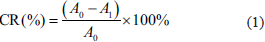Abstract
Background
Curcuma zedoaria is a common traditional herbal medicine that contains mainly curcumin and volatile oils with antioxidant, anti-inflammatory, and antitumor properties.
Objectives
This study aimed to investigate the effects of C. zedoaria extract on the biological behavior of ovarian cancer (OC) cells.
Materials and Methods
Active compounds were extracted from C. zedoaria, and the content of different active compounds was quantified. Their scavenging abilities against DPPH and -OH free radicals were analyzed, and the minimum inhibitory concentrations (MICs) against different bacterial strains were determined. The human OC cell line SK-OV-3 was divided into four groups and cultured in media containing C. zedoaria extract at concentrations of 0.0, 0.3, 0.6, and 1.2 g/L. The proliferation, inhibition, migration, and apoptosis of primary OC cells were then assessed using the methyl tetrazolium (MTT) assay, the scratch-healing assay, and flow cytometry. Tumor cell invasion and clonogenicity were also assessed. Finally, the status of MEK/ERK signaling in the cells was examined by protein imprinting.
Results
C. zedoaria extract contained 0.18% curcumin, 0.04% demethoxycurcumin, and 0.02% bisdemethoxycurcumin. When the concentration of C. zedoaria extract was ≥10 mg/mL, it exhibited a scavenging rate of over 85% for DPPH and -OH free radicals. In addition, C. zedoaria extract effectively inhibited the growth of Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus, and Staphylococcus epidermidis, with lower MICs observed for gram-positive bacteria. MTT cell proliferation and scratch assay results showed significant inhibition of SK-OV-3 cell proliferation and migration by C. zedoaria extract. Flow cytometry results indicated that the extract strongly promoted apoptosis in SK-OV-3 cells. Compared with the control group, the invasive and clonogenic abilities of SK-OV-3 cells decreased with increasing concentration of the extract. Protein imprinting results showed that treatment with C. zedoaria extract led to a concentration-dependent decrease in p-MEK1/2 and p-ERK1/2 protein levels in SK-OV-3 cells.
Conclusion
C. zedoaria extract possesses potent antioxidant, antibacterial, and antitumor properties. In addition, C. zedoaria extract can suppress the activation of MEK/ERK signaling, thereby inhibiting the proliferation and migration of SK-OV-3 cells, inducing apoptosis, and reducing the invasive and clonogenic potential of SK-OV-3 cells. It provides a theoretical basis for the discovery of novel drugs using C. zedoaria extract and its clinical applications in the treatment of OC.
Introduction
Ovarian cancer (OC) is a common malignancy in gynecology, and inhibiting tumor spread and metastasis to improve patient survival is a current focus of research (Choi et al., 2007).
Chemotherapy is currently the primary treatment modality for OC; however, it not only targets tumor cells but also causes damage to normal cells (van Stein et al., 2021). Traditional Chinese medicine (TCM) in the treatment of OC has demonstrated efficacy in alleviating clinical symptoms, reducing recurrence rates, and prolonging overall survival. As a result, it has received increasing attention from scientists (Yang et al., 2022).
Curcuma zedoaria, the dried rhizomes of plants in the Curcuma genus of the Zingiberaceae family, is a traditional medicinal herb known for its blood-activating and congestion-relieving properties (Yamasaki et al., 2022). The primary active ingredient in C. zedoaria extract is curcumin. Modern pharmacological research has confirmed that curcumin in C. zedoaria extract has potent antioxidant properties, effectively neutralizing free radicals in the body. This, in turn, reduces oxidative stress-induced damage to cells, helping to prevent or mitigate the development of various diseases (Gagliardi et al., 2020; Lin et al., 2019; Salehi et al., 2020). Curcumin in C. zedoaria extract has potent anti-inflammatory effects by inhibiting inflammatory responses and reducing the production and release of inflammatory mediators (Simeonova et al., 2022).
Curcumin effectively alleviates inflammatory symptoms and reduces inflammation-induced damage (Saadati et al., 2019). In addition, it has excellent antitumor properties, effectively inhibiting cancer cell proliferation and metastasis while promoting apoptosis in cancer cells, thereby inhibiting tumor growth and development (Giordano & Tommonaro, 2019; Mortezaee et al., 2019). Chronic inflammatory responses are closely associated with cancer progression, and the anti-inflammatory effects of curcumin can inhibit the generation and release of inflammatory factors, thereby reducing the damage caused to cells by inflammatory responses. Curcumin plays a role in the prevention and treatment of cancer (Zhang et al., 2022). Curcumin can also inhibit the growth and proliferation of cancer cells, induce apoptosis (programmed cell death), block tumor cell migration and invasion, and suppress tumor angiogenesis (Shakeri et al., 2019). In addition, studies showed that curcumin increases the sensitivity of tumor cells to chemotherapy, reducing its toxicity to normal cells and improving treatment outcomes (Abadi et al., 2022). Other active constituents of C. zedoaria extract, such as sesquiterpene compounds, also exhibit pharmacological effects with potential anticancer properties. Their use as an adjunct therapy for cancer may improve the effectiveness of treatment. However, further research is needed to validate the specific therapeutic efficacy of C. zedoaria extract.
In this study, the OC cell line SK-OV-3 was cultured to investigate the effects of C. zedoaria extract on cell proliferation, inhibition, migration, and apoptosis. This study aimed to understand the influence of C. zedoaria extract on the biological behavior of OC cells and to gain insight into its in vitro anti-OC effects.
