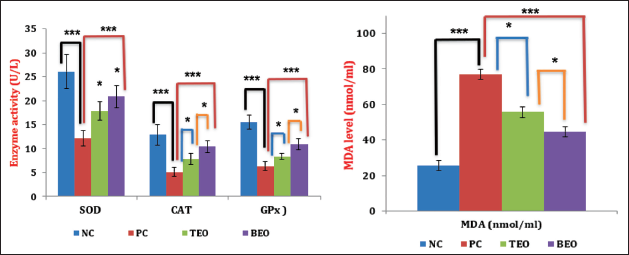
TEO and BEO Ameliorate H2O2-induced Inflammation in the Liver
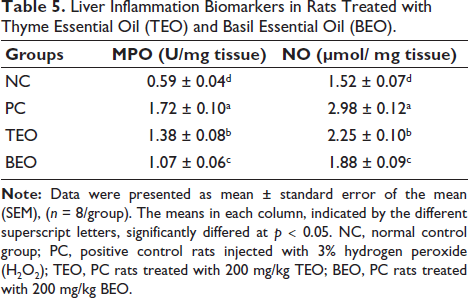
The PC group exhibited a significant (p < 0.05) elevation in MPO activity (1.72 U/mg tissue) and nitrite level, (NO level (2.78 µmol/mg tissue) in the liver compared with those in the NC group (0.59 U/mg tissue and 1.52 µmol/mg tissue, respectively) (Table 5). In contrast, rats administered TEO or BEO showed a significantly lower NO level (2.25 and 1.88 µmol/mg tissue, respectively) and MPO activity (1.38 and 1.07 U/mg tissue, respectively) in their livers than the PC group. The results also indicate that treatment with BEO resulted in significantly better improvement than treatment with the TEO group.
Liver Inflammation Biomarkers in Rats Treated with Thyme Essential Oil (TEO) and Basil Essential Oil (BEO).
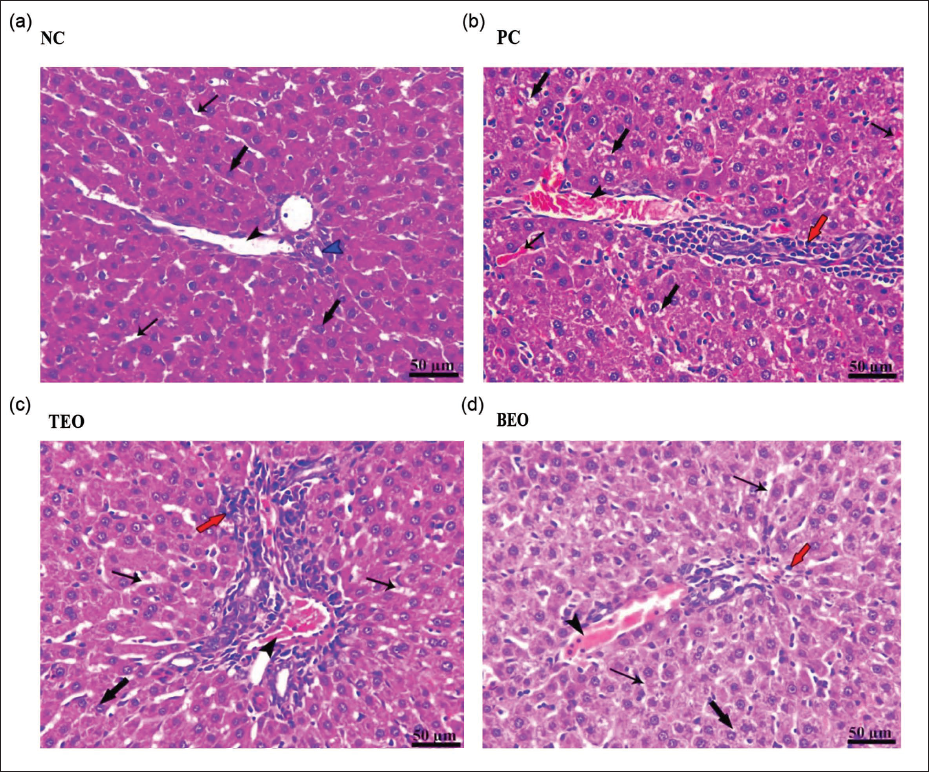
Histopathological Examination
In the present study, histopathological observations were made on liver tissue samples stained with H&E to examine the effects of TEO and BEO treatments on liver structure and cellular morphology as depicted in Figure 5. The histological analysis indicates that the NC group maintained intact liver architecture, with no observable signs of cellular damage or inflammatory infiltration. In contrast, the control positive (PC) group exhibited significant liver injury, with congestion, vacuolar degeneration, and mononuclear cell infiltration, indicating extensive hepatic stress and immune response. The TEO group showed moderate protection, with reduced congestion and inflammation relative to the PC group, though some vacuolar degeneration persisted. The BEO group demonstrated the highest level of hepatoprotection, with minimal congestion, intact hepatocytes, and mild cellular changes, suggesting that BEO may offer effective support for maintaining liver health under stress conditions.
