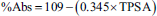Abstract
Background
Historical accounts suggest that, throughout antiquity, medicinal plants and their constituent parts have been used to treat a variety of ailments. Asparagus racemosus, this plant exhibits a wide range of potential uses in medicine.
Objectives
The purpose of this study is to identify any potentially bioactive ingredients in the methanolic extract of A. racemosus.
Materials and Methods
A variety of compounds were detected using gas chromatography, and their molecular structures were confirmed by Fourier transform infrared (FTIR) analysis and mass spectrometry. Using in silico approaches, the molecule’s potential as a therapeutic candidate, biological activity, and projected targets were carefully examined.
Results
The molecule 4-(((9-(5-(ethylcarbamoyl)-3,4-dihydroxy tetrahydrofuran-2-yl)-9H-purin-6-yl)amino)methyl)benzoic acid was found in the highest concentration (27.37%). The identified molecule is purine alkaloid in nature. The promising drug-like properties and therapeutic efficacy for the investigated molecule.
Conclusion
It could serve as a viable therapeutic option for addressing cardiovascular disorders.
Introduction
Many individuals exhibit reluctance toward the abundant synthetic and chemical medications available today, primarily due to their perceived adverse effects (Galeano et al., 2020). The traditional herbs gain popularity due to minimal toxicity (Sahoo & Manchikanti, 2013). Although there are many effective modern synthetic medications available, individuals are choosing plant-based natural remedies (Yuan et al., 2016). This inclination arises from the diverse phytoconstituents present in various plant parts, which can treat and cure diseases (Anand et al., 2019; Semwal et al., 2019). The practice of using plants as medicine is deeply rooted in the Indian medical tradition, with evidence indicating its prevalence since ancient times (Pandey et al., 2013).
More than 25% of the active compounds found in contemporary prescription drugs come from medicinal plants (Malongane et al., 2017). These plants’ bioactive compounds have been linked to a variety of pharmacological effects, including anti-inflammatory, anticancer, antifungal, and antioxidant qualities (Yadav, Khare et al., 2017). Consequently, it is critical to assess these bioactive chemicals’ potential to understand their feasibility in treating various illnesses (Juszczak et al., 2019).
The bioactive chemicals that are isolated and identified from medicinal plants are the source of many very effective drugs (Satapute et al., 2019). Chromatographic and spectrophotometric methods are crucial for comprehending the chemical and pharmacological properties of these herbs (Fan et al., 2018; Razack et al., 2015). An improved method for tracking biomolecules in herbal extracts is the application of hyphenated methods (Krishnaveni & Mirunalini, 2010). For this reason, we have used the same methodology to analyze the medicinal herb Asparagus racemosus, also referred to as Shatavari.
Asian in origin, A. racemosus belongs to the Liliaceae family and grows at lower altitudes. This plant is used in medicine with its dried roots (Vishwakarma & Goswami, 2013). These roots are said to have ulcer-healing qualities, which are probably attained by boosting mucosal resistance or offering cytoprotection (Kamat et al., 2000). Its efficacy in mitigating acquired immunodeficiency syndrome (AIDS) symptoms has also been demonstrated by recent research, and Ayurvedic practitioners have traditionally utilized it to treat neurological disorders. It has been used for a variety of purposes, but there is not any scientific data to support these claims. However, other studies indicate that A. racemosus root extracts have advantageous qualities such as teratogenicity, immunomodulatory, antihepatotoxic, antilithiatic, and galactagogue (Sliwoski et al., 2014).
The research on various treatments for Parkinson’s disease (PD) reveals promising insights into their potential cardioprotective and neuroprotective activities. Immune control and neuroinflammation are critical factors in PD progression. Neuroinflammation, driven by misfolded α-synuclein and subsequent chronic activation of glial cells, accelerates nigrostriatal degeneration, leading to the characteristic motor symptoms of PD (Rai, Birla, Singh et al., 2017).
Mucuna pruriens (Mp), a traditional Ayurvedic medicine, shows potential in this context due to its anti-inflammatory and antioxidant properties. Mp contains bioactive compounds like
Similarly, Withania somnifera (Ws), another Ayurvedic herb, demonstrates neuroprotective effects against PD induced by environmental toxins like Maneb (MB) and Paraquat (PQ). Ws treatment improves locomotor activity, dopamine levels, and oxidative damage markers. It reduces oxidative stress and modulates apoptotic pathways by increasing Bcl-2 and decreasing Bax protein levels, alongside reducing GFAP expression (Prakash et al., 2014)
Both Mp and Ws highlight the potential of herbal remedies with anti-inflammatory and antioxidant properties in treating neurodegenerative diseases. Their effects on immune modulation, oxidative stress, and apoptosis underscore their role in offering neuroprotection and potentially contributing to cardioprotective activity by maintaining overall brain health and reducing neuroinflammation (Yadav, Rai et al., 2017).
