Abstract
Hemostatic disorders are the leading cause of morbidity and even mortality worldwide. Therefore, there is a growing tendency to develop novel, effective, and safer agents for the management of hemostatic diseases. Current hemostatic agents come with several drawbacks, such as unavoidable adverse effects, instability in oral administration, lack of biodegradability, poor safety profile in long-term uses, and high costs. Therefore, herbal remedies with better safety profiles have become a popular alternative therapeutic option for the prevention and treatment of coagulopathies. Aesculus hippocastanum is a commonly found decorative plant with significant medicinal importance. Its various organs contain valuable phytochemicals, and extensive research has demonstrated the effectiveness of A. hippocastanum-based treatments for venous disorders. This review explores the impact of A. hippocastanum on inflammation, oxidative stress, platelet aggregation, and the endothelium of blood vessels. Early studies on the pharmacological properties of A. hippocastanum indicate that it is an exceptional astringent and hemostatic medicinal plant. Its main bioactive compounds, including escin, esculetin, kaempferol, quercetin, and (‒)-epicatechin, not only exhibit vasoconstriction effects but also demonstrate strong anti-inflammatory, antiplatelet, and antioxidant properties. A. hippocastanum exerts these effects by modulating targets such as P-selectin, Nrf2/HO-1, NF-κB, 5HT2A, P2RY1, P2RX1, and cPLA2. Nevertheless, further research is necessary to thoroughly confirm A. hippocastanum’s effectiveness as a hemostatic agent.
Introduction
Uncontrolled hemorrhaging is recognized as the leading factor in both civilian and war-related fatalities prior to receiving medical treatment. Additionally, it is noted for impeding the process of wound healing. Severe trauma is responsible for around 5.8 million fatalities annually, with 40% of these deaths being caused by significant blood loss (Montazerian et al., 2022; Spahn et al., 2019). Hemostasis refers to the intricately controlled initial physiological response of the body to stop bleeding at a site of injury. The mechanism underlying hemostasis is regarded as a crucial stage, utilizing various blood constituents to promptly close the puncture or injury site (Sang et al., 2021). The initiation of blood coagulation begins with platelet adhesion at the injured area, leading to the formation of a temporary platelet plug to seal the breach temporarily. Subsequently, a sequence of biochemical reactions involving numerous clotting factors enables the transformation of fibrinogen into the insoluble fibrin clot. The concluding phase of hemostasis encompasses fibrinolysis and tissue restoration. In instances of significant trauma, the innate hemostatic process frequently falls short of achieving optimal hemorrhage management (Sharma et al., 2024). Consequently, there is a pressing need for the creation of versatile hemostatic materials. These materials should demonstrate efficacy in clot formation, possess a notable absorption capacity, exhibit antimicrobial characteristics, promote expedited healing, and effectively manage such exigent circumstances. In contemporary clinical practice, a variety of hemostatic materials are utilized; however, they often present limitations such as inadequate biocompatibility, toxicity, and poor biodegradability (Jiao et al., 2023).
The utilization of plants as therapeutic agents for a variety of illnesses has laid the groundwork for modern medical sciences. As per the World Health Organization, roughly 80% of the populations in Asia and Africa rely on traditional medicine as a primary healthcare approach for managing diseases, including those related to blood disorders (Cordier & Steenkamp, 2012). Aesculus hippocastanum, generally referred to as horse chestnut, is a relict species from the Paleogene period and is primarily found in small, limited regions of the Balkan Peninsula. It falls under the Aesculus genus’s section Aesculus and stands as the sole indicative of this genus in Europe. Since its introduction as an ornamental tree in Europe in the late 16th century, it has become a ubiquitous presence in parks, gardens, and urban avenues due to the aesthetic appeal of its leaves and flowers. Beyond its ornamental value, horse chestnut is also esteemed as a valuable medicinal plant, with various parts used in both traditional and modern medical practices (Owczarek-Januszkiewicz et al., 2023).
In vitro research has demonstrated that A. hippocastanum extracts (HSCEs) exhibit anti-inflammatory, antioxidant, and vasoprotective effects. These properties have been effective in lowering vascular permeability and inflammation, suggesting its potential as a treatment for conditions like chronic venous insufficiency (CVI). In vivo studies in animal models have further validated the efficacy of A. hippocastanum, showing improvements in blood circulation and reductions in swelling. However, some studies have reported possible adverse effects, including gastrointestinal discomfort and hepatotoxicity, in certain instances (Idris et al., 2020; Owczarek et al., 2021). Given what has been said, the present study aims to review potential hemostatic effects of A. hippocastanum.
