Abstract
Background
Inflammatory illnesses and severe tissue injuries are frequently accompanied by pain. A widespread global health issue that has a significant negative impact on people’s quality of life everywhere is inflammation and the pain it causes.
Objectives
In this study, we sought to evaluate Ganoderic acid A’s antinociceptive and anti-inflammatory properties in mice models.
Materials and Methods
The current study aims to investigate the influence of Ganoderic acid A on animal models of inflammation and nociceptive response produced by heat and chemicals. Ganoderic acid A’s antinociceptive activity was examined using acetic acid-induced writhing, formalin and capsaicin-induced nociception, the tail immersion test, and the hot plate method. Ganoderic acid A’s anti-inflammatory effects were investigated utilizing carrageenan-induced inflammation, as well as the assessment of pro-inflammatory cytokine levels in mice. Additionally, the open-field test was used to examine behavioral changes in the mice.
Results
The findings of this work unequivocally showed that Ganoderic acid A significantly reduced the various thermally- and chemically-induced nociception in mice. Ganoderic acid A considerably increased the delay time in the hot plate and tail immersion tests. Capsaicin and formalin-stimulated models showed better licking times, and acetic acid-induced nociception was considerably reduced. Ganoderic acid A also effectively suppressed carrageenan-induced inflammation and decreased the synthesis of pro-inflammatory regulators including tumor necrosis factor-alpha, interleukin (IL)-1beta, and IL-6 in the experimental animals. However, it demonstrated only minor improvements in the behavioral assessment.
Conclusion
Thus, these findings, when paired with future investigations, will provide a better knowledge of Ganoderic acid A’s antinociceptive and anti-inflammatory capabilities, allowing for the development of a novel analgesic drug. To corroborate the preclinical findings, however, rigorously planned clinical investigations are desperately needed.
Introduction
Pain is a physiological and psychological phenomenon that is associated with tissue harm, either real or potential (Tadege et al., 2023). One of the main reactions and signs of tissue damage is pain (Li et al., 2021). Various body parts, such as the stomach, head, muscles, limbs, and joints, can experience pain. Insomnia, worry, exhaustion, decreased appetite, and even limb dysfunctions may accompany it (Gao et al., 2021). Numerous forms of pain have been identified, and they are all brought on by different factors. Severe physical discomfort is associated with these aches, which can have an adverse effect on daily activities. The reaction to inflammation brought on by strong and harmful stimuli that have the ability to trigger tissue injury is what distinguishes pain (Li et al., 2021). The sensation that alerts the tissue damage or indicates damage that has already been done as a result of a disease or injury is known as nociception associated with pain. Patients exhibit a lower pain threshold and a higher sensitivity to stimulation, leading in greater nociception (Alabi et al., 2019).
The processing of information about the internal or external environment by the central nervous system (CNS) as a result of nociceptors being activated is known as nociception. Information is received by the dorsal horn of the spinal cord or the nucleus caudalis, the trigeminal counterpart of the spinal cord, from nociceptors located in peripheral tissues when they are stimulated by noxious stimuli, such as tissue injury. The information is subsequently routed to the brainstem and, finally, the cerebral cortex, where it is translated into pain perception. Higher brain center processing is the source of pain; however, nociception can also exist in the absence of pain.
For instance, even after a total spinal cord transection, a person’s spinal cord can still process information from nociceptors, but it is unlikely that this information will generate stimulus-evoked pain because it cannot be conveyed past the transection (NRC, 2010). Nociceptors are mainly found in muscles, tissues, blood vessels, and bones. There are various mechanisms involved in regulating pain, including receptors, regulators, and neurotransmitters, that are associated with central and peripheral sensitization. The role of pain is to protect and warn the body to prevent further damage (Gao et al., 2021).
Pain serves as a universal marker of inflammation, signifying the release of diverse chemical mediators through molecular cascades. These include potassium ions and prostaglandins, which have the direct ability to activate pain receptors (Li et al., 2021), and it triggers a defense mechanism in response to the distressing sensory experiences linked to tissue injury (Gao et al., 2021). Inflammation is an immune process that occurs in reaction to the body’s protective systems triggered by a variety of unpleasant and harmful stimuli (Li et al., 2021). It provides a defense reaction to allergens, harmful substances, infections, and cell damage, resulting in redness, swelling, asthma, fever, pain, and other symptoms in the affected area (Zhang et al., 2022). Inflammation is the most frequent adaptive reaction of the body. Complex biological processes, like enzyme activation, the production of extravagating fluid and inflammatory markers, cell movement, and the breakdown and regeneration of tissue, are involved in both discomfort and inflammation (Tadege et al., 2023).
Inflammation is differentiated by a variety of factors that include increased blood flow, vascular permeability, and inflammatory cell infiltration into the injured location (Gao et al., 2021). To control central sensitization and pain, immune cells such as macrophages produce inflammatory regulators such as tumor necrosis factor (TNF)-alpha, interleukin (IL)-1beta, and IL-6, which interact with nociceptive neurons. In this situation, inflammation and pain are inextricably linked because nociceptive neurons influence inflammatory responses (Gao et al., 2021). There are two types of inflammation: acute and chronic. Acute inflammation is a transitory condition that can be resolved by removing the triggering stimuli. However, chronic acute inflammatory responses frequently result in arthritis, atherosclerosis, and bowel illness (Zhang et al., 2022).