Satavari offers potential benefits that align with the cardioprotective and neuroprotective effects observed in M. pruriens and W. somnifera through its impact on immune modulation, oxidative stress, and apoptosis. Its ability to reduce inflammation, combat oxidative stress, and regulate apoptotic processes underscores its role in maintaining overall brain health and potentially providing cardiovascular protection. By integrating these properties, Satavari may serve as a valuable therapeutic agent in addressing both neurodegenerative and cardiovascular conditions (Kaur et al., 2020).
The use of computer-aided technology in the discovery of novel medications has increased recently. According to Lee and Kim (2019), the process of separating active molecules from phytochemicals in medicinal plants has become more efficient. The use of computer-aided technology in the creation of novel medications has increased recently (Lamers & Feys, 2014). Senthil et al. (2022a) state that in silico research in conjunction with target prediction is a useful tool for target prediction and illness identification methods.
To determine the bioactive components of the methanolic extract made from A. racemosus, gas chromatography–mass spectrometry (GC–MS) analysis was performed. After that, putative bioactive chemicals were investigated further using in silico techniques and computer-aided molecular analysis.
Materials and Methods
Procurement of Plant Material
Fresh A. racemosus (Shatavari) stems were gathered from Ibrahimpatnam, Telangana’s Lingampally hamlet. The Department of Botany at Hyderabad University in Hyderabad verified the authenticity of the plant materials.
Preparation of Extracts
The stems were let to air dry in a shaded area at 38 ± 5°C. The dried material was ground into a coarse powder using the Crompton TRET500 India electric grinder. After adding methanol to the powdered material, a Soxhlet equipment was used to extract the substance. One hundred grams or more of stem powder were put into a muslin cloth bag with 100 mesh size. Petroleum ether was used to defat the stem powder. One liter of methanol was used in the extraction procedure, which continued until a colorless liquid was obtained. It was recycled using a rotary flash evaporator (Aditya Scientific RE-3A rotary evaporator, Hyderabad, India) that ran at 75°C, 55 rpm, and lower pressure. The dry residue kept in the desiccator was used to calculate the yield percentage (Morris & Lim-Wilby, 2008).
Phytochemical Examination
Phytochemical analysis of the extract was conducted using a standard approach (Kokate, 2001).
GC–MS Analysis
Weighing 100 mg of extract, it was dissolved in 100 mL of high-pressure liquid chromatography (HPLC)-grade methanol. After that, the resulting mixture was diluted to achieve a 30 µg/mL concentration. The GC program was executed using the Agilent 7890A GC System, paired with a JEOL AccuTOF GCv/JMS-T100GCv mass spectrometer detector. The analysis utilized an HP5 column with dimensions of 30 m in length, 0.25 mm internal diameter, and a 0.25 µm film thickness, made from polysiloxane material. Helium served as the carrier gas at a flow rate of 1 mL/min. The oven temperature was set to 280°C, and the injection was performed at 70°C with a volume of 1 µL. With a split ratio of 1:10, the sample flow rate was kept constant at 1 mL/min, and the run took 50 min in total. The JEOL AccuTOF GCV model was utilized in the positive mode of electron ionization (EI) for the MS procedure. The mass range that was examined was 35–800 amu. The temperature of the ion source was kept at 220°C, and the split ratio was adjusted to 1:10. There was a 4-min solvent delay added, and the entire MS run time was 50 min. By comparing the mass spectra of the separated and obtained compounds with those in the NIST library database, the compounds were identified during the study (Sen et al., 2022). The concentration of compounds was determined based on peak area and calculated using the formula:
In Silico Study
The Osiris Data Warrior software and the Swiss ADME tool, which can be found at
Drug Likeliness and Pharmacokinetic Potential
Swiss ADME was used to forecast the drug’s likelihood as well as its pharmacokinetic and toxicity profiles (
Target Prediction
Swiss target prediction was used to forecast the identified compounds’ therapeutic efficacy (
Results
The aim of this research was to pinpoint various nonpolar biomolecules present in the methanolic extract derived from A. racemosus. Utilizing chromatographic analysis, the different kinds of compounds found in the extract were identified. The selected chemicals’ structures are validated using spectral analysis. The various extract constituents’ drug-likeliness behavior was validated by an in silico analysis and target prediction. There are several known medical uses for the selected plant. To determine the plant extract’s molecular identification and potential therapeutic benefit, this study concentrated on evaluating the extract’s target potential.
