Abstract
Background
Metyrapone inhibits 11 β-hydroxylase, contributing to adrenal insufficiency (AI). This study aims to explore the efficacy of Tribulus terrestris fruit extract (TTFE) and an isolated compound (hecogenin) on metyrapone-induced AI.
Objectives
Adrenal glands manage processes such as metabolism, blood pressure, and stress response by generating hormones from their two main components, the cortex and medulla. The adrenal cortex generates glucocorticoids (GC) and mineralocorticoids, while the medulla produces adrenaline and noradrenaline. Reduced production of GC, mineralocorticoids, and adrenal androgens as a result of the adrenal gland’s destruction or lack of stimulation is the hallmark of AI. Primary, secondary, and tertiary forms of AI are prevalent conditions with various etiologies. If left untreated, they lead to an adrenal crisis. Synthetic GC (prednisolone and hydrocortisone) and mineralocorticoids (fludrocortisone) are the only drugs that help treat AI.
Materials and Methods
The Wistar rats were selected for the study and treated with T. terrestris extract and an isolated compound (hecogenin) in the presence of metyrapone. Behavioral and hormonal assessments were performed. Corticosterone, aldosterone, sodium, and potassium were measured.
Results
The findings showed that exposure to metyrapone showed a drastic reduction of cortisol, aldosterone, and locomotor scores. Additionally, metyrapone decreased locomotor score and serum cortisol. However, treatment with Tribulus extract and hecogenin showed a significant rise in the levels of aldosterone, corticosterone, sodium, and potassium.
Conclusion
400 and 800 mg/kg of TTFE and hecogenin 50 mg/kg showed significant increases in the levels of corticosterone, aldosterone, sodium, and potassium levels in Wistar rats.
Introduction
Adrenal glands manage processes such as metabolism, blood pressure, and stress response by generating hormones from their two main components, the cortex and medulla. The adrenal cortex generates glucocorticoids (GC) (cortisol and corticosterone), mineralocorticoids (aldosterone), and adrenal androgens (dehydroepiandrosterone and androstenedione), while the medulla produces adrenaline and noradrenaline, also known as catecholamines (Turcu et al., 2014; Wolff et al., 2023). Reduced production of GC, mineralocorticoids, and adrenal androgens as a result of the adrenal gland’s destruction or lack of stimulation is the hallmark of adrenal insufficiency (AI) (Kumar & Wassif, 2022). Primary, secondary, and tertiary forms of AI are prevalent conditions with various underlying causes (Husebye et al., 2021). Primary AI is an uncommon and potentially lethal condition that can result from a variety of factors. Hemorrhage, infection, autoimmune Addison’s disease, infiltrative illnesses or metastases, and bleeding are the most common causes (Buonocore & Achermann, 2020). The most prevalent subtype of AI is secondary AI, which is seldom linked to specific germ-line mutations and is brought on by several drugs and pituitary damage (Gruber & Bancos, 2022). The intrinsic hypothalamic–pituitary–adrenal (HPA) axis may be suppressed by prolonged exposure to GC therapy at doses above physiological levels or roughly equal to a daily dose of 5 mg of prednisolone. This condition is known as tertiary AI, and it might manifest as an adrenal crisis that could be lethal after withdrawal from long-term exogenous GC ingestion (Sagar et al., 2021). Although the introduction of synthetic GCs (hydrocortisone, prednisolone, and dexamethasone) and mineralocorticoids (fludrocortisone) have contributed to managing AI, there is a need for new and safer drugs. There is ample evidence of the beneficial effects of various categories of secondary metabolites, especially saponins, and most of these have served as the basis for the development of novel medications (Santos Cerqueira et al., 2012). Tribulus terrestris, an herbaceous plant indigenous to India, falls under the Zygophyllaceae family, encompassing about 20 species worldwide. It is widely recognized as “Tribulus” and is also known by numerous other names like “puncture vine,” “caltrop,” “goat head,” “puncture weed,” “Mexican sandbur,” “Texas sandbur,” or “bullhead.” T. terrestris (TT) primarily flourishes in Mediterranean nations and subtropical areas across the globe, which is represented in Figure 1 (Fernández-Lázaro et al., 2022). Secondary metabolites found in T. terrestris fruits include saponins, polyphenol chemicals, and alkaloids. The steroidal saponins are predominantly furostanol and spirostanol in nature. Furostanol saponins are thought to be biogenetic antecedents to the Spiro analogs (Ștefănescu et al., 2020). T. terrestris includes a steroidal saponin called “hecogenin,” which is employed as a starting material in the synthesis of steroidal medications; also, diosgenin is a key precursor of the same plant employed in pharmaceutical companies for producing synthetic steroids (Borges et al., 2023; Chen et al., 2015). Treatment involved in AI is the life-long administration of GCs and mineralocorticoids. Continued usage of synthetic steroids has many side effects. This study is an attempt to treat metyrapone-induced AI with the T. terrestris fruit extract (TTFE) and with an isolated compound (hecogenin).