While pain serves as the body’s defense mechanism in the face of harmful stimuli, it can also have a negative impact on an individual’s psyche, behavior, and social life in addition to posing a socio-economic burden on society. If pain is not properly managed, it can result in lower productivity and a lower quality of life (Alabi et al., 2019). The basic goal of pain management is to alleviate or eliminate pain and discomfort with no side effects. Usually, the goal of administering analgesic and anti-inflammatory drugs is to decrease pain. Opiates, such as morphine, and nonsteroidal anti-inflammatory drugs (NSAIDs), such as diclofenac and indomethacin, are presently available pain remedies. The pain is momentarily relieved by both opioids and NSAIDs (Li et al., 2021) and frequently has a variety of adverse effects, such as hepatotoxicities, heart problems, gastrointestinal damage, and respiratory exhaustion (Gao et al., 2021). Opioids can result in tolerance and reliance, whereas NSAIDs can induce stomach ulcers (Tadege et al., 2023).
Inflammatory pain lacks effective treatment, so researchers are exploring plant-based therapies to alleviate it. They are seeking alternative NSAIDs and opiate replacements with minimal side effects from plant-derived substances, which show promise for developing new analgesic agents. Ganoderic acid A is the primary bioactive compound found in Ganoderma lucidum. This bioactive compound possesses substantial medicinal importance and a broad spectrum of pharmacological activities. It demonstrates noteworthy cardioprotective (Zhang et al., 2020), anticancer (Wu et al., 2013), anti-hyperlipidemia (Guo et al., 2020), renoprotective (Meng et al., 2020), anti-lung fibrosis (Wen et al., 2020), and neuroprotective (Zhang et al., 2021) properties. The antinociceptive and anti-inflammatory properties of Ganoderic acid A have not been assessed yet. While Ganoderic acid A’s anti-inflammatory role is well-documented, its potential antinociceptive actions remain largely unexplored in scientific literature. As a result, this work aims to not only assess the antinociceptive effect of Ganoderic acid A but also to delve into its anti-inflammatory activity. In many behavioral tests, reflex reactions to painful stimuli, such as heating the tail or the hind paw to cause a short withdrawal response that is usually applied at threshold or slightly above threshold intensities, are assessed (NRC, 2010). The animal’s response (such as a nociceptive withdrawal reflex), which is thoroughly examined in this study, limits the stimulus duration, making these the main tests of nociceptive processing.
Materials and Methods
Chemicals
Prior to the start of the trial, all chemicals used in this study, including Ganoderic acid A (purity ≥98%), morphine, naloxone (NLX), carrageenan, diclofenac, formalin, and others, were acquired from Sigma Aldrich, USA.
Animals
The applicable national regulations and institutional policies for animal use and care were followed, and mice were acquired from the Approved Animal Center with the appropriate ethical approval for animal experimentation study. All the experimental study protocols were used by adopting ARRIVE guidelines (Percie du Sert et al., 2020).
The 6–7 weeks aged male Swiss albino mice weighing above 26 ± 5 g were acquired for the study. The animals were caged in polycarbonate cages and kept under-regulated room temperature of 23–25°C and 55%–60% relative humidity, with a 12-hour light–dark cycle. They had free access to both standard rodent chow pellets (Bio-Serv, Flemington, USA) and drinking water. Mice were handled with the greatest care and consideration. Animals were acclimated for a week before commencing the studies.
Effect of Ganoderic Acid A on the Hot Plate-induced Nociception in Mice
The hot plate experimental test is a widely used method to assess a compound’s antinociceptive activity by subjecting animals to stimuli that cause thermal discomfort and timing the onset of neuropathic pain reaction as a proxy for acute pain threshold (Tadege et al., 2023). Different chemicals were introduced, both singly and in combination, into six groups of mice. Every animal was fasting for the whole night. One group served as the control, while the other was given oral morphine at a dose of 5 mg/kg as a positive drug. The test sample, Ganoderic acid A, was given to the other group at a dose of 15 mg/kg. The chemicals were administered in varied combinations and at different doses to the remaining three groups. A combination of 2 mg/kg NLX, an opioid antagonist, and control was given to the fourth group; 15 mg/kg Ganoderic acid A and 2 mg/kg NLX were given to the fifth group; and approximately 5 mg/kg morphine and 2 mg/kg NLX were given to the last group. Animals were placed within the heated surface of the acrylic cylinder (55 ± 0.2ºC) to demonstrate the experiment. The amount of time that elapsed after the animals were placed on the platform before their hind paws licked, shook, or leaped was meticulously recorded as their latency of response (Gao et al., 2021). The animals’ rate of reaction to the temperature change was measured by response latency (Asiri et al., 2024). The animals were given the various doses as previously described 30 minutes before the heat shock in a hot plate (Gao et al., 2021). Mice were individually placed on a hot plate that was set at a specific temperature before the trials began. Those mice that did not lick their hind paw or leap (nociceptive responses) within 30 seconds of being placed on the hot plate were eliminated (He et al., 2022). To prevent tissue damage, a 30-second cutoff duration was established. All mice in the group had their latency times assessed prior to and following sample injections at 30, 60, 90, and 120 minutes (Li et al., 2021). Analogies were drawn between the increasing latency periods in the groups receiving treatment (Tadege et al., 2023).
Ganoderic Acid A’s Effect on Mice’s Tail Immersion-induced Nociception
The hot tail immersion method was also utilized to assess the test drugs’ antinociceptive impact against thermal pain. The tail immersion method was used to screen mice for hot water sensitivity (Azim et al., 2021). Under strict supervision, the experimental animals were allowed to submerge one-third of their tails in a water bath sustained at 55 ± 0.5ºC. The sample and reference medications used in the hot plate test were administered to mice for this assay. The mice had their tails immersed in hot water for around 3 cm. The mice exhibited signs of distress, and their tails were swiftly withdrawn from the scalding water. The time the tail was immersed in the hot water and the time it was removed were recorded 30 minutes before and after treatments at 30, 60, 90, and 120 minutes. To prevent tissue damage, the maximum immersion time was set at 15 seconds. “An extended immersion time showed that Ganoderic acid A has an analgesic effect” (Gao et al., 2021).