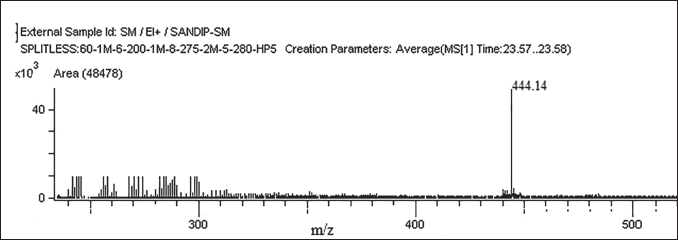
Yield Proportion and Plant Compound Examination
The methanolic extract exhibited a yield of 24.8%. Subsequent analysis unveiled the existence of terpenoids, phenolic compounds, flavonoids, steroids, alkaloids, and aliphatic compounds within the extract.
GC–MS Analysis
The maximum percentage peak areas 27.37% (retention time: 19.54) were traced for compound number 9. The other compounds that were traced were molecule-7 (24.41, RT—12.64) and molecule-2 (11.83, RT—6.41) (Figure 1 and Table 1). Mass spectroscopy was used to confirm the compounds 9 structures (Figure 2). Thirteen compounds were identified in the methanolic extract (Figure 3).
Chromatographic Profile of the Methanolic Extract from Asparagus racemosus Obtained via GC.
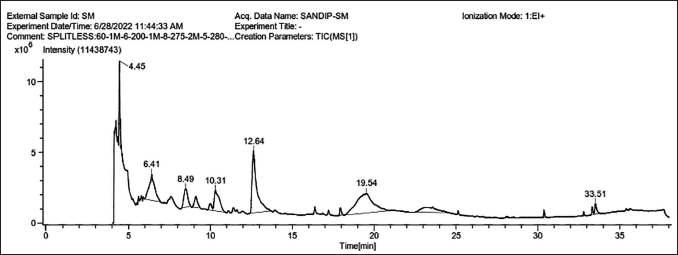
Gas Chromatography Analysis of Methanolic Extract from
Asparagus racemosus
.
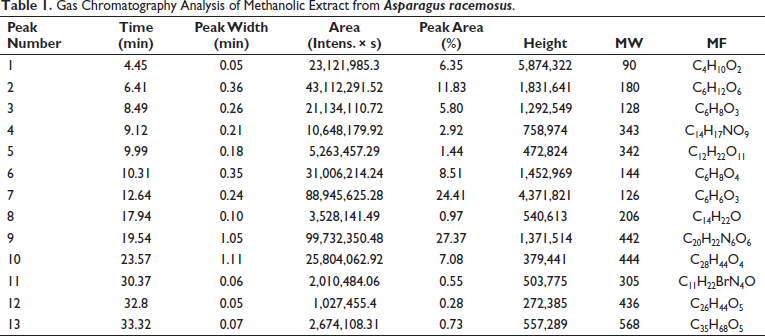
Mass Spectrum of Molecule-9 Identified in the Methanolic Extract Asparagus racemosus.
Molecular Structures Identified in the Methanolic Extract of Asparagus racemosus.