Physiology of Hemostasis
Hemostasis occurs through the equilibrium of procoagulant and anticoagulant factors. After vascular injury, various components within the vascular system including cellular elements and noncellular constituents collaborate closely to halt bleeding and subsequently dissolve the formed clot from circulation. These hemostatic elements also play crucial roles in processes such as wound healing and angiogenesis. The response of the hemostatic system to vascular damage is rapid and finely regulated. Hemostasis, the process of stopping bleeding, requires a sequence of coordinated actions such as vasoconstriction, the development of a platelet plug, and blood coagulation (Weitz & Fredenburgh, 2013).
Vascular injury triggers vasoconstriction as the initial response of the vascular system. This constriction arises from both direct damage to smooth muscle cells and the release of endothelin-1 from endothelial cells (ECs) and platelets. Consequently, blood flow diminishes in the injured area, effectively reducing blood loss. Subsequently, platelets adhere to the exposed subendothelium at the site of injury, a process termed adhesion (Khazaei et al., 2008). During adhesion, platelets undergo shape changes and release granules, initiating their activation. This activation prompts further platelet recruitment and initiates platelet aggregation, culminating in the formation of a platelet plug. Notably, vascular injury exposes subendothelial elements such as collagen I and III, laminin, and microfibrils to the circulating blood, facilitating platelet adhesion, activation, and secretion. Among these elements, collagen plays a pivotal role in platelet adhesion, representing the primary subendothelial component responsible for initiating the hemostatic response. Indeed, platelets adhere to the exposed subendothelial matrix of the damaged vessel wall primarily through interactions involving von Willebrand factor (vWF) and collagen. Upon activation, platelets recruit and aggregate additional platelets, leading to the formation of a provisional platelet plug at the site of injury. This process is crucial in initiating primary hemostasis and halting bleeding following vascular injury (Scridon, 2022).
After platelet plug formation, the coagulation cascade is initiated, leading to the activation of inactive proenzymes and clotting factors into active enzymes. This cascade, comprising a series of proteolytic reactions, ultimately generates thrombin, a pivotal enzyme in hemostasis. Thrombin plays a central role by converting soluble fibrinogen into insoluble fibrin strands. These fibrin strands intertwine with the platelet plug, strengthening it and forming a stable blood clot. As the clot matures, platelets undergo contraction, drawing the fibrin strands closer together in a process known as clot retraction. This retraction serves to consolidate the clot and aids in wound stabilization, promoting the initial phases of tissue repair. Subsequently, as tissue healing progresses, the clot must be dissolved to restore normal blood flow. Fibrinolysis, the process responsible for clot dissolution, involves the activation of plasminogen to plasmin. Plasmin, in turn, cleaves fibrin strands, leading to the breakdown of the clot into soluble fibrin degradation products. This controlled fibrinolysis is vital in resolving the clot and restoring vascular patency, thereby concluding the hemostatic response (Chapin & Hajjar, 2015; Smith et al., 2015).
A. hippocastanum Composition
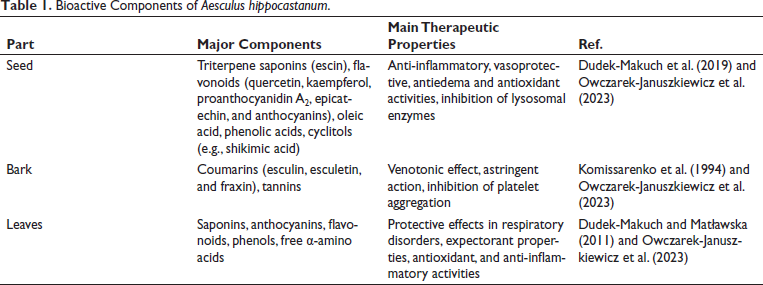
The active components derived from different parts of the A. hippocastanum offer a spectrum of pharmacological properties ranging from anti-inflammatory and vasoprotective actions to astringency and respiratory support. Understanding these bioactive constituents and their properties provides insights into the traditional and potential modern therapeutic applications of A. hippocastanum-derived preparations (Idris et al., 2020).
