Abstract
Background
Cedrin is a major bioactive flavonoid compound present in the Cedrus deodara plant and has been reported to have several biological activities. Diabetes mellitus is an endocrine and metabolic disease marked by high blood sugar levels leading to reduced insulin production and/or decreased insulin efficacy. Hyperglycemia leads to chronic health complications and organ dysfunction, specifically affecting the nerves, blood vessels, kidneys, eyes, liver, and heart.
Objectives
The aim of this work is to study the effect of cedrin on glycemic control, hyperlipidemia, liver function markers, and biochemical markers of streptozotocin (STZ)-induced diabetes in rats.
Materials and Methods
The STZ was utilized to initiate diabetes in experimental rats, and after the onset of diabetes, the rats were treated with cedrin for 30 days. After the treatment periods, the experimental rats’ glucose levels, food and water uptake, and body and organ weights were assessed. The biochemical markers, including insulin, hexokinase, glycated hemoglobin, and so on, were investigated using diagnostic kits. The concentrations of oxidative marker levels, inflammatory cytokines, and lipid marker levels were assessed using diagnostic kits.
Results
Cedrin treatment effectively mitigated the glucose level, food and water consumption, organ weights, and glycated hemoglobin (HbA1c) while increasing the body weight and hexokinase levels of the STZ-induced rats. In the STZ-induced rats, the cedrin treatment remarkably elevated the antioxidant levels, reduced oxidative stress biomarkers, and decreased the inflammatory cytokine levels in their serum.
Conclusion
The current study demonstrates that cedrin treatment effectively regulated dyslipidemia, inflammatory response, and oxidative stress in rats with STZ-induced diabetes. Therefore, it can be a potential salutary agent to treat diabetes in the future.
Introduction
Diabetes mellitus is a metabolic condition defined by a deficiency of insulin due to malfunctioning pancreatic beta cells, leading to impaired carbohydrate metabolism (Roussel et al., 2021). An estimated 463 million adults are currently affected by diabetes mellitus, and this case is expected to increase to 700 million by the year 2045 (Smyth, 2021). Type-1 diabetes arises when the body fails to manufacture insulin, necessitating the administration of insulin by injection. Type-2 diabetes refers to a state of elevated fasting blood sugar levels, even when insulin is present (Cho et al., 2018; Mohanta et al., 2023). The etiology of type-2 diabetes is defined by impaired production of insulin because of the death of the insulin-secreting β-cells, as well as reduced insulin activity caused by insulin resistance (Miao et al., 2018). Hyperglycemia is a foremost cause of oxidative stress, which in turn results in the generation of reactive oxygen species (ROS). In addition, elevated levels of ROS caused by high blood sugar disturb the insulin pathways and promote the onset of insulin resistance (Gurzov et al., 2014). Furthermore, abnormal lipid levels are common in diabetes as a result of metabolic disruptions that impact crucial pathways involved in lipid metabolism. Dyslipidemia is often characterized by elevated serum cholesterol levels and decreased levels of high-density lipoprotein (HDL) (Nowotny et al., 2015). Streptozotocin (STZ) is a frequently employed agent for inducing diabetes in rats. The STZ induces the demise of pancreatic cells by chemically modifying the deoxyribonucleic acid (DNA) by alkylation. This alteration results in a reduction in the release of insulin (Kakimoto et al., 2013). The STZ treatment has been shown to cause insulin resistance and impede insulin production from β cells. This induces type 2 (noninsulin-dependent) diabetes mellitus in rats (Golson et al., 2014; Qinna & Badwan, 2015). The primary aim of treatment is to achieve glycemic control to prevent the occurrence of microvascular, macrovascular, and neurological problems associated with diabetes (Rodríguez-Gutiérrez et al., 2021). Currently, there is a wide range of antidiabetic medications available on the market to treat high blood sugar levels. However, both metformin and sulfonylureas, which are types of antidiabetic medicines, have various undesirable side effects (Baynes, 2015). Therefore, the need for effective and safer alternatives to treat hyperglycemia has been of utmost importance in recent times.