Materials and Methods
Preparation of C. zedoaria Extract
Twenty grams of C. zedoaria medicinal herbs (San Yuan Tianyu Biotech Co., Ltd., China) were ground and sieved. They were then soaked in a 95% ethanol solution (Hubei Chengfeng Chemical Co., Ltd., China) in a ratio of 1:12 for 30 min, followed by ultrasonic extraction three times for 20 min each. The ethanol was evaporated by heating at 80°C, and the extract was concentrated to 1 mL, filtered through a 0.22 µm membrane filter, and stored refrigerated at 4°C. CLC-3200 high-performance liquid chromatography (HPLC; Qingdao Shenghan Chromatography Technology Co., Ltd., China) was used to analyze the active constituents in the extract, using acetic acid–acetonitrile–water (pH = 2.5, 1%) as the mobile phase, a flow rate of 1 mL/min, a wavelength of 420 nm or 214 nm, a column temperature of 25°C, an injection volume of 5 µL, and quantification of the major active constituents.
Determination of the Antioxidant Activity of C. zedoaria Extract
DPPH· is a relatively stable free radical. According to the instructions of DPPH· radical scavenging reagent (Sigma–Aldrich, USA), a solution of C. zedoaria extract was mixed with 1 mL DPPH· methanol solution (0.2 mM), and after a light-protected reaction for 30 min using the HBS-1096A Microplate Reader (Nanjing Detie Laboratory Equipment Co., Ltd., China) absorbance was measured at 517 nm. According to the instructions of ·OH radical scavenging reagents (Shanghai National Medicine Group Chemical Reagent Co., Ltd., China), a mixture of 0.2 mL of 3 mM FeSO4-EDTA, followed by 0.5 mL of 5 mM deoxyribose, 1.5 mL of C. zedoaria extract, 0.6 mL of 20 mM phosphate-buffered saline (PBS) (pH = 7.4), and 0.2 mL of 3 mM H2O2 was uniformly blended. This mixture was incubated at 37°C for 1 h. Subsequently, 1 mL of 2.8% (w/v) trichloroacetic acid solution and 1 mL of 1% trichloroacetic acid solution were applied, followed by a 15-min boil in a water bath. After cooling, using the HBS-1096A Microplate Reader (Nanjing Detie Laboratory Equipment Co., Ltd., China) absorbance was measured at 532 nm. Pure extract was used as the blank control, the measurements were performed three times, and the mean values were used to calculate the clearance rate (CR) for both DPPH· and ·OH free radicals using the following equation:
In the above equation, A0 and A1 are absorbances of blank control and extract, respectively.
Determination of the Antibacterial Activity of C. zedoaria Extract
C. zedoaria extract was mixed with Mueller–Hinton Broth culture medium (M8556, Beijing Solarbio Technology Co., Ltd., China) containing 1% Tween-80 to prepare a dilution gradient ranging from 50.0 to 0.1 mg/mL. Escherichia coli (ATCC25922, E. coli), Pseudomonas aeruginosa (ATCC9027, P. aeruginosa), Staphylococcus aureus (ATCCM8550, S. aureus), and Staphylococcus epidermidis (ATCC12228, S. epidermidis) were inoculated into fresh Mueller media (China Microbial Cell Resource Bank Preservation Center, China) at 37°C for 24 h. After activation, bacterial colonies were inoculated into sterilized physiological saline to prepare a suspension, and the turbidity was adjusted to a 0.5 McFarland standard using a turbidimeter (Shanghai Yidi Electronic Scientific Instruments Co., Ltd., China). Two hundred microliters of the diluted C. zedoaria extract were applied to a sterile 96-well plate, with 50 µL bacterial suspension applied then. Incubation was at 37°C for 24 h, and the minimum inhibitory concentration (MIC) was determined by evaluating the state of bacterial growth inhibition after complete suppression
Determination of the Anticancer Activity of C. zedoaria Extract
Cell Culture
Human OC cell line SK-OV-3 (Hefei Wanwu Biotechnology Co., Ltd., China) was cultured in RPMI 1640 media (Beijing Solarbio Technology Co., Ltd., China) containing 10% fetal bovine serum (Gibco, USA) and 1% penicillin–streptomycin (Beijing Solarbio Technology Co., Ltd., China). They were cultured in a CO2 incubator (C170, Eppendorf, Germany) with saturated humidity of 37℃ and 5% CO2. Monolayer culture cells were repeatedly digested with 0.25% trypsin (Beijing Solarbio Technology Co., Ltd., China), and the cells were inoculated into a 96-well plate. The cell culture status was observed with an inverted microscope (Tianjin Micrometer Photoelectric Co., Ltd., China).
Cell Proliferation
According to the instructions of the methyl tetrazolium (MTT) cell proliferation detection kit (Sigma–Aldrich, USA), SK-OV-3 cells (logarithmic growth phase) were seeded into a 96-well plate. Subsequently, cell culture media containing C. zedoaria extract at concentrations of 0.0, 0.3, 0.6, and 1.2 g/L were applied to each well. The cells containing 0.0 g/L C. zedoaria extract were used as a positive control group. The cells were then cultured for 12, 24, 48, 72, and 96 h, after which 10 µL of MTT reagent was applied to each well and further incubated for 4 h. Following this, 100 µL of dimethyl sulfoxide reagent was applied to each well to terminate the culture. The plate was shaken for 10 min, and optical density (OD) was measured at 490 nm employing an enzyme marker (FC, Thermo Fisher Scientific, USA). The cell proliferation inhibition rate was calculated: Inhibition rate (%) = (1 – OD Experimental Group/OD Control Group) ×100%. Since there is relatively little research on the application of C. zedoaria extract, the concentration dose selection was established based on the concentration range that has no cytotoxic effect, as determined in our previous studies. Then, the groups were set based on the trend of multiplying the concentration dose, which can provide a reference for the selection of doses in future in vivo studies.