As listed in Table 1, the seeds of A. hippocastanum contain several active compounds, with escin being the most prominent. Escin exhibits anti-inflammatory properties and is recognized for its vasoprotective effects. Some studies have also evaluated escin potential as a cytotoxic agent, and it exhibited cytotoxic effects on certain cancer cell lines. However, its impact on normal cells is more nuanced. For instance, Celep et al. (2012) found that the bark extract of A. hippocastanum besides the high antioxidant capacity could cause cytotoxicity in both cancer and normal cells when used in high concentrations. On the other hand, Dudek-Makuch et al. (2019) have shown that treating ECs with A. hippocastanum does not affect viability of these cells. Escin is also widely utilized in the management of CVI due to its ability to reduce vascular permeability and edema by stabilizing capillary walls (Gallelli, 2019; Idris et al., 2020). Additionally, horse chestnut seeds contain flavonoids such as quercetin and kaempferol, which contribute antioxidant and anti-inflammatory actions, further enhancing the therapeutic potential of A. hippocastanum seed extracts (Oszmiański et al., 2014). The bark of the A. hippocastanum is a source of Coumarins, mainly esculin and fraxin, and tannins, compounds known for their astringent properties (Komissarenko et al., 1994). Tannins exert their effects by binding to and precipitating proteins, leading to tissue tightening and toning (Morimoto et al., 1987). This astringent action finds utility in traditional medicine for addressing conditions such as diarrhea, where tannins can help reduce intestinal inflammation and fluid loss. A. hippocastanum leaves harbor saponins, which are glycosides with diverse pharmacological activities. These compounds possess expectorant properties, aiding in the clearance of respiratory secretions and suppression of cough reflexes. In addition to saponins, A. hippocastanum leaves also contain flavonoids, contributing to their antioxidant and anti-inflammatory characteristics. These bioactive constituents collectively support the therapeutic potential of A. hippocastanum leaf extracts in respiratory ailments such as bronchitis and asthma (Idris et al., 2020; Owczarek-Januszkiewicz et al., 2023).
Bioactive Components of Aesculus hippocastanum.
Effects of A. hippocastanum on Vascular Endothelium
The vascular endothelium, the most inner and greatly heterogeneous monolayer cells lining the vasculature system, serves a variety of pivotal passive and active roles in hemostasis, including physical barrier function and contribution to the secretory, immunologic, and metabolic processes of blood vessels (Rajendran et al., 2013). Numerous pathological circumstances, such as acute or chronic inflammatory disorders, tend to result in a deleterious inflammatory cascade that may transform the vascular endothelium structure and lead to deregulated coagulation, impaired vascular tonicity, increased endothelial permeability, and increased inflammatory cell infiltration into the affected site (Marcu et al., 2018; Norooznezhad & Mansouri, 2021). Considering its crucial function in the inflammatory reaction, the secretion of proinflammatory mediators, and the well-organized operation of the clotting process and recovery of injured organs, the endothelium is a pivotal and versatile element in different grades of inflammatory disorders and coagulopathies and, accordingly, a valuable target for therapeutic modifications (Pablo-Moreno et al., 2022).