Cedrin is a bioactive flavonoid compound that is found extensively in the Cedrus deodara plant and has been reported to have several pharmacological properties (Bisht et al., 2021). Cedrin showed effective antitumor (Shi et al., 2016), neurotoxicity (Zhao et al., 2018), alpha-amylase inhibition (Xu et al., 2017), antimicrobial (Zeng et al., 2012), and antioxidant (Zeng et al., 2014) properties. Nevertheless, the comprehensive antidiabetic potential of cedrin in animal diabetic models has not been documented. The goal of this work is to study the effect of cedrin on glycemic control, hyperlipidemia, liver function markers, and biochemical markers of STZ-induced diabetes in rats.
Materials and Methods
Experimental Rats
The present work was performed on 8-week-old male Wistar rats with an average body weight of 230 ± 10 g. The rats were kept in a controlled laboratory setting, with meticulous care given to their welfare. The conditions were regulated at 22–26°C, with air moisture levels ranging from 40 to 70% and a 12-h cycle of darkness and light. During the study period, the rats were provided with drinking water and a standard rat diet. The animals were given 7 days to relax to alleviate any stress caused by transit. All the protocols for the animal experimentations were verified and approved by the institutional animal ethics committee.
Experimental Groups
The rats were categorized into four groups (I–IV), with each group consisting of six rats (n = 6). Group I was control and provided only with a standard diet. Group II was a diabetes-induced group, which is induced by STZ. The fresh STZ solution was synthesized using 0.05 M sodium citrate buffer and administered intraperitoneally to the rats at 55 mg/kg to induce diabetes. To establish diabetes mellitus induction, we utilized glucose strips (AccuChek, Roche) to analyze the glucose levels of the experimental rats. The study only included diabetic rats with blood glucose levels over 250 mg/dL. Group III rats were STZ-induced and treated with cedrin (25 mg/kg) for 30 days. The group IV rats were STZ-induced and treated with glibenclamide (5 mg/kg) for 30 days.
Analysis of Body Weight, Organ Weight, and Food/Water Uptake Levels
The body weight, food, and water uptake of the experimental rats were regularly checked, and the results were tabulated. After the completion of treatments, the rats were euthanized by cervical dislocation, and organs, such as liver and kidneys, were excised. The organs were then weighed to measure the absolute organ weight index.
Analysis of Biochemical Marker Levels
The glycated hemoglobin (HbA1c) and insulin levels in the serum were assessed using diagnostic kits following the manufacturer’s guidelines (Abcam, USA). The commercial diagnostic kits were utilized to study the hexokinase, glucose-6-phosphatase (Glu-6-P), fructose-1,6-bisphosphatase (Fru-1,6-BP), total protein, and creatinine levels in the serum (Elabscience, USA). The experiments were conducted as per the manufacturer’s guidelines to assess these marker levels.
Analysis of Liver Marker Enzymes
Using the commercial assay kits, the liver function marker enzyme activities like alanine transaminase (ALT), aspartate transaminase (AST), alkaline phosphatase (ALP), and lactate dehydrogenase (LDH) enzymes were analyzed in the serum. The assays were performed according to the specifications of the manufacturer (Abcam, USA).
Analysis of Lipid Marker Levels
In order to assess the triglycerides (TG), total cholesterol (TC), low-density lipoprotein (LDL), high-density lipoprotein (HDL), apolipoprotein A-I (apo A-I), and apo B levels in the serum, commercially procured assay kits were utilized. All assays were conducted in triplicates as per the manufacturer’s specifications (Elabscience, USA).
Analysis of Oxidative Stress Marker Levels
The lipid hydroperoxides, TBARS, catalase (CAT), superoxide dismutase (SOD), glutathione (GSH), and glutathione peroxidase (GPx) levels in the serum were analyzed using diagnostic kits and following the instructions provided by the manufacturer (Abcam, USA).
Analysis of Proinflammatory Cytokine Levels
The serum tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and interleukin-1 beta (IL-1β) levels were quantified using commercial kits and following the indicated protocols by the manufacturer (Elabscience, USA). The assays were conducted in an independent manner.
Statistical Analysis
The data are studied using the GraphPad Prism software (Version 10; GraphPad Software Inc., Boston, USA). The results are illustrated as a mean ± standard deviation (SD) of three replicate assays. They were studied using a one-way analysis of variance (ANOVA) and Duncan’s post hoc assay. The significance was determined using a threshold p < 0.05.
