Abstract
Background and Purpose
In the present work, chitosan is furnished with tin dioxide (CsSnO2) nanoparticles (NPs) synthesized using the green route using Moringa oleifera (M. oleifera) flower extract.
Materials and Methods
These preparation methods are low-cost, easily reproducible, nontoxic, and eco-friendly. X-ray diffraction (XRD), dynamic light scattering (DLS), field emission scanning electron microscope (FESEM), energy dispersive X-ray, Fourier-transform infrared spectroscopy (FT-IR), and photoluminescence (PL) analysis were used to learn more about the CsSnO2 NPs. The antibacterial property of CsSnO2 NPs was assessed by the disc diffusion method against various pathogens. The MTT assay was done to assess the cytotoxicity of CsSnO2 NPs in MCF-7 cells. The expressions of the apoptotic proteins were examined using assay kits.
Results
XRD results demonstrated that the formulated CsSnO2 NPs exhibit a tetragonal structure. From the FT-IR spectra, the Sn–O stretching peak was observed at 579 cm–1. A FESEM image shows that the CsSnO2 NPs were formed into a spherical structure. Surface defects, such as tin and oxygen vacancies, are visible in the PL spectra, and the average particle size of CsSnO2 NPs is 32 nm. Thus, surface defects are essential parameters for biocidal properties. The antimicrobial property of CsSnO2 NPs and amoxicillin was investigated against bacteria and human fungal pathogens. CsSnO2 NPs exhibit similar antimicrobial properties as compared to amoxicillin. Also, the dosage of CsSnO2 NPs elevated their antimicrobial activity. The anticancer property of the green-synthesized CsSnO2 NPs against MCF-7 cells was further analyzed to study their ability to cytotoxicity and apoptosis induction, like caspase-3, -8, and -9 activation.
Conclusion
The study’s outcome revealed that the green synthesized CsSnO2 NPs are an outstanding candidate for cancer therapy.
Introduction
Cancer is one of the important reasons for mortality globally. Moreover, its burden has increased dramatically for several reasons, such as aging, a growing population, and accelerated socio-economic development (Cao et al., 2021). Breast cancer is a prominent global health issue that mostly affects women and is characterized by a high occurrence rate and substantial death rate. The reported number of recorded cases in 2020 was 2.3 million, resulting in 685,000 deaths worldwide (Wilkinson & Gathani, 2022). Furthermore, breast cancer has ranked second among all cancer types, accounting for approximately 12% of the overall cases. Nonetheless, the management of breast cancer entails various challenges, such as evaluating risk factors, predicting diagnostics, inhibiting metastasis, selecting proper therapy, and ensuring cost-effective methods (Mazurakova et al., 2022). According to the American Society for Infectious Diseases, multidrug-resistant (MDR) microbes pose a grave threat to the human population (Van Duin & Paterson, 2016). Therefore, developing a therapeutic with anticancer and antibacterial properties is crucial. Consequently, researchers have paid much attention to nanotechnology because it can kill bacteria and fight cancer.
Tin oxide (SnO2) is an essential n-type metal oxide semiconductor with a broadband gap (Eg = 3.6 eV) and has antimicrobial activity against several microbes like viruses, fungi, and bacteria, and antioxidant and photocatalytic properties (Dheyab et al., 2022; Patil et al., 2012). Khanom et al. (2018) examined the antibiotic property of SnO2 nanoparticles (NPs) in a concentration-dependent manner against numerous pathogens. Synthesized Co-SnO2 NPs showed that their large surface area and their size are reasons for increased antibacterial activity (Qamar et al., 2017). According to Ahamed et al.’s (2018) research, SnO2 NPs significantly increase apoptosis in breast cancer cells by inducing oxidative stress. They studied the anticancer properties of SnO2 NPs against human cancer cells like colorectal (HCT116), lung (A549), and hepatocellular (HepG2) cancer cells. The requirement for environmental sustainability has led to a great deal of focus on developing eco-friendly manufacturing of new, nontoxic, biodegradable materials used to control hospital-mediated cross-contamination by pathogens. As a result, nanocomposites with nanometer size scale, based on biodegradable polymers and organic or inorganic fillers, have developed as a developing area in a variety of medical usages (Roopan et al., 2015; Tammina et al., 2017).