Horse chestnut seed extract contains bioactive flavonoids and saponins, which have the capability of reducing the permeability of blood vessels, reducing the activities of hyaluronidase, elastase, and lysosomal enzymes, decreasing the exudation of fluids from blood vessels into peripheral tissues, and preventing the breakdown of mucopolysaccharides in capillary walls through its stimulatory effect on prostaglandin F2 alpha (PGF2α) production. In addition, escin impedes disarrangement of the regular distribution and expression of the platelet endothelial cell adhesion molecule-1 (PECAM-1) and vascular cell adhesion molecule 1 (VCAM-1) adhesion molecules in hypoxic conditions, which may aid in elucidating its protective function concerning vascular permeability (Crinnion, 2009; Gallelli, 2019). From a pathophysiological and biochemical point of view, proinflammatory mediators, including reactive oxygen species (ROS), tumor necrosis factor-alpha (TNF-α), monocyte chemoattractant protein-1 (MCP-1), interferon-gamma (IFN-γ), interleukins (ILs), matrix metalloproteinases (MMPs), phospholipase A2 (PLA-2), cyclooxygenase (COX), lipoxygenase (LOX), and leukotrienes, are liberated from the vascular ECs during traumatic injuries, resulting in the accumulation of immune cells and platelets at the inflamed site. Then, the vascular endothelium expresses certain extracellular matrixes (ECMs) and cell adhesion molecules (CAMs), such as P-selectin, E-selectin, and vWf, which in turn form bonds with platelets and neutrophils, and instigate an inflammatory reaction, alter the flow properties of blood, and ultimately result in thrombosis (Gwozdzinski et al., 2024; Puhr-Westerheide et al., 2019). It has been found that β-escin, an active isomer of escin, induces biosynthesis of cholesterol in vascular endothelium, which is promptly followed by a noticeable reduction in the integrity of actin filaments. These biochemical and morphological alterations in cells lead to notably reduced reactions to TNF-α stimulation. This includes diminished migration, attenuated vascular endothelium permeability, and inhibited nuclear factor kappa B (NF-κB)-dependent signal transduction leading to the downexpression of effector proteins induced by TNF-α (Domanski et al., 2016). As reported in previous investigations, escin inhibits two key steps: The hypoxia-induced stimulation of ECs and subsequently the secretion of CAMs and inflammatory mediators. These two main steps include a reduction in ATP concentration (Rajendran et al., 2013), which is the initiation phase of the coagulation cascade, and a decrease in the enzymatic activity of PLA-2 (Arnould et al., 1996; Dudek-Makuch & Studzińska-Sroka, 2015; Norooznezhad & Mansouri, 2021). In other studies, it has been shown that β-escin reduces vascular EC response to histamine and also alleviates capillary wall hyperpermeability caused by serotonin (Rasheed et al., 2021). In an in vitro study conducted on cytokine mixture-induced human umbilical vein endothelial cells (HUVEC) cells, it has been reported that quercetin and kaempferol, two main flavonoid compounds found in A. hippocastanum, significantly reduce the levels of E-selectin, VCAM-1, intercellular adhesion molecule 1 (ICAM-1), cyclooxygenase-2 (COX-2), and inducible NO synthase (iNOS) and block activator protein 1 (AP-1) as well as NF-κB signaling molecules in a dose-dependent manner. In this study, it was found that the inhibitory effect of kaempferol on the CAMs downregulation is significantly greater in kaempferol-exposed cells than in cells treated with quercetin, and on the other hand, the inhibitory effect on COX-2, iNOS, AP-1, and NF-κB was higher in quercetin-exposed cells as compared to kaempferol-treated cells (Crespo et al., 2008). Ding et al. reported that escin amends the elevated levels of rho-associated kinase (ROCK), RhoA, nuclear NF-κB, and the downregulated IκBα, cytoplasmic NF-κB, tight junction proteins (claudin-5 and occludin) in the intracerebral hemorrhagic mice model. These effects resulted in an improvement in blood–brain barrier (BBB) integrity and, as a consequence, an improvement in neurological function (Ding et al., 2021). In another study, it was found that escin reverses recombinant tissue plasminogen activator (rt-PA)-induced hemorrhagic transformation in ischemic stroke via protecting BBB and cerebrovascular ECs through the regulation of AMPK/Cav-1/MMP-9 signaling pathway (Sun et al., 2023). Taken together, these mentioned mechanisms make A. hippocastanum a valuable natural agent for targeting endothelial-related vascular disorders as well as coagulopathies.