Results
Effect of Cedrin on the Body Weight, Organ Weights, and Food and Water Uptake Levels of the Experimental Rats
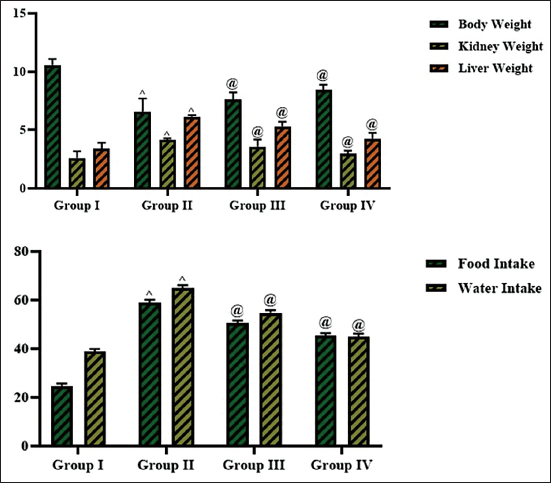
The levels of body weight, organ weights (liver and kidney), and food and water uptake of the experimental rats were tested, and the data are shown in Figure 1. The STZ induction drastically decreased (p < 0.01) the body weight (6.59 ± 0.2), increased the liver (6.13 ± 0.6 g), and kidney (4.13 ± 0.7 g) weights, and elevated the water (64.90 ± 0.5 mL) and food consumption (58.97 ± 0.6 g) of the experimental rats in comparison to the control. Remarkably, the 25 mg/kg of cedrin treatment led to a considerable elevation (p < 0.05) in body weight (7.59 ± 0.4), a diminution in water (54.76 ± 0.4 mL) and food consumption (50.66 ± 0.5 g), and decreased organ weights in the diabetic rats. These findings align with the outcomes of the glibenclamide treatment. Therefore, it is clear that cedrin treatment effectively mitigated the diabetes-induced physiological changes in the experimental rats.
Effect of Cedrin on the Blood Glucose, Plasma Insulin, and HbA1c Levels in the Experimental Rats
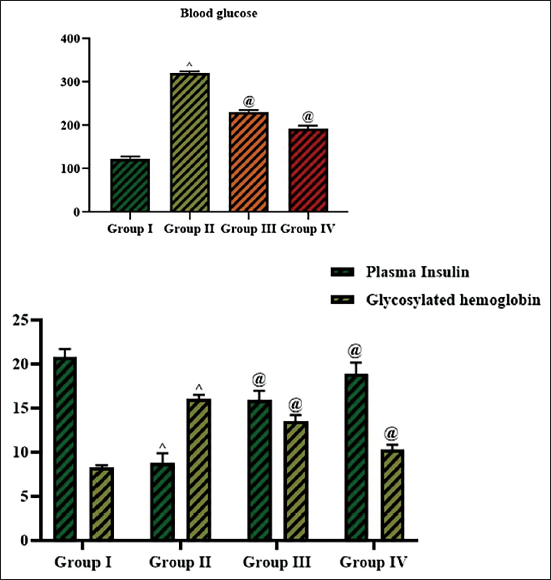
The regulating effects of cedrin on abnormal blood glucose, insulin, and HbA1c levels are depicted in Figure 2. The STZ-induced rats were shown to have increased (p < 0.01) HbA1c (16 ± 07%) and blood glucose (320.52 ± 0.6 mg/dL) while decreasing plasma insulin (8.81 ± 0.4 pM/L) levels when compared to the control. Considerably, the cedrin (25 mg/kg) treatment markedly lowered (p < 0.05) the levels of HbA1c (13.50 ± 0.3%) and blood glucose (231.44 ± 0.6 mg/dL) while boosting the insulin (15.94 ± 0.4 pM/L) levels in the STZ-induced rats. These findings are in line with the findings of the standard drug glibenclamide treatment. The present findings highlighted the beneficial effects of cedrin on mitigating the blood glucose and HbA1c rise and subsequent drop of insulin in diabetic rats.