Effect of Ganoderic Acid A on the Carrageenan-induced Paw Edema in Mice
The paw edema was utilized to evaluate the anti-inflammatory properties of the compounds under investigation. The mice were distributed into three groups: the mice from the positive control group received 10 mg/kg of indomethacin, the mice from the negative control group received carrageenan alone, and the mice from treatment group received Ganoderic acid A at 15 mg/kg of concentration. The animals’ right paw’s sub-plantar space was injected with 100 µL of carrageenan suspension (1% w/v) 60 minutes after the samples were administered intraperitoneally (Hajhashemi et al., 2024). Before any treatments were administered, the basal right hind paw volume was measured and the animals’ paw edema was evaluated using a plethysmometer. Each animal’s paw volume was measured prior to, during, and 4 hours following the inflammatory provocation (Gao et al., 2021).
Investigating the Impact of Ganoderic Acid A on Acetic Acid-induced Nociception in Mice
The pain-relieving potential of Ganoderic acid A was examined using acetic acid-induced writhing in mice. The mice were distributed into three groups. The first group received regular water (control) (Li et al., 2021). The second and third groups were given Ganoderic acid A and diclofenac (a positive control), respectively, 30 minutes before being exposed to acetic acid (Gao et al., 2021). Following a 5-minute acetic acid challenge, each mice was examined independently, and over the course of 25 minutes, the writhing motions in their abdomens were recorded (Gao et al., 2021; Li et al., 2021). Following the acetic acid injection, the mice displayed various writhing behaviors, including abdominal contractions, limb extension, elongation, and trunk twisting (Asiri et al., 2024). The outcomes were depicted as the percentage of inhibition of writhing (Li et al., 2021).
Ganoderic Acid A’s Impact on Mice’s Capsaicin-induced Nociception
Using the capsaicin-triggered nociceptive test method, the impact of Ganoderic acid A on neuropathic nociception was ascertained. The sample and reference drugs were given to all mice in accordance with the acetic acid test protocol 1 hour before the intraplantar injection of capsaicin (20 mL, 1.6 mg/paw) (Gao et al., 2021). The subject animals were kept in the examination area for 5 minutes after induction, and the quantity of time they spent licking the capsaicin-administered paw was noted (Li et al., 2021).
Ganoderic Acid A’s Impact on Mice’s Formalin-induced Nociception
In this work, experimental mice were used to investigate Ganoderic acid A’s antinociceptive properties utilizing the formalin test method (Gao et al., 2021). Three sets of experimental mice were created. The first set of animals received normal water, which served as a control. The standard concentration of Ganoderic acid A was given to groups II and III as a test sample and morphine was added as a positive control. Mice were placed on transparent monitoring booths and 1% formalin in 0.9% saline was applied subcutaneously (50 µL/paw) to the hind paw surface of the animals (i.p.) 60 minutes after the last injection (Gao et al., 2021). The amount of time in seconds that an animal spent biting or licking its hind paw after receiving a formalin injection was thought to indicate a pain response (Li et al., 2021). The formalin test contains two phases: the neurogenic phase, which lasts for 0–5 minutes, and the inflammatory phase, which lasts for 15–30 minutes and is when the animal exhibits painful behaviors (Asiri et al., 2024). An interphase, a quiet period that occurs between these phases, occurs during which nociceptive responses are either completely absent or drastically reduced. The first phase (first 5 minutes) and the inflammatory phase (late phase, 15–60 minutes) mean nociceptive scores were recorded (Balali-Dehkordi et al., 2024).
Impact of Ganoderic Acid A on Pro-inflammatory Markers in the Model of Air Pouch Mice Caused by Carrageenan
Using the carrageenan-triggered air pouch model, the effects of Ganoderic acid A on the pro-inflammatory markers of the animals were investigated using the following methodology. The mice’s backs were shaved after they were given an anesthetic. Next, over the course of 3 days, pure air was injected twice into the mice’s backs to develop the air pouch (Gao et al., 2021). Following that, the animals with the pouches were divided into four factions. To elicit an inflammatory reaction, 0.5 mL of carrageenan were given to animals carrying pouches. The first group received 10 mL/kg of 1% Tween 80 as a control. Group IV was treated with dexamethasone, while group III received Ganoderic acid A. Carrageenan, 0.5 mL, was given to the second group. Following an hour of testing, the animals were killed, and the pouch was split apart, 2 mL of saline solution were injected into the cavity, and the contents were then drawn back out to gather the cells. The resultant suspension was centrifuged using the appropriate assay kits, and the resulting pellet was measured for the TNF-α, IL-1beta, and IL-6 levels (Gao et al., 2021; Li et al., 2021).
Impact of Ganoderic Acid A on the Alterations in Mice Behavior Evaluated by an Open-field Test (OFT)
The OFT was designed to measure emotionality in animals (Belovicova et al., 2017). The OFT was used to determine how Ganoderic acid A affected the natural locomotor activity of the experimental animals using the procedures described below. The animals were given the sample and standard drugs in accordance with the protocol of the formalin test for this purpose. After an hour of treatment, each mouse was placed in a different square on an open field setup that measured 50 × 50 × 50 cm (Gao et al., 2021). A 40-minute video recording of mouse behavior was captured using an above camera. Software called Smart 3.0 video tracking was used to analyze the videos (Yeung et al., 2021). It was possible to observe the animals’ altered behavior (Gao et al., 2021). Immobility was noted during the session as the square numbers passed by each mice’s paw (Li et al., 2021).
