Abstract
Background
Nociception, often described as the body’s ability to sense and respond to harmful stimuli, represents a global discomfort that profoundly impacts individuals across the globe. It functions as a fundamental warning process, alerting organisms against potential threats and promoting protective behaviors. However, when dysregulated or chronic, nociception can become a significant burden, leading to persistent pain and diminished quality of life. Various analgesic drugs play a crucial role in alleviating pain and improving the quality of life for millions of people worldwide. While analgesic drugs provide significant relief for acute and chronic pain conditions, they can cause side effects ranging from mild gastrointestinal disturbances, drowsiness, and constipation to major complications such as cardiovascular damage and nephrotoxicity.
Objectives
Hence, we investigated the potency of a phytochemical tomatidine against various noxious stimuli-induced nociception.
Materials and Methods
Tomatidine pain-relieving potency has been assessed against heat stimuli and chemical-induced visceral pain. The neuropathic pain-relieving potency of tomatidine was examined with glutamate and capsaicin-induced mice models.
Results
Tomatidine potency against intense pain was examined with a formalin-induced biphasic nociception model. Carrageenan induced an inflammatory response, peritoneal leukocyte infiltration, and air pouch model were performed to analyze the anti-inflammatory effect of tomatidine against inflammatory cytokines-induced nociception. The antisedative potency of tomatidine was confirmed with open-field test analysis. Our results confirm that tomatidine effectively inhibited nociception induced by thermal and chemical stimuli. It also alleviates the inflammatory cytokines stimulated pain and does not render any sedative effect in mice models.
Conclusion
To conclude tomatidine is a potent nonsedative antinociceptive drug that can be formulated as an analgesic.
Keywords
Introduction
Analgesia refers to a vaguely defined, unpleasant sensation triggered by either external or internal stimuli. It is a crucial indication that acts as an alarm to protect in the initial stage itself (Armstrong & Herr, 2023; Raja et al., 2020). Based on the stimuli and the pathophysiology, the analgesia has been broadly classified into acute, nociceptive, chronic, and neuropathic pain (Deshmukh et al., 2014). Nociceptors were activated when exposed to noxious stimuli such as extreme thermal conditions, mechanical stress, harmful chemicals, and others. When these stimuli persist long enough, acute pain transitions to inflammatory pain as nociceptive neurons release proinflammatory markers, sensitizing or activating surrounding cells (Falk & Dickenson, 2014; Woller et al., 2017). In contrast to nociceptive pain, neuropathic pain stems from diseases such as postherpetic neuralgia, chronic disease-induced neuropathies, and others, which directly affect the sensory nerves and cause pain (Ang et al., 2017; Mitsi & Zachariou, 2016).
Even though analgesia acts as an alert mechanism, fundamentally protective in nature, it can also lead to significant discomfort and suffering, which may be unbearable and incapacitating. The interaction between nociception and persistent inflammation appears to extend the pain perceptions (Wei et al., 2016). Chronic pain, in its various forms, affects about 10–20% of adults at some point in life. Twin studies indicate that approximately one-third of the susceptibility to chronic pain is heritable (Hocking et al., 2012), with some of this susceptibility having a direct genetic basis (Diatchenko et al., 2005). The deregulation of cytokine expression and stimulation of nuclear factor kappa B (NF-κB) activation are associated with pain. Furthermore, post-traumatic stress disorder (PTSD) that occurs following the trauma conditions can both result from and contribute to pain (Hoofwijk et al., 2016).
Chronic pain goes beyond mere physical discomfort, significantly impacting a patient’s mental health, day-to-day activities, and social life. Those suffering from chronic nociception may experience discriminative and a diminished livelihood, which can exacerbate emotional well-being (Dueñas et al., 2016). Relying on pain medications and their associated adverse reactions can further impact overall health. Moreover, managing chronic nociception, including healthcare costs and potential income loss, adds to the financial strain (Cohen et al., 2021).
Phytochemicals derived from medicinal plants form the foundation of many medicines used to treat various health conditions and are valuable sources for potent drug discovery (Asmamaw & Achamyeleh, 2018; Bachheti et al., 2020). Medicinal plant extracts are widely recognized as effective folk remedies with anti-inflammatory properties. These plants are abundant in phytochemicals that serve as novel drugs and provide a solid basis for the development of new inflammation-reducing medications (Bunte et al., 2019; Kiani et al., 2019). Tomatidine is one such steroidal alkaloid compound present in the leaves and stems of plants belonging to the Solanaceae family such as Lycopersicon esculentum and Solanum cathayanum (Bailly, 2021). It possesses varied medicinal properties, including inflammation reducing property (Yan et al., 2013) anticancer (Chiu & Lin, 2008), antimicrobial (Guay et al., 2018; Lamontagne et al., 2018), muscle-preserving, cardioprotective, neuroprotective, metabolic, and bone health benefits (Yu et al., 2020). Its multifaceted therapeutic potential makes it an interesting subject for further research and development in various medical fields. The study aimed to assess the antinociceptive potency of tomatidine against various noxious stimuli in a mouse model.
