Abstract
Background
Cardiovascular disease continues to be a predominant source of morbidity and mortality globally. Commiphora gileadensis L. is a tree under the genus Commiphora.
Objectives
To investigate the anti-inflammatory effect of C. gileadensis L. on cardiovascular and its action in maintaining cardiovascular integrity in Type 1 diabetic mice.
Materials and Methods
Fifty male Bagg albino (BALB/c) mice are classified into five groups: control, untreated diabetic, diabetic C. gileadensis L. sap-treated, methanol extract-treated, and acetone extract-treated. For each mouse in five groups body weight, glycated hemoglobin (HbA1c), sodium, potassium, chloride, urea, creatinine, alanine aminotransferase (ALT), lipid profile, aspartate aminotransferase (AST), lactate dehydrogenase (LD), total creatine kinase (CK), creatine kinase-myocardial band (CK-MB), adropin, nitric oxide (NO), endothelin-1, and vascular endothelial growth factor (VEGF) were measured. In addition, total lymphocytes, CD3+, CD4+, CD8+, CD4+ CD25+, and CD8+ CD25+ were measured in all groups.
Results
Compared to the untreated diabetic group, diabetic groups treated with C. gileadensis L. had significantly increased body weight after 6 months (p < 0.05). Diabetic mice treated with C. gileadensis L. extracts had significantly lower HbA1c levels than the diabetic untreated group (p < 0.01). Furthermore, C. gileadensis L. extracts lowered serum urea and creatinine in diabetic mice (p < 0.05). Triglyceride, cholesterol, and low-density lipoprotein (LDL) were reduced in mice treated with C. gileadensis L. extracts, whereas high-density lipoprotein (HDL) was elevated compared with diabetic untreated mice (p < 0.01). Serum AST and endothelin-1 were significantly decreased in diabetic groups treated with C. gileadensis L. extracts (p < 0.05), while adropin and NO increased (p < 0.01). Diabetic mice treated with C. gileadensis L. extracts had significantly lower LD, total CK, CK-MB, and VEGF levels than the untreated diabetic group (p < 0.01). In addition, C. gileadensis L. reduces CD3+, CD4+, CD8+, while increases CD4+ CD25+, and CD8+ CD25+ subsets in diabetic mice (p < 0.0001).
Conclusion
C. gileadensis L. induces the levels of cardiovascular integrity markers (adropin and NO) and reduces cardiovascular malfunction markers [endothelin-1 (ET-1) and VEGF] in diabetic mice. The observed properties could be attributed to the antioxidant activity of C. gileadensis L. Furthermore, C. gileadensis L. may help in the prevention of atheroma formation by reducing CD4+ and CD8+ and increasing T regulatory cells (CD4+ CD25+ and CD8+ CD25+).
Introduction
The cardiovascular system is controlled in cellular activity and homeostasis by many hormones (Cooper-DeHoff & Pepine, 2007; Montani et al., 2011). Adropin is a peptide hormone comprising 76 amino acids, encoded by the energy homeostasis-associated gene (ENHO). The amino acid sequence is similar among humans, mice, and rats (Kumar et al., 2008). Adropin expression is predominantly observed in the liver and brain. It is essential for sustaining energy balance and controlling metabolism (Altamimi et al., 2019; Thapa et al., 2019). Adropin has systemic effects on insulin sensitivity and glucose metabolism across various organs, including the heart, liver, and skeletal muscle. Several studies have demonstrated the significant cardiovascular roles of adropin and its protective role in preventing damage to vascular endothelial cells (Lovren et al., 2010; Sato et al., 2018). These previous results indicate that the activation of adropin signaling may have advantageous effects on cardiovascular function. Ongoing research is being conducted to better understand the specific molecular mechanisms that contribute to these observed outcomes. Nitric oxide (NO) is a vital free radical in the circulatory system, particularly in microcirculation, related to its vasodilatory characteristics and its role in sustaining and regulating blood flow in healthy tissues (Stein et al., 2016; Wong et al., 2014). NO is produced via the enzymatic reaction of l-arginine and oxygen, facilitated by the enzyme nitric oxide synthase (NOS). Three isoforms of NOS exist: endothelial nitric oxide synthase (eNOS), neuronal nitric oxide synthase (nNOS), and inducible nitric oxide synthase (iNOS) (Llorens et al., 2002). eNOS and nNOS have been detected in the myocytes of the left ventricle. nNOS, specifically, is the constitutive isoform of the myocardium responsible for regulating cardiac inotropy and relaxation through the mediation of NO (Martin et al., 2006). In the vascular system, NO regulates blood flow, vascular tone, and platelet aggregation. These actions contribute to its antihypertensive, antithrombotic, and atherosclerotic effects (Förstermann, 2010; Li et al., 2014; Moncada & Higgs, 2006). Additionally, it