Discussion
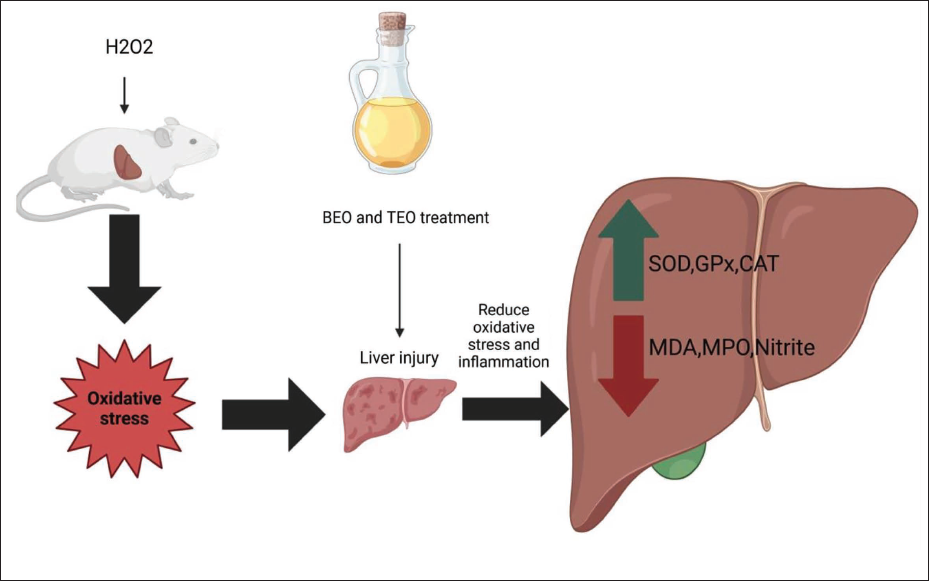
Different toxins and molecules that are consumed in excess can attack liver tissue, causing it to malfunction and impair the ability of the body to sustain life. But there are natural products, like EO, that can control this disorder. Thus, it was demonstrated that various EO have antioxidant properties that are crucial for reducing the oxidative stress brought on by hepatotoxins such as H2O2 (Daoudi &Bnouham, 2020). The current investigation demonstrated that BEO and TEO have promising preventive potential in the liver, protecting against liver damage brought on by exposure to H2O2. Figure 6 summarizes the mechanisms by which BEO and TEO do this effect.
Schematic Diagram shows the Antioxidant Mechanisms by Which Basil Essential Oils (BEO) and Thyme Essential Oil (TEO) could Attenuate Hydrogen Peroxide (H2O2)-induced Hepatic Damage in Rats.
GC–MS findings suggested that the predominant constituents of TEO were p-cymene and thymol, whereas the principal constituent of BEO was β-linalool. This was corroborated by findings from other researchers who examined the constituents of BEO (Hussain et al., 2008; Politeo et al., 2007) and TEO (Masada, 1976; Ramadan et al., 2022). In general, the components and their percentages in EO vary as a result of many factors such as the plant variety, climate, as well as the method of extraction. However, the EO of a single plant species are distinguished by the presence of main compounds largely responsible for its EO’s biological properties.
The antioxidant activity of both TEO and BEO was tested using DPPH, H2O2, ORAC, and NO scavenging activity assays. Due to its lower IC50 values, BEO was found to be the most effective antioxidant in both methods (DPPH, H2O2, and NO scavenging activity assays) when compared to TEO. BEO also showed higher ORAC than TEO. Previous studies have explained the antioxidant activity of TEO as a result of its high content of phenolic substances as well as terpenoids (Kadri et al., 2011; Lacroix et al., 1997). We also found a high content of the active compounds p-cymene and Thymol in TEO. The efficiency of BEO as a natural antioxidant compared to synthetic substances such as butylated hydroxytoluene (BHT), showed lower IC50 values of BEO than for BHT (Bozin et al., 2006; Hussain et al., 2008; Politeo et al., 2007).