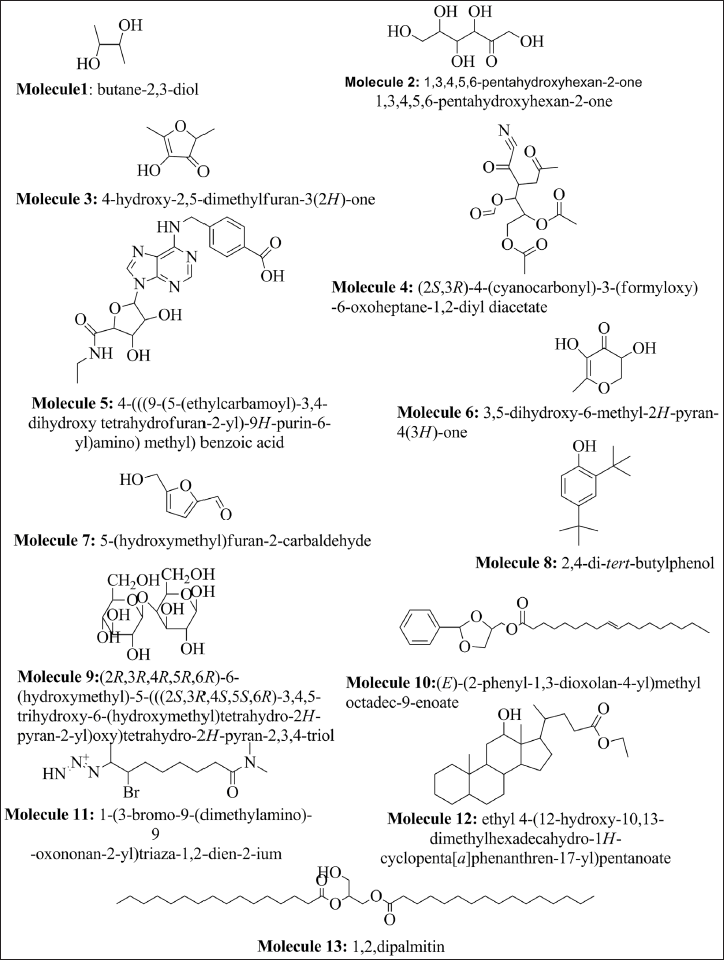
Molecular Property
The efficiency of a drug’s action and its ability to attach to a receptor are greatly impacted by factors including molecular structure, flexibility, and complexity. It is generally believed that molecules having a spherical shape exhibit better absorption (Nicholls et al., 2010). “Efficient receptor binding is usually favored by increased flexibility and decreased molecular complexity” (Méndez-Lucio et al., 2017; Rohs et al., 2005). Table 2 provides a summary of these results. Compounds 4, 8, and 9 show spherical shapes, while compounds 3, 5, 6, 7, and 12 show minimal flexibility while the other molecules show linear shapes. Furthermore, every molecule—aside from molecule 11—shows an increase in molecular complexity.
Molecular Properties of the Compounds Recognized by Gas Chromatography–Mass Spectrometry (GC–MS) Analysis.
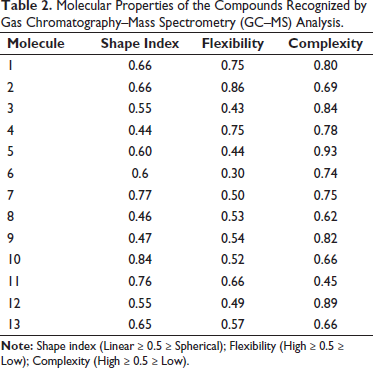
Physicochemical Properties
The likelihood of a molecule being a medicine is greatly influenced by its physicochemical characteristics (Benet et al., 2016; Prasanna & Doerksen, 2009). Table 3 and Figure 4 provide a summary of these findings. The cLogP values of molecules 3, 7, 8, 11, and 12 range from 0.06 to 5.71. High toxicity is indicated by values 7.91 and 27.24 for molecules 10 and 13. Every molecule, except for molecules 1, 3, 6, 7, 8, 10, and 12, has more than five hydrogen acceptor sites. Every molecule, except for one, has fewer than 10 hydrogen donor sites. The molar refractive indices of molecules 4, 5, 8, 9, 10, 11, and 12 range from 40 to 130. Similarly, all molecules except molecules 5 and 9 have total polar surface area (TPSA) values between 90 and 140.
Physical and Chemical Traits of the Molecules Identified through Gas Chromatography (GC) Analysis.
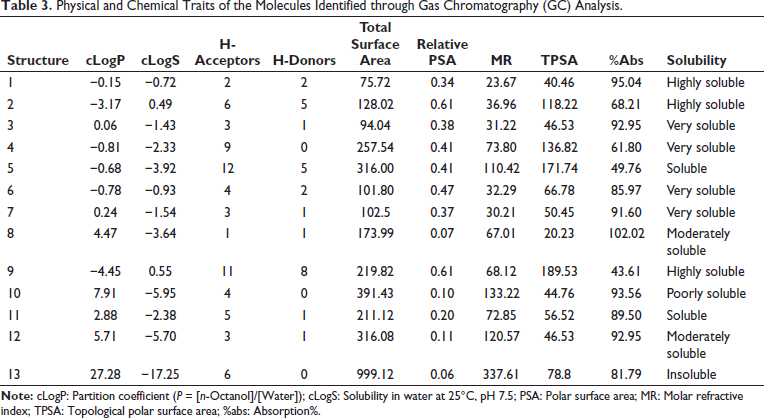
Bioactivity Rader for Molecules Inmethanolic Extract of Asparagus racemosus.