Wounding Healing Assay
SK-OV-3 cells were seeded as described above. When cell confluence reached approximately 90%, the medium was replaced with a serum-free medium containing C. zedoaria extract at concentrations of 0.0, 0.3, 0.6, and 1.2 g/L. The cells containing 0.0 g/L C. zedoaria extract were used as a positive control group. Scratch wounds were made on the bottom of the culture dish using a sterile 10 µL pipette tip. The cell culture lasted for 24 h, and the healing of the scratch wounds was visualized and photographed.
Cell Apoptosis
According to the instructions of the Annexin V-FITC/PI assay kit (Beijing Takara Bio Medical Biotechnology Co., Ltd., China), SK-OV-3 cells were seeded (as described above). Subsequently, cell culture media containing C. zedoaria extract at concentrations of 0.0, 0.3, 0.6, and 1.2 g/L were applied, and cells were further incubated for 72 h, rinsed twice with PBS, centrifugated at 1,000 rpm for 10 min, resuspended in 500 µL binding buffer, and then Annexin V-FITC reagent (5 µL) and PI reagent (10 µL) were applied. The cells containing 0.0 g/L C. zedoaria extract were used as a positive control group. After thorough mixing, the cell suspension was incubated in the dark for 5 min. Cell apoptosis rates were immediately determined using a flow cytometer (CytoFLEX, Beckman Kurt, USA).
Detection of Cell Invasiveness
A total of 100 µL matrix adhesive diluted in the ratio of 1:8 (Sigma–Aldrich, Germany) was coated onto the upper chamber of a 24-well Transwell cell culture plate (Corning, China). After the matrix gel had solidified, cell culture media containing C. zedoaria extract at concentrations of 0.0, 0.3, 0.6, and 1.2 g/L were applied, and the cells were further incubated for 72 h. The cells containing 0.0 g/L C. zedoaria extract were used as a positive control group. Subsequently, cells (4 × 104 per well) were seeded onto the matrix gel, and they were cultured in serum-free media in the upper chamber, while 500 µL of complete culture medium was applied to the lower chamber. After 48 h culture, cells in the upper chamber were wiped off, and then they were fixed with 4% paraformaldehyde (Mindray, China), stained with 1% crystal violet (Shanghai MackLin Biochemical Technology Co., Ltd., China), rinsed with phosphate buffer solution (PBS; Shanghai Biyuntian Biotechnology Co., Ltd., China) to remove excess dye, and observed under a microscope. Cell counts were performed in three different fields at 100× magnification.
Detection of Cell Cloning Ability
SK-OV-3 cells (logarithmic growth phase) were uniformly seeded at 1,000 cells/well in a 6-well plate. After overnight incubation, cell culture media containing C. zedoaria extract at concentrations of 0.0, 0.3, 0.6, and 1.2 g/L were applied, and the cells were further cultured for 72 h. The cells containing 0.0 g/L C. zedoaria extract were used as a positive control group. With the supernatant discarded, cells were rinsed twice with PBS. They were then fixed with 100% methanol (Tianjin Xidian Chemical Technology Co., Ltd., China) for 30 min and stained with 1% crystal violet (Tianjin Xidian Chemical Technology Co., Ltd., China) dye for 20 min. Excess dye was washed off with distilled water, and then photographs were taken to visualize the formation of clones.
Western Blotting
SK-OV-3 cells were seeded as previously described, followed by the addition of cell culture media containing C. zedoaria extract at concentrations of 0.0, 0.3, 0.6, and 1.2 g/L. The cells containing 0.0 g/L C. zedoaria extract were used as a positive control group. The cells were further cultured for 72 h. After that, they were rinsed twice with PBS, centrifugated at 1,000 rpm for 10 min, and then lysed with radioimmunoprecipitation assay reagent (Shanghai Biyuntian Biotechnology Co., Ltd., China). Protein concentration in extracts was determined using the bicinchoninic acid assay (Shanghai Biyuntian Biotechnology Co., Ltd., China). A total of 50 µg of protein was subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis to separate proteins, then transferred onto a polyvinylidene fluoride membrane (Thermo Fisher Scientific, USA). The membrane was blocked with a blocking solution for 1 h. Primary antibodies against MEK1/2, p-MEK1/2, ERK1/2, p-ERK1/2, and GAPDH (Cell signaling, USA) were applied at a 1:1,000 dilution and incubated overnight at 4°C. Subsequently, a secondary horseradish peroxidase-conjugated immunoglobulin G (IgG) antibody (Cell signaling, USA), diluted at 1:2,000, was applied and incubated at 25°C for 1 h. After membrane washing, it was developed. Images were captured under the Gel Doc XR Gel Imaging System (Bio-Rad, USA). The relative levels of MEK1/2, p-MEK1/2, ERK1/2, and p-ERK1/2 proteins were calculated utilizing GAPDH as an internal reference.
Statistical Analysis
Group comparisons were conducted using one-way analysis of variance (ANOVA), with data presented as the mean ± standard deviation (x̿ ± s), and a p value less than 0.05 was considered statistically significant. Data analysis and processing were performed employing SPSS 19.0 (IMB, USA).