plays a role in the activation of endothelial progenitor cells (EPCs) and smooth muscle cells (SMCs) proliferation (Fleissner & Thum, 2011). Endothelin-1 (ET-1) is a 21-amino acid peptide produced by endothelial cells in blood vessels and various other cells in the body (Rautureau & Schiffrin, 2012). ET-1 exerts a paracrine effect on the underlying vascular smooth muscle cells (VSMCs), leading to a significant vasoconstrictive response (Li et al., 2013). Vascular endothelial growth factors (VEGF) are glycoproteins that regulate angiogenesis by facilitating endothelial cell differentiation. Elevated VEGF activity has been linked to the presence of inflammation, heightened blood pressure, and the development of atherosclerotic lesions, ultimately contributing to the onset of coronary heart disease (O’Sullivan et al., 2022). There is growing evidence that both CD4 and CD8 T lymphocytes have a role in the development of atherosclerosis (Pan et al., 2017). Commiphora gileadensis L., also referred to as the Arabian balsam, is a tree classified under the genus Commiphora. It is indigenous to the Arabian Peninsula and southern Egypt. The wood, sap, bark, and seeds of the tree exhibit notable therapeutic characteristics (Al-sieni, 2014). Traditional Arabian medicine has used the tree in the treatment of inflammatory disorders, constipation, abdominal pain, arthralgia, and cephalalgia. Multiple research has recorded the tree’s antimicrobial characteristics (Bouslama et al., 2019). Moreover, the sap of C. gileadensis L. has been employed as an antibacterial agent in numerous in vivo and in vitro investigations (Alhazmi et al., 2020). Furthermore, a previous study indicated that the methanolic extract from C. gileadensis L. possesses antibacterial activities and aids in wound healing (Alhazmi et al., 2022). This study aimed to investigate the anti-inflammatory effect of C. gileadensis L. on cardiovascular and its action in maintaining cardiovascular integrity in Type 1 diabetic mice.
Materials and Methods
This analytical investigation was carried out at the Faculty of Applied Medical Sciences from February 2023 to August 2023.
Collection of C. gileadensis L.
The C. gileadensis L. sample was obtained from the Alaab Valley, a mountainous region near Al-Madinah city in western Saudi Arabia. In February 2022 (the optimal time for sap yield), the leaves and fallen branches of the tree were gathered.
Preparation of C. gileadensis L. sap
The uppermost sections of the C. gileadensis L. branches were trimmed, with a 5-mm gap remaining from the tips, and the sap that was released as a result of the cuts was immediately gathered. Following the addition of an equal volume of ethanol, the sap underwent centrifugation at a speed of 10,000 revolutions per minute for a length of 10 min, followed by agitation for 15 min at room temperature. Following that, the liquid portion was preserved at −20°C until it was exposed to further examination (Iluz et al., 2010).
Preparation of C. gileadensis L. Methanol Extract
The leaves and branches of C. gileadensis L. were cleaned with tap water and dried at 40°C in a hot-air oven. After drying, the material was processed into a fine powder. To remove contaminants, the material was sifted. After that, a 10 g sample of the powder was macerated for 24 h in a sterile funnel. The maceration process used 100 mL of 100% pure methanol. The funnel was vigorously agitated before filtering using sterile filter paper. Drying C. gileadensis L. extract in a 40°C water bath produced a concentrated extract. After 14 days in a refrigerator at 4°C, the specimen was stored at −20°C for evaluation (Eitan et al., 2015).
Preparation of C. gileadensis L. Acetone Extract
In a vacuum oven, the C. gileadensis L. leaves and branches were dried at 60°C for 6 h. After drying, a razor blade was used to cut the plant material into tiny pieces, creating a powder. Then, 10 g of unprocessed C. gileadensis L. pieces were immersed in 200 mL of acetone for 3 days at room temperature. The magnetic-stirrer-homogenized acetone solution was refreshed daily. The extracted material was dried with a rotary evaporator to remove any leftover acetone. After that, the sample was stored at −20°C until analysis (Sulimani, 2015).
Experiment Design
A total of 50 male BALB/c mice, averaging 2 months in age and weighing between 20 and 25 g, were purchased from the animal facility. The mice were housed in a standard rodent habitat equipped with woodchip bedding. The cage was situated in a well-ventilated, wide space, according to a 12-hour light/dark cycle, and was regularly regulated at a temperature of 25°C. During the trial, the mice were provided with standard rodent chow and tap water. After a 2-week acclimatization period, all 50 mice were randomly assigned into five groups, each including 10 animals.