Statistical Analysis
Three separate tests yielded data that was given as mean ± standard deviation (SD). GraphPad Prism software (Version 10; GraphPad Software Inc., Boston, USA) was utilized to statistically analyze the values. Dunnett’s multiple comparison test was used to guide the two-way analysis of variance (ANOVA) used for the statistical analysis of the data. p < 0.05 was used to denote significance between the induced and control animal groups (Gao et al., 2021; Li et al., 2021).
Results
Ganoderic Acid A’s Antinociceptive Effects on Mice’s Hot Plate-induced Nociception
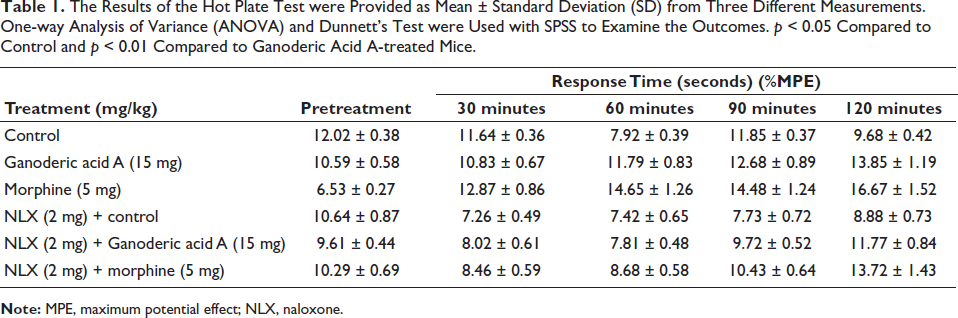
The hot plate test was used to assess how Ganoderic acid A affected the mice’s nociception, and Table 1 lists the effects of Ganoderic acid A on pain relief that were noted. Ganoderic acid A at 15 mg/kg significantly (p < 0.01) increased inhibitory effects after 90 and 120 minutes, with response durations of 12.68 ± 0.89 seconds and 13.85 ±1.19 seconds, respectively. The maximum potential effect (MPE) was assigned to this effect. Compared to the animals given Ganoderic acid A, the control group showed faster response times. The treatment of 5 mg/kg of morphine significantly (p < 0.01) inhibited nociception in 120 minutes, with a reaction time of 16.67 ± 1.52 seconds being the maximum. The combination of morphine (5 mg/kg) and NLX (2 mg/kg) had an initial reaction time of 8.46 ± 0.59 seconds at 30 minutes and 13.72 ± 1.43 seconds after 120 minutes. Mice on the hot plate responded faster to Ganoderic acid A treatment when coupled with NLX. The obtained outcomes of the hot plate-induced nociception revealed that Ganoderic acid A significantly increased the delay time.
The Results of the Hot Plate Test were Provided as Mean ± Standard Deviation (SD) from Three Different Measurements. One-way Analysis of Variance (ANOVA) and Dunnett’s Test were Used with SPSS to Examine the Outcomes. p < 0.05 Compared to Control and p < 0.01 Compared to Ganoderic Acid A-treated Mice.
Ganoderic Acid A’s Antinociceptive Effects on Mice’s Tail Immersion-induced Nociception
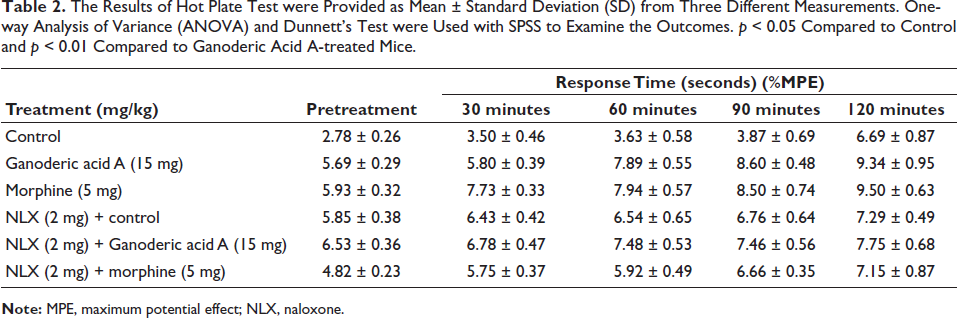
Ganoderic acid A’s antinociceptive activities were tested using a tail immersion test, and the results are shown in Table 2. Ganoderic acid A at 15 mg/kg substantially (p < 0.01) enhanced reaction time to thermal pain stimuli in 90 minutes (8.60 ± 0.48 seconds) and 120 minutes (9.34 ± 0.95 seconds). When compared to the control, the Ganoderic acid A’s response time improved significantly (p < 0.01) with tail immersion in hot water. Compared to the mice who received Ganoderic acid A treatment, the untreated control animals showed a slower response time. Similar to morphine therapy, the tail immersion reaction time was also improved by Ganoderic acid A administration. The fact that the Ganoderic acid A and morphine treatments enhanced the tail immersion reaction time even in the presence of NLX suggests that Ganoderic acid A has antinociceptive properties. Ganoderic acid A has elevated the reaction time to the thermal stimuli, according to the results of the tail immersion-induced nociception method.