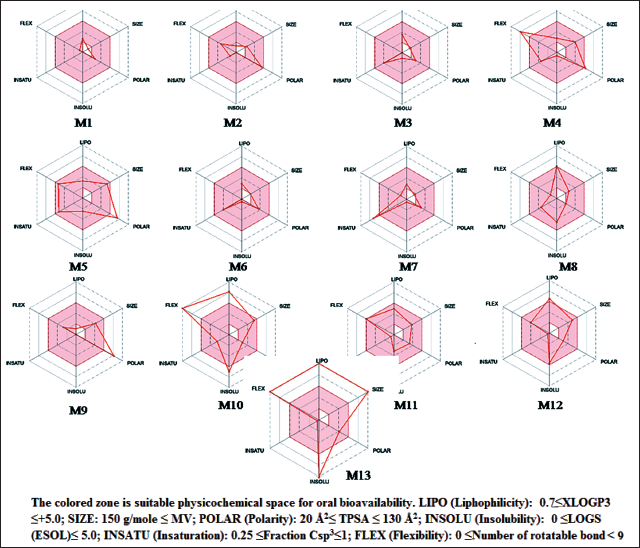
Druglikeness
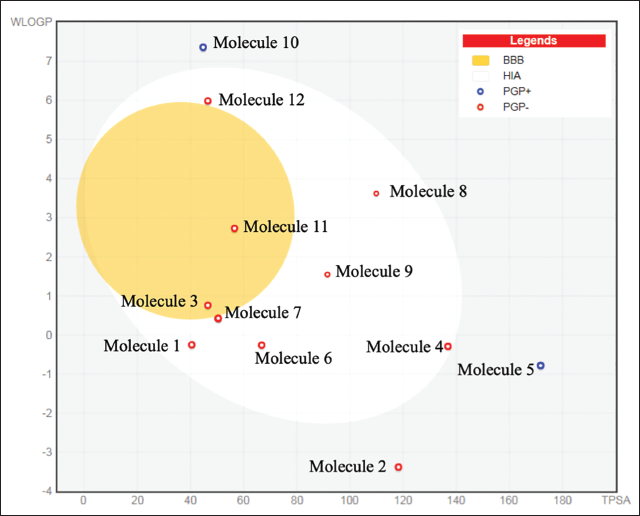
With the exception of molecules 5, 9, 10, 12, and 13, all identified molecules adhered to the Lipinski rule. The Ghose rule was only obeyed by molecules 4, 8, and 11. The molecules 4, 5, 9, 10, 12, and 13 also adhered to Veber and Egan’s guidelines. In a similar vein, only molecules 4 and 11 adhered to the Muegge criteria. Only molecules 2 and 10 exhibit drug-likeliness behavior, according to a thorough investigation, with positive druglikeness values of 1.58 and 2.74, respectively. For every molecule except for 5, 9, and 13 (0.11–0.17), the bioavailability score was found to be 0.55. These results show that molecule-8 has advantageous drug-like characteristics (Table 4 and Figure 5).
Druglikeness of the Molecules Detected by Gas Chromatography–Mass Spectrometry (GC–MS) Analysis.
Schematic Representation of Molecules Recognized in the Methanolic Extract of Asparagus racemosus, Akin to a Boiled Egg Diagram.
Bioactivity Score and Toxicity Profiles
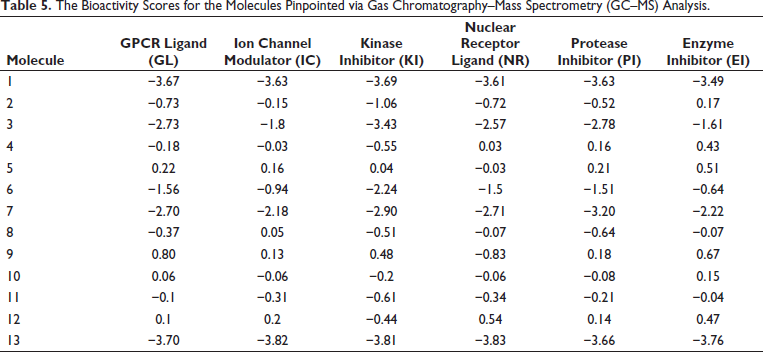
For compounds that bind to target receptors, the hierarchy of bioactivity is as follows: EI > IC > GL > PI > NR > KI, as Table 5 illustrates. Molecule 9 demonstrated binding affinity for G-protein-coupled receptors and showed multiple receptors bioactivity scores greater than zero. In 2016, Husain et al. (2016) argued that there were no hazardous effects shown by molecule 9 (Table 6).