Multimetallic nanocomposites synthesized using different shapes and sizes are more advantageous as they overtake the role of simple metallic NPs concerning function, structural stability, and specificity. The integrated NPs exhibit individual or combined beneficial properties, and incorporating biopolymers such as chitosan adds valuable properties to these synthetic NPs. Various types of NPs synthesized using chitosan have been used for targeting tumor cells in drug delivery. At present, titanium dioxide (TiO2) is mainly being utilized together with natural polymers like chitosan to synthesize nanocomposites with numerous beneficial properties (Basavegowda et al., 2017; Dobrucka et al., 2021; Wrońska et al., 2021).
Chitosan is commonly utilized in various fields to encapsulate active compounds, enzyme immobilization, and as a medication delivery vehicle. Biodegradability, biocompatibility, non-toxicity, and polycationic nature are noteworthy features of chitosan (Divya & Jisha, 2018). Chitosan can also speed up the process of cell death, revealing the interaction of negatively and positively charged chitosan molecules in the bacterial cell wall that results in the release of microorganisms that die as a result of intracellular components (Karthikeyan et al., 2022). It is also used to functionalize metal NPs, improving their biocidal efficacy. However, there has never been a report on the biocidal effects of chitosan SnO2 in green processes. This includes the fact that it can kill bacteria and fungi and, most importantly, fight cancer.
Tin oxide (SnO2) and tin oxide nanoparticles (SnO2NPs) have developed as the most significant metal oxide NPs due to their properties, including their antioxidant and antibacterial properties (Haq et al., 2021; Vidhu & Philip, 2015), biocompatibility, and low operating temperatures. SnO2 is a transparent conductor due to its stability, high optical transparency, and low electrical resistance, while SnO2 is an insulator (Kuang et al., 2018; Mientus et al., 2020). NPs of SnO2 have many applications, including photocatalysis, lithium batteries, solar panels, supercapacitors, catalysis, and gas sensors (Pandit & Ahmad, 2022; Pusawale et al., 2011; Rehman et al., 2021; Zhao et al., 2015).
Moringa oleifera is the most widely used herbal medicine for treating cancer. Its efficacy is attributed to polyphenols such as tannins, flavonoids, and terpenoids present in the plant (Wisitpongpun et al., 2020). The plant is used to treat numerous ailments conventionally and stimulate good well-being. The beneficial parts of the plant include leaves, flowers, seeds, roots, gum, and bark. It is utilized in conventional treatment treating infections and numerous illnesses. It possesses antimicrobial, antidiabetic, anti-inflammatory, anticancer, cardioprotective, neuroprotective, and antioxidant properties (Madi et al., 2016). M. oleifera can lower cholesterol, modulate the immune system, is hepatoprotective, increases wound healing, is antihypertensive, and is also a diuretic agent (Ravani et al., 2017). It also improves liver and kidney functionality (Vergara-Jimenez et al., 2017). The NPs and phytochemicals extracted from the plant naturally exhibit potent antibacterial and anticancer properties. Combining NPs with a natural polymer, such as chitosan, might help develop a treatment strategy that could produce better results (Elderdery et al., 2022).
M. oleifera extract has antimicrobial properties, including inhibiting bacterial strains and antifungal activity against fungal pathogens. It may also have antiviral properties against certain viruses (Wisitpongpun et al., 2020). Chitosan-coated tin dioxide NPs show potential antimicrobial properties against bacterial and fungal pathogens. They have potential applications in biomedicine, food packaging, and water treatment. However, safety concerns and environmental impact need further evaluation. Further research is needed to optimize the synthesis and characterize the antimicrobial activity.
In the present study, chitosan-decoded SnO2 (CsSnO2) NPs are prepared using the M. oleifera flower extract method. The resulting CsSnO2 NPs were characterized for their optical, structural, and antimicrobial activities. We also evaluated the synergistic anticancer effect of CsSnO2 NPs synthesized using M. oleifera flower extract against breast cancer (MCF-7) cells.