Effects of A. hippocastanum on Platelet
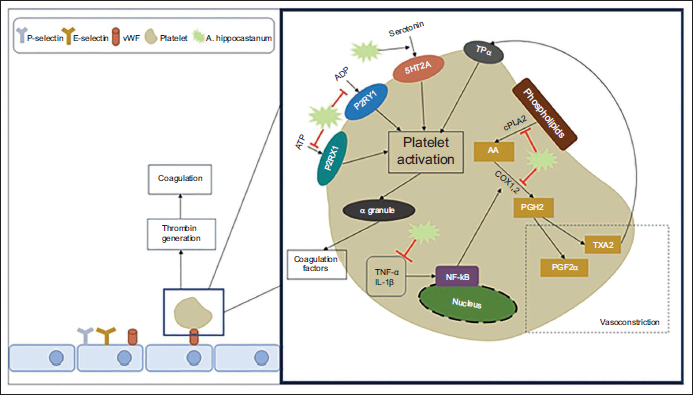
Despite platelet’s critical role in normal hemostasis, its hyperactivity often correlates with low- to high-grade vascular dysfunction and, in some cases, life-threatening complications (Yeung et al., 2018). Conventional pharmacological targets for hindering platelet stimulation have mainly been limited to integrin αIIbβ3, COX-1, and certain purinoceptors. Adenosine diphosphate (ADP) instigates platelets through three subtypes of purinergic receptors: Gi-coupled P2Y12 receptor, the Gq-coupled P2Y1 receptor, and the ligand-gated P2X1 cation channel (Marcu et al., 2018; Norooznezhad & Mansouri, 2021; Rajendran et al., 2013). ADP purinoceptors located on platelets are therefore promising therapeutic targets for antiplatelet agents (Yeung et al., 2018). Platelets contain a wide range of granules enriched with a myriad of mediators and adhesion molecules (e.g., P selectin, vWF, fibrinogen, TGF-β, CXCL chemokines, TLRs, MCP3, bivalent cations, serotonin, histamine, and so on), which contribute to interaction between leukocytes, platelets, circulating proteins in plasma, and the vascular endothelium, and as a consequence, promote blood clot formation (Jenne et al., 2013). Recently determined signaling molecules modulating platelet function have made it conceivable to provide novel attitudes for therapeutic mediation in the blood to prevent platelet hyperactivity.
In this regard, Felixsson et al. (2010) explored the biological effects of HSCE in an in vitro study. It has been found that its mode of action is through the ADP receptors. They showed that the horse chestnut tree-derived extract reversed platelet aggregation induced by ADP in human platelet-rich plasma (PRP). Furthermore, when an antagonist of the 5HT2A receptor, ketanserin, was combined with HSCE-exposed platelets, a reduction of platelets was observed. Accordingly, they proposed that inhibition of platelet aggregation by HSCE appears to be the sum of both stimulatory, induced by the 5HT2A receptor, and inhibitory, mediated by HSCE-derived flavonoids, effects. It can be concluded from this investigation that HSCE exhibits an inhibitory effect on human platelet aggregation induced by ADP (Felixsson et al., 2010). However, this anticoagulation effect was also requested to be investigated, along with potential interactions with other agents like other commonly used antiplatelet medicines in vascular disorders. So, the clinical application of HSCE as an antiplatelet agent ADP receptor antagonist remains to be more and more studied (Felixsson et al., 2010).
Esculetin, a coumarin compound found in HSCE, conveys thrilling pharmacological effects in several in vitro and in vivo models. For example, in an investigation, the effect of esculetin was studied in human platelet activation and animal models. In this study, esculetin impeded platelet aggregation stimulated by arachidonic acid and collagen in washed human platelets. The modulatory effects of esculetin on the platelet function in U46619, an analog of thromboxane A2-induced human platelets, were investigated. It has been reported that esculetin inhibits multiple cellular events, including release of ATP, formation of OH• radical, P-selectin expression, Akt phosphorylation, and PLC-γ2/PKC, but it did not reduce mitogen-activated protein kinase (MAPK) expression in human platelets stimulated by collagen. Analysis of platelet function revealed that esculetin considerably extended the blood closure time. In animal models, esculetin significantly increased the duration of blood clot formation and decreased acute pulmonary thromboembolism (PTE)-associated mortalities. Hence, esculetin may be employed as a useful therapeutic agent for managing thromboembolic complications (Park & Lee, 2021).
In another investigation, Hsia et al. (2019) indicated that esculetin shows antiplatelet properties by inhibiting the activation of PLC-γ2/PKC and Akt cascades and hindering OH• radical formation. Furthermore, they investigated the participation of cAMP, cGMP, integrin αIIbβ3, vasodilator-stimulated phosphoprotein (VASP), and NF-κB, since cAMP and cGMP nucleotides decrease VASP phosphorylation and instigate NF-κB, followed by triggering αIIbβ3 that considerably contributes to inhibitory pathways of platelet. They found that esculetin did not remarkably affect platelet aggregation induced by fibrinogen in elastase-exposed platelets; nonetheless, it significantly obstructed the activation of integrin αIIbβ3 by interfering with the binding of FITC-labeled procaspase-activating compound 1 (PAC-1). In addition, neither ODQ, a guanylate cyclase inhibitor, nor SQ22536, an adenylate cyclase inhibitor, markedly blocked esculetin-associated antiplatelet activity stimulated by collagen. Furthermore, esculetin outstandingly diminished the phosphorylation of p65 and IκBα and also prevented IκBα breakdown in platelets induced by collagen. These findings highlight that the NF-κB-related inhibitory effect of esculetin on αIIbβ3 may offer a new feedback mechanism to modulate the physiological functioning of platelets (Hsia et al., 2019).