Materials and Methods
Experimental Animals
Healthy young adult mice, aged 8–10 weeks and weighing between 30 and 40 g were utilized for the study. These animals were kept in a hygienic laboratory environment at 22–25°C, 55–60% relative humidity, and a 12-h light/dark cycle. Cages and bedding are regularly changed to prevent infection. Laboratory pellet diet and water were fed to the rodents without any limit restriction, and food was restricted only overnight before behavioral analyses.
Nociceptive Stimuli with the Hot Plate Model
To ensure accurate results, only healthy mice with sensitivity to heat were utilized for the test. These thermally sensitive mice were grouped into 10. 1% Tween 80 was treated to control mice. 10, 25, and 50 mg/kg body weight of tomatidine doses were given to II, III, and IV group mice, respectively. Groups V and VI were treated with 5 mg/kg body weight of morphine and 2 mg/kg body weight of naloxone. To evaluate the antinociceptive efficacy of tomatidine, Groups VII, VIII, and IX were treated with both naloxone (2 mg/kg body weight) and varying doses of tomatidine (10, 25, and 50 mg/kg body weight, respectively). Group X received both naloxone (2 mg/kg body weight) and morphine (5 mg/kg body weight). Eddy’s hot plate test, following Gunn et al. (2011) [24] protocol was conducted for 30 min, and the mice were kept on the hot plate upheld at 50°C for 20 s, and the first signs of nociceptive behavior licking paw were recorded for 2 h and the % maximum possible effect (MPE) was calculated.
Nociceptive Stimuli with the Tail Immersion Model
Mice displaying a tail deflection time between 1.5 and 2.5 s, indicative of thermal sensitivity, are subjected to testing. After 30 min of drug treatment, the mice tails of each group were immersed 4 cm deep in hot water upheld at 55°C, and tail withdrawal time was noted. Maximum of 20 s the test was performed for individual mice (Olaleye et al., 2000). The experiment was conducted for up to 2 h post-treatment, with measurements taken at 30-min intervals. %MPE was calculated for all the groups.
Nociceptive Stimuli with Acetic Acid Stimulated Writhing Model
Tomatidine potential to inhibit acetic acid-stimulated writhing was examined with a nociception test based on the method by Koster et al. (1959). Mice were divided into five groups and were treated with Tween 80, 10 mg/kg body weight of diclofenac sodium, and 10, 25, and 50 mg/kg body weight of tomatidine, respectively. Each mouse, after being treated with acetic acid and the respective drug was placed in a monitoring enclosure for 30 min, and the writhes performed by mice were recorded for further analysis.
Nociceptive Stimuli with Glutamate
Analogous groupings of acetic acid-induced nociception test were also used for the glutamate-stimulated pain test and the test was performed as per Beirith et al. (2002) method. Each group of mice received treatment with Tween 80, various concentrations of tomatidine and diclofenac sodium. Glutamate, at a concentration of 10 µM was then injected into the ventral surface of the right hind paw of each mouse. Subsequently, the mice were placed individually inside a monitoring enclosure for 15 min, and the licks made by the mice were noted.
Nociceptive Stimuli with Capsaicin
Neuropathic pain was induced in mice through the capsaicin injection into the paws of mice, as described by Luiz et al. (2007). Prior 30 min to the start of the experiment, mice were pretreated with Tween 80, different concentrations of tomatidine and diclofenac sodium. The capsaicin solution was prepared by dissolving capsaicin in phosphate-buffered saline (PBS) at a concentration of 1.6 µg per 20 µL, with a potential of hydrogen (pH) of 7.4. Each mouse received an injection of 20 µL of the capsaicin solution into its paw and was then observed for 5 min. The number of licking behaviors exhibited by both the control and drug-treated mice was recorded.
Nociceptive Stimuli with Biphasic Formalin Model
Nociception was induced in the mice model with formalin, using the protocol of Mohamad et al. (2010). The mice were grouped into five, and control rats were treated with 1% Tween 80, three different concentrations of tomatidine, and a standard drug (5 mg/kg body weight). 20 µL of 2.5% v/v formalin solution was injected subcutaneously into the ventral surface of the mice’s right hind paws. The mice were subsequently placed in an observation chamber for 35 min. In two phases: the initial phase or nociceptive phase (0–5 min) and the final phase or inflammatory phase (15–35 min) the number of lickings performed by the mice was observed.
Evaluation of Carrageenan Stimulated Inflammation
Inflammation was induced in mice’s right paws with carrageenan by Chung et al. (2019) method. Tween 80, three different concentrations of tomatidine, and the 10 mg/kg body weight of customary anti-inflammatory medication indomethacin respectively were given to animals an hour before the experiment. 1% carrageenan was injected into the right paw and 0.9% saline into the left paw of the animals. Subsequently, 4 h edema size was measured with a plethysmometer for every hour. The measurements were documented to calculate %MPE.