Results
Effective Component Detection of C. zedoaria Extract
Analysis of the active constituents in the C. zedoaria extract, as shown in Figure 1, indicates that the major active constituents in the extract are curcumin, demethoxycurcumin, and bisdemethoxycurcumin, which make up 0.18%, 0.04%, and 0.02% of the extract, respectively.
Proportion of Main Active Ingredients in Curcuma zedoaria Extract.
The Scavenging Ability of C. zedoaria Extract on DPPH· and ·OH Radicals
In this study, the scavenging capacity of C. zedoaria extract on DPPH free radicals was evaluated at different concentrations.
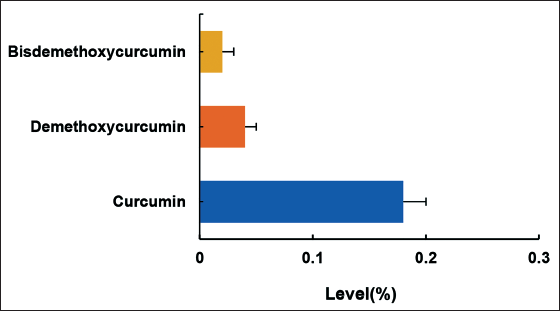
Figure 2A shows that with increasing concentrations of C. zedoaria extract in the range of 0.0–10.0 mg/mL, the CR of DPPH free radicals increased rapidly. However, at concentrations of C. zedoaria extract ≥10.0 mg/mL, the CR of DPPH free radicals gradually reached a plateau. The scavenging capacity of C. zedoaria extract on -OH free radicals was also evaluated. In Figure 2B, C. zedoaria extract showed a similar effect on the CR of -OH free radicals as that observed for DPPH free radicals.
Curcuma zedoaria Extract Scavenging Rate Curve for Free Radicals. A: DPPH· Free Radical Scavenging Rate; B: ·OH Free Radical Scavenging Rate.
MICs of C. zedoaria Extract
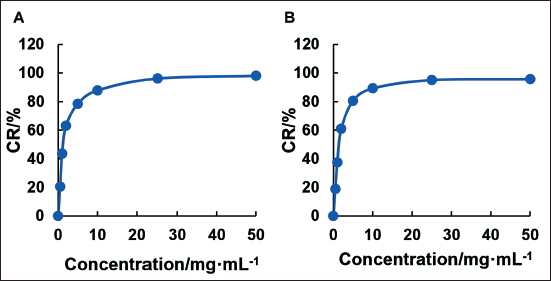
The assessment of MICs of C. zedoaria extract against various bacterial strains is presented in Figure 3. The MICs of C. zedoaria extract for E. coli, P. aeruginosa, S. aureus, and S. epidermidis were found to be 50.00 mg/mL, 6.25 mg/mL, 3.13 mg/mL, and 1.56 mg/mL, respectively.
Minimum Inhibitory Concentrations (MICs) of Curcuma zedoaria Extract.
Effect of C. zedoaria Extract on Ovarian Tumor Cells
Effects on the Proliferation of Ovarian Tumor Cells
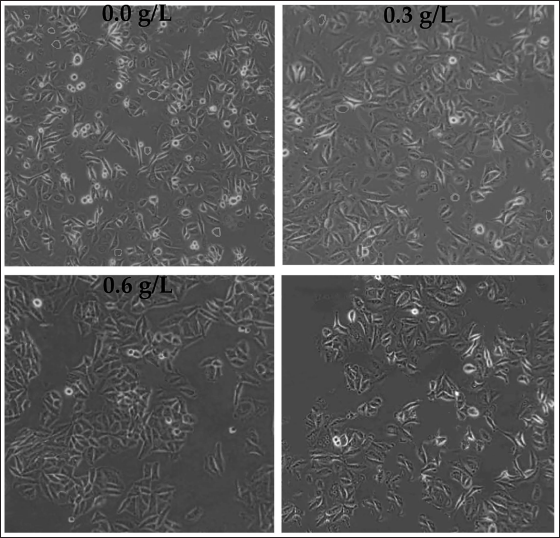
The effect of different concentrations of C. zedoaria extract on the growth of SK-OV-3 cells was evaluated. Figure 4 shows that the number of adherent SK-OV-3 cells decreased significantly with increasing concentrations of C. zedoaria extract.
Observation of Ovarian Tumor Cell Growth with Different Concentrations of Curcuma zedoaria Extract (×200).
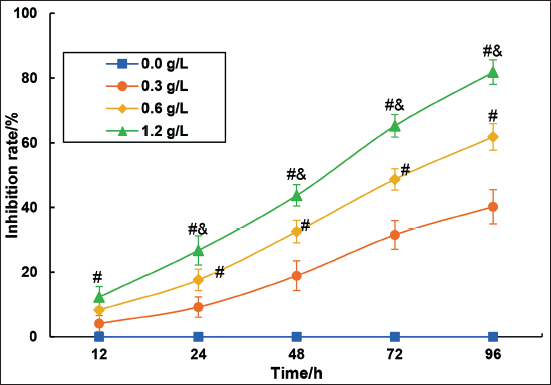
The effect of different concentrations of C. zedoaria extract on the proliferation of SK-OV-3 cells was verified using the MTT assay (Figure 5).