It has been clarified that esculetin downregulates the predominant protein kinases that are responsible for platelet activation and its cellular processes, including PI3K/Akt and MAPK [p38 MAPK, extracellular signal-regulated kinase (ERK), and c-Jun N-terminal kinase (c-Jun JNK)]. In addition, esculetin decreases TXA2 synthesis, PLA2 activation, and release of cytosolic granules (e.g., ATP, serotonin, P selectin, histamine, and so on), ultimately leading to the prevention of overall platelet aggregation (Lee, 2021).
In a study, Choi et al. (2015) showed that kaempferol, a flavonoid compound found in HSCE, markedly reversed the activity of activated factor X (FXa) and thrombin. Kaempferol also prevented fibrin polymerization in turbidity. Kaempferol completely reduced phosphorylation of ERK 1/2, p38 MAPK, c-Jun JNK 1/2, and PI3K/PKB (AKT) in thrombin-activated platelets and prolonged clotting time in an epinephrine/collagen-induced platelet activation assay (Choi et al., 2015; Lee, 2021).
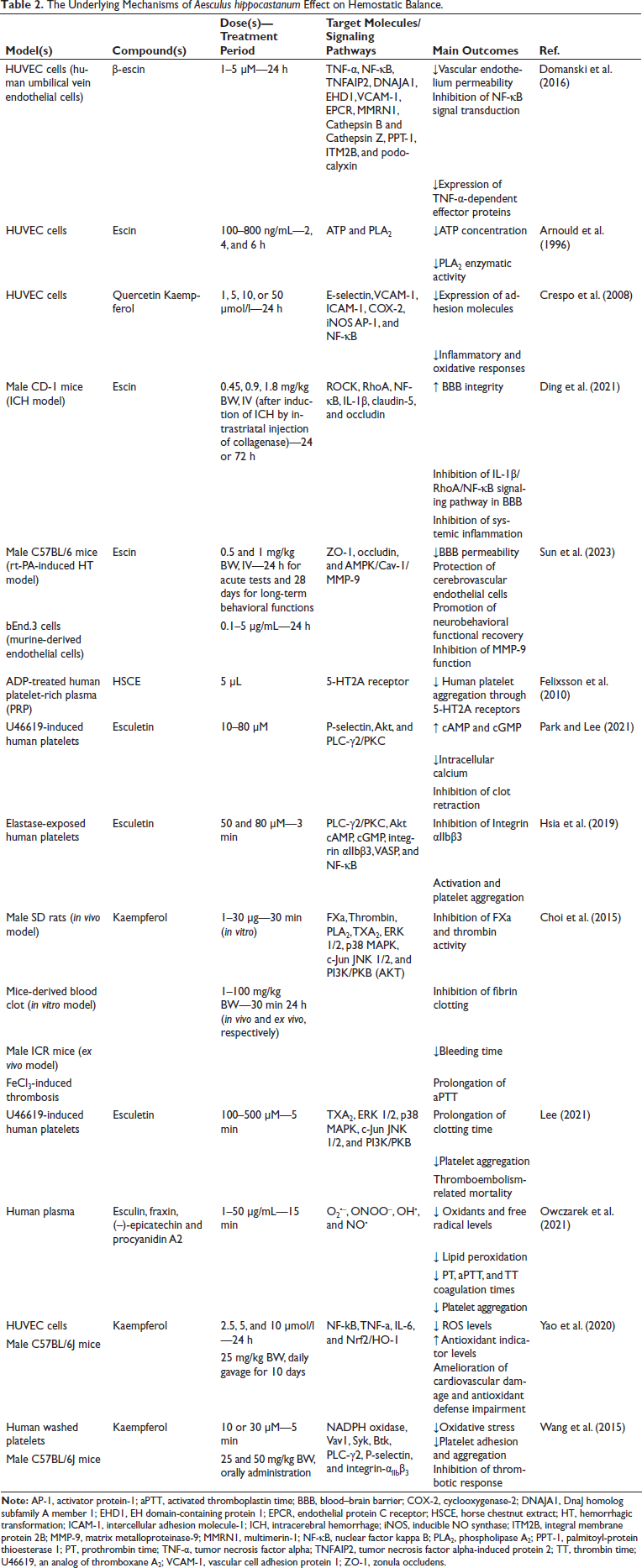
Taken together, horse chestnut has exhibited effective antiplatelet properties, making it a potentially valuable pharmacological candidate to combat platelet-associated thrombosis and cardiovascular disorders due to its bioactive ingredients. The underlying mechanisms have been outlined in Table 2 and Figure 1.