Assessment of Peritoneal Leukocyte Infiltration Induced by Carrageenan
One hour after administration of different doses of tomatidine, 0.2% Tween 80, diclofenac sodium, and peritoneal injection of 500 µL of carrageenan were given to the animals to stimulate leukocyte exodus into the peritoneum (Melo et al., 2011). Mice were humanely euthanized through cervical dislocation 4 h after receiving the carrageenan injection. Subsequently, a PBS solution containing ethylenediaminetetraacetic acid (EDTA) (1 mM, i.p., 10 mL) was administered. Following this, the abdomen was gently massaged to facilitate further fluid collection, which was then centrifuged at 2,500 rpm for 5 min. The resulting supernatant was discarded, and the pellet was dissolved in 1 mL of PBS. A 10-µL sample of this solution was diluted in 200 µL of Turk solution, and total cells, as well as mononuclear and polymorphonuclear cells were enumerated using a Neubauer chamber under optical microscopy. The results were reported as the number of cells per cubic millimeter.
Quantification of Inflammation-inducing Cytokines by Air Pouch Mice Model
An air pouch was created in the mice by injecting 5 mL of sterile air, followed by 2 mL of sterile air for 3 days on alternative days. An air pouch was induced in the mice in a sterile lab environment, and the mice were anesthetized and shaved at the injecting site before the induction of the air pouch (Fehrenbacher & McCarson, 2021). The mice developed with air pouch were grouped into six and injected with 1% Tween 80, 0.5 mL carrageenan alone, and other three groups were treated with carrageenan and different doses of tomatidine, and the sixth group was treated with carrageenan and dexamethasone. The mice were sacrificed an hour after the treatment of drugs and carrageenan, 2 mL of saline was injected into the air pouch and the exudates were ejected using the sterile needle. The collected pouch exudates were then centrifuged and the pellets were subjected to quantification of proinflammatory cytokines.
Behavioral Analysis by Open-Field Test
Animals were subjected to treatment with three different doses of tomatidine and morphine before an hour of the experiment. The control and drug-treated mice were relocated from the animal housing labs to the behavioral analysis laboratory 30 min prior initiation of the experiment. A 50 cm (length) × 50 cm (width) × 38 cm (height) measuring chamber made up of white high-density and nonporous plastic material was used for the study. The chamber was wiped with 95% ethanol completely to avoid scent clues. The mice were placed gently into the middle of the chamber by grasping the tail and allowed to explore the chamber without any interruptions (Seibenhener & Wooten, 2015). The movement of the mice was recorded to count squares explored by the animals.
Statistical Analysis
Results acquired from each experiment were analyzed statistically with the software GraphPad Prism. Data were assessed through one-way analysis of variance (ANOVA) followed by Dunnett’s post hoc test. Each bar represents mean ± standard error of the mean (SEM) of six animals, #, *represent statistical significance between control vs. other groups at p < 0.05, p < 0.01.
Results
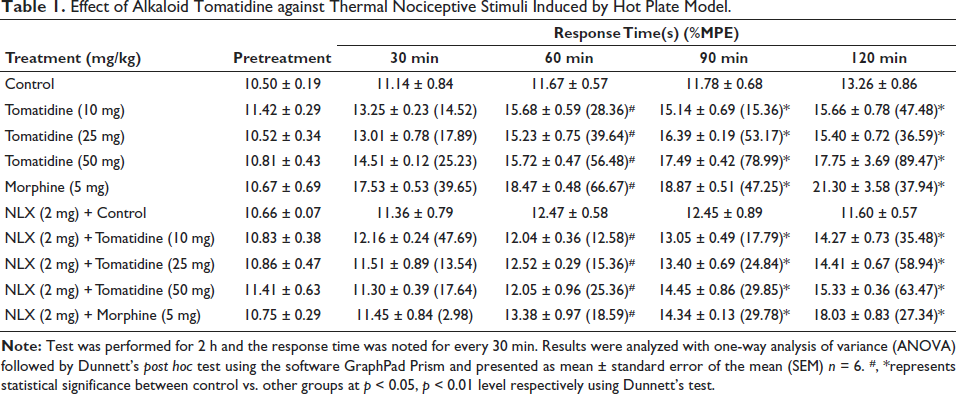
Effect of Alkaloid Tomatidine against Thermal Nociceptive Stimuli Induced by Hot Plate Model
The analgesic effect of tomatidine was assessed against the thermal nociceptive stimulus induced by the hot plate and the results were tabulated in Table 1. The % of MPE obtained after 2 h of treatment confirmed the efficacy of tomatidine in relieving thermal stress. 50 mg tomatidine treatment mice maximum of 89.47% of MPE, and it was considerably decreased to 63.47% of MPE with the opioid antagonist naloxone treatment. These results were significantly higher than the results obtained with standard opioid analgesic drug morphine, which exhibited 37.94% of MPE when treated alone and along with naloxone treatment, the % of MPE was 27.34. In the presence of opioid antagonist naloxone, the analgesic effect of tomatidine was observed in dose-dependent manner.