The first group was assigned as a negative control; no therapy was given to the mice in this group.
The second group was the diabetic-untreated group. Streptozotocin was delivered at 55 mg/kg body weight intraperitoneally into these mice for 5 days to induce diabetes. Streptozotocin prevents deoxyribonucleic acid (DNA) synthesis in mammalian cells and causes cell death. Three days later, Streptozotocin produces pancreatic swelling, ultimately leading to the degeneration of β-cells in the Langerhans islets inducing experimental diabetes (Akbarzadeh et al., 2007).
The third group was assigned as the diabetic C. gileadensis L. sap-treated group. After 5 days of streptozotocin administration (after diabetic induction), all mice in this group received 200 mg/kg C. gileadensis L. sap orally (Sulimani, 2015).
The fourth group was a diabetic group treated with C. gileadensis L. methanolic extract. Following the induction of diabetes mellitus, the mice in this particular group were treated with 200 mg/kg C. gileadensis L. methanol extract as the third group.
The fifth group was referred to as the C. gileadensis L. acetone-extract-treated group. After the induction of diabetes, all mice in this group were treated with 200 mg/kg C. gileadensis L. acetone extract orally.
Measurement of Body Weight
The body weight of each mouse that participated in this study was measured every week throughout the trial using a digital balance (OHAUS, Scout Pro SPU601, China).
Collection of Blood Specimens
Blood samples were taken from the retro-orbital venous plexus into plain tubes after 5 days of streptozotocin administration and throughout the trial for random blood-glucose estimation. Furthermore, at the end of the experiment (after 6 months), blood specimens were collected in plain tubes and centrifuged at 2,500 rpm for 15 min, and then sera were kept at −80°C until analysis.
Estimation of Random Blood-Glucose Levels and Glycated Hemoglobin (HbA1c)
Random blood glucose levels were estimated by colorimetric assay. Furthermore, HbA1c was measured for all mice included in this study at the end of the experiment (after 6 months) using a glycohemoglobin kit (POINTE Scientific Inc., United States) by cation exchange resin. Hemolyzed whole blood is combined with a mild binding cation-exchange resin for 5 min. HbA1c binds resin during this moment. After mixing, a filter separates glycohemoglobin-containing supernatant from resin. By measuring the glycohemoglobin fraction and total hemoglobin fraction absorbance at 415 nm (405–420 nm acceptable), the percent glycohemoglobin is calculated. Using the two absorbances, the glycohemoglobin percent is calculated (Emeribe et al., 2015).
Estimation of Renal and Liver Function Tests
The serum sodium (Na+), potassium (K+), chloride (Cl−), urea, creatinine, and ALT levels were estimated using kits purchased from the Beckman Coulter company and the Beckman Coulter AU 480 bio-machine.
Lipid Profiles Estimation
At the end of the experiment, the triglycerides, total cholesterol, and high-density lipoprotein (HDL) were estimated by colorimetric assay for each mouse included in the present study, while the low-density lipoprotein (LDL) was calculated.
Estimation of Cardiac Biomarkers
Serum total creatine kinase (CK), creatine kinase-myocardial band (CK-MB) isoform, and lactate dehydrogenase (LD) were estimated using kits purchased from the Beckman Coulter company.
Estimation of Adropin, NO, ET-1, and VEGF
Serum adropin, NO, ET-1, and VEGF were estimated using the quantitative competitive enzyme-linked immunosorbent assay (ELISA) kits from the Thermo Fisher company. The serum levels of these parameters were measured using the Varioskan™ LUX instrument (Thermo Fisher Scientific, United States).
Estimation of CD3+, CD4+, CD8+, and CD25+
Sample Preparation
The blood samples collected in ethylenediaminetetraacetic acid (EDTA) tubes were carefully placed on top of a Ficoll-Paque density gradient medium in a 15 mL conical tube. The samples were continuously centrifuged at a speed of 400 × g for 30 min at a low temperature of 4°C. The layer containing mononuclear cells was meticulously extracted and transferred to a fresh tube. The cells were rinsed twice with phosphate-buffered saline (PBS) and then suspended in a staining solution consisting of PBS with 2% fetal bovine serum and 0.1% sodium azide.
Flow Cytometry Staining and Analysis
The cell suspension was divided into 100 µL in four tubes and labeled with the fluorochrome-conjugated monoclonal antibodies (CD4+, CD8+, CD25+, and CD3+). The staining procedure was conducted for 30 min at a temperature of 4°C while ensuring the absence of light. Following the staining process, the cells were rinsed with a staining buffer and subsequently treated with a 1% paraformaldehyde solution for fixation. Subsequently, all samples were incubated for 30 min at a temperature of 4°C while being shielded from light. Following this, the samples were washed and subsequently resuspended in fluorescence-activated cell sorting (FACS) buffer. Then, the samples underwent analysis using the flow cytometer (Le Saout et al., 2008).