One of the main causes of human cell and liver damage is lipid peroxidation, which is brought on by many oxidants such as H2O2, one of the ROS. Cell membranes become corrupted as a result of lipid peroxidation (Elkeiy et al., 2020). As compared to NC, liver enzyme (AST, ALT, GGT, and ALP) activities and total bilirubin levels significantly increase in the H2O2 treatment group (PC). On the contrary, the levels of both albumin and total protein in plasma significantly decreased, which are indicators of damage to liver cells. These findings are generally in line with those of earlier research that examined the detrimental effects of H2O2 treatment in drinking water on liver biomarkers in rats (Abozid &El-Sayed, 2013; Abozid et al., 2018; Kaplowitz et al., 1986; Sevanian &Hochstein, 1985). Both TEO and BEO, on the other hand, may have a hepatoprotective effect in the rats treated with H2O2, as evidenced by the fact that liver enzymes (ALT, AST, GGT, and ALP) activities and total bilirubin levels in TEO and BEO-treated groups decreased significantly after their intervention. These findings are largely in line with the findings of earlier studies, which demonstrated the protective effect of thyme and basil EO on liver cells in rats by enhancing a number of liver function indicators (Mossa et al., 2013; Rostami et al., 2022; Sobhy et al., 2020; Teofilović et al., 2021). TEO and BEO have a high concentration of natural antioxidants, which may explain why liver cells are shielded from H2O2 when both EO are used. The results of the estimated antioxidant activity (in vitro) of these two EO can be linked to their protective effect on liver cells. Furthermore, when compared to TEO, BEO performed better in vitro antioxidant activity with enhanced in vivo liver functions.
According to numerous studies, H2O2 is a powerful substance in order to cause oxidative stress in laboratory animals (Duan et al., 2016; Ganie et al., 2016). Its detrimental effect is that it generates other free radicals, such as the hydroxide radical, that are highly reactive with biological molecules. Cell membrane polyunsaturated fatty acids are destroyed via the lipid peroxidation process by the autocatalytic, free radical-mediated process to produce lipid hydroperoxides, particularly MDA (El-Magd, Khamis et al., 2017; Shi et al., 2010). A common indicator of oxidative damage is MDA. As a result, the elevated levels of MDA found in the PC group suggest that there may have been an increased level of free radical production and lipid peroxidation. The rise in MDA suggests that lipid peroxidation caused by H2O2 worsens liver oxidative damage. One possible explanation for the notable reduction in SOD and CAT activity could be the generation of ROS following H2O2 treatment (Ganie et al., 2011). GPx is capable of effectively scavenging H2O2, so its declining activity is a reflection of how H2O2 exposure has disrupted the body’s normal oxidative balance (Lan et al., 2019). Free radicals are neutralized by activating antioxidant enzyme systems like SOD, CAT, and GPx (Abdelhady et al., 2017; El-Magd, Abdo et al., 2017; Fang et al., 2002). Thus, TEO and BEO initiated antioxidant defenses against oxidative stress caused by H2O2. The antioxidant activity of phytochemical compounds in EO has been related to the hydroxyl group attached to the aromatic ring, which has the ability to donate hydrogen atoms with electrons and neutralize free radicals (Radha Krishnan et al., 2014). Important terpenes and phenolic compounds found in BEO protect cells from free radicals and keep them from oxidation by different substances that produce free radicals (Thirugnanasampandan &Jayakumar, 2011). BEO contains powerful antioxidants like β-linalool and
Oxidative stress is facilitated by the release of MPO, one of the most important molecules that occurs after phagocytes are recruited and activated. Furthermore, pro-inflammatory cytokines stimulate NO during hepatic damage, leading to abnormal NO production, which greatly aids in the etiology and progression of hepatic damage (Oluwafemi Adetuyi et al., 2020). The results of this study indicate a noticeable rise in the activity of MPO and level of NO in the PC group as compared to the NC group. Nonetheless, TEO and BEO treatments resulted in a lower level of NO and MPO, suggesting that these oils may lessen liver inflammation in rats. The results of TEO are consistent with previous studies (El-Boshy et al., 2019; Soliman et al., 2021), which supported the protective effect of thyme, especially in improving various inflammation biomarkers. Our results also aligned with earlier research that provided support for the liver-protective effect of the basil plant extracts (Jebur et al., 2021; Teofilović et al., 2021; Yacout et al., 2012).