Effect of Alkaloid Tomatidine against Thermal Nociceptive Stimuli Induced by Hot Plate Model.
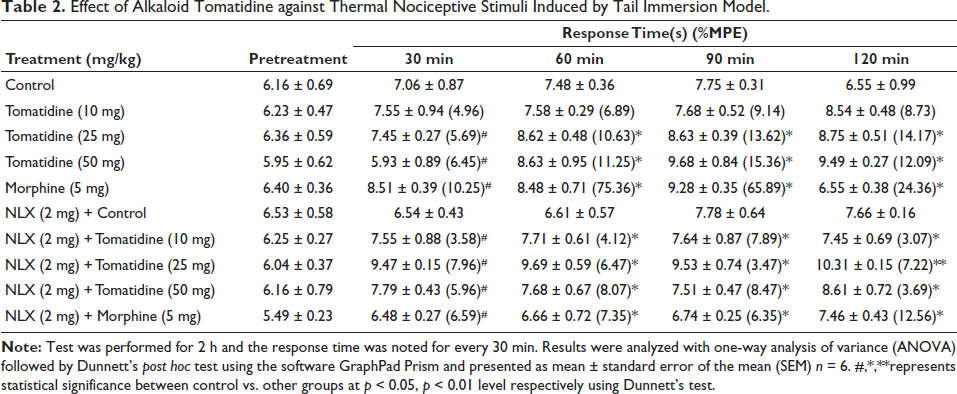
Effect of Alkaloid Tomatidine against Thermal Nociceptive Stimuli Induced by Tail Immersion Model
Table 2 illustrates the results % of MPE exhibited by the control and experimental mice. The 50 mg/kg tomatidine-treated mice showed a significant increase % of MPE of 12.09 in the absence of naloxone, and in the presence of naloxone 25 mg/kg tomatidine treated exhibited an increased level of %MPE (7.22) compared to the 50 mg/kg tomatidine treated mice (7.22). The % of MPE exhibited by the standard drug morphine-treated mice is higher than the percentage of tomatidine-treated mice.
Effect of Alkaloid Tomatidine against Thermal Nociceptive Stimuli Induced by Tail Immersion Model.
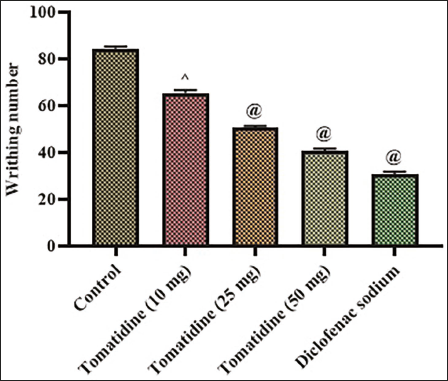
Effect of Alkaloid Tomatidine against Visceral Nociceptive Stimuli Induced by Acetic Acid
Figure 1 depicts the number of writhes recorded in control and tomatidine-treated mice, which were subjected to acetic acid-induced visceral nociception. Abdominal writhes count was considerably decreased in the tomatidine-treated mice than the control mice. Mice injected with 50 mg/kg of tomatidine exhibited 39 ± 0.6 writhes, which were comparable to abdominal writhes counts produced by standard analgesic drug diclofenac sodium (32 ± 0.07 writhes) treated animals. The untreated control mice exhibited a significantly increased number of abdominal writhes (83 ± 0.8 writhes) compared to the mice treated with 10 mg/kg (71 ± 1.2 writhes) and 25 mg/kg of tomatidine (56 ± 0.4 writhes).
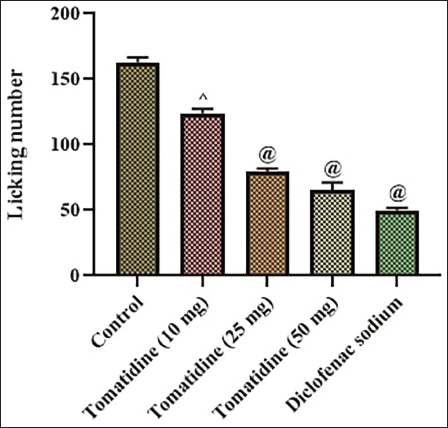
Effect of Alkaloid Tomatidine against Nociceptive Stimuli Induced by Neurotransmitter Glutamate
The analgesic effect of tomatidine against pain transmission and sensitization in the peripheral and central nervous systems induced by the neurotransmitter glutamate was assessed, the results are depicted in Figure 2. Control animals exhibited a significantly increased licking number of 162 ± 1.4 compared to the tomatidine-treated mice, which had shown 131 ± 1.6, 84 ± 0.9, and 71 ± 1.8 licking numbers with 10, 25, and 50 mg/kg tomatidine mice, respectively. Diclofenac sodium-treated mice were exposed to a considerably decreased number of licking (49 ± 0.9) compared to the other group mice.