Statistical Analysis
Prism10 was used for statistical analysis. All data were presented as mean ± standard deviation (SD). The comparison for body weight, HbA1c, sodium, potassium, chloride, urea, creatinine, ALT, lipid profile, AST, LD, total CK, CK-MB, adropin, NO, endothelin-1, VEGF, total lymphocytes, and CD3+, CD4+, CD8+, CD4+ CD25+, and CD8+ CD25+ populations between all groups was used one-way analysis of variance (ANOVA) and the significance level was chosen at p < 0.05. The flow data were analyzed using the FlowJo software (Tree Star, Ashland, and Oregon) for flow cytometry.
Results
Body Weight
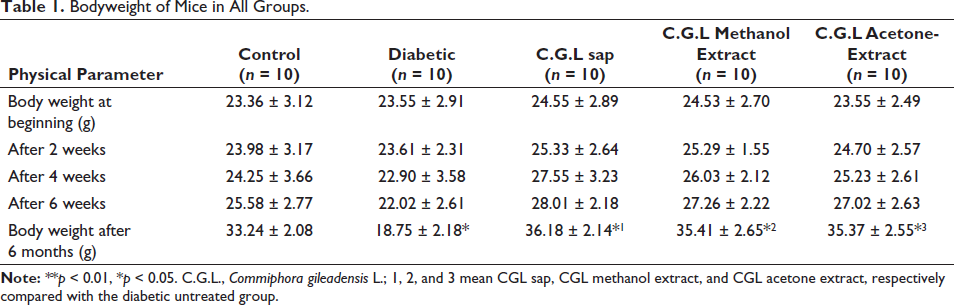
Table 1 displays the body weight of all groups of mice through a 6-month period. After 6 months, the diabetic mice treated with C. gileadensis L. sap (36.18 ± 2.14), methanol (35.41 ± 2.65), and acetone (35.37 ± 2.55) extracts exhibited significantly greater body weights in comparison to the diabetic mice that did not receive any treatment (18.75 ± 2.18, p < 0.05).
Bodyweight of Mice in All Groups.
HbA1c
Following the administration of streptozotocin for 5 days, it was observed that random blood glucose levels had increased in all groups. The HbA1c was significantly reduced in diabetic mice treated with C. gileadensis L. sap (6.11 ± 0.83), methanol (6.33 ± 1), and acetone (6.21 ± 1.1) extracts compared with the untreated diabetic group (13.1 ± 2.4, p < 0.01) (Table 2).
Glycated HbA1c of Mice in All Groups After 6 Months.

Renal and Liver Function Tests
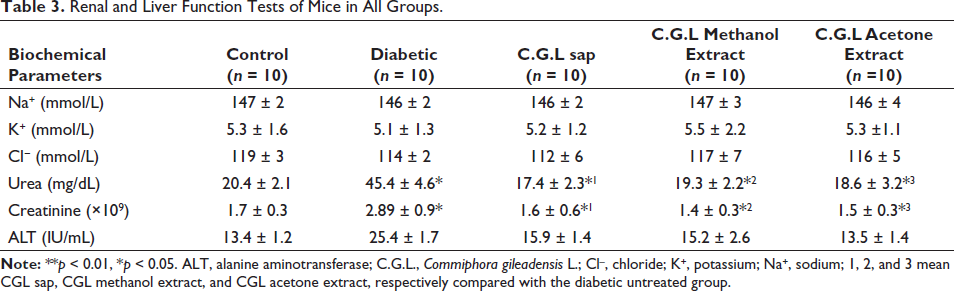
Renal and liver function tests are represented in Table 3. Serum urea and creatinine levels were significantly lower in diabetic mice treated with C. gileadensis L. sap (17.4 ± 2.3 and 1.6 ± 0.6, respectively), methanol (19.3 ± 2.2 and 1.4 ± 0.3, respectively), and acetone extracts (18.6 ± 3.2 and 1.5 ± 0.3, respectively) compared with untreated diabetic mice (45.4 ± 4.6 and 2.89 ± 0.9, respectively, p < 0.05).
Renal and Liver Function Tests of Mice in All Groups.