Effect of Cedrin on the Hexokinase, Glu-6-P, and Fru-1,6-BP Levels in the Experimental Rats
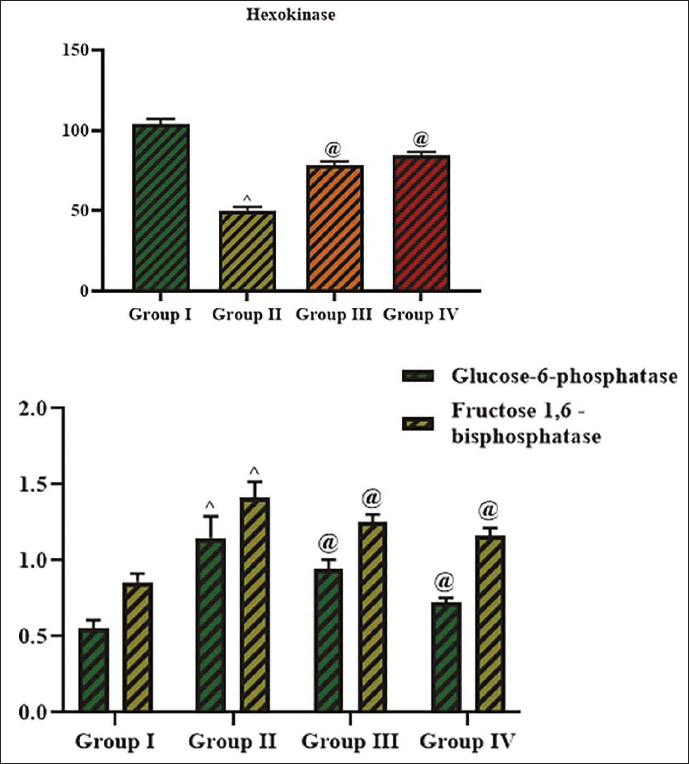
Figure 3 presents the measurements of hexokinase, Glu-6-P, and Fru-1,6-BP in the serum of experimental rats. The current findings indicate that the levels of Glu-6-P (1.14 ± 0.8 nM/mL) and Fru-1,6-BP (1.41 ± 0.2 nM/mL) were increased (p < 0.01), while the hexokinase (50.07 ± 0.4 nM/mL) levels were reduced in the serum of diabetic rats. However, the cedrin treatment at 25 mg/kg concentration led to a considerable reduction in the Glu-6-P (0.94 ± 0.1 nM/mL) and Fru-1,6-BP (1.25 ± 0.6 nM/mL) levels while increasing (p < 0.05) the hexokinase (78.22 ± 0.4 nM/mL) levels in the serum of diabetic rats. Additionally, the results of glibenclamide-treated rats supported the antidiabetic activity of cedrin.
Effect of Cedrin on the Total Protein and Creatinine Levels
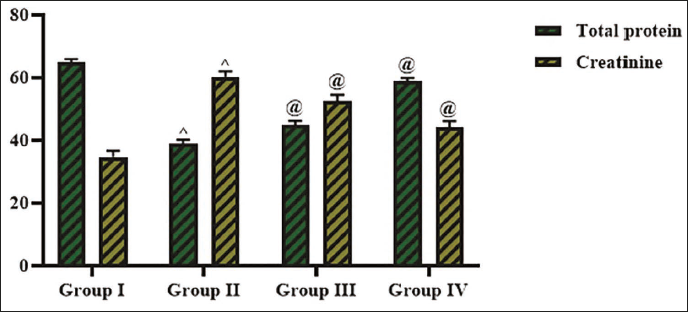
The creatinine and protein levels in the serum of experimental rats were studied, and the outcomes are given in Figure 4. The present findings indicate that the creatinine (60.36 ± 0.1 g/dL) was increased, and total protein (38.99 ± 0.4 g/dL) levels were reduced in the diabetic rats. But the treatment with cedrin demonstrated a remarkable increase in the total protein (45.04 ± 0.7 g/dL) and decrease in creatinine (52.56 ± 0.3 g/dL) levels in the diabetic rats. Additionally, these results are also found similar to the findings of the standard drug glibenclamide treatment.