Materials and Methods
Drugs and Chemicals
Metyrapone (MTP), hecogenin (HG), and hydrocortisone (HC) were purchased from Merck, Bangalore. All other chemicals and reagents utilized in this experiment were procured from local suppliers.
Collection and Processing of Plant Material
T. terrestris was collected and authenticated. T. terrestris fruits were separated from the plant, powdered, extracted and isolated hecogenin using Santos and Branco (2014).
Tribulus terrestris.

Experimental Animals
Healthy Wistar rats (180–220) were procured and maintained in animal house conditions with 24 ± 1°C temperature, 45–50% relative humidity, 12 h light and dark cycle, fed with standard pellet diet and water ad libitum. All the animals were acclimatized to animal house environment for 7 days prior to the start of experiment.
Grouping of Animals and Induction of AI
The rats were divided into seven groups (n = 6) as follows:
Group I: Normal control; vehicle-treated;
Group II: Disease control; metyrapone treated 150 mg/kg/oral;
Group III: TTFE; 200 mg/kg/oral;
Group IV: TTFE; 400 mg/kg/oral;
Group V: TTFE; 800 mg/kg/oral;
Group VI: Hecogenin (HG); 50 mg/kg/oral;
Group VII: Hydrocortisone (HC); 124 mg/kg/oral.
Metyrapone was selected to induce AI in rats. The dosing for all the groups was started from day 1 once daily per oral. The hormonal (corticosterone and aldosterone) levels were evaluated on days 1, 14, and 28 after treatment with extracts and hecogenin. Blood was withdrawn by the tail vein for the hormonal assessments. On the 28th day, animals were sacrificed to study the histopathology of the adrenal cortex and brain.
Biochemical Assays
Collection of Blood Sample
1.5 mL of blood was collected from the lateral tail vein using a 23 G needle attached to the syringe. Rats were placed in restrainers individually to collect the blood (Parasuraman et al., 2010). The blood samples were collected and sent to the laboratory of Adichunchanagiri Hospital and Research Centre for the analysis of corticosterone, aldosterone, sodium, and potassium. Enzyme-linked immunosorbent assay (ELISA) was performed for the hormonal analysis, which is a sensitive, reliable, replicable, specialized, and speedy method (Jameel et al., 2014; Nassiri et al., 2017; Zhou et al., 2021). Sodium and potassium were measured by spectrophotometer (Farhadi & Dizaye, 2019).
Behavioural Assessments
Locomotor Activity
The locomotor assessment of the animals was measured by Digital Actophotometer (Inco). A metallic square arena with a light source and photocells for measuring the locomotion. During animal locomotion, the light passing from the photocells from one end to the other will be blocked, counted, and recorded for 10 min (Sanawar et al., 2022).
Dark and Light Test
The apparatus consists of two chambers. One white chamber and one dark chamber partitioned and connected to each other with a removable door. The animal was placed in the dark chamber facing towards the door. The animal was allowed to freely for 5 min. Latency time was noted along with many transitions. Time spent in each chamber was recorded. After every use, both chambers were cleaned with a hypochlorous solution (Campos-Cardoso et al., 2023).
Histopathology
The adrenal glands were fixed using 10% formalin, processed. The processed tissues were embedded in paraffin blocks and were cut about 5 mm thickness using a rotary microtome. These sections were stained with hematoxylin and eosin using routine procedures. Using the light microscope, the tissue slides were examined for any pathological changes.
Statistical Analysis
Data were expressed as mean ± standard deviation (SD), and statistical comparisons were carried out using analysis of variance (ANOVA) followed by Dunnett’s test. The data were analyzed by using GraphPad Prism 8.0.1 software. p values < 0.05 were considered significant.
