Abstract
Background and Purpose
We investigated the effectiveness of Vaccinium arctostaphylos L. fruit extract (VAFE) in protecting hepatocytes against N-diethylnitrosamine (DEN)-related hepatocellular carcinoma (HCC) by exploring its impact on biochemical, molecular, and antioxidant pathways, as well as its role in regulating the HOTAIR/miR-124/Notch1 axis pathway.
Materials and Methods
In the present study, 50 Wistar rats in 5 groups (n = 10/group) of normal, HCC (80 mg/kg DEN), HCC rats receiving 200 and 400 mg/kg DEN + VAFE, and normal rats receiving 400 mg/kg VAFE were divided. The serum concentrations of liver function indices (bilirubin, total protein, albumin, alanine aminotransferase, aspartate aminotransferase, C-reactive protein, and alkaline phosphatase), inflammatory cytokines (interleukin (IL)-1β, IL-10, tumor necrosis factor-α, and IL-6) and oxidants parameters (catalase, superoxide dismutase, and glutathione peroxidase enzyme activity and nitric oxide levels) were measured. Liver tissue HOTAIR, miR-124, Notch1, and Jagged1 genes and proteins were measured. p53-positive cells of liver hepatocytes were evaluated by immunohistochemical technique.
Results
DEN induced significant changes in body and liver weight, serum liver enzymes, antioxidant levels, inflammatory markers, and HOTAIR/miR-124/Notch1 axis genes/proteins expression related to liver pathways. VAFE demonstrated dose-dependent effects in countering these changes, significantly improving weight, liver health, antioxidant capacity, and inflammatory responses, especially at 400 mg/kg. The histopathological evaluation showed improvements in liver tissue structure with VAFE treatment.
Conclusion
Overall, our study demonstrates the administration of VAFE can anticancer effects of DEN-induced HCC.
Introduction
Hepatocellular carcinoma (HCC) stands as the predominant form of primary liver cancer, with an annual incidence ranging from 500,000 to 1,000,000 new cases and resulting in approximately 600,000 deaths annually (Singal et al., 2023). Tumor suppressors (PTEN, p53, and CDKN2B) and oncogenes (MET, MYC, and FGF19) profoundly influence the metabolic processes, cell cycle regulation, repair mechanisms, angiogenesis, and immune responses within hepatocytes and other liver parenchymal cells, ultimately driving tumorigenesis (Garcia-Lezana et al., 2021). Additionally, pro-inflammatory cytokines play significant roles in fostering a conducive tumor microenvironment that fosters the development of liver tumors (Thylur et al., 2020).
Nitrosamines represent a class of organic carcinogenic compounds characterized by the chemical formula R2N−N=O. These substances emerge through the interaction of nitrous acid (HNO2) with secondary amines. Nitrosamines are commonly found in tobacco, vehicle exhaust, cosmetics, canned food items, and foods subjected to cooking or frying processes, such as meats, bread, and vegetables (Stanfill et al., 2023). Various nitrosamine variants, including N-nitrosoanabasine, N-nitrosodimethylamine, N-nitrosonornicotine, and N-diethylnitrosamine (DEN), 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol have been identified in food products. Alkylating-methylating nitrosamines are particularly concerning due to their ability to induce deoxyribonucleic acid (DNA) damage in normal cells. Studies have demonstrated that rats exposed to nitrosamines derived from tobacco developed tumors in various organs, including the colon, nose, lung, mouth, liver, esophagus, pancreas, and breast (Moazeni et al., 2020; Tang et al., 2020). DEN undergoes biotransformation, yielding alkylating metabolites that induce DNA damage in hepatocytes. Reactive oxygen species (ROS) and reactive nitrogen species (RNS) generated during DEN metabolism disrupt the balance between free radical scavenging and production, inhibiting endogenous antioxidant enzymes and causing damage to cellular macromolecules, including proteins and polyunsaturated fatty acids (Memon et al., 2020).