Materials and Methods
Materials
The chitosan (200 kDa, 87% deacetylation degree) and SnCl4 (98%) were purchased from Sigma Aldrich, USA.
Flowers Extract Preparation
The fresh M. oleifera flowers were obtained from commercial sources and 10 g of fresh M. oleifera flowers were boiled with 100 mL of double distilled water and agitated with a magnetic stirrer for 1 h at 80°C. The color of the solution turned from watery to a light white-yellowish color. After cooling to 37°C, the solution was filtered using Whatman No. 1 filter paper.
Preparation of Chitosan-encapsulated SnO2 NPs
At room temperature (RT), 0.1 M tin chloride solute and 1% acetic acid solution of chitosan were mixed with 100 mL of M. oleifera flower aqueous extract. The homogenous reaction chitosan-Sn metal ion mixture solution was heated on a hot plate at 80°C for 4 h with magnetic stirring. The final solution was cooled to 37°C and centrifuged at 8,000 rpm for 15 min and then dried at 120°C for 2 h. The resultant CsSnO2 NPs were annealed in the atmosphere for 5 hours at 800°C before being used for further characterization.
Characterization Studies
The CsSnO2 NPs were characterized using an X-ray meter (X’PERT PRO Analytical). CsSnO2 NPs diffraction peaks were noted in a 20°–80° range at a wavelength of 1.54 Å. The CsSnO2 NPs were examined using a field emission scanning electron microscope (Carl Zeiss Ultra 55 FESEM) with energy dispersive X-ray (EDX) spectrometry (model: Inca). The Nano-Plus instrument was used to measure particle size’s dynamic light scattering (DLS). A Perkin-Elmer spectrometer in the 400–4,000 cm–1 range was utilized to obtain the Fourier-transform infrared spectroscopy (FT-IR) spectra. The photoluminescence spectrum (PLS) was measured at 37°C using a luminescence spectrophotometer (Perkin-Elmer LS-5513) and a xenon lamp.
Antimicrobial Activity
The antibacterial properties of the CsSnO2 NPs were assessed using the well diffusion technique. The CsSnO2 NPs were dissolved using a 1 mL solution of 5% dimethyl sulfoxide (DMSO) as a stock solution. Pathogen species such as strains like Staphylococcus aureus (S. aureus), Streptococcus pneumoniae (S. pneumoniae), Bacillus subtilis (B.subtilis), Klebsiella pneumoniae (K. pneumoniae), Shigella dysenteriae (S. dysenteriae), Escherichia coli (E. coli) and fungal activity Candida albicans (C. albicans) were cultured on agar plates, and then the 6 mm wells were developed on the agar plate’s surface. These wells were then filled with the synthesized CsSnO2 NPs. Following the 24-h incubation, the inhibition zones around wells were evaluated, and the data were tabulated (Hameed et al., 2013, 2015).
Cell Culture
The MCF-7 and 3T3 (embryonic fibroblasts from mouse) cells were procured from ATCC, USA. The cells were kept at 37°C and cultivated using a Dulbecco’s Modified Eagle Medium (DMEM) containing 10% fetal bovine serum (FBS) and 1% antimycotic mixtures in an incubator with 5% CO2.
MTT Analysis
Cytotoxicity of CsSnO2 NPs was evaluated against MCF-7 and 3T3 cells using MTT analysis. The cells were added on 96-well plates and treated using varying nanocomposite dosages (2, 4, 16, 32, 64, 128, and 256 µg/mL) for 24 h. Later, about 20 µL of MTT was mixed and incubated for 4 h. The formazan deposits formed were mixed with DMSO (150 µL). Absorbance was taken in a spectrophotometer at 570 nm. The percentage of cell viability was noted. The results were derived from triplicate analysis. Cellular morphology was studied under the optical microscope at (20×) magnification.
Measurement of Activities of Caspase 3, 8, and 9
Control cells and IC25 and IC50 concentrations of CsSnO2 NPs prepared using M. oleifera flower extract-treated cells were kept for 24 h of incubation. The activity of caspases was determined using the manufacturer’s instructions for the assay kits. The kit contains specific substrates tagged with chromophore p-nitroanilide (pNA), which is later released during caspase activity and is studied at 405 nm (Iyappan et al., 2021).