With increasing incubation time, it was observed that the inhibitory effect on SK-OV-3 cell proliferation gradually intensified with 0.3 g/L, 0.6 g/L, and 1.2 g/L concentrations of C. zedoaria extract. Compared to the 0.3 g/L concentration, the proliferation inhibition of SK-OV-3 cells was significantly enhanced with 0.6 g/L and 1.2 g/L concentrations of C. zedoaria extract (p < 0.05). Furthermore, compared to the 0.6 g/L concentration, the inhibition of proliferation of SK-OV-3 cells was significantly enhanced with 1.2 g/L and 1.2 g/L concentrations of C. zedoaria extract (p < 0.05).
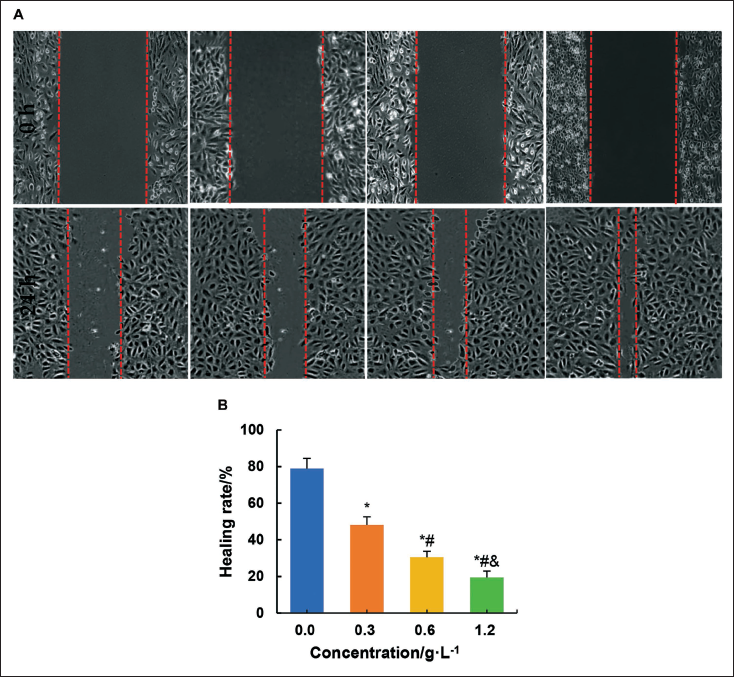
Effect on the Migration of Ovarian Tumor Cells
In this study, the influence of different concentrations of C. zedoaria extract on SK-OV-3 cell migration was evaluated.
As shown in Figure 6, the migration rate of SK-OV-3 cells after culture with 0.3 g/L, 0.6 g/L, and 1.2 g/L C. zedoaria extract was significantly decreased compared to the 0.0 g/L concentration of C. zedoaria extract (p < 0.05). Furthermore, the migration rate of SK-OV-3 cells was drastically reduced with 0.6 g/L and 1.2 g/L C. zedoaria extract compared to 0.3 g/L C. zedoaria extract concentration (p < 0.05). Similarly, the migration rate of SK-OV-3 cells was significantly reduced with 1.2 g/L C. zedoaria extract compared to 0.6 g/L C. zedoaria extract (p < 0.05).
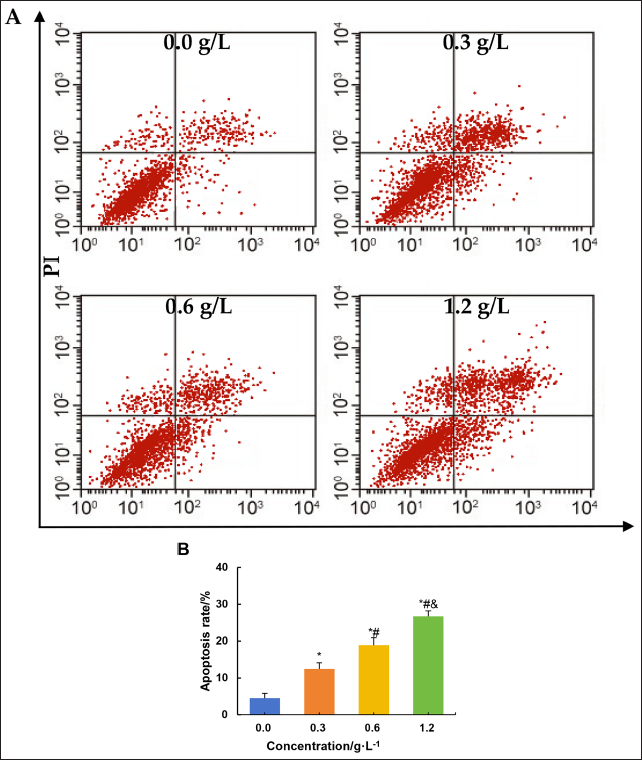
Effect on Apoptosis of Ovarian Tumor Cells
The effect of different concentrations of C. zedoaria extract on apoptosis in SK-OV-3 cells was evaluated by flow cytometry (Figure 7). Relative to the 0.0 g/L concentration of C. zedoaria extract, the apoptotic rate of SK-OV-3 cells increased significantly with 0.3 g/L, 0.6 g/L, and 1.2 g/L concentrations of the extract (p < 0.05). Compared to the 0.3 g/L concentration, both the 0.6 g/L and 1.2 g/L concentrations of C. zedoaria extract significantly increased the apoptotic rate of SK-OV-3 cells (p < 0.05). Furthermore, compared to the 0.6 g/L concentration, both the 1.2 g/L and 1.2 g/L concentrations of C. zedoaria extract significantly increased the apoptotic rate of SK-OV-3 cells (p < 0.05).