Results
Measurement of Corticosterone
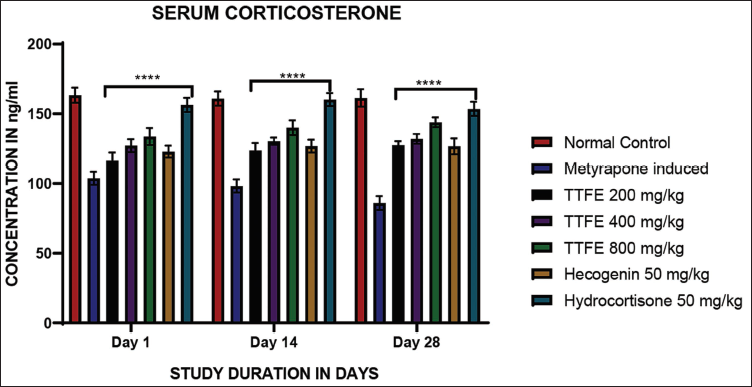
Corticosterone was measured weekly for four consecutive weeks using ELISA. The results were significant compared to the metyrapone-induced group on days 1, 14, and 28. An 800 mg/kg dose of TTFE showed higher corticosterone levels than a 50 mg/kg dose of hecogenin, as shown in Figure 2. The extract increased corticosterone concentrations in a dose-dependent manner.
Measurement of Aldosterone
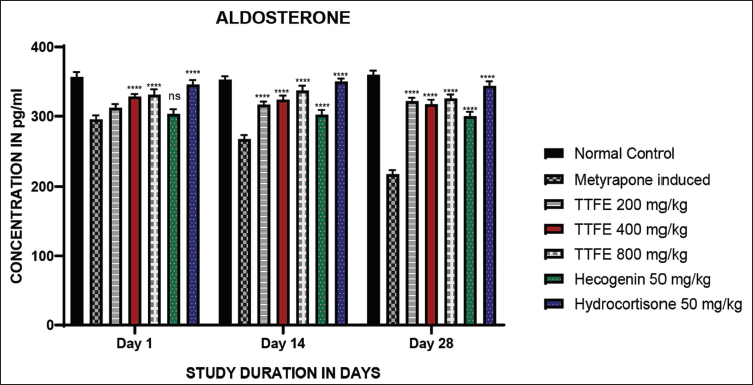
Aldosterone was measured using ELISA. Aldosterone levels increased significantly in a dose-dependent manner compared to the metyrapone-induced group. A 50 mg/kg dose of hecogenin did not show significant differences compared to the metyrapone-induced group on day 1. However, on days 14 and 28, aldosterone levels were significantly higher compared to the metyrapone-induced group. The results are shown in Figure 3.
Estimation of Sodium
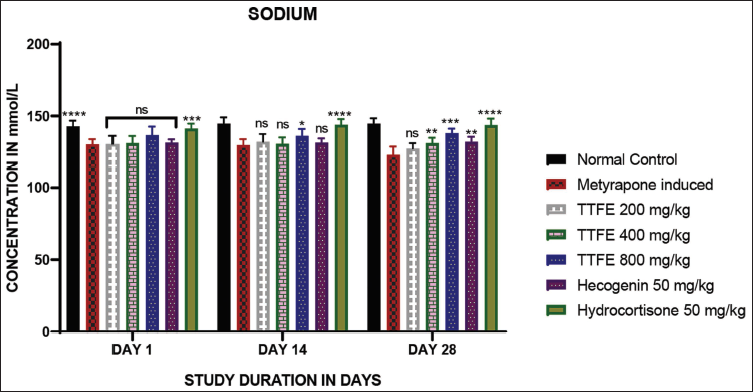
Sodium levels were estimated using a spectrofluorometer. There was no significant increase in sodium levels on days 1 and 14, except for the 800 mg/kg dose of TTFE compared to the metyrapone-induced group on day 14, and the 200 mg/kg dose of TTFE compared to the metyrapone-induced group on day 28. The results are shown in Figure 4.
Estimation of Potassium
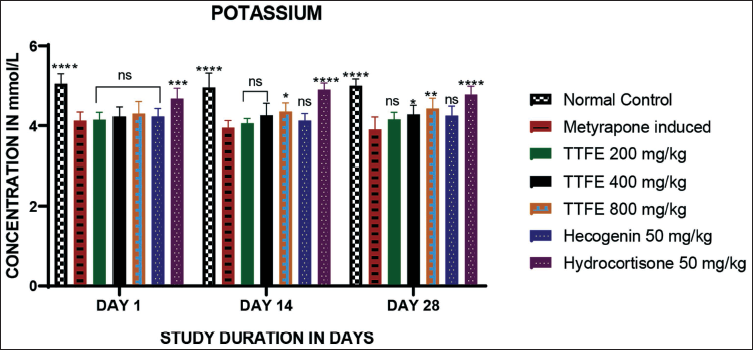
Potassium levels were measured using a spectrofluorometer. No increase in potassium levels was observed on day 1 in the group treated with TTFE at various doses compared to the metyrapone-induced group. On day 14, the 800 mg/kg dose of TTFE showed a slight increase in potassium levels compared to the metyrapone-induced group. Similarly, on day 28, both the 400 and 800 mg/kg doses of TTFE showed an increase in potassium levels compared to the metyrapone-induced group. The 50 mg/kg dose of hecogenin showed no increase in potassium levels from days 1 to 28. The results are shown in Figure 5.