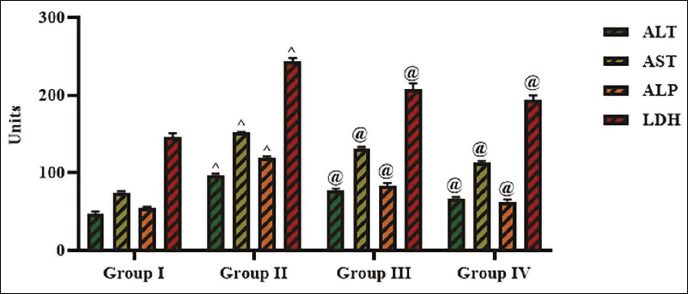
Effect of Cedrin on the Liver Marker Enzyme Levels in Experimental Rats
Figure 5 depicts the hepatic marker enzyme activities in the serum of experimental rats. In the serum of STZ-induced rats, there was a drastic elevation (p < 0.01) in the ALT (97.18 ± 0.5 IU/L), ALP (119.39 ± 0.1 IU/L), AST (151.66 ± 0.3 IU/L), and LDH (243.64 ± 0.1 IU/L) activities in contrast to the control. Remarkably, the cedrin treatment at a 25 mg/kg concentration substantially decreased (p < 0.05) these enzyme activity levels in the STZ-induced rats. The results of the standard drug glibenclamide further supported the hepatoprotective activity of cedrin.
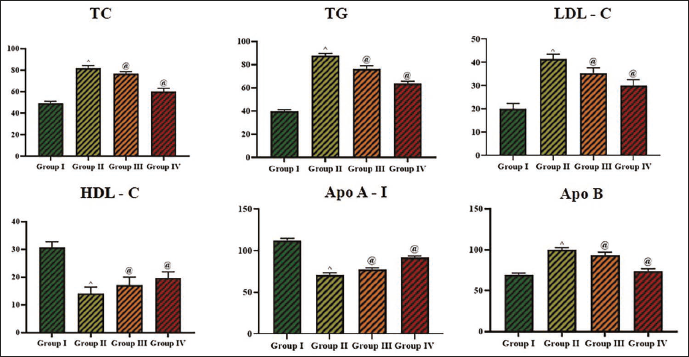
Effect of Cedrin on the Lipoprotein Marker Levels
The TG, TC, LDL, HDL, apo A-I, and apo B levels in the serum of experimental rats were studied, and the levels are illustrated in Figure 6. The TG (87.63 ± 0.4 mg/L), TC (81.97 ± 0.3 mg/L), LDL (41.59 ± 0.1 mg/L), and apo B (99.94 ± 0.3 mg/L) levels were remarkably elevated (p < 0.01 mg/L), while HDL (14.11 ± 0.8 mg/L) and apo A-I (71.23 ± 0.3 mg/L) was diminished (p < 0.01) in the STZ-induced rats. Surprisingly, the cedrin treatment at 25 mg/kg concentration significantly (p < 0.05) reduced the TG (76.38 ± 0.2 mg/L), TC (76.78 ± 0.2 mg/L), LDL (35.45 ± 0.2 mg/L), and apo B (93.35 ± 0.2 mg/L) levels while boosting (p < 0.05) the HDL (17.08 ± 0.4 mg/L) and apo A-I (77.82 ± 0.4 mg/L) levels in the serum of diabetic rats. These antilipidemic activities of the cedrin were further supported by the standard drug glibenclamide treatment.
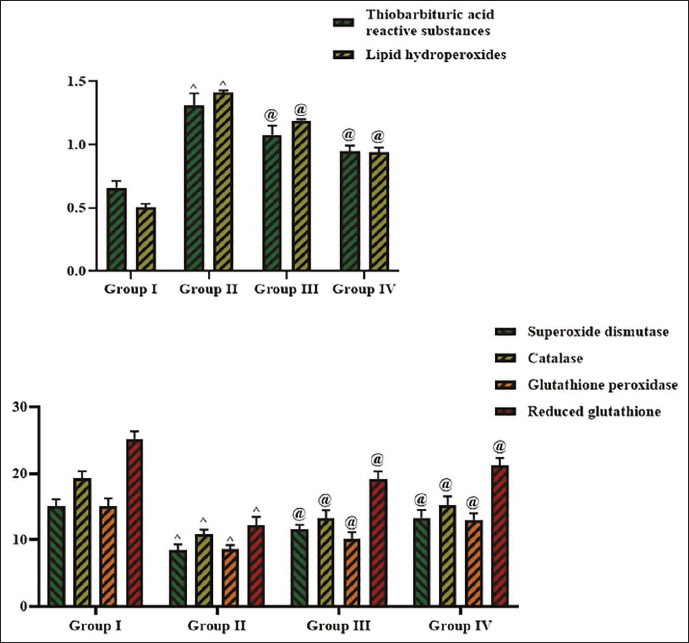
Effect of Cedrin on the Oxidative Stress Marker Levels
Figure 7 depicts the effects of cedrin treatment on the oxidative stress marker levels in the serum. The STZ-induced rats showed elevated levels of lipid peroxides (1.41 ± 0.1 nM/L) and TBARS (1.30 ± 0.2 nM/L) while reducing SOD (8.46 ± 0.4 IU/L), GSH (12.23 ± 0.6 nM/L), CAT (10.82 ± 0.6 IU/L), and GPx (8.58 ± 0.4 IU/L) levels (p < 0.01) compared with control. Remarkably, the 25 mg/kg of cedrin treatment successfully diminished the lipid peroxides (1.19 ± 0.1 nM/L) and TBARS (1.07 ± 0.5 nM/L) while boosting (p < 0.05) the SOD (11.61 ± 0.3 IU/L), GSH (19.07 ± 0.4 nM/L), CAT (13.25 ± 0.5 IU/L), and GPx (10.08 ± 0.6 IU/L) levels in the STZ-induced rats. This supports the antioxidant activities of the cedrin.