HOTAIR, a long non-coding RNA (lncRNA) associated with various cancers, including liver cancer, regulates gene expression through interactions with chromatin-modifying complexes, impacting cell epigenetics (Price et al., 2021). In the realm of liver cancer, miR-124, a microRNA, modulates cellular processes like proliferation, invasion, and metastasis, with its dysregulation linked to disease progression. The transmembrane receptor Notch1, pivotal in cell signaling for proliferation, differentiation, and apoptosis, has been implicated in liver cancer development through aberrant pathway activation. Similarly, Jagged1, a Notch receptor ligand, contributes to liver cancer by activating the Notch pathway and promoting tumor growth, metastasis, and poor patient prognosis. Ongoing research into how these molecules interact and function in liver cancer unveils complex molecular mechanisms that could potentially offer new targets for therapeutic strategies (Chen & Kong, 2022).
The fruit extract of Vaccinium arctostaphylos L. (VA) belongs to the Ericaceae family, which is distributed globally but originates from Eastern Europe, Asia, and Northern Africa. Flourishing in humid to semi-humid regions, this plant blossoms with pinkish-white flowers between July and September. Vaccinium arctostaphylos L. fruit extract (VAFE) is composed of approximately 25–35% starch, 5% mucilage, 10% saccharose, 11% pectins, and various polyphenols (such as catechin, daidzein, caffeic acid, pcoumaric acid, kaempferol, genistein, and isoquercitrin), alongside phytosterols, coumarins, asparagine, tannins, and scopoletin (Khudier et al., 2023). In contemporary medicine, VAFE and its bioactive constituents are utilized in treating cardiovascular diseases, exerting antimicrobial effects in respiratory tract infections, preventing urolithiasis, and modulating immunity and estrogenic effects (Saliani et al., 2023). Studies suggest that in the prostate cancer cell line (PC-3), VAFE inhibited proliferation and metastasis by reducing DNA hypermethylation (Gorbanzadeh & Zaefizadeh, 2017). Moreover, noteworthy is VAFE’s ability to augment the expressions of TIMP-1 and TIMP-2, thereby inhibiting MMP-2 and MMP-9, consequently restraining the survival and metastasis of AGS and MKN-45 cells (Matsushima et al., 2013). In this study, we aim to evaluate the anticancer properties of VAFE on DEN-induced HCC in Wistar rats. We will specifically investigate its impact on antioxidative, anti-inflammatory, mitochondrial apoptosis, and HOTAIR/miR-124/Notch1 signaling pathways.
Materials and Methods
Preparation of VAFE
The freshly harvested VA fruit (3,500 g) underwent drying at 35°C in a dark environment, following confirmation by a botanist. Subsequently, the leaves were pulverized using a soil grinder, yielding a powder, which was then combined with 70% ethanol (30:70 distilled water (DW)/ethanol). After incubation (35°C/72 hours), the mixture underwent filtration through a paper filter, followed by compression using a rotary evaporator. The resulting extract (200 g) was maintained at 4°C (Perrino et al., 2023).
Experimental Design
Fifty male Wistar rats (220 ± 20 g) were randomly allocated into 5 groups (n = 10 per group) as outlined in Table 1. Prior to commencing the study, a 72-hour adaptation period was provided for the rats to acclimate to the study conditions, including temperature, food, and water. The rats were housed in propylene cages kept at 20 ± 5°C, with a relative humidity of 40 ± 10%, and subjected to a 12/12 dark/light cycle. They had access to standard pellets and tap water. All procedures related to the care and euthanasia of the rats were conducted under the oversight and approval of the ethics committee at Jinan City People’s Hospital, adhering strictly to established protocols for the housing and handling of laboratory animals. The healthy control group was treated with an intraperitoneal injection of 0.5 ml phosphate-buffered saline (PBS), while the HCC group received an intraperitoneal injection of 80 mg/kg DEN dissolved in PBS (500 µl). The co-treatment groups (HCC + 200 and HCC + 400 VAFE) were orally administered DEN along with 200 and 400 mg/kg of VAFE, respectively. Furthermore, the healthy + 400 VAFE group received 400 mg/kg of VAFE orally. The LD50 technique was employed to determine the effective dose of the nontoxic treatment, along with preliminary studies and existing research on CT and VAFE. Administration of DEN and VAFE occurred daily at fixed times, 9 am and 3 pm, respectively (Bazm et al., 2018).