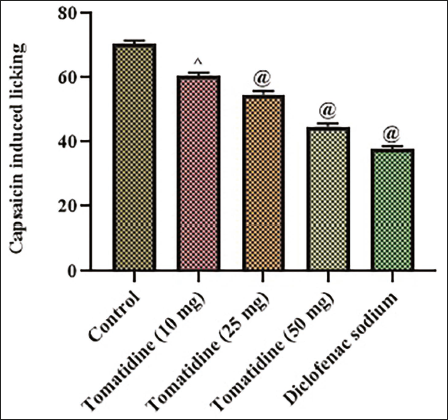
Effect of Alkaloid Tomatidine against Neuropathic Nociceptive Stimuli Induced by Capsaicin
Figure 3 depicts the analgesic potency of tomatidine against the neuropathic nociceptive stimuli induced by the irritant capsaicin. 50 mg/kg tomatidine treated mice and the diclofenac sodium treated mice revealed moderately decreased licking counts of 48 ± 0.07, 37 ± 0.06, respectively. Both 10 and 25 mg tomatidine-treated mice exhibited 59 ± 0.06, 56 ± 0.08 number of lickings, which were significantly lesser than the licking number of control mice (73 ± 0.06).
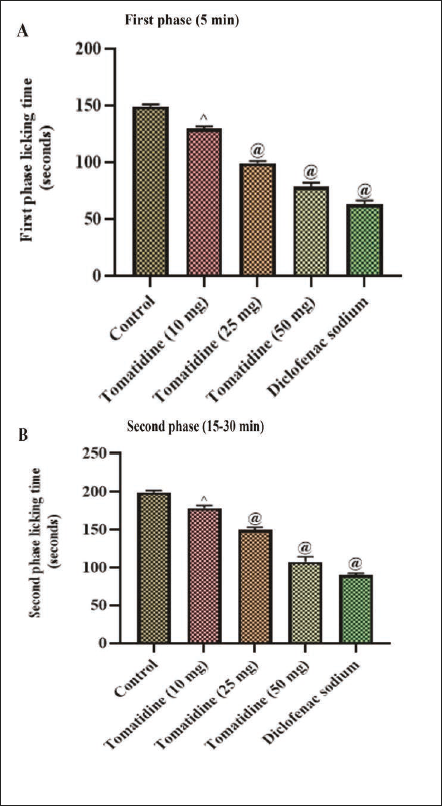
Effect of Alkaloid Tomatidine against Biphasic Nociceptive Stimuli Induced by Formalin
Figure 4 depicts the tomatidine analgesic effect against the acute and chronic pain induced by formalin. The tomatidine treatment in mice significantly decreased pain induction both in the first and second phases of the experiment. During the initial phase, the tomatidine-treated rats exhibited 132 ± 0.08, 98 ± 0.02, and 81 ± 0.04 s licking time with 10, 25, and 50 mg/kg tomatidine-treated mice, respectively, whereas it was increased to 178 ± 1.2, 148 ± 0.09, and 118 ± 1.6 s in the final phase. The same pattern was observed with diclofenac sodium-treated mice which exhibited 66 ± 0.06 s of licking time during the initial phase and 87 ± 0.08 at the final phase. Control mice exhibited higher licking time of 148 ± 0.04 and 19 ± 0.08 s, respectively, at the initial phase and final phase.
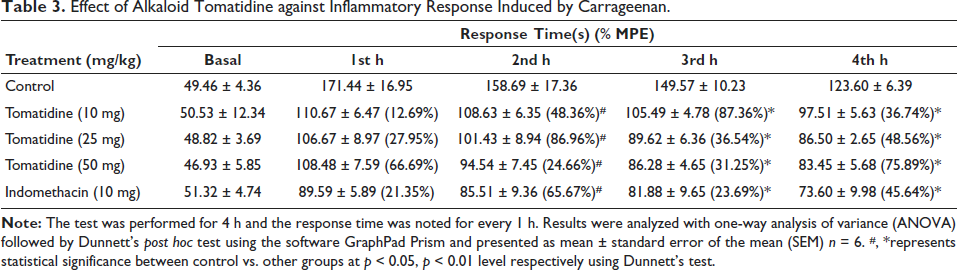
Effect of Alkaloid Tomatidine against Inflammatory Response Induced by Carrageenan
Table 3 represents the % of MPE exhibited by the control and drug-treated rats for the subsequent 4 h after induction of the inflammatory response with carrageenan. The % of MPE was significantly decreased with tomatidine treatment in a dose-dependent manner. Compared to the control mice, the tomatidine and indomethacin-given mice presented a significantly decreased % of MPE.