The Bioactivity Scores for the Molecules Pinpointed via Gas Chromatography–Mass Spectrometry (GC–MS) Analysis.
Assessment of the Toxicity Potential of the Molecules Detected by Gas Chromatography–Mass Spectrometry (GC–MS) Analysis.

Pharmacokinetics Profiles
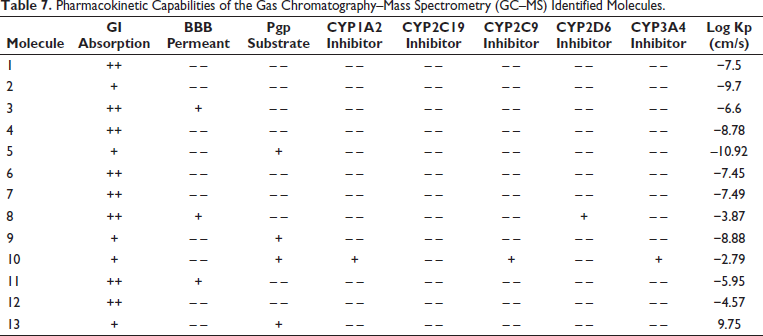
Diffusion is the main mechanism by which the active molecule is absorbed. Human intestinal absorption (HIA) and gastrointestinal (GI) absorptivity are important factors to consider when dealing with bimolecular compounds (Prescott, 1974; Wessel et al., 1998). Compared to the stomach, the small intestine provides a larger surface area for medication absorption. Moreover, the blood–brain barrier (BBB) prevents cytotoxic effects by regulating the entry of drug molecules into the central nervous system (Pardridge, 2012).
Drug distribution and excretion depend heavily on P-glycoprotein (PGP) (Tanigawara, 2000). It stops pharmaceuticals from building up in the brain by inhibiting the BBB and oral bioavailability of medications. PGP inhibition may result in increased drug accumulation in the brain and medication interactions (Lin & Lu, 1998). Enzymes known as cytochrome P450 (CYP) are essential for drug metabolism. Certain medications may reduce drug metabolism and other metabolic processes if they inhibit certain enzymes. For medications meant for topical application, skin permeability (KSP) is a crucial consideration (Potts & Guy, 1992).
There is a low GI absorptivity for 2, 5, 9, 10, and 13. There was evidence of BBB penetrability for 3, 8, and 11. For 1, 3, 4, 7, and 8, a higher HIA capacity was traced. Except for molecule 10, none of the other molecules showed any CYP inhibitory action; only molecules 5, 9, 10, and 13 showed the PGP efflux effect. Furthermore, Table 7 and Figure 4 show that the molecules’ skin permeability falls within a reasonable range.
Pharmacokinetic Capabilities of the Gas Chromatography–Mass Spectrometry (GC–MS) Identified Molecules.
Prediction and Analysis of Molecular Targets
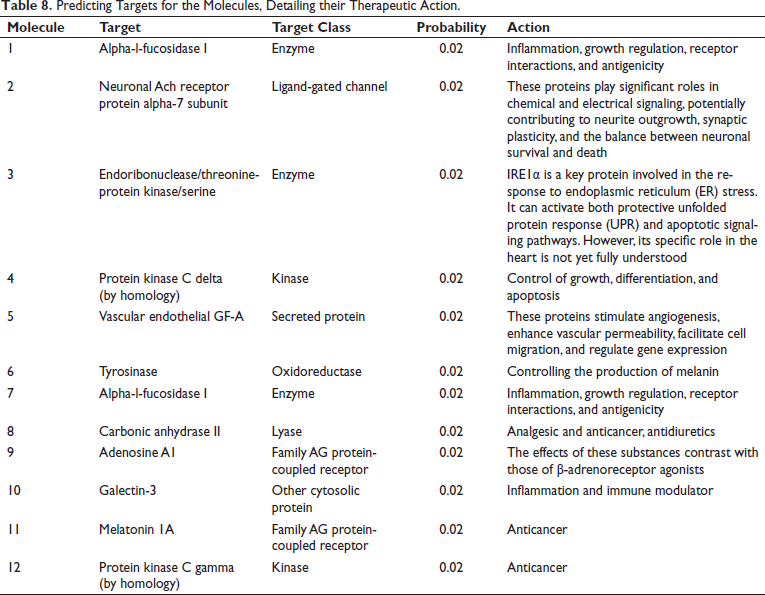
Target prediction analysis was performed for each molecule based on the bioactivity score; the findings are shown in Table 8 and were connected with the outcomes of the GC–MS and in silico investigations. Molecule 9 targets the G-protein coupled beta adreno receptor, which suggests that it may be used as a cardioprotective drug, according to the analysis. Their behavior is contrary to that of β-adrenoreceptor agonists. For example, adenosine inhibits β-mediated tachycardia and lipolysis. According to molecule 9 from Satavari, it may therefore be an important biomolecule for the treatment of cardiovascular problems (Gfeller et al., 2014). The purine alkaloids, such as theophylline, are diuretics, muscle relaxants, vasodilators, and heart stimulants (Zeng et al., 2022). Other important compounds, such as molecules 2 and 7, have been employed as anti-inflammatory drugs and to preserve the equilibrium between neuronal death and survival.