The Underlying Mechanisms of Aesculus hippocastanum Effect on Hemostatic Balance.
Antioxidant Effects of A. hippocastanum
Understanding the interconnected mechanisms of vascular endothelial dysfunction, oxidative stress, and inflammation is crucial in comprehending the pathophysiology of hemostasis disorders. Vascular endothelial dysfunction initiates a cascade involving oxidative stress and inflammation within vessel walls, creating a detrimental cycle that exacerbates hemostasis dysfunction. Additionally, oxidative stress and hemostasis disorders, such as heightened platelet reactivity and activation of the coagulation cascade, often accompany inflammation. These factors are linked to venous insufficiency and may contribute to disease etiology, progression, and symptom aggravation (Higashi, 2022; Nowak et al., 2010).
ROS have a dual role in vascular signaling, where their balanced levels are crucial for cellular functions, but excess ROS can harm vascular health. For instance, the superoxide anion (O2•–) is necessary for cell growth, yet its overproduction can react with nitric oxide (NO•), leading to vasoconstriction and endothelial dysfunction. This reaction also forms peroxynitrite (ONOO−), a highly destructive species (Buetler et al., 2004). Similarly, physiological levels of hydrogen peroxide (H2O2) regulate EC functions like vasorelaxation and proliferation, but elevated levels may hinder proliferation or trigger vessel muscle hypertrophy (Bretón-Romero & Lamas, 2014). In conditions such as CVI, inflammatory processes can elevate oxidative stress, exacerbating ROS generation in affected veins and potentially worsening the disease progression. Understanding these intricate relationships between ROS and vascular health is crucial for developing targeted interventions to mitigate oxidative stress-related vascular damage (Owczarek-Januszkiewicz et al., 2023).
Esculin has been demonstrated in vitro to possess the ability to directly scavenge ROS, helping to reduce oxidative stress. Additionally, in animal models, esculin has been found to enhance antioxidant enzyme activity like superoxide dismutase (SOD) and glutathione peroxidase (GSH-PX), further contributing to its antioxidant effects. Esculin anti-inflammatory properties primarily manifest through its ability to inhibit the NF-κB signaling pathway, a key regulator of inflammation, and to reduce the expression of proinflammatory cytokines such as IL-6 and TNF-α (Li et al., 2022).
Excessive ROS can lead to oxidative and nitrative harm to large molecules, particularly proteins within the coagulation cascade. Recent research findings emphasize, for instance, the significance of nitrated fibrinogen in the possible onset of coagulation abnormalities (Martinez et al., 2012). HSCE has demonstrated the ability to shield fibrinogen from nitrative damage caused by peroxynitrite (ONOO–), potentially mitigating abnormal clotting tendencies to some extent. Flavan-3-ols derivatives were found to be more effective than coumarins in this regard, aligning with their superior scavenging capabilities. Notably, (–)-epicatechin exhibited high activity, consistent with previous findings for this compound. It was observed that (–)-epicatechin antinitration properties can reduce the prothrombotic impact on fibrinogen clotting behavior induced by nitration, highlighting its potential therapeutic role in preventing clotting disorders (Owczarek et al., 2021).
In a study by Yao et al. (2020), kaempferol protected vascular endothelium against oxidative stress. These results indicated that the observed protective effect of kaempferol was mediated by the Nrf2/HO-1 signaling pathway. In addition, kaempferol diminished IL-6 and TNF-α levels and also repressed the activation of NF-kB and its downstream inflammatory mediators in mice aortic tissues and HUVEC cells. Further analysis indicated that kaempferol decreased H2O2– and paraquat-mediated inflammation and oxidative stress in in vitro and in vivo, respectively (Yao et al., 2020).