Statistical Analysis
The GraphPad Prism software was utilized for the statistical analyses, and the values are illustrated as the mean ± SD of triplicate assays (n = 3). The values are analyzed by one-way analysis of variance (ANOVA) and Tukey’s post hoc assay.
Results
X-ray Diffraction (XRD) and DLS Analysis of the CsSnO2 NPs
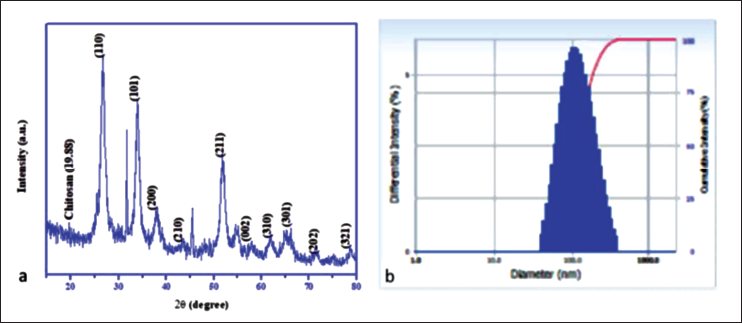
The XRD diffraction peaks of the CsSnO2 NPs are illustrated in Figure 1a. The diffraction peaks (2θ) are positioned at 26.59°, 33.81°, 37.93°, 43.94°, 51.75°, 54.52°, 58.09°, 61.93°, 64.80°, 71.56°, and 78.80° with corresponding (hkl) planes of (110), (101), (200), (210), (211), (220), (002), (310), and (301), respectively. The particle size distribution of CsSnO2 NPs in an aqueous solution was investigated using the DLS method (Figure 1b). The hydrodynamic diameter of the CsSnO2 NPs was observed at 114 nm, which may be CsSnO2 NPs surrounded by aqueous molecules.
X-ray Diffraction (XRD) Patterns of Chitosan-decoded SnO2 (CsSnO2) Nanoparticles (NPs) (a) and Dynamic Light Scattering (DLS) Spectrum of CsSnO2 NPs (b).
SEM and EDX Analysis of the CsSnO2 NPs
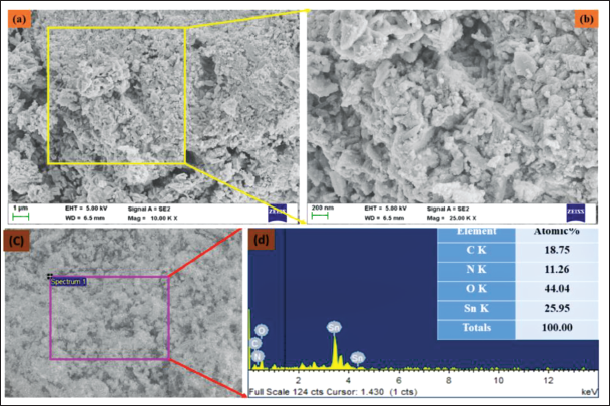
The lower- and higher-magnification field emission scanning electron microscopy (FE-SEM) images of CsSnO2 NPs are shown in Figure 2a–c. The CsSnO2 NPs are formed into a spherical structure with an aggregation formation. In addition, the CsSnO2 NPs surface resembles a sharper edge with a porous structure, which might originate from the accumulation of chitosan molecules on their surface. The EDX analysis of CsSnO2 NPs revealed an atomic percentage of 18.75% of carbon (C), 11.26% of nitrogen (N), 44.04% of oxygen (O), and 29.95% of tin (Sn) (Figure 2d).
Scanning Electron Microscopy (SEM) and Energy Dispersive X-ray (EDX) Analysis of the Synthesized Chitosan-decoded SnO2 (CsSnO2) Nanoparticles (NPs). Lower (a,c) and Higher (b) Magnification Field Emission Scanning Electron Microscopy (FE-SEM) Microphotographs and EDAX (d) Analysis of CsSnO2 NPs.