Predicting Targets for the Molecules, Detailing their Therapeutic Action.
Discussion
Shatavari (A. racemosus) is an Ayurvedic herb traditionally known for its adaptogenic and reproductive health benefits. While it is most associated with supporting the female reproductive system and promoting hormonal balance, research has also explored its potential cardiovascular properties (Visavadiya & Narasimhacharya, 2005). It has been demonstrated that Shatavari lowers cholesterol, especially low-density lipoprotein (LDL) cholesterol, which is frequently referred to as “bad cholesterol.” Reducing LDL cholesterol is essential for lowering the risk of heart disease and atherosclerosis. The herb may have a hypotensive (blood pressure-lowering) effect. This is likely due to its diuretic properties and its ability to promote vasodilation (widening of blood vessels), which can reduce the workload on the heart and lower blood pressure (Ziauddin et al., 1996). Shatavari has several properties that may be beneficial for cardiovascular health, including its antioxidant, cholesterol-lowering, blood pressure-regulating, and anti-inflammatory effects. These properties suggest that Shatavari could be supportive in maintaining heart health and preventing cardiovascular diseases. Purine alkaloids are a broad class of naturally occurring substances that are present in many kinds of plants. They are particularly well-known for their stimulating properties, and their presence in various plants reflects a significant aspect of chemical biodiversity. Purine alkaloids, particularly caffeine, theobromine, and theophylline, have significant cardiovascular effects ranging from increased heart rate and blood pressure to potential antiarrhythmic properties (Barnes, 2013). These effects depend on the specific alkaloid, its dosage, and the individual’s underlying health conditions. Caffeine tends to have the most potent and variable effects, while theobromine and theophylline exhibit milder actions with some therapeutic benefits (Molfino & Nannini, 2002). It has been observed that alcoholic root extract of A. racemosus causes cardiac collapse at higher dosages and has positive ionotropic and chronotropic effects on the frog heart. Cats exposed to the extract experienced hypotension, which atropine prevented, suggesting a cholinergic mode of action. Additionally, after the extract was administered intravenously to rabbits, there was a modest increase in bleeding time and no effect on clotting time, as well as congestion and a full standstill of blood flow in the mesenteric veins of mice and rats (Roy et al., 1971). The GC–MS analysis traced molecule-9 as a principal constituent. A study’s 27.37% concentration of the purine alkaloid is high compared to typical bioactive compounds in medicinal plants, which rarely exceed 10–15%. Such a high concentration suggests the plant has therapeutic potential but may pose safety concerns. For example, studies on caffeine in coffee beans report concentrations of only 2–4%. The molecular properties of the compound exhibited a linear shape (0.47) with moderate flexibility (0.57) and high complexity (0.82). The physicochemical property exhibited good druglikeness behavior (2.74) but with a percentage of absorption and bioavailability. The positive bioactive score showed multireceptor binding affinity, but more binding affinity was found for the G-protein coupled receptor. The pharmacokinetic potential predicted no toxicity with moderate GI-absorptivity with good skin permeability. No CYP-inhibition was found, but a positive PGP substrate inhibitor was found, which indicated that it inhibits the BBB and oral bioavailability of drugs, preventing drug accumulation in the brain.
The predicted target for the reported molecule is the Adenosine A1 receptor. A1Rs control neurotransmitter release, neuronal excitability, blood vessel dilation, and heart rate. A1Rs induce cardiac depression, such as bradycardia and negative cardiac inotropy, and vasoconstriction and inhibit renin release (Ribeiro et al., 2002).
The reported molecule is complex and resembles a type of nucleoside analog. These compounds are often used in antiviral, anticancer, and immunomodulatory therapies.