In another study, the potential effects of kaempferol on the activation of NADPH oxidase (NOX), which has a crucial part in platelet activation and aggregation, were explored. Collagen-induced O2•– production was remarkably suppressed by kaempferol in a dose-dependent mode. Furthermore, kaempferol effectively interacted with the primary component of NOX, p47phox, and significantly hindered the collagen-induced phosphorylation of p47phox, hence preventing the activation of NOX.
Following the suppression of NOX, ROS-dependent inactivation of SHP-2 was effectively preserved by kaempferol. Eventually, the tyrosine kinase-mediated phosphorylation process of key elements (Vav1, Syk, Btk, and PLC-γ2) for the signaling pathways of collagen receptors was reduced by kaempferol. Kaempferol also suppressed the subsequent events, such as increased cytoplasmic calcium, P-selectin overexpression, and activation of integrin-αIIbβ3. Collectively, kaempferol reversed adhesion and aggregation in collagen-exposed platelets in vitro and also impeded the in vivo thrombotic responses (Wang et al., 2015).
Conclusion
Herbal remedies are often seen as safer and more effective than synthetic alternatives. Plants are increasingly valued as sources of natural remedies that can prevent and treat diseases by boosting antioxidant activity, inhibiting bacterial growth, or influencing genetic pathways. Even today, medicinal herbs remain crucial as a source of biomolecules for developing new medicines. In this study, the effects of A. hippocastanum on vascular endothelium, platelets, and antioxidant defense in the context of hemostasis were reviewed. According to the literature, it has been verified that the pharmacological effects of A. hippocastanum are attributed to its active compounds. Esin, esculetin, flavonoids, coumarins, and tannins are the major components of A. hippocastanum useful for hemostatic effects. These active compounds can influence the factors involved in platelet activation, such as 5HT2A, P2RY1, purinergic receptor P2X1 (P2RX1), cPLA2, and the NF-kB pathway. Additionally, HSCE affects the vascular endothelium and its permeability through targets like P-selectin, E-selectin, VCAM-1, hyaluronidase, and PECAM-1. Furthermore, it can prevent damage to the vascular walls caused by oxidative stress both directly by targeting produced free radicals and indirectly by increasing the activity of antioxidant enzymes and by affecting pathways like Nrf2/HO-1 and NF-kB. Based on the mentioned information, this plant is a good candidate for designing mechanistic studies aimed at achieving effective agents for hemostasis disorders.
Footnotes
Abbreviations
5HT2A: 5-Hydroxytryptamine 2A receptor; ADP: Adenosine diphosphate; aPTT: Activated partial thromboplastin time; BBB: Blood–brain barrier; BW: Body weight; CAM: Cell adhesion molecule; COX: Cyclooxygenase; cPLA2: Cytosolic phospholipase A2; ECM: Extracellular matrix; ERK: Extracellular signal-regulated kinase; FXa: Activated factor X; GSH-PX: Glutathione peroxidase; HUVEC: Human umbilical vein endothelial cells; ICAM-1: Intercellular adhesion molecule-1; ICR: Institute of Cancer Research; IFN-γ: Interferon-gamma; IL: Interleukin; IV: Intravenous; JNK: c-Jun N-terminal kinase; LOX: Lipoxygenase; MAPK: Mitogen-activated protein kinase; MCP-1: Monocyte chemoattractant protein-1; MMP: Matrix metalloproteinase; NF-κB: Nuclear factor kappa B; ONOO-: Peroxynitrite; P2RX1: Purinergic receptor P2X1; P2RY1: Purinergic receptor P2Y1; PAC-1: Procaspase-activating compound 1; PECAM-1: Platelet endothelial cell adhesion molecule-1; PI3K: Phosphoinositide 3-kinase; PKB: Protein kinase B; PLA-2: Phospholipase A2; PRP: Platelet-rich plasma; PT: Prothrombin time; PTE: Pulmonary thromboembolism; ROCK: Rho-associated protein kinase; ROS: Reactive oxygen species; SD: Sprague–Dawley; SOD: Superoxide dismutase; TNF-α: Tumor necrosis factor-alpha; TT: Thrombin time; VASP: Vasodilator-stimulated phosphoprotein; VCAM-1: Vascular cell adhesion molecule-1; vWF: von Willebrand factor.
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest regarding the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