FT-IR and PL Analysis of the CsSnO2 NPs
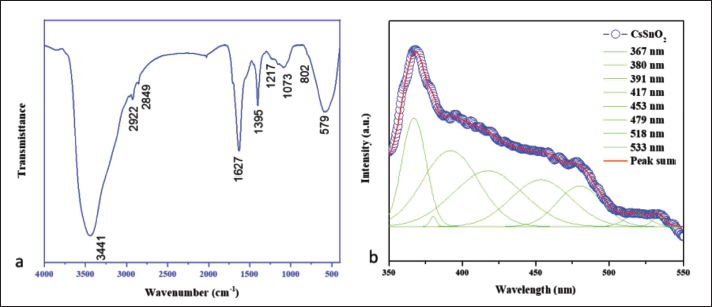
The FT-IR spectrum of the M. oleifera flower extract-derived CsSnO2 NPs is depicted in Figure 3a. The observed broad-OH and -NH stretching peaks at 3,456 cm–1 correspond to biomolecules and chitosan molecules. Small intense peaks at 2,922 and 2,849 cm–1 could be caused by asymmetric and symmetric C–H stretching. Figure 3b shows the PLS of CsSnO2 NPs at a wavelength of 325 nm. The UV emission was noted at 391 nm, 380 nm, and 367 nm, corresponding to the near band edge (NBE) emission.
(a) Fourier-transform Infrared Spectroscopy (FT-IR) Analysis of the Synthesized Chitosan-decoded SnO2 (CsSnO2) Nanoparticles (NPs). The Fourier-transform Infrared Spectroscopy (FT-IR) Spectrum of the CsSnO2 NPs Shows Several Peaks, Which Indicate the Presence of the Stretching and Bonding in the NPs and Photoluminescence (PL) Spectrum of CsSnO2 NPs (b).
Antibacterial Activity of CsSnO2 NPs
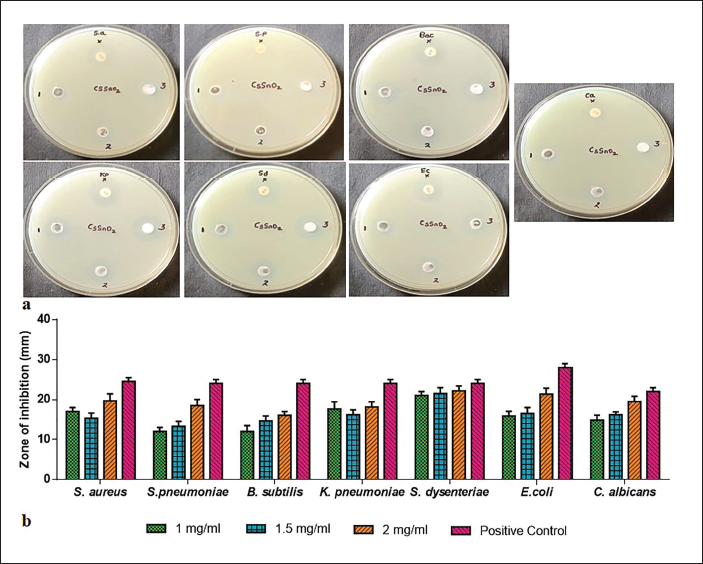
The antimicrobial effects of CsSnO2 NPs were tested against S. aureus, S. pneumoniae, B.subtilis, K. pneumoniae, S. dysenteriae, E. coli and fungal activity C. albicans using the well diffusion technique, as illustrated in Figure 4a. Antimicrobial activity can be seen in the zone of inhibition of CsSnO2 NPs and traditional antibiotics like amoxicillin (Figure 4b). To elevate the concentration of CsSnO2 NPs while augmenting the potential antimicrobial activity.
Antibacterial Activity of Chitosan-decoded SnO2 (CsSnO2) Nanoparticles (NPs) Tested Against S. aureus, S. pneumoniae, B. subtilis, K. pneumoniae, S. dysenteriae, E. coli, and fungal activity C. albicans of the Human Pathogen (a) and Graph Indicates the Zone of Inhibition (ZOI) of CsSnO2 NPs and Amoxicillin Samples (b).