Effect on the Invasive Ability of Ovarian Tumor Cells
The invasive ability of SK-OV-3 cells under the influence of different concentrations of C. zedoaria extract was assessed (Figure 8).
The number of SK-OV-3 cells decreased significantly (p < 0.05) after cultivation with 0.3 g/L, 0.6 g/L, and 1.2 g/L extract concentrations compared to 0.0 g/L, and the invasive ability decreased with increasing extract concentration.
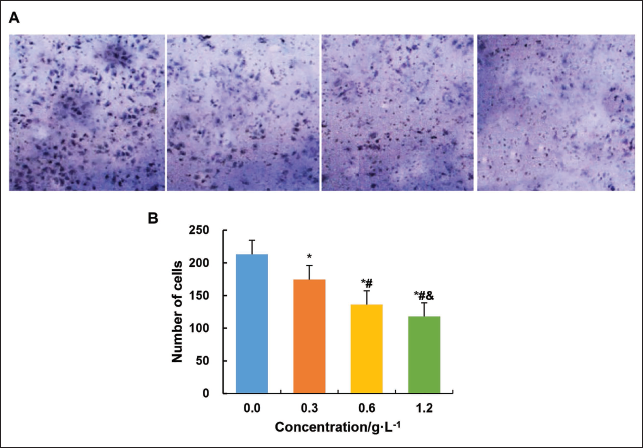
Effect on the Cloning Ability of Ovarian Tumor Cells
The invasive ability of SK-OV-3 cells under the influence of different concentrations of C. zedoaria extract was evaluated.
As shown in Figure 9, with increasing concentration of C. zedoaria extract, the number of visible cells gradually decreased. This indicates that C. zedoaria extract may reduce the clonogenic ability of tumor cells by inhibiting their proliferation, disrupting tumor cell division, and growth.
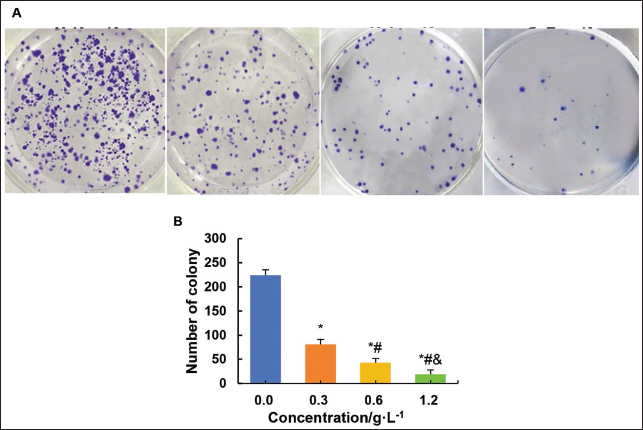
Effect on the MEK/ERK Signaling in Ovarian Tumor Cells
Western blotting experiments were conducted to assess the impact of various concentrations of C. zedoaria extract on MEK/ERK signaling-related proteins in ovarian tumor cells (Figure 10). Relative to the 0.0 g/L concentration of C. zedoaria extract, SK-OV-3 cells cultured with 0.3 g/L, 0.6 g/L, and 1.2 g/L concentrations of C. zedoaria extract showed a drastic reduction in p-MEK1/2 and p-ERK1/2 level (p < 0.05). Relative to the 0.3 g/L concentration of C. zedoaria extract, the 0.6 g/L and 1.2 g/L concentrations of C. zedoaria extract greatly decreased p-MEK1/2 and p-ERK1/2 in SK-OV-3 cells (p < 0.05). Likewise, relative to the 0.6 g/L concentration of C. zedoaria extract, the 1.2 g/L concentration of C. zedoaria extract also significantly reduced p-MEK1/2 and p-ERK1/2 levels in SK-OV-3 cells (p < 0.05).
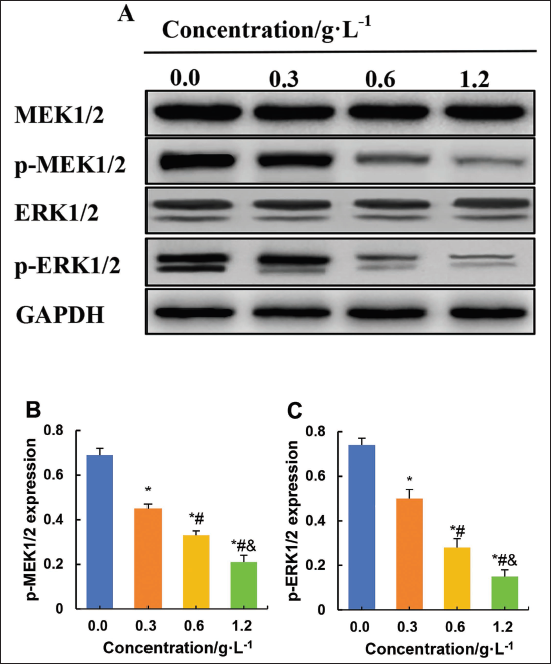
Discussion
Ovarian tumors are neoplasms that develop within the ovarian tissue and can be classified as benign or malignant.