It may exhibit cardiovascular effects due to its structural similarity to other purine derivatives and nucleoside analogs. This compound could affect the cardiovascular system by acting as a vasodilator. Adenosine is known to cause vasodilation, particularly in the coronary arteries, improving blood flow to the heart. If this compound has adenosine-like activity, it could promote vasodilation and improve cardiac function (Belardinelli et al., 1995).
Adenosine is used to treat supraventricular tachycardia (SVT). It could exhibit antiarrhythmic properties if the compound interacts with adenosine receptors or similar pathways. It might lower blood pressure by promoting relaxation of the vascular smooth muscles or increase blood pressure if it has sympathomimetic effects (stimulating the sympathetic nervous system). This compound could either enhance or diminish the force of cardiac contractions, depending on its interaction with cellular pathways related to calcium handling and signaling within the heart muscle (Rubin & Kron, 1994). Further studies would be required to determine the exact cardiovascular profile of this specific compound.
Comparing the therapeutic potential of purine alkaloids to existing cardiovascular drugs involves evaluating their mechanisms of action, efficacy, safety, and overall therapeutic value. The identified purine alkaloid has potential as a cardioprotective agent, mainly due to its mild vasodilatory and diuretic effects. However, it is generally less potent and less specific in action compared to established cardioprotective drugs. While it might be helpful as an adjunct therapy or in mild cases, its broader and less predictable effects, especially at higher doses, mean it does not match the efficacy or safety of existing cardiovascular medications designed to target specific conditions with precision and reliability (Green & Jones, 2002).
There have been studies on the cardioprotective effects of purine alkaloids, particularly caffeine and theobromine. Studies suggest that moderate coffee intake may be linked to better cardiovascular health and a decreased risk of heart disease. Similarly, theobromine, found in cocoa, has been linked to reduced blood pressure and better blood vessel function. Clinical trials and observational studies support these findings, though results can vary based on individual health and consumption levels. Even though the data points to possible advantages, it is best to speak with medical professionals before making big dietary adjustments because of these impacts (Miller & McDonald, 2006).
From the above discussion, it can be concluded that molecule-9 can be considered as potential cardiovascular agent. The exact mechanism can be described by in vivo and in vitro experimental models. The in silico study predicted poor absorption and bioavailability of the reported compound, which can be improved by formulation development and nanoparticle design. Future investigations will focus on the molecule’s isolation and structural conformation. After confirmation of the structural analysis, the therapeutic efficacy of the molecule must be established, followed by improvement of oral bioavailability.
Conclusion
The study identified eleven molecules through gas chromatographic analysis, with their structures confirmed by spectral analysis. The highest concentration was observed for 4-(((9-(5-(ethylcarbamoyl)-3,4-dihydroxytetrahydrofuran-2-yl)-9H-purin-6-yl)amino)methyl)benzoic acid. It has a greater capacity for HIA and displays good drug-likeliness behavior without any toxicity. Target prediction analysis confirmed therapeutic potential as a cardioprotective agent. These findings highlight the success of the reported molecule as a potential therapeutic agent for the treatment of multiple cardiovascular disorders.
Footnotes
Abbreviations
% Abs: Percentage absorption; °C: Degree celsius; µL: Microliter; A1Rs: Adenosine A1 receptor; ADME: Absorption distribution metabolism and excretion; Bax protein: Bcl-2-associated X protein; BBB: Blood–brain barrier; Bcl-2: B-cell lymphoma-2; cLogP: Partition coefficient; CYP: Cytochrome P-450; EI: Enzyme inhibitor; GC–MS: Gas chromatography–mass spectroscopy; GFAP: Glial fibrillary acidic protein; GI: Gastrointestinal; GL: G-Protein ligand; HIA: Human intestinal absorption; HPLC: High-pressure liquid chromatography; IC: Ion channel; KI: Kinase inhibitor; LDL: Low-density lipoprotein; MF: Molecular formula; MR: Molar refractive index; MW: Molecular weight; NF-κB: Nuclear factor kappa B; NR: Nuclear receptor; pAkt1: AKT serine/threonine kinase 1; PD: Parkinson diseases; PGP: P-glycoprotein; PI: Protease inhibitor; PSA: Polar surface area; SVT: Supraventricular tachycardia; TNF-α: Tumor necrosis factor-alpha; TPSA: Topological polar surface area.
Acknowledgments
We express our sincere appreciation to SAIF and IIT Bombay for their assistance with the GC–MS analysis. Additionally, we extend our heartfelt thanks to the School of Pharmacy, Guru Nanak Institutions Technical Campus for generously providing the necessary facilities for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Deanship of Graduate Studies and Scientific Research, Jazan University, Saudi Arabia, through project number: (RG24-M019).