In vitro Study’s
CsSnO2 NPs Decrease the Viability of the MCF-7 Cell Line
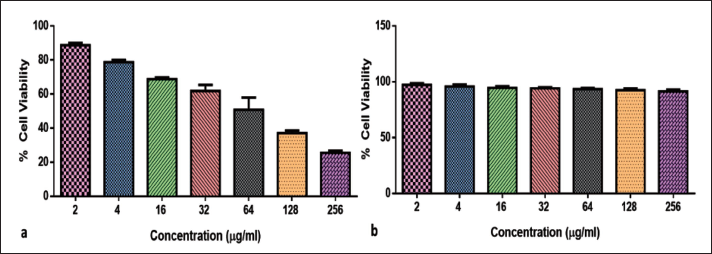
MTT analysis was performed to determine the cytotoxic efficacy of CsSnO2 NPs against MCF-7 and 3T3 cells (Figure 5a and b). CsSnO2 NPs-treated breast cancer cells displayed a higher percentage of cytotoxicity, whereas normal mouse fibroblast cells showed (Figure 5b) higher viability rates compared to MCF-7. The higher concentration displayed more cytotoxicity and thus reduced the number of viable cells. The CsSnO2 NPs induces cytotoxicity IC50 concentration at 29.8 µg/mL against MCF-7 cells
Chitosan-decoded SnO2 (CsSnO2) Nanoparticles (NPs) Inhibited the MCF-7 (a) and 3T3 (b) cells. Cells were Supplemented with Varying Concentrations of CsSnO2 NPs for 24 h. The Cells were Then Investigated Using MTT Analysis. The Data were Given as Mean ± SD of Triplicates.
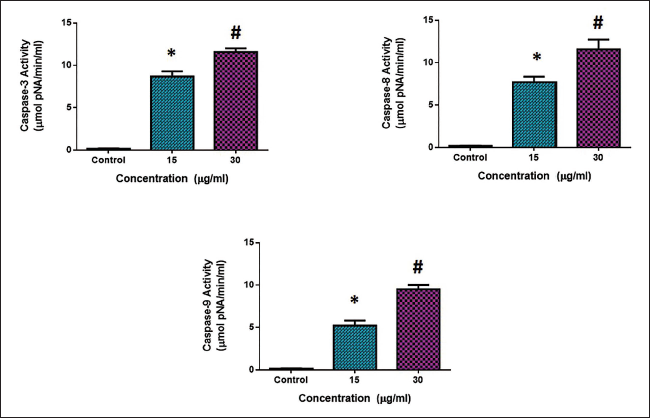
CsSnO2 NPs Increase Apoptotic Protein Expressions in the MCF-7 Cells
Apoptotic protein expressions were analyzed in both the control and CsSnO2 NPs-treated MCF-7 cells using corresponding assay kits (Figure 6). In comparison to the untreated control cells, the caspase-3, -8, and -9 expressions were decreased; however, the CsSnO2 NPs-treated expression was increased in the MCF-7 cells. Hence, it was apparent that CsSnO2 NPs induce apoptosis in the MCF-7 cells.