Lipid Profiles
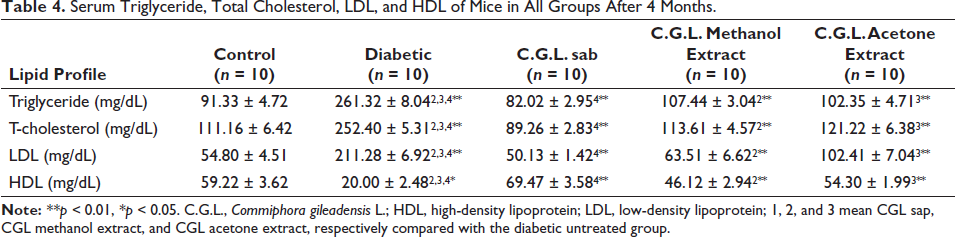
Lipid profiles for all groups are represented in Table 4. The lowest triglyceride levels were present in the diabetic group treated with C. gileadensis L. sap (82.02 ± 2.95). The triglyceride levels in the diabetic mice treated with C. gileadensis L. sap (82.02 ± 2.95), methanolic extract (107.44 ± 3.04), and acetone extract (102.35 ± 4.71) were significantly lower compared with the untreated diabetic group (261.32 ± 8.04) (p < 0.01). According to total cholesterol levels, the diabetic C. gileadensis L. sap-treated group had the lowest level among all groups (89.26 ± 2.83). Compared with the diabetic group, those diabetic mice treated with C. gileadensis L. sap (89.26 ± 2.83) and its methanolic (113.61 ± 4.57) and acetone (121.22 ± 6.38) extracts had significantly lower total cholesterol levels than those in the untreated diabetic groups (252.40 ± 5.31) (p < 0.01). LDL levels were significantly decreased in the diabetic C. gileadensis L. sap (50.13 ± 1.42), methanol extract (63.51 ± 6.62), and acetone (102.41 ± 7.04) extract-treated groups compared with the untreated diabetic group (211.28 ± 6.92) (p < 0.01). Regarding HDL, the diabetic C. gileadensis L. sap (69.47 ± 3.58), methanol extract (46.12 ± 2.94), and acetone extract-treated (54.30 ± 1.99) groups had significantly higher levels than the untreated diabetic group (20.00 ± 2.48) (p < 0.01).
Serum Triglyceride, Total Cholesterol, LDL, and HDL of Mice in All Groups After 4 Months.
Cardiovascular Integrity Markers
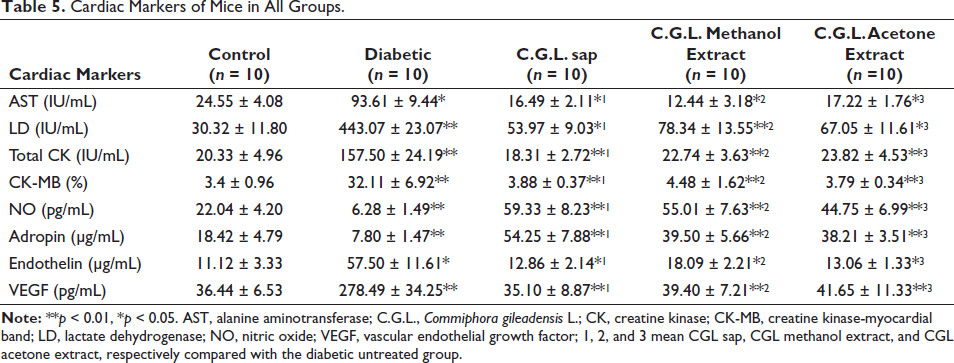
The results of cardiovascular integrity markers are displayed in Table 5. The AST activity was shown to be significantly reduced in diabetic mice that were treated with sap, methanol, and acetone extracts of C. gileadensis L. (16.49 ± 2.11, 12.44 ± 3.18, and 17.22 ± 1.76, respectively) compared to diabetic mice that did not receive any treatment (93.61 ± 9.44, p < 0.05). The LD activity was shown to be considerably decreased in diabetic mice that received treatment with sap, methanol, and acetone extracts of C. gileadensis L. (53.97 ± 9.03, 78.34 ± 13.55, and 67.05 ± 11.61, respectively) compared to diabetic mice that did not receive any treatment (443.07 ± 23.07, p < 0.01). Furthermore, the study observed a substantial decrease in total CK activity and CK-MB levels in diabetic mice that were treated with sap (18.31 ± 2.72 and 3.88 ± 0.37, respectively), methanol (22.74 ± 3.63 and 4.48 ± 1.62, respectively), and acetone (23.82 ± 4.53 and 3.79 ± 0.34, respectively) extracts when compared to the untreated group (157.50 ± 24.19 and 32.11 ± 6.92, respectively, p < 0.01). C. gileadensis L. sap (59.33 ± 8.23), methanol (55.01 ± 7.63), and acetone (44.75 ± 6.99) extracts significantly induced a positive cardiovascular integrity marker, NO, in diabetic mice compared with the diabetic untreated mice (7.80 ± 1.47, p < 0.01). The second cardiovascular integrity marker, adropin, was considerably elevated in diabetic mice that were treated with C. gileadensis L. sap, methanol, and acetone extracts. The mean adropin levels for the treated groups were 54.25 ± 7.88, 39.50 ± 5.66, and 38.21 ± 3.51, respectively, whereas the untreated group had a mean adropin level of 7.80 ± 1.47. This difference in adropin levels was found to be statistically significant (p < 0.01). Interestingly, the levels of the cardiovascular malfunction markers ET-1 and VEGF were significantly reduced in diabetic mice that received treatment with sap (12.86 ± 2.14 and 35.10 ± 8.87, respectively), methanol (18.09 ± 2.21 and 39.40 ± 7.21, respectively), and acetone (13.06 ± 1.33 and 41.65 ± 11.33, respectively) extracts compared to the group of diabetic mice that did not receive any treatment (57.50 ± 11.61, p < 0.05, and 278.49 ± 34.25, p < 0.01, respectively).