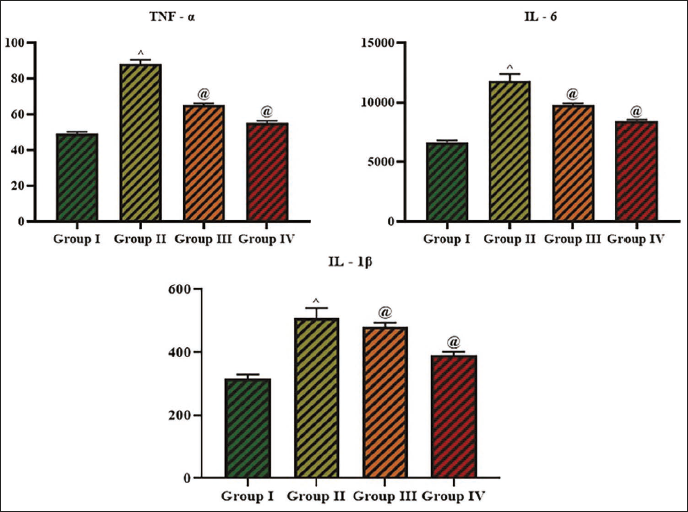
Effect of Cedrin on Inflammatory Cytokine Levels
The IL-6 (11,789.93 ± 21 pg/mL), IL-1β (508.84 ± 62 pg/mL), and TNF-α (88.24 ± 28 pg/mL) levels in the serum of the experimental rats are shown in Figure 8. Significantly (p < 0.01) higher amounts of these inflammatory cytokines were seen in the STZ-induced rats when compared to control. In contrast, the 25 mg/kg of cedrin treatment led to a considerable decrease (p < 0.05) in the IL-6 (9,770.61 ± 43 pg/mL), TNF-α (65.05 ± 41 pg/mL), and IL-1β (480.83 ± 52 pg/mL) levels in the serum of diabetic rats (Figure 8). The glibenclamide treatment also demonstrated a considerable diminution in these inflammatory marker levels, which also supports the anti-inflammatory properties of cedrin.
Discussion
Diabetes is a group of endocrine and metabolic disorders marked by high blood sugar levels, leading to a reduced insulin production and/or decreased insulin efficacy. Hyperglycemia leads to chronic health complications and various organ dysfunctions (Sneha & Gangil, 2019). Diabetes-induced hyperglycemia disrupts multiple biochemical and metabolic processes, hence impairing the operation of critical organs. Consistently lowering blood glucose levels will reduce the likelihood of microvascular problems (Kolb & Martin, 2017). Various methods are employed for the treatment of diabetes, including the consumption of nutritious food and dietary regulation, the administration of insulin injections, or the use of conventional hypoglycemic medications. These substances enhance the viability of pancreatic islets and promote β-cell regeneration via islet neogenesis-associated proteins (Krentz, 2006). Multiple oral antidiabetic medications with different modes of action for insulin stimulation are currently available on the market. Consequently, we have employed various hypoglycemic drugs to manage diabetic mellitus; however, they exhibit significant detrimental side effects, such as hepatic complications, lactic acidosis, and diarrhea (Gupta et al., 2011). Therefore, the need to explore potential and safer therapeutic strategies to treat diabetes is of great interest. The current study explored the antidiabetic effects of cedrin on STZ-induced rats.