Locomotor Scores
Locomotor scores were assessed using an Actophotometer. The locomotor scores for all groups were significant from days 1 to 28, except for the 200 mg/kg TTFE group, which did not show significance compared to the metyrapone-induced group on day 1, as shown in Table 1.
Locomotor Scores for 28 Days.
Dark and Light Test
The time spent in the dark chamber was recorded. Significant results were observed for all groups, except the 200 and 400 mg/kg. TTFE groups in comparison to the metyrapone-induced group on day 1. Additionally, the 200 mg/kg extract group showed no significant difference from the metyrapone-induced group on days 14 and 28, as detailed in Table 2.
Latency Time (Time Spent in the Dark Chamber) for 28 Days.
Histopathology
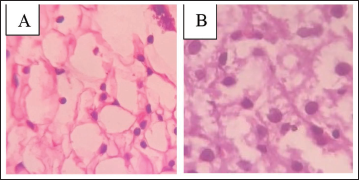
The examination of the adrenal glands revealed no detectable abnormalities in the cortex. The vacuoles appeared normal, with no evidence of enlargement or excessive growth of tissue. There were no signs indicating hypertrophy or hyperplasia within the glandular structure. The overall condition of the cortex remained healthy and unremarkable. This lack of pathological changes suggests stability in adrenal function. The findings are depicted in Figure 6.
Histopathology of Rats’ Adrenal Gland Showing No Abnormalities in the Cortex. Vacuoles are Normal, with No Signs of Hypertrophy or Hyperplasia in Both the Groups. (A) Normal Rat, (B) Metyrapone Induced.
Discussion
The therapeutic management of AI includes two classes of drugs, such as GCs and mineralocorticoids. However, there are many side effects on continued usage of synthetic corticosteroids including diabetes (Descours & Rigalleau, 2023; Li & Cummins, 2022), osteoporosis (Chen et al., 2023; Humphrey et al., 2023), myopathy (Coutinho et al., 2023), polymyalgia rheumatic (Espígol-Frigolé et al., 2023), glaucoma (AlObaida et al., 2022; Kawabe & Uesawa, 2023) and diabetes (Li & Cummins, 2022; Shah et al., 2022). The present study demonstrates, for the first time, corticosterone and aldosterone elevating properties of hecogenin from T. terrestris on metyrapone-induced AI in Wistar rats. We showed that hecogenin was effective in metyrapone-induced AI. Hecogenin at 50 mg/kg showed a significant increase in corticosterone and aldosterone compared to the disease-induced group and the value near to hydrocortisone group.
Conclusion
The study was designed to evaluate the effects of TTFE and an isolated compound (hecogenin) on metyrapone-induced AI. Administration of metyrapone produced reduction of corticosterone, and aldosterone levels in the Wistar rats. The 400 and 800 mg/kg of TTFE and hecogenin 50 mg/kg showed a significant increase in the levels of corticosterone and aldosterone respectively.
Summary
T. terrestris treatment leads to a significant increase in corticosterone and aldosterone in adrenal cortex. Hecogenin and Tribulus extract results in a notable increase in locomotor scores of rats. Treatment with the Tribulus extract and hecogenin leads to a concentration-dependent improvement in sodium and potassium levels.
Footnotes
Abbreviations
AI: Adrenal insufficiency; ELISA: Enzyme-linked immunosorbent assay; GC: Glucocorticoids; HC: Hydrocortisone; HG: Hecogenin; HPA axis: Hypothalamic–pituitary–adrenal axis; MC: Mineralocorticoids; MTP: Metyrapone; TTFE: Tribulus terrestris fruits extract.
Acknowledgments
The authors acknowledge Sri Adichunchanagiri College of Pharmacy and Vidya Siri College of Pharmacy, Bangalore for providing support to carry out the present work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The ethical approval for the study has been obtained from the ethics committee (SACCP-IAEC/2022-02/59).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia [Grant No. KFU242624].