Cardiac Markers of Mice in All Groups.
Peripheral CD3+, CD4+, CD8+, and CD25+
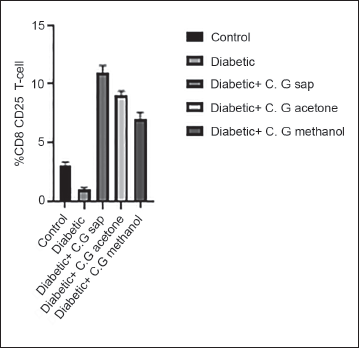
The data revealed a reduction in total lymphocytes in diabetes groups that were treated with C. gileadensis L. sap, methanol, and acetone extracts, as compared to the untreated diabetic group (p < 0.0001) (Figure 1). The diabetic mice treated with C. gileadensis L. sap, methanol, and acetone extracts showed a significant decline in CD4+ T cells compared with the untreated diabetic group (p < 0.0001) (Figure 2). The diabetes groups treated with C. gileadensis L. sap, methanol, and acetone extracts showed a significant decrease in CD8+ T lymphocytes compared to the untreated diabetic group (p < 0.0001) (Figure 3). The data demonstrated that CD3+ levels were dramatically diminished in diabetic mice treated with C. gileadensis L. sap, methanol, and acetone extracts compared to the untreated diabetic groups (p < 0.0001) (Figure 4). The CD4+ CD25+ cell population demonstrated a considerable increase in all diabetic groups administered C. gileadensis L. relative to the untreated diabetic groups (p < 0.0001) (Figure 5). The population of CD8+ CD25+ cells was shown to be higher in diabetic mice that received treatment with C. gileadensis L. sap, methanol, and acetone extracts as compared to the diabetic mice that did not get any treatment (p < 0.0001) (Figure 6).
Total Lymphocytes of Mice in All Groups.
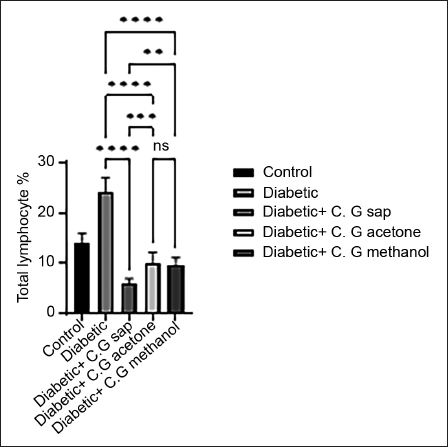
CD3+ Subset of Mice in All Groups.
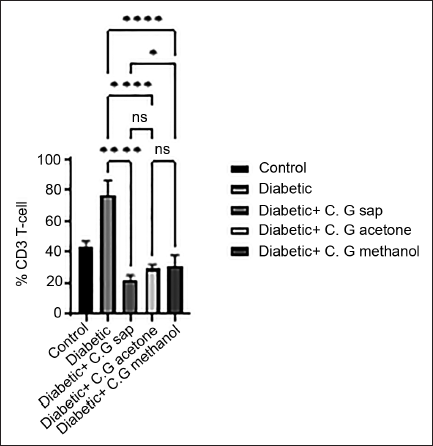
CD4+ Subset of Mice in All Groups.