Discussion
In a green process, M. oleifera flower extract was used to synthesize chitosan-decorated SnO2 NPs. The chitosan molecule was dissolved in acetic acid, and tin chloride solute and M. oleifera flower extract were added. Tin cations (Sn2+) were formed from this metal salt. The Sn2+ ions are bonded to the chitosan bridge network of hydroxyl (OH) and amine (NH) groups. M. oleifera flower extract contains many phytocomponents, such as alkaloids, steroids, flavonoids, tannins, saponins, carbohydrates, and proteins (Saini et al., 2016). Plant extracts were utilized as reducing and stabilizing agents in the green formulation, and chitosan-modified engineered tin dioxide NPs are well dispersed by reduced graphene oxide (Tu et al., 2023). They include many phytochemical constituents, which play a pivotal role as reducing and capping agents of the Sn2+ ions needed to form the SnO2 NPs. CsSnO2 NPs were constructed and converted due to the Sn-chitosan nucleation solution (Gebreslassie & Gebretnsae, 2021). The XRD diffraction peaks of the CsSnO2 NPs are illustrated in Figure 1a. The diffraction peaks with corresponding (hkl) planes of (110), (101), (200), (210), (211), (220), (002), (310), and (301), respectively, matched the tetragonal structure of SnO2 NPs (JCPDS No. 88-0287) (Ragupathy et al., 2022; Wongsaprom et al., 2018). Furthermore, weak chitosan diffraction of 2θ value was observed at 19.88°. These results confirm that chitosan and SnO2 hybridization exhibit intermolecular interactions and synergistic effects. Using the Debye–Scherrer equation, the average particle size of CsSnO2 NPs is 32 nm (Holzwarth & Gibson, 2011). The particle size distribution of CsSnO2 NPs in the DLS method (Figure 1b) showed that the hydrodynamic diameter of the CsSnO2 NPs was observed at 114 nm, which may be CsSnO2 NPs surrounded by aqueous molecules.
The lower- and higher-magnification FE-SEM images of CsSnO2 NPs are shown in Figure 2a–c. Due to a biopolymer component (chitosan), the characters lacked a well-defined crystalline structure in this instance. Nevertheless, the SnO2 NPs have been decorated onto the chitosan surface matrix. The EDX analysis of CsSnO2 NPs revealed an atomic percentage of 18.75% of carbon (C), 11.26% of nitrogen (N), 44.04% of oxygen (O), and 29.95% of tin (Sn) (Figure 2d).
The FT-IR spectrum of the M. oleifera flower extract-derived CsSnO2 NPs are the observed broad-OH and -NH stretching peaks at 3,456 cm–1, corresponding to biomolecules and chitosan molecules (Mishra et al., 2015). Small intense peaks at 2,922 and 2,849 cm–1 could be caused by asymmetric and symmetric C–H stretching (Figure 3a). The antioxidants’ –C=C and –C–O stretch vibrations in M. oleifera flowers are assigned to the bands at 1,397 and 1,653 cm–1. When the reduction occurs on the surface of the particles, these reducing agents can donate electron pairs to the metals (Sn). Because polyphenols have a negative charge on the surface of the reduced metal, they act as capping agents. The typical chitosan peaks at 1,217, 1,073, and 802 cm–1 denote C–O–C stretching in the glucose circle, C–O stretching, and C–H in-plane bending. A metal and oxide (M–O) absorption peak at 579 cm–1 wavenumber position was also noted and attributed to Sn–O stretching (Ma et al., 2020; Manohari et al., 2022; Talouki & Ghorbani, 2017).
Figure 3b shows that UV emission was noted at 391, 380, and 367 nm, corresponding to the NBE emission. The violet emission peak occurs at 417 nm and is attributable to the electron transition regulated by defects in the band gap. At 453 and 479 nm, two blue emissions correlate to deep-level emissions, like oxygen vacancies and Sn interstitials. Two green emissions (518 and 533 nm) were also observed, accredited to the singly ionized oxygen vacancy. The oxygen vacancies and Sn interstitial defects on the CsSnO2 NPs’ surface are significant to their ability to kill living things.
To elevate the concentration of CsSnO2 NPs while augmenting the potential antimicrobial activity (Figure 4a and b), CsSnO2 NPs biocidal properties can be attributed to one of the following mechanisms: The interaction of chitosan with metal ions (Sn4+) results in increased chitosan adsorption with a positive charge density onto the negatively charged bacterial cell membrane, which leads to more growth inhibition (Amininezhad et al., 2015; Mostafa & Mwafy, 2020). Furthermore, binding the Cs with Sn4+ complex formation to the cell DNA blocks protein formation. The high surface area of NPs causes more oxidative stress by reactive oxygen species (ROS) generation (Zhang et al., 2018), which is accountable for their augmented chemical and biological effects. In the present work, from the FESEM results, the CsSnO2 NPs surface resembles a sharp edge with a porous structure, which might originate from the accumulation of chitosan molecules on their surface. CsSnO2 NPs with irregular surfaces and rough edges were discovered to adhere to the bacteria and trigger cell membrane damage. As a result, microbes are dead.