The Results of Hot Plate Test were Provided as Mean ± Standard Deviation (SD) from Three Different Measurements. One-way Analysis of Variance (ANOVA) and Dunnett’s Test were Used with SPSS to Examine the Outcomes. p < 0.05 Compared to Control and p < 0.01 Compared to Ganoderic Acid A-treated Mice.
Ganoderic Acid A’s Anti-inflammatory Effects on Mice’s Paw Edema Caused by Carrageenan
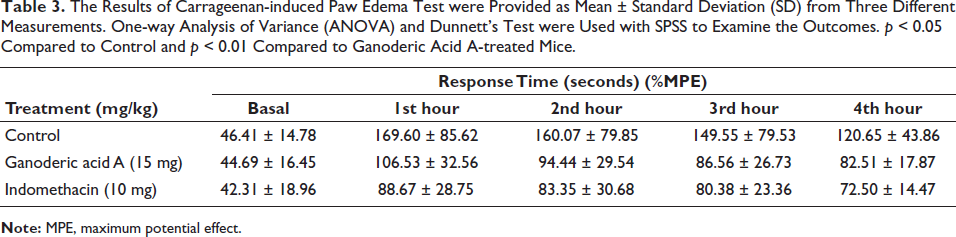
The anti-inflammatory effect of Ganoderic acid A and indomethacin (the conventional medicine) was examined using the carrageenan test. The results of an investigation into Ganoderic acid A’s inhibitory effects against paw edema that had been induced are shown in Table 3. A substantial increase in paw volume was observed in vehicle (distilled water) mice upon induction of acute inflammation. This rise started 1 hour after the carrageenan injection and peaked 4 hours later. From the first to the fourth hours following carrageenan induction, the Ganoderic acid A sample at a concentration of 15 mg/kg exhibited a decrease (p < 0.01) in paw edema in contrast to the control group. This sample was also more active than the usual medication, indomethacin. Thus, pretreatment with Ganoderic acid A and indomethacin dramatically (p < 0.01) decreased edema in the carrageenan-injected paw. Ganoderic acid A demonstrated excellent anti-inflammatory action against carrageenan-induced inflammation.
The Results of Carrageenan-induced Paw Edema Test were Provided as Mean ± Standard Deviation (SD) from Three Different Measurements. One-way Analysis of Variance (ANOVA) and Dunnett’s Test were Used with SPSS to Examine the Outcomes. p < 0.05 Compared to Control and p < 0.01 Compared to Ganoderic Acid A-treated Mice.
Ganoderic Acid A’s Antinociceptive Impact on Mice’s Acetic Acid-induced Nociception
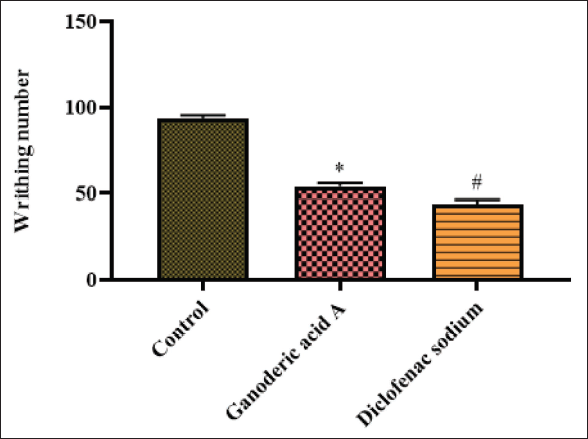
When given Ganoderic acid A and contrasted with the control group, the animals induced with acetic acid showed writhing numbers, as seen in Figure 1. Both Ganoderic acid A and diclofenac sodium significantly (p < 0.01) reduced acetic acid-induced writhing in mice when compared with control. Ganoderic acid A decreased (p < 0.01) acetic acid-triggered nociception in experimental mice.
The Effect of Ganoderic Acid A on the Acetic Acid-induced Nociception in Mice is Represented as Mean ± Standard Deviation (SD) Obtained from Three Separate Measurements. One-way Analysis of Variance (ANOVA) and Dunnett’s Test were Used with SPSS to Examine the Outcomes. p < 0.05 Compared to Control and p < 0.01 Compared to Ganoderic Acid A-treated Mice.
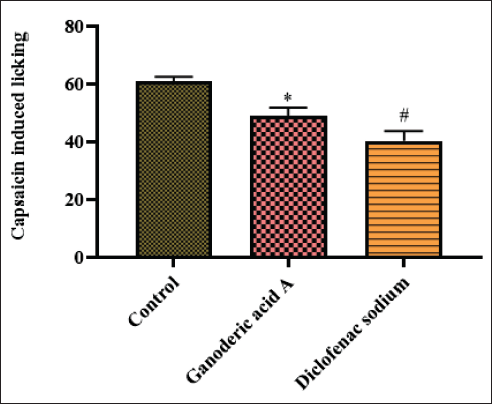
Antinociceptive Activity of Ganoderic Acid A on the Capsaicin-induced Nociception in Mice
The capsaicin challenge dramatically boosted the mice’s licking behavior, as seen in Figure 2. Ganoderic acid A administration, in contrast to the control, significantly (p < 0.01) decreased the mice’s capsaicin-induced licking behavior. Like Ganoderic acid A therapy, diclofenac sodium medication also decreased the mice’s licking behavior when exposed to capsaicin. This suggests that Ganoderic acid A demonstrated significant (p < 0.01) antinociceptive action against capsaicin-induced pain in the experimental animals.
The Effect of Ganoderic Acid A on the Capsaicin-induced Nociception in Mice is Represented as Mean ± Standard Deviation (SD) Obtained from Three Separate Measurements. One-way Analysis of Variance (ANOVA) and Dunnett’s Test were Used with SPSS to Examine the Outcomes. p < 0.05 Compared to Control and p < 0.01 Compared to Ganoderic Acid A-treated Mice.
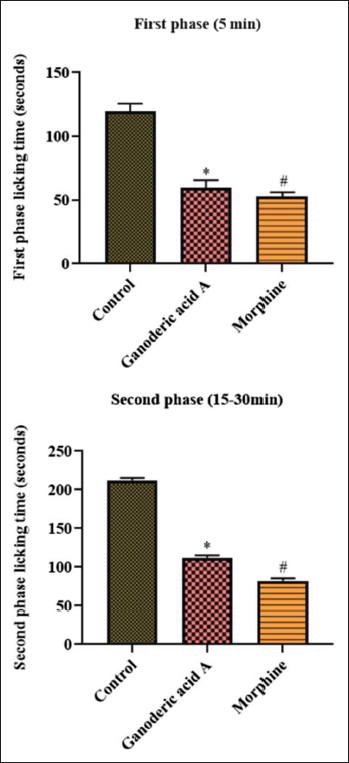
Antinociceptive Activity of Ganoderic Acid A on the Formalin-induced Nociception in Mice
Figure 3 illustrates how Ganoderic acid A affects mice’s formalin-induced biphasic nociception. Animals given formalin alone showed an increase in licking, indicating nociception. In the negative control group, paw licking was approximately greater than 100 seconds (0–5 minutes) in the early phase, and roughly 200 seconds (15–30 minutes) in the late phase. Ganoderic acid A reduced licking than the control in both the first and second phases. The decrease in licking is consistent with the antinociceptive qualities of Ganoderic acid A. Both phases’ formalin-provoked licking rates were likewise reduced by the morphine treatment.
The Effect of Ganoderic Acid A on the Formalin-induced Nociception in Mice is Represented as Mean ± Standard Deviation (SD) Obtained from Three Separate Measurements. One-way Analysis of Variance (ANOVA) and Dunnett’s Test were Used with SPSS to Examine the Outcomes. p < 0.05 Compared to Control and p < 0.01 Compared to Ganoderic Acid A-treated Mice.
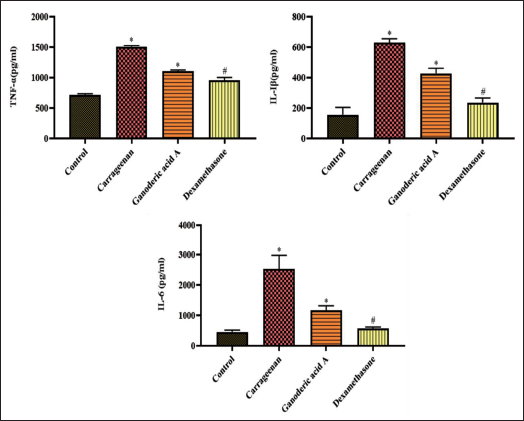
Ganoderic Acid A’s Anti-inflammatory Efficacy Against Pro-inflammatory Markers in a Carrageenan-induced Air Pouch Animal Model
In contrast to the control group, the carrageenan-provoked air pouch mice exhibited elevated TNF-α, IL-1beta, and IL-6, as seen in Figure 4. In contrast, the Ganoderic acid A therapy significantly reduced these elevations. Ganoderic acid A treatment significantly (p < 0.01) reduced TNF-α, IL-1beta, and IL-6 levels in the air pouches of mice challenged with carrageenan. TNF-α, IL-1beta, and IL-6 levels were all reduced by the dexamethasone treatment, in line with the administration of Ganoderic acid A, which demonstrated its remarkable anti-inflammatory properties.
The Effect of Ganoderic Acid A on the Pro-inflammatory Cytokines in the Carrageenan-induced Air Pouch Mice Model is Represented as Mean ± Standard Deviation (SD) Obtained from Three Separate Measurements. One-way Analysis of Variance (ANOVA) and Dunnett’s Test were Used with SPSS to Examine the Outcomes. p < 0.05 Compared to Control and p < 0.01 Compared to Ganoderic Acid A-treated Mice.
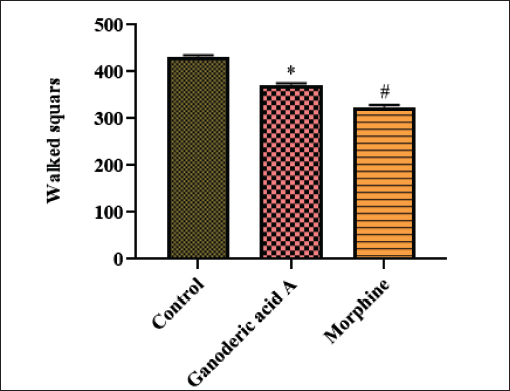
Impact of Ganoderic Acid A on the Alterations in Mice Behavior Evaluated by an OFT
Using an OFT, the sedative effect of Ganoderic acid A was assessed; the outcomes are revealed in Figure 5. Animals given Ganoderic acid A supplementation showed comparatively (p < 0.01) fewer walking squares than the control group, suggesting some mild sedation. Morphine administration resulted in mildly decreased motility when compared with control. Overall, the mice’s movement was only slightly reduced.
The Effect of Ganoderic Acid A on the Behavioral Changes in the Mice Assessed by Open-field Test is Represented as Mean ± Standard Deviation (SD) Obtained from Three Separate Measurements. One-way Analysis of Variance (ANOVA) and Dunnett’s Test were Used with SPSS to Examine the Outcomes. p < 0.05 Compared to Control and p < 0.01 Compared to Ganoderic Acid A-treated Mice.