Effect of Alkaloid Tomatidine against Inflammatory Response Induced by Carrageenan.
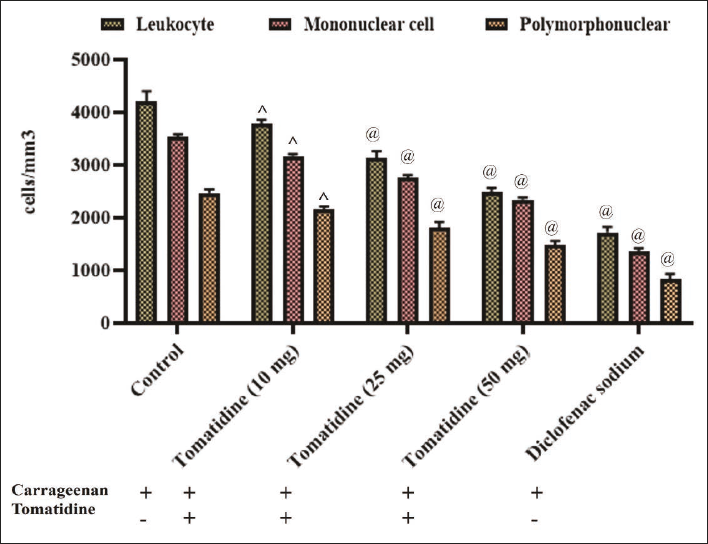
Effect of Alkaloid Tomatidine against the Peritoneal Leukocyte Infiltration Induced by Carrageenan
Leukocyte infiltration count in the peritoneum of carrageenan-injected control and drug-treated mice was measured, and the results are illustrated in Figure 5. The leukocyte infiltration was considerably increased in the control animals than the drug-treated mice. Tomatidine treatment significantly decreased the leukocyte infiltration in the peritoneal cavity than the control mice. The inhibition of leukocyte infiltration in tomatidine-treated mice was perceived in a dose-dependent manner. Both the mononuclear and polymorphonuclear cell counts were decreased in the tomatidine-treated mice peritoneal exudates than in the control mice. Diclofenac sodium-treated mice presented a considerably decreased number of leukocyte infiltration than the control and tomatidine-treated animals.
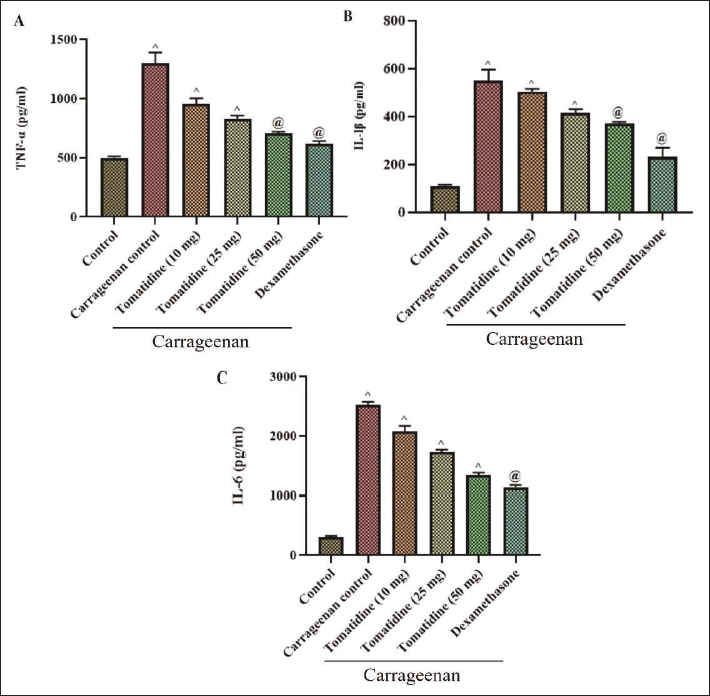
Effect of Alkaloid Tomatidine against the Inflammation-inducing Cytokines Induced by Air Pouch Model
Figure 6 exhibits inflammation-inducing cytokine levels quantified from the exudates collected from the air pouch induced. The inflammation-inducing cytokines tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), and interleukin-6 (IL-6) levels were considerably increased in the control mice than in the tomatidine and dexamethasone-injected mice. The IL-6 levels were comparatively increased in the control mice induced with carrageenan compared to the TNF-α and IL-1β levels. Tomatidine treatment significantly decreased the levels of all three cytokines TNF-α, IL-1β, and IL-6 in a dose-dependent manner. Carrageenan alone treated mice showed significantly increased levels of inflammatory cytokines confirming the induction of an inflammatory response. The standard drug dexamethasone treatment comparatively decreased the levels of inflammation-inducing cytokines.