Animal Grouping.
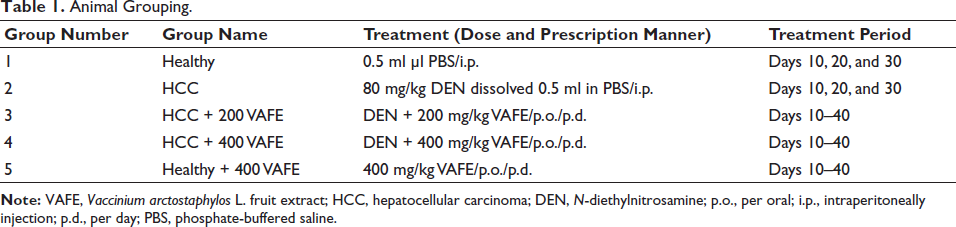
LD50 for VAFE
The LD50 of VAFE was determined using Lork’s two-step method. First, 9 animals were allocated to three groups and administered VAFE at 20, 200, and 2,000 mg/kg, respectively. Subsequently, these rats were closely observed for any signs of mortality or toxicity within 24 hours. Following this, another set of 3 rats per group received VAFE at 100, 500, and 5,000 mg/kg, and similarly monitored within the same timeframe. The LD50 was calculated utilizing Lork’s formula, incorporating the lowest dose and the highest safe dose (D safe) resulting in mortality (toxic dose, D toxic) observed during the monitoring period.
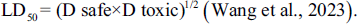
Serum levels of aspartate aminotransferase (AST), C-reactive protein (CRP), alkaline phosphatase (ALP), total protein (TP), albumin (ALB), alanine aminotransferase (ALT), and bilirubin (BIL).
Upon completion of the research, rats were euthanized via preanesthesia and anesthesia, facilitated by administering xylazine 2% (50 mg/kg) and ketamine 10% (30 mg/kg). Blood specimens were then samples from the heart, and serum was isolated using a centrifuge. Subsequently, serum levels of BIL, AST, CRP, ALP, ALT, TP, and ALB were assessed by commercial enzyme-linked immunosorbent assay (ELISA) kits as instructed (Wang et al., 2023).
Glutathione Peroxidase (GPx), Catalase (CAT), and Superoxide Dismutase (SOD) Serum Activity
We utilized a sandwich-based ELISA kit specifically designed for rodents, sourced from Cusabio company in China, to quantify the serum activity of GPx (Cat. No.: CSB-E12146r), CAT (Cat. No.: CSB-E13439r), and SOD (Cat. No.: CSB-EL022397RA), as instructed (Wang et al., 2023).
Serum Levels of Nitric Oxide (NO)
The Griess colorimetric approach evaluated serum NO levels, which serve as crucial indicators of lipid peroxidation (LPO) and oxidative stress (OS). In summary, 500 µl of serum specimens were mixed with 6 mg of zinc oxide, thoroughly combined, and then centrifuged at 10,000 g for 15 minutes. The resulting supernatant was added to the Griess solution (500 µl). Following incubation for 60 minutes at 37°C, the mixture absorbance was measured using a Stat Fax ELISA reader (303 microwell readers, Awareness Technology company, United States) at wavelengths of 540 nm and 630 nm (Vargas-Maya et al., 2021).
Liver Tissue LPO Levels (Thiobarbituric Acid Reactive Substances (TBARS) Levels), Total Antioxidant Capacity (Ferric-reducing Antioxidant Power (FRAP) Levels), and Thiol Levels
One method utilized to assess total antioxidant capacity involves FRAP evaluation. In brief, 100 mg of liver tissue was homogenized at 4°C and then mixed with cold PBS (200 µl). From this mixture, 100 µl was transferred to 10 µl of FRAP solution. Following an incubation period of 15 minutes at 25°C and subsequent centrifugation (12,000 g/10 minutes), the supernatant absorbance was assessed at 593 nm by a Stat Fax ELISA reader (303 microwell readers, Awareness Technology company, United States).
Another method to measure LPO levels involves assessing TBARS in liver tissue. Initially, the homogenized intestinal tissue mixture (100 µl) was mixed with TBARS solution