The excessive proliferation of ovarian tumor cells leads to the formation of masses that cause pelvic swelling and discomfort (Morand et al., 2021). In addition, the location and size of ovarian masses can exert pressure on and affect adjacent organs or tissues (Liu et al., 2019). TCM has been widely used in the treatment of cancer. Compared with surgery, chemotherapy, and radiotherapy, TCM has the advantages of low toxicity, improved physical fitness of patients, and inhibition of tumor cell growth in the treatment of cancer. C. zedoaria is a plant belonging to the ginger family and is a common medicinal herb (Zhou et al., 2024). C. zedoaria belongs to the category of blood-activating and congestive drugs, which have various pharmacological effects such as anticoagulation, improvement of blood rheology, antitumor, antitissue fibrosis, analgesic, antibacterial and anti-inflammatory, and antiviral. This study prepared the extract of C. zedoaria and found that the main active ingredients in the extract were curcumin, demethoxycurcumin, and bisdemethoxycurcumin. Demethoxycurcumin and bisdemethoxycurcumin, also known as dimethoxycurcumin, are derivatives of curcumin and are the active constituents of C. zedoaria extract and have pharmacological properties similar to curcumin (Hatamipour et al., 2019). Curcumin and its derivatives exhibit various pharmacological activities, such as antioxidant, anti-inflammatory, antitumor, and antimicrobial properties (Chueh et al., 2020).
In addition, C. zedoaria extract contains other active constituents, including aromatic and phenolic compounds in the volatile oil fraction and fatty acids and polysaccharides in the nonvolatile fraction. The cellular damage and inflammatory response caused by oxidative stress are closely linked to the development of cancer. Therefore, the antioxidant capacity of the C. zedoaria extract was further investigated in this study. DPPH- is a relatively stable nitrogen-centered free radical. -OH is known as the most reactive and highly toxic reactive oxygen species, with the ability to attack biological substrates such as deoxyribonucleic acid (DNA) and polyunsaturated fatty acids, leading to lipid peroxidation reactions and damage (Schlosser et al., 2024). This study found that, with increasing concentration of C. zedoaria extract, the CRs of DPPH and OH radicals gradually increased, with CRs of 87.8% and 89.2%, respectively, at concentrations ≥10 ng/mL. This indicates that C. zedoaria extract can effectively eliminate both DPPH and -OH free radicals, demonstrating excellent antioxidant capabilities. This study further investigated the antibacterial ability of C. zedoaria extract and found that it can effectively eliminate E. coli, P. aeruginosa, S. aureus, and S. epidermidis, with a higher MICs CR for E. coli. This suggests that C. zedoaria extract has superior antibacterial activity against gram-positive bacteria compared to negative bacteria.
The rapid proliferation of malignant ovarian tumor cells can lead to the disruption of ovarian tissue structure, resulting in abnormalities in follicular development and ovulation, ultimately affecting female fertility (Nacarelli et al., 2020).
In addition, the rapid growth of malignant ovarian tumor cells can lead to complications, such as tumor necrosis and cyst formation (Lomnytska et al., 2021). Subsequently, C. zedoaria extract was used to treat SK-OV-3 OC cells and was found to be effective in inhibiting the growth and proliferation of SK-OV-3. The main active constituent of C. zedoaria extract is curcumin, which is known for its antitumor activity. Studies by Barinda et al. (2022) showed that curcumin can increase the sensitivity of OC animal models to the chemotherapeutic drug cisplatin, while also ameliorating chemotherapy-induced renal fibrosis and promoting tumor cell proliferation. Curcumin, the main active constituent of C. zedoaria extract, has potent antioxidant properties and helps to scavenge free radicals in the body. This action helps to alleviate oxidative stress-induced damage to cells, thus helping to prevent cancer (Ashrafizadeh et al., 2020). Studies conducted by Bian et al. (2022) showed that the medicinal serum containing C. zedoaria in combination with Astragalus membranaceus (Huangqi) could inhibit the signal transduction of Wnt5/β-catenin signaling, thereby suppressing the growth of colorectal cancer xenografts.
Furthermore, Zhu et al. (2021) found that C. zedoaria medicinal serum mediated the inhibition of proliferation, invasion, and migration of breast cancer MDA-MB-231 cells by modulating PI3K/Akt/mTOR signaling. The combined use of C. zedoaria with Carapax Trionycis showed enhanced therapeutic effects compared to monotherapy (Zhu et al., 2021). This suggests that C. zedoaria extract may inhibit the Pro of OC cells, thereby exerting anticancer effects. However, the exact mechanisms of action require further investigation.
Cancer cell migration refers to the process by which these cells move from the primary lesion to other parts of the body.
Malignant ovarian tumor cells can invade surrounding tissues and organs, leading to local spread and distant metastasis, affecting the function of other organs and reducing both patient survival and the likelihood of cure (Wu et al., 2018). The migration of malignant tumor cells often indicates the deterioration and progression of cancer, leading to an exacerbation of the disease process and increased complexity in the course of the disease (Gingold et al., 2022). The prognosis of cancer patients is closely related to the presence of distant metastases, and patients with migratory cancer cells are typically in advanced stages of the disease, resulting in a poorer prognosis (Ma et al., 2021; Neuhausser et al., 2020; Zulet et al., 2023). This study found that the use of C. zedoaria extract can significantly inhibit the migration, invasion, and cloning ability of SK-OV-3 OC cells, with concentration-dependent properties. Zhang et al. (2017) demonstrated that curcumin can induce demethylation of the MEG3 gene promoter region, thereby inhibiting the proliferation and drug resistance of OC cells. Curcumin and paclitaxel-prepared coloaded mixed micelles could inhibit the proliferation of SK-OV-3 cells without increasing the paclitaxel resistance of cells (Abouzeid et al., 2014). Curcumin, the main active component of C. zedoaria extract, was shown to be effective in inhibiting the proliferation of ovarian tumor cells. This may be related to factors such as abnormal gene expression or epigenetic changes, which require further validation. C. zedoaria extract, particularly curcumin, exhibits remarkable antitumor properties. In addition, this study found that the use of C. zedoaria extract can significantly promote apoptosis of SK-OV-3 OC cells and exhibit concentration-dependent characteristics. Curcumin can promote cancer cell apoptosis through various mechanisms, including cell cycle regulation, mitochondrial apoptotic pathways, modulation of pro and antioncogenic gene expression, and changes in signaling (Karunagaran et al., 2005). Xu et al. (2018) demonstrated that the combination of Astragalus extract (Astragalus polysaccharide) with C. zedoaria extract (curcumin) can regulate apoptosis-related proteins Bax, Caspase-3, and Bcl-2 to initiate and promote apoptosis in lung cancer cells (A549).