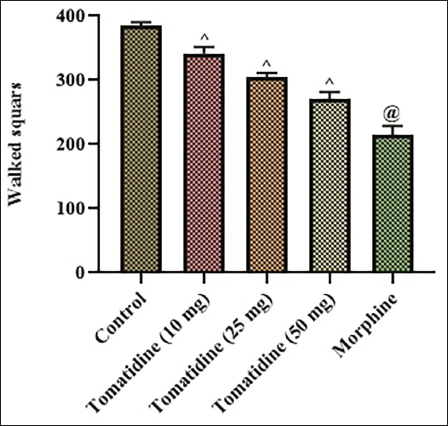
Effect of Alkaloid Tomatidine on Motor Function and Anxiety Levels
The common side effects of most analgesic drugs are impaired motor function and anxiety; hence, these parameters were analyzed through open-field tests in the tomatidine-treated mice, and the data are illustrated in Figure 7. The control mice exhibited increased exploratory behavior by crossing 395 ± 1.2 squares compared to the other group mice. The exploratory behavior in tomatidine mice was decreased in a dose-dependent manner. 10, 25, and 50 mg tomatidine mice crossed 347 ± 1.8, 297 ± 1.4, and 274 ± 1.8 squares, respectively, which was greater than the squares crossed by morphine-treated mice (227 ± 2.1 squares).
Discussion
Pain management has emerged as a significant global concern, with an estimated one in five adults worldwide experiencing pain (Sovaila et al., 2024). Hyperalgesia, a condition characterized by heightened sensitivity to pain is often perpetuated by inflammatory mediators that sensitize nociceptors and somatosensory neurons, prolonging pain perception (Liu & Kelliher, 2022). To address this, nonsteroidal anti-inflammatory drugs (NSAIDs) are commonly prescribed globally to ease nociception and reduce inflammation, which is the major contributor to pain symptoms. However, prolonged use of analgesic medications like NSAIDs can lead to adverse side effects (Queremel Milani & Davis, 2023). Thus, there is a pressing need for the formulation of cost-effective inflammation-reducing medication with minimal side effects.
Medicinal plant products remain the readily available and cost-effective drug for primary healthcare in developing nations (Gumisiriza et al., 2019). The advantage of utilizing plant-based remedies lies in their phytochemistry, offering minimal side effects and a high acceptance rate within communities for treating various human ailments (Kerdar et al., 2019). Consequently, the use of phytochemicals as substitute drugs is rapidly gaining traction. The World Health Organization (WHO) inventory lists over 20,000 species of medicinal plants, highlighting the vast potential they offer (Vaou et al., 2021). Information about therapeutic florae yield secondary metabolites with healing applications, with approximately 50% of recent medications derived from these sources (Sharma et al., 2021). Therefore, we assessed the efficacy of the phytochemical tomatidine in preventing various noxious stimuli-induced nociception.
Nociception is a multifaceted and varied phenomenon that differs significantly depending on the affected tissue as well as the mechanism of injury, including thermal, mechanical, inflammatory, or neuropathic causes (DeSantana & Sluka, 2008; Schmidt et al., 2010). Over time, varied in vivo models have been developed to enhance our understanding of pain across various disease states, encompassing both acute and chronic conditions (Gregory et al., 2013). In our study, we evaluated the efficacy of tomatidine with varied nociceptive mice models, hence confirming its analgesic potency to treat different types of analgesia. Reflex pain tests measure behavioral responses to various stimuli such as heat, cold, mechanical pressure, and electrical input. It activates nociceptive receptors at the application site and elicits specific motor reactions. Reflexive behavioral assessments of these responses meticulously simulate human studies of increased sensitivity to painful stimuli, known as hyperalgesia (Coronado et al., 2011). Therefore, these tests can offer valuable insights into human pain responses. The use of thermal stimuli forms the foundation of many widely used tests to detect either hyperalgesia or analgesic effects. Some of the earliest methods for thermal testing include the Tail Flick Test and the hot plate test (D’Amour & Smith, 1991; Gregory et al., 2013; Woolfe & MacDonald, 1944). Treatment with tomatidine significantly increased the time of response in both thermal stimuli mice model hot plate test and tail immersion test confirming its pain-relieving potency. Tomatidine also exerts its pain-relieving effect in the presence of opioid antagonist naloxone.
The visceral pain in animal models is typically induced by chemical acetic acid and it is also induced by mechanical and electrical trauma (Ness & Gebhart, 1990). The organs of the pelvic and abdomen region or the peritoneum are irritated with the acetic acid to analyze the visceral pain behavior, which is exhibited with the abdominal writhes. The abdominal writhes induced by acetic acid are due to its prostaglandin-stimulatory effect, which leads to activation of sensory polymodal neurons (Gawade, 2012). Tomatidine treatment effectively inhibited the acetic acid-induced abdominal writhes, which proves its potency in alleviating visceral nociception.