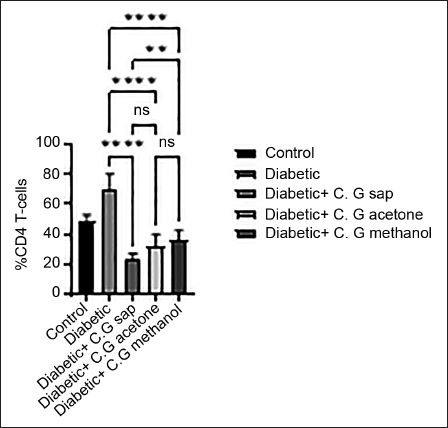
CD8+ Subset of Mice in All Groups.
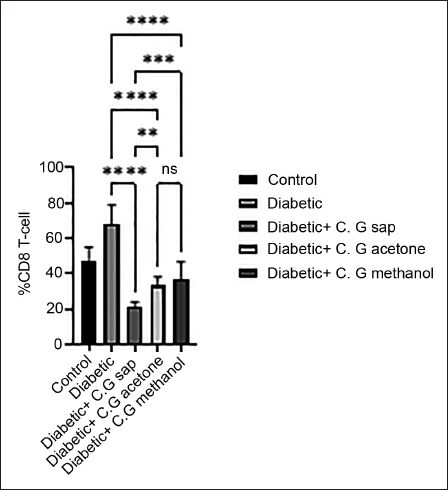
CD4+ CD25+ Subset of Mice in All Groups.
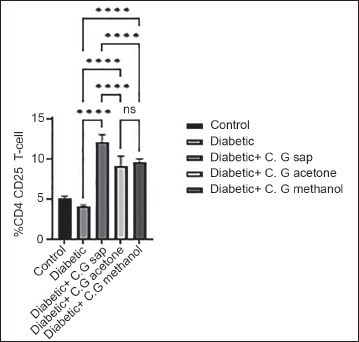
CD8+ CD25+ Subset of Mice in All Groups.
Discussion
In spite of contemporary medical interventions, cardiovascular disease persists as a prevalent chronic and primary contributor to both illness and death on a global scale. However, the prognosis of cardiovascular disease can be significantly enhanced by means of early therapy, which is facilitated by the precise and timely diagnosis of the condition. Therefore, it is essential to investigate new creative risk factors and diagnostic criteria to fulfill the prerequisites for early intervention (Fuster & Kovacic, 2014). In the conventional approach, a variety of proteins, including enzymes, have been employed for the purpose of diagnosing cardiovascular disease. These proteins’ levels increase after the occurrence of an acute myocardial infarction but do not increase prior to the event and return to their normal levels after a few days. Therefore, it is imperative to thoroughly investigate and identify novel biomarkers capable of detecting cardiovascular abnormalities in advance of their manifestation. Adropin is a newly discovered protein that has previously demonstrated its protective function on endothelial cells (Kuloglu & Aydin, 2014). A previous investigation revealed notable disparities in the plasma adropin concentration among individuals diagnosed with various cardiac diseases, including stable angina pectoris, acute myocardial infarction, cardiac Syndrome X, and atherosclerosis as compared to a group of healthy individuals. Furthermore, a negative correlation is shown between serum adropin levels and the angiographic severity of coronary atherosclerosis (Aydin et al., 2016). The measurement of serum adropin levels has emerged as a potential biomarker for the assessment and monitoring of patients with cardiovascular diseases (Sayın et al., 2014). NO is essential for controlling vascular tone and cardiac contractility. Moreover, it also contributes to the maintenance of blood-vessel homeostasis. The lack of NO has been identified as a predisposing factor for hypertension and atherosclerosis (Dahlöf, 2010). ET-1 is a powerful and enduring vasoconstrictor and appears to be elevated among individuals diagnosed with systemic arterial hypertension (Gupta et al., 2017). These biomarkers (adropin, NO, and ET-1) can be used as early diagnostic criteria for ischemic heart diseases. Previous studies found that the C. gileadensis L. reduces blood pressure in hypertensive patients, so the current study was done to evaluate the effect of C. gileadensis L. on cardiac biomarkers. The current study demonstrated a reduction in blood glucose levels and HbA1c levels in diabetic mice following treatment with extracts derived from C. gileadensis L. Moreover, after 6 months, diabetic mice that did not receive any treatment and had an unrestricted diet showed a decline in their renal function. The levels of blood urea and creatinine were found to be higher in this particular group of mice. Even though the diabetic mice treated with C. gileadensis L. extracts were also subjected to a free-diet regimen, it was observed that these groups had normal levels of urea and creatinine. A prior study demonstrated that an aqueous C. gileadensis L. extract normalized diabetic rats’ triglycerides, total cholesterol, and LDL. In the current study, C. gileadensis L. sap, methanolic extract, and acetone extract also reduce triglycerides, total cholesterol, and LDL while boosting HDL. The current study showed that the untreated diabetic group exhibits elevated levels of cardiac enzymes (AST, LD, total CK, and CK-MB). These enzymes may have arisen as consequences of prolonged elevation of blood glucose, total cholesterol, and LDL levels. Also, impaired renal function in this group may play a role. These enzymes appear within the normal range in diabetic mice treated with C. gileadensis L. sap, methanol, and acetone extracts. Moreover, untreated diabetic mice exhibited a reduction in adropin and NO (positive cardiovascular integrity markers) levels. These markers are induced in diabetic mice that had been administered C. gileadensis L. sap and methanol and