An inability of the body to effectively use glucose during a diabetic condition results in increased utilization of proteins and subsequent weight loss. The reduction in body weight is a result of the deterioration of connective tissues due to STZ (Kumar et al., 2021). Significant weight loss is a prominent feature of STZ-induced diabetes, mostly caused by heightened protein loss and the lack of glucose for energy utilization (Fakhruddin et al., 2017). The current work demonstrated a drastic reduction in the body weight of diabetic rats. However, the cedrin treatment remarkably elevated the body weight of diabetic rats, which shows its salutary properties.
Hyperglycemia occurs when there is an excessive synthesis of glucose and a reduced use of glucose by the body’s tissues. The current study observed elevated FBG levels and reduced insulin levels, which are indicative of successful induction of diabetes in rats (Campos, 2018). Increased HbA1c levels and the presence of decreased blood insulin are dependable markers for evaluating the diabetic state. A rise in HbA1c levels considerably raises the probability of acquiring microvascular problems associated with diabetes. Furthermore, there is a significant correlation between HbA1c levels and diabetes. Diabetes is a metabolic condition that impacts several vital organs, including the liver and kidneys (Khan et al., 2020). The results of this work revealed that the cedrin treatment remarkably reduced the liver and kidney weights, FBG, and HbA1c levels while boosting the insulin levels in the diabetic rats. These findings highlighted that cedrin can alleviate diabetes-related complications in diabetic rats.
In diabetic conditions, there is an overproduction of serum fatty acids, which are then transformed into phospholipids and cholesterol in the liver. The liver can simultaneously release these two, together with any extra TGs, into the bloodstream. The elevated levels of serum lipoproteins in diabetes are mostly caused by an elevation in the release of free fatty acids due to the reduced lipase activity (Heeren & Scheja, 2021). The diabetic condition resulted in changes to their lipoprotein and lipid profiles. The higher blood glucose status was accompanied by alterations in the serum lipoproteins of the diabetic rats (Andallu et al., 2009). The lack of insulin during diabetes conditions leads to the inactivation of the lipoprotein lipase enzyme, resulting in secondary problems arising from increased levels of TGs and TC in the blood (Grice & Elmendorf, 2017). Diabetes-mediated hyperlipidemia occurs when there is an excessive migration of fat from adipose tissue because of insufficient use of glucose. Uncontrolled diabetes is well acknowledged to cause elevated levels of TG and LDL while reducing HDL, which contributes to the progression of coronary artery diseases. Moreover, research has demonstrated that a lack of insulin in diabetes mellitus results in many disruptions in metabolic and regulatory functions, ultimately causing the buildup of lipids such as TGs and TC in individuals with diabetes (Elberry et al., 2011). The lipid biomarkers of diabetic rats in the current work exhibited elevated TG, TC, LDL, and apo B status, while HDL and apo A-I levels were reduced. Also, the treatment of cedrin led to reduced lipid markers, including TC, TG, and LDL, and considerably raised the HDL and apo A-I levels. These findings highlighted the antilipidemic properties of cedrin.
Oxidative stress has a significant effect on the onset of diabetes. Diabetes independently enhances the generation of ROS that harm tissues by glucose auto-oxidation, which hinders enzymatic protein glycosylation. Diabetes has an impact on the levels of antioxidant enzymes, leading to an elevation in oxidative stress (Singh et al., 2022). Oxidative stress, which results in lipid peroxidation and chemical alterations in cellular components, is a primary cause of diabetes mellitus. Lipid peroxidation is a crucial contributor to the onset of diabetes mellitus (Rehman & Akash, 2017). CAT serves as the primary regulator of H2O2 and counteracts its effects by catalytically transforming it into water and oxygen. CAT deficiency increases the vulnerability of pancreatic islet cells to excessive ROS accumulation and oxidative stress. This ultimately results in the malfunction of the pancreatic islets and the progression of diabetes mellitus (Asmat et al., 2016). SOD and GPx are crucial enzyme antioxidants that play a pivotal role in avoiding oxidative stress and participate in cellular defense processes, hence safeguarding tissues from oxidative injury. Furthermore, earlier studies have indicated an elevation in pancreatic MDA content in rats with hyperglycemia (Greene et al., 1999). In the current work, we noted a notable elevation in lipid peroxides and TBARS levels and a decrease in antioxidants like SOD, CAT, GSH, and GPx levels in the serum of diabetic rats. Nevertheless, the cedrin treatment exhibited a decrease in TBARS and lipid peroxides and restoration of antioxidant levels that were close to normal. These findings supported the fact that cedrin has potential antioxidant properties.