In addition to curcumin, C. zedoaria extract contains other active compounds with antioxidant, antitumor, immunomodulatory, and antimetastatic properties (Li et al., 2022). Previous research has shown that curcumin, a component of C. zedoaria extract, can affect several biological processes of tumor cells, including cell cycle, apoptosis, and invasive ability (Calibasi-Kocal et al., 2019). This study found that C. zedoaria extract can induce apoptosis in ovarian tumor cells and that this effect is concentration-dependent. These findings highlight the importance of C. zedoaria extract in the treatment of ovarian tumors and metastatic cases. This supports the investigation of C. zedoaria as a potential anticancer therapeutic agent.
Aberrant activation of the Ras/Raf/MEK/ERK signaling pathway has been implicated in the initiation, development, and chemoresistance of OC. This signaling pathway plays a critical role in various aspects of tumor cells, including proliferation, migration, invasion, apoptosis, and resistance to tumor chemotherapy. Aberrant activation of the Ras/Raf/MEK/ERK signaling pathway can lead to impaired apoptotic functions, excessive proliferation, and increased levels of angiogenesis, thereby contributing to the malignant transformation of cells. This study found that the use of C. zedoaria extract can significantly inhibit the phosphorylation levels of MEK1/2 and ERK1/2 in SK-OV-3 OC cells and exhibit concentration-dependent characteristics. p-ERK represents the active form of ERK, and the sustained activation of ERK signaling may promote the transformation of normal cells into a tumorigenic phenotype. The C. zedoaria extract could inhibit the ERK signaling pathway by affecting the protein expression of ERK1/2, p-MEK, and p-c-Raf (Yuan et al., 2021). This study revealed that C. zedoaria extract can inhibit the activation of MEK/ERK signaling, which acts in the occurrence of OC.
Currently, drug resistance to chemotherapy and its side effects are key factors affecting the therapeutic outcomes and prognosis of cancers such as OC. The results of this study confirm that C. zedoaria extract can significantly inhibit the proliferation, migration, cloning, and invasion of OC SK-OV-3 cells, and promote apoptosis. This is very important for the development of new drugs that address the aforementioned issues, with low or no toxic side effects and excellent therapeutic efficacy.
This study solely focuses on the analysis of the anticancer effects of C. zedoaria extract using OC SK-OV-3 cells. It can be observed that after treatment with C. zedoaria extract, there is a reduction in cell growth density, but no obvious issues, such as cell membrane rupture, are found. Therefore, it is speculated that its cytotoxic effects may be relatively low. However, whether C. zedoaria extract possesses cytotoxic effects depends on various factors, including the specific components of the extract, concentration, duration of action, cell type, and so on. Thus, future studies will still require a comparative analysis of the cytotoxic effects of C. zedoaria extract through multicell experiments.
The challenges brought by the low bioavailability of curcumin in vivo are multifaceted, including low absorption efficiency, rapid metabolism, limited distribution, difficulty in ensuring efficacy, and limitations of solutions. To overcome these challenges, it needs to continue to explore the pharmacokinetic properties of curcumin, develop more efficient, safe, and economical drug delivery systems, and fully consider the impact of individual differences on drug efficacy. However, it is important to note that this study only evaluated the effect of C. zedoaria extract on the biological characteristics of OC cells in vitro. Future research should investigate the effects on the inhibition of ovarian tumor growth and angiogenesis in vivo, as well as dissect the mechanisms underlying its therapeutic effects in OC.
Conclusion
C. zedoaria extract contains active compounds from the ginger family, including curcumin, demethoxycurcumin, and bisdemethoxycurcumin, which exhibit potent antioxidant and antibacterial properties.
In addition, C. zedoaria extract demonstrated concentration-dependent inhibition of SK-OV-3 cell proliferation and migration, while promoting apoptosis and suppressing the invasive and clonogenic capabilities of tumor cells. This effect is primarily achieved by inhibiting the activation of the MEK/ERK signaling pathway. In conclusion, this study may provide valuable insights for the development of therapeutic agents for the treatment of OC.
Footnotes
Abbreviations
CR: Clearance rate; HPLC: High-performance liquid chromatography; MICs: Minimum inhibitory concentrations; MTT: Methyl tetrazolium; OC: Ovarian cancer; PBS: Phosphate-buffered saline; TCM: Traditional Chinese medicine.
Authors’ Contribution
Conception and design: Na Song; performing the experiments and analyzing the data: Xiuyun Wang and Mingxing Yuan; analyzing the data and writing and revising the manuscript: Na Song and Xiuyun Wang. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.