Discussion
The present study was conducted to investigate the pain-relieving and anti-inflammatory effects of Ganoderic acid A using several animal models of pain and inflammation. Prior research has demonstrated the extensive anti-inflammatory effects of Ganoderic acid A (Gao et al., 2021). The medicinal antinociceptive properties of this substance have not been much validated by science. Therefore, the current study focused on exploring this further. Nociceptor neurons are specialized primary afferent neurons that identify pain-inducing events. These cells react to a variety of chemical and physical stimuli, such as pressure, heat, and cold, as well as irritants and inflammatory mediators, but only at levels sufficient to cause damage to surrounding tissue. The compounds that give nociceptors their unique characteristics are poorly understood (Caterina et al., 2000). Pain can be categorized as neuropathic, inflammatory, or physiological and is produced in multiple ways. Ignoring discomfort can have a lot of long-term effects. The inflammatory cell activation and the release of pro-inflammatory markers have an impact on nociceptive responses (Gao et al., 2021). The majority of research on pain is conducted through animal-based experimental tests, which can help in both medication development and understanding the physiopathology of the condition (López-Cano et al., 2017).
The pain-relieving effect of Ganoderic acid A was examined using tail immersion, hot plate, capsaicin, acetic acid, and formalin-provoked methods. The anti-inflammatory properties of Ganoderic acid A were determined by inducing inflammation with carrageenan and measuring the levels of pro-inflammatory regulators in the air pouch model. These experiments involved applying mechanical, thermal, or electrical stimuli. Additionally, the animals’ overall behavior during exposure to these stimuli was observed using an OFT. Our findings from the hot plate and tail immersion tests indicated that Ganoderic acid A lowered the delay time. In the tests using formalin, acetic acid, and capsaicin, it also reduces the sense of pain. Additionally, it decreased the rats’ paw thickness in the carrageenan test, demonstrating an anti-inflammatory action. Suppression of pro-inflammatory cytokines in the carrageenan air pouch paradigm was another observation. Additionally, it demonstrated little effect on behavioral consent. According to the current study, Ganoderic acid A has previously been shown to have anti-inflammatory and antinociceptive properties at various doses (Gao et al., 2021). However, the current study took the highest concentration and provided supporting data to enhance the formulation of Ganoderic acid A as analgesics. In comparison to the control group, Ganoderic acid A (15 mg) and morphine (5 mg/kg) had a longer delay time in the hot plate evaluation. The hot plate approach is thought to be suitable for drugs that have a central effect. The hot plate test, which evaluates the complex sensitivity to an acute, noninflammatory nociceptive stimulus, is one of the paradigms frequently utilized to examine central nociceptive activity. Any medication that increases the delay time on this test must primarily affect the CNS, according to a well-accepted procedure (Tadege et al., 2023). Therefore, Ganoderic acid A needs to have a primary function.
The tail immersion-induced nociception paradigm is a frequently employed and highly valuable technique for characterizing the antinociceptive properties of new pharmaceuticals and plant extracts. Opiod-like and marginal painkillers can be distinguished from one another using the tail immersion-induced pain paradigm. The tail immersion assay incorporates the spinal and supraspinal systems based on heat activation intensity. A correlation exists between the tail immersion assay’s latency time prolongation and the central analgesic effect of controlled drugs (Gao et al., 2021). When compared to the control in the earlier investigation, the dieckol therapy significantly increased the delay time (Li et al., 2021). A further study discovered that mice given Ganoderic acid A had increased latency times in the tail immersion, supporting the study data and demonstrating the antinociceptive effects of Ganoderic acid A (Gao et al., 2021).
Using an animal model of carrageenan-induced paw edema, the study verified Ganoderic acid A’s anti-inflammatory properties. When compared to mice that were stimulated with carrageenan, animals treated with Ganoderic acid A showed a notable decrease in paw volume, suggesting that Ganoderic acid A had efficient anti-inflammatory properties. When carrageenan is administered into the experimental animals’ rear feet, it can increase the inflammatory response acutely by causing paw edema. Prior research demonstrated that dieckol therapy dramatically decreased and prevented inflammation in experimental animals caused by carrageenan (Li et al., 2021). It is unclear exactly how carrageenan triggers inflammation, although it has been revealed to lower the amount of epithelial glycoproteins in the paw and to prevent macrophages and lymphocytes from interacting (Tadege et al., 2023).
One common pain model used to examine the antinociceptive properties of new painkillers and plant extracts is the acetic acid-induced writhing of the abdomen (Li et al., 2021). Assessing the effect of peripheral analgesics on visceral and inflammatory nociception is regarded as a sensitive technique (Gao et al., 2021). In the current investigation, Ganoderic acid A administration significantly reduced the number of writhing mice in the experimental group as compared to the control group. Innovative medications may have this form of antinociceptive impact because they directly inhibit the receptors that mediate pain, which has an antinociceptive effect (Li et al., 2021). This theory states that pain associated with inflammation is produced by capillary permeability, which is brought on by the activation of the nociceptive fibers in the nerve terminal. This decreases the nociceptive threshold. Acetic acid raised the release of prostaglandin E2 (PGE2) and prostaglandin F2 (PGF2) at peritoneal receptors, which may be responsible for pain (Tadege et al., 2023). Therefore, these nociceptive targets may be interfering with Ganoderic acid A’s antinociceptive action. In a prior investigation, we found that Ganoderic acid A supplementation decreased the occurrences of abdominal writhing in mice given acetic acid, indicating that it has an antinociceptive effect and corroborated the current findings (Gao et al., 2021).