MTT results proved that the MCF-7 cell growth was markedly declined by treatment with the green synthesized CsSnO2 NPs (Figure 5). This might be because of the upstream result of cytotoxicity initiated by increased ROS production (Zhang et al., 2018). Hence, the results substantiate that the CsSnO2 NPs have a potent cytotoxicity, which correlates with the earlier findings (De Marzi et al., 3013). The tumor cell viability was decreased to 50% when exposed to 37.33 µg/mL concentration of green synthesized CsSnO2 NPs and the cultures were subjected to further analysis.
Apoptosis can be induced in cancer cells by triggering apoptotic signaling via the death receptor-mediated (extrinsic) and/or the mitochondria-mediated (intrinsic) pathways (Park et al., 2014). The activation of the intrinsic pathway occurs via the involvement of Bcl2 family proteins, including antiapoptotic proteins like Bcl-2 and Bcl-XL, as well as proapoptotic proteins such as Bax and Bak. Caspase is an enzyme stimulated by caspase 3, which further enhances the caspase-8 and caspase-9 activations (Herr, 2018). Thus, the nanocomposites significantly augmented the activities of numerous proapoptotic proteins and activated caspase 3, 8, and 9. MCF-7 cells administered with the green synthesized CsSnO2 NPs exhibited more caspase-3, -8, and -9 activities when compared with the control (Figure 6). The study’s findings displayed that green synthesized CsSnO2 NPs prepared using M. oleifera flower extract triggered apoptosis in MCF-7 cells by caspase-dependent signaling. Treatment with the green synthesized CsSnO2 NPs prepared using M. oleifera flower extract increased the proapoptotic marker expressions.
The potential cytotoxicity of SnO2 NPs on breast cancer cells has been demonstrated using many different synthesized SnO2 NPs from various methods and sources, but the mechanism underlying this cytotoxicity has not been fully explored. It was already highlighted in earlier work that SnO2 NPs activate the mitochondrial apoptosis signaling by overexpressing Bax/Bcl-2 and downregulating PI3K/AKT/mTOR pathway (Guo et al., 2021). Based on these data, we can speculate as to how SnO2 NPs might produce their anticancer properties. The antiproliferative properties of SnO2 NPs prepared by hydrothermal technique against K562 cells were examined (Ahmadabad et al., 2021). Cell growth, migration, and oxidative stress could be blocked, and apoptosis could be induced by SnO2 NPs in oral tumor cells (Li et al., 2020).
Conclusion
The present study found that the chitosan is decorated with tin dioxide (CsSnO2) NPs exhibit antimicrobial and anticancer effects on reducing cell viability and inducing apoptosis. The results of the various characterization analyses showed the formation of metallic CsSnO2 NPs with spherical structure, narrowly distributed NPs with an average size at 114 nm, which may be CsSnO2 NPs surrounded by aqueous molecules. The antimicrobial properties of the CsSnO2 NPs and amoxicillin were investigated in human pathogens. CsSnO2 NPs exhibit similar antimicrobial properties as compared with amoxicillin. The treatment of CsSnO2 NPs significantly decreased cell viability, increased apoptotic protein expressions, and thereby induced apoptosis in the MCF-7 cells. Hence, the present findings indicate that CsSnO2 NPs hold the potential to be a highly effective candidate for breast cancer treatment. Furthermore, future research should be conducted to fully comprehend the specific therapeutic mechanisms of CsSnO2 NPs in breast cancer treatment.
Footnotes
Abbreviations
NPs: Nanoparticles; MDR: Multidrug-resistant; TiO2: Titanium dioxide; CsSnO2: Chitosan-decoded SnO2; MCF-7: Breast cancer; FESEM: Field emission scanning electron microscope; EDX: Energy dispersive X-ray; DLS: Dynamic light scattering; PLS: Photoluminescence spectrum; ZOI: Zone of inhibition.
Declaration of Competing Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deputyship for Research & Innovation, Ministry of Education in Saudi Arabia [Project number ISP23-99].