acetone extracts. Furthermore, it was observed that untreated diabetic mice exhibited elevated levels of negative cardiovascular integrity markers ET-1 and VEGF. C. gileadensis L. seemed to decrease the concentrations of ET-1 and VEGF in diabetic groups. A previous study found that C. gileadensis L. extract had antioxidant activity due to its content of flavonoids, volatile oils, saponins, triterpenes, and sterols that work as free radical scavengers (Al-Seeni, 2017). These substances may give C. gileadensis L. an antioxidant activity that reduces oxidative stress by scavenging free radicals and maintains cardiovascular integrity. Immune reaction plays a critical role in atherosclerotic heart diseases. The blood CD4+, CD8+, and CD4+ CD25+ cells and CD4+/CD8+ ratio may be indicators of coronary artery disease and could be a therapeutic target for cardiovascular diseases (Rajib et al., 2019). Both CD4+ and CD8+ had previously shown a strong association with coronary heart disease (O’Sullivan et al., 2022). Activated CD4+ T cells in response to oxidized LDL-antigen build and propagate atheroma (Rajib et al., 2019). In patients with symptomatic atherosclerosis, the arterial intima and adventitia exhibited a greater abundance of CD8+ T lymphocytes compared to the arterial media. Furthermore, there was a direct correlation between the severity of atherosclerotic lesions and the elevated quantity of intimal CD8+ T cells (Allahverdian et al., 2014). The current study found that C. gileadensis L. reduces CD4+ and CD8+ in diabetic mice. This means that the C. gileadensis L. may prevent immunologic reactions by reducing both CD4+ and CD8+ populations. Moreover, C. gileadensis L. increases CD4+ CD25+ population, which is known as T regulatory cells. This cell preserves the cardiovascular system integrity by regulating the immune-inflammatory responses of target T cells and antigen-presenting cells (APCs). This regulation restricts the expansion of T cells and the generation of cytokines. Tregs exert control over immune-inflammatory responses by releasing inhibitory cytokines, depleting interleukin-2 (IL-2), or modulating receptor-ligand interactions, which can lead to apoptotic induction or functional changes in APCs. Totally, T regulatory cells play a significant role in protection against cardiovascular disease (CVD) (Xiao et al., 2016). The CD8+ CD25+ population is another T regulatory cell that immune balance and their dysfunction causing critical immunologic disorders (Redouane et al., 2019). In the current study, C. gileadensis L. increases the CD8+ CD25+ subset in diabetic mice.
Recommendation and Conclusion
The findings of this study suggest that C. gileadensis L. induces the levels of cardiovascular integrity markers (adropin and NO) and reduces cardiovascular malfunction markers (ET-1 and VEGF) in diabetic mice. The observed properties could potentially be attributed to the antioxidant activity of C. gileadensis L. Furthermore, C. gileadensis L. may help in the prevention of atheroma formation by reducing CD4+ and CD8+ and increasing T regulatory cells (CD4+ CD25+ and CD8+ CD25+). A further in vitro study is recommended to elucidate the mechanism by which C. gileadensis L. preserves cardiovascular homeostasis.
Study Limitation
The study lacks a computed tomography (CT) scan of the heart that determines the presence of atheroma in the coronary artery.
Footnotes
Abbreviations
APCs: Antigen-presenting cells; AST: Aspartate aminotransferase; C. gileadensis L.: Commiphora gileadensis L.; CK: Creatine kinase; CVD: Cardiovascular disease; ET1: Endothelin-1; HbA1c: Glycated hemoglobin; HDL: High-density lipoprotein; LD: Lactate dehydrogenase; LDL: Low-density lipoprotein; NO: Nitric oxide; NOS: Nitric oxide synthase; VEGF: Vascular endothelial growth factor.
Acknowledgments
The author extends their appreciation to Taif University, Saudi Arabia, for supporting this work through project number (TU-DSPP-2024-270).
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The animal study protocol was accredited by the National Committee for Bioethics at Taif University (protocol code HAO-02-T-105), and the Committee considered that the proposal fulfills the requirements.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Taif University, Saudi Arabia, Project No. TU-DSPP-2024-270.