Neuropathic nociception originates from intricate physiological changes that occur after peripheral nerve injury. It implicates various ion channels, receptors, and proteins, leading to nerve cell dysfunction and increased excitability (Smith, 2020). One significant factor in the development of neuropathic pain is the dysfunction of voltage-gated sodium channels, which contributes to increased neuronal firing. Furthermore, following injury, the release of glutamate neurotransmitters becomes more pronounced, activating pain pathways and amplifying pain perception (Bennett et al., 2019). Capsaicin, a phytochemical targets the transient receptor potential cation channel vanilloid subfamily member 1 (TRPV1), which plays a role in peripheral inflammation, both somatic and visceral, and is crucial in modulating nociceptive signals to the spinal cord and brain stem centers (Frias & Merighi, 2016). To evaluate the effect of tomatidine against neuropathic pain, we performed both the glutamate-induced pain model and capsaicin-induced pain model. In both the models, tomatidine effectively reduced the licking number, which correlates with our study, where the NSAID blocks prostaglandin and the calcium channels present on the nociceptive afferent nerves, thereby inhibiting glutamate discharge and the nociception (Johnson et al., 2011).
The formalin test is regarded as a highly effective test for the development of new analgesic drugs (Mogil, 2009). This test enables the examination of two distinct types of pain: (a) acute peripheral pain, which is facilitated by the direct activation of nociceptors via TRPA1 channels, and (b) inflammatory and central nociceptive sensitization (López-Cano et al., 2017). Tomatidine treatment significantly inhibited both the acute peripheral and central nociception pain induced with the formalin nociception mice model.
Inflammation is characterized by an increased presence of mediators that are either transported through the bloodstream or released at the site of inflammation by endothelial cells, resident cells, or migrated leukocytes. Many of these mediators can induce pain by activating specialized primary afferent nerve endings known as nociceptors (Rittner & Brack, 2007; Zhang & An, 2007). Tumor necrosis factor-beta (TNF-β) triggers a signaling cascade that activates IL-6, interleukin-8 (IL-8), IL-1β, nerve growth factor,prostaglandins, and sympathetic amines, which acts on nociceptors. (Sommer & White, 2010). Antisera against TNF-β or knockouts of TNFR1 have been shown to alleviate inflammatory pain. The hyperalgesic effects of IL-1β have been demonstrated through intraplantar or intraneural injections, involving mechanisms of action via prostaglandins, nitric oxide, and bradykinin. Neutralizing IL-6 or IL-6 knockouts has been reported to inhibit inflammatory pain (Ayoub et al., 2020; Pinto et al., 2021).
Since inflammation plays a vital role in nociception, we evaluated the analgesic potency of tomatidine with various carrageenan-induced inflammatory mice model. Tomatidine, which has been reported to inhibit inflammation in lipopolysaccharide (LPS)-stimulated murine macrophages (Chiu & Lin, 2008) and primary chondrocytes (Chu et al., 2020), via suppressing NF-κB signaling pathways and eventually inhibits the proinflammatory cytokines synthesis. In our study, tomatidine treatment significantly prevented the peritoneal leukocytes infiltration induced by the carrageenan and also inhibited the synthesis of proinflammatory cytokines production. Finally, the sedative effect, which is the prime side effect of analgesics was assessed in the tomatidine-treated mice with an open-field test. The results of open field analysis confirm that tomatidine treatment does not render any sedative effect in mice.
Conclusion
Analgesic medications, opioids, NSAIDs, and adjuvant therapies are essential for nociception management, but their prolonged use has led to significant health crises. Therefore, developing an analgesic drug without side effects is a global necessity. We evaluated the efficacy of a steroidal alkaloid, tomatidine, to inhibit nociception in mice. Given the variability in pain physiology among individuals, we assessed tomatidine efficacy using various nociception models in mice. Tomatidine treatment effectively inhibited reflex pain induced by thermal stimuli, visceral nociception stimulated with acetic acid, neuropathic pain induced by glutamate and capsaicin, biphasic nociception stimulated by formalin, and inflammatory nociception stimulated by carrageenan. Additionally, tomatidine did not produce any sedative effects in the mice. The results from these pain model analyses indicate that tomatidine possesses potent antinociceptive properties and holds promise as a potential analgesic drug.
Abbreviations
ANOVA: One-way analysis of variance; EDTA: Ethylenediaminetetraacetic acid; NSAIDs: Nonsteroidal anti-inflammatory drugs; PBS: Phosphate-buffered saline; PTSD: Post-traumatic stress disorder; TNF-β: Tumor necrosis factor-beta; TRPV1: Transient receptor potential cation channel vanilloid subfamily member 1; WHO: World Health Organization.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This work was approved by the Institutional Ethical Committee, Shanghai University of Traditional Chinese Medicine, Shanghai 200082, China (Ethical number: 2023/74).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