Capsaicin is a well-known agonist of pain receptors that has the ability to activate nociceptive fibers. These functions are also controlled by the release of numerous neurotransmitters, which also play a role in the start of nociception (Gao et al., 2021). The spicy component of red peppers, capsaicin, is known to excite the vanilloid receptor (transient receptor potential cation channel V1 or TRPV1) present on polymodal C-fibers and in other tissues when applied topically or injected into the skin of people or experimental animals. A complicated series of processes, including receptor desensitization, neuronal toxicity, and the release of pro-inflammatory mediators, are triggered by this stimulation. A burning sensation is produced when capsaicin excites sensory neurons. This is followed by a refractory state, which functions as an analgesic against nociceptive stimulation (Caterina et al., 2000). Our findings unequivocally showed that Ganoderic acid A supplementation, when combined with capsaicin, significantly decreased the number of mouse-licking events. Thus, it was evident that Ganoderic acid A might lessen the nociception that mice experienced when exposed to capsaicin. The formalin test is widely used in pharmaceutical research as a reliable and sensitive way to evaluate pain (Hajhashemi et al., 2024). The formalin-provoked nociceptive experiment was used to extract Ganoderic acid A’s peripheral and central antinociceptive effects. Mice that receive subcutaneous formalin injection are susceptible to a biphasic nociceptive response, which typically consists of a first phase and a second phase. The way formalin affects sensory fibers results in neurogenic pain, which is directly correlated with the first phase of nociception. Bradykinin, prostaglandins, and serotonin are among the nociceptive mediators released during the inflammatory phase, which is often referred to as the second phase of nociception. A mouse experiences pain when its hind paw is injected with formalin, which activates the pain C fibers in the paw (Hajhashemi et al., 2024). Paw-licking time serves as a proxy for pain intensity in this test. In the current investigation, the administration of Ganoderic acid A significantly reduced the formalin-stimulated nociception in both the first and second phases. The formalin-provoked nociceptive assay results showed that Ganoderic acid A significantly decreased the incidence of mouse licking in both stages, demonstrating Ganoderic acid A’s antinociceptive properties.
The subcutaneous injection of carrageenan into the animals may promote the production of pro-inflammatory mediators that may trigger an inflammatory signal (Li et al., 2021). This idea explains why carrageenan is found to promote inflammation in the majority of animal studies (Tadege et al., 2023). The TNF-α, IL-1beta, and IL-6 that may be produced when carrageenan is injected into experimental animals have the potential to cause harmful inflammation (Li et al., 2021). In our investigation, Ganoderic acid A treatment on carrageenan-induced air pouch in mice significantly reduced TNF-α, IL-6, and IL-1beta levels. Effective pro-inflammatory regulators including TNF-α, IL-1beta, and IL-6 are secreted by spinal glial cells, and these cytokines have the capacity to activate pain receptors (Li et al., 2021).
Lastly, behavioral testing is a crucial component of cutting-edge drug development studies. Nonetheless, it is critical to remember that unique circumstances may have an impact on an animal’s behavior (Belovicova et al., 2017). It is well accepted that in lab animals that resemble humans, pain can cause affective-motivational problems. Nonetheless, other research indicates that certain chronic pain conditions might not affect the psychology of rodents (Yeung et al., 2021). The OFT method can be used to assess the antinociceptive medications or extracts. This approach proved beneficial in examining the potential nonspecific disruptions caused by new painkillers on the movement of the test subjects (Li et al., 2021). In the current investigation, the addition of Ganoderic acid A showed very slight alterations in the experimental animals’ locomotion, evidencing the accuracy of the data that is already accessible. Therefore, the information gathered from this study offers a foundation for additional investigation into mechanisms of action, which should help in the future creation of innovative, enhanced therapeutic formulations made with Ganoderic acid A to address inflammation and nociception.
Conclusion
In mice, Ganoderic acid A successfully decreased the nociception elicited by hot plates, tail immersion, acetic acid, and capsaicin. Furthermore, the findings of the current investigation revealed that Ganoderic acid A has promising anti-inflammatory efficacy in animal models. Ganoderic acid A therapy decreased the various levels of edema caused by carrageenan and was found to be significantly more effective. It decreases pro-inflammatory cytokines, contributing to its outstanding anti-inflammatory properties. Our aggregate research findings clearly indicate Ganoderic acid A’s antinociceptive and anti-inflammatory properties, providing a theoretical foundation for future pharmacological research. The present study still has some limitations, like it failed to unveil the exact molecular mechanisms underlying the antinociceptive and anti-inflammatory action of the Ganoderic acid A, which needs to be further clarified in the future. Furthermore, additional clinical trials are required to establish its safety and efficacy so that it can be quickly deployed for human usage in the future.
Summary
Overall, the research demonstrated Ganoderic acid A’s antinociceptive and anti-inflammatory capabilities in several kinds of thermally- and chemically-produced pain and inflammatory models.
Footnotes
Abbreviations
ANOVA: Analysis of variance; CNS: Central nervous system; IL: Interleukin; MPE: Maximum potential effect; NLX: Naloxone; NSAID: Nonsteroidal anti-inflammatory drug; OFT: Open-field test, PGE2: Prostaglandin E2: PGF2: Prostaglandin F2; SD: Standard deviation; TNF: Tumor necrosis factor; TRPV1: Transient receptor potential cation channel V1.
Acknowledgments
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The applicable national regulations and institutional policies for animal use and care were followed, and mice were acquired from the Approved Animal Center with the appropriate ethical approval for animal experimentation study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