The ALT, ALP, LDH, and AST are reliable indicators of hepatotoxicity, as increased activities of these enzymes suggest liver injury. The higher activities of these enzymes in diabetic rats are a consequence of injury to the liver cells and promote the production of glucose and ketones (Ghimire et al., 2018). The hepatotoxic effects of STZ were seen to result in the release of liver cytosolic enzymes into the bloodstream. Prior research has demonstrated the heightened levels of these enzymes in diabetic individuals, including those with STZ-induced diabetes, in experimental animals (Lozano-Paniagua et al., 2021). Elevated creatinine and urea levels in diabetic rats suggest renal impairment caused by the inability of insulin and glucose to enhance gluconeogenesis (Mohamed et al., 2016). The current findings highlighted that cedrin effectively decreased these markers enzyme levels in the diabetic rats. In addition, the cedrin treatment also diminished the creatinine and total protein levels in the diabetic rats. These outcomes highlight that the cedrin treatment successfully protected the liver as well as kidney tissues from the diabetic conditions.
Diabetes mellitus is associated with reduced physical activity and a sedentary lifestyle, which is connected to higher levels of chronic inflammation. Hyperglycemia-induced inflammation is well acknowledged as a key factor in the progression of diabetes (Lei et al., 2019). Diabetes mellitus is linked to the development of inflammation, which can lead to malfunction in multiple organs. The development of diabetic inflammation is facilitated by a sequence of interrelated processes that include the synthesis of chemokines, followed by the release of inflammatory markers. In diabetes, IL-1β, IL-6, and TNF-α increase insulin signaling. In addition, certain inflammatory markers can stimulate insulin resistance in adipose tissues and the liver by blocking insulin signal transduction (Odegaard et al., 2007). In this circumstance, the release of these cytokines occurs in the bloodstream and certain organs, leading to metabolic inflammation (Esser et al., 2014). The findings of the current work highlighted that the diabetic rats demonstrated an upsurge in the cytokine levels in their serum. Interestingly, the cedrin treatment successfully decreased these cytokine levels. These findings proved the anti-inflammatory properties of cedrin.
Conclusion
The current study demonstrates that cedrin treatment effectively regulates dyslipidemia, inflammatory response, oxidative stress, and other diabetic-related complications in rats with diabetes. The present results indicate that cedrin has the ability to alleviate diabetic conditions produced by STZ in rats. The main limitation of the present study is that it lacks in-depth molecular studies to unveil the underlying molecular mechanisms of cedrin’s antidiabetic activity, which needs to be addressed in the future. Additional research conducted in this specific area could improve our comprehension of the fundamental mechanisms that contribute to the beneficial effects of cedrin in treating diabetes.
Footnotes
Abbreviations
ALP: Alkaline phosphatase; ALT: Alanine transaminase; apo A-I: Apolipoprotein A-I; apo B: Apolipoprotein B; AST: Aspartate transaminase; CAT: Catalase; Fru-1,6-BP: Fructose-1,6-bisphosphatase; Glu-6-P: Glucose-6-phosphatase; GPx: Glutathione peroxidase; GSH: Glutathione; HbA1c: Glycated hemoglobin; HDL: High-density lipoprotein; IL-6: Interleukin-6; IL-1β: Interleukin-1 beta; LDH: Lactate dehydrogenase; LDL: Low-density lipoprotein; ROS: Reactive oxygen species; SOD: Superoxide dismutase; STZ: Streptozotocin; TBARS: Thiobarbituric acid reactive substances; TC: Total cholesterol; TG: Triglycerides; TNF-α: Tumor necrosis factor-alpha.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This work was approved by the Institutional Animal Ethical Committee by The First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