The histopathological findings in this study provide valuable insights into the protective effects of TEO and BEO on liver tissue under conditions likely to induce hepatic stress or injury. The results highlight distinct differences in hepatic structure across treatment groups, underscoring the potential therapeutic benefits of these EO in preserving liver health. The PC group exhibited severe pathological changes, including pronounced congestion in both the portal vein and hepatic sinusoids, extensive vacuolar degeneration of hepatocytes, and significant perivascular mononuclear cell infiltration. These changes suggest acute hepatic stress, likely triggered by exposure to a hepatotoxic agent or condition. The vacuolar degeneration observed in hepatocytes is indicative of lipid accumulation or cellular injury, often associated with oxidative stress or inflammation, which can impair liver function and eventually lead to more severe liver diseases (Diab et al., 2024; El-Magd et al., 2022). The infiltration of mononuclear cells around blood vessels signals an immune response, likely aimed at counteracting the underlying hepatic damage. These findings are consistent with prior studies showing that liver congestion and hepatocyte degeneration are common markers of liver injury and inflammation under stress conditions (Abozid &El-Sayed, 2013; Abozid et al., 2018). In the TEO group, moderate portal vein congestion and hepatic sinusoids, along with milder vacuolar degeneration of hepatocytes, were observed. Compared to the PC group, TEO administration appears to have provided partial protection against liver injury, as evidenced by reduced congestion and a less intense inflammatory response. Thymol, a primary component of TEO, has demonstrated antioxidant and anti-inflammatory properties in prior studies, which may help mitigate oxidative damage and reduce the severity of liver injury (Daoudi &Bnouham, 2020; Salehi et al., 2018). The BEO group demonstrated the most significant hepatoprotective effects, showing only mild congestion in the portal vein and hepatic sinusoids, with minimal signs of vacuolar degeneration and a well-preserved hepatic cord structure. Hepatocytes appeared largely intact, with only slight microsteatosis in a few cells, and there was a minimal immune response, indicated by mild mononuclear cell infiltration. These results suggest that BEO offers a robust protective effect against hepatic stress, likely due to the presence of potent antioxidants such as linalool and eugenol, which have been shown to counteract oxidative stress and enhance cellular resilience in liver tissues (Ademiluyi et al., 2016; Bentayeb et al., 2014). The cord-like arrangement of hepatocytes and the preserved sinusoids in the BEO group resembles the histological structure observed in healthy liver tissue, further supporting BEO’s potential as a hepatoprotective agent. The findings in this group are consistent with prior studies that have highlighted the efficacy of BEO in reducing hepatic oxidative stress and inflammation, potentially through upregulation of endogenous antioxidant defenses and inhibition of inflammatory pathways (Ademiluyi et al., 2016; Hussain et al., 2008; Politeo et al., 2007; Ramadan et al., 2022).
Strengths of this study include its use of natural products (BEO and TEO) to treat liver problems caused by harmful substances like H2O2, which makes their use safer than that of manufactured medications. Additionally, the amounts of these products can be taken with regular meals. However, the study’s small sample size and the potential need to isolate the various components of each EO and examine their individual effects to identify the most effective compounds are the study’s two main limitations.
Conclusion
The study’s findings revealed that exposure to H2O2 leads to significant deterioration in antioxidant markers and a corresponding rise in liver function markers. Thyme and BEOs are protective because they can restore antioxidant and liver function status. However, BEO had a significantly stronger impact than TEO. It is evident that the food and pharmaceutical industries could find value in using EO of basil and thyme as a readily available source of naturally occurring bioactive compounds, which have positive health effects.
Footnotes
Abbreviations
ALP: Alkaline phosphatase; ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; BEO: Basil essential oil; CAT: Catalase; DPPH: 2,2-Diphenyl-1-picrylhydrazyl; GGT: Gamma-glutamyl transferase; GPx: Glutathione peroxidase; H2O2: Hydrogen peroxide; MDA: Malondialdehyde; MPO: Myeloperoxidase; ROS: Reactive oxygen species; SOD: Superoxide dismutase; TEO: Thyme essential oil.
Acknowledgments
The author would like to thank Professor Ashraf B. Abdel-Naim, Department of Pharmacology and Toxicology, Faculty of Pharmacy, King Abdulaziz University, Professor of Pharmacology and Toxicology, and Professor. Mohamed A. El-Moselhy, Clinical Pharmacy and Pharmacology Department, Ibn Sina National College for Medical Studies, Professor of Pharmacology, for their invaluable input and support throughout the research process.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The animal study was reviewed and approved by the IRRB-ER/EC committee and was performed following the guidelines of the Implementing Regulations of the Law of Ethics of Research on Living Creatures in the Kingdom of Saudi Arabia and other applicable national and international regulations (Research Ethics Committee Reference Number IRRB-ER/EC-20102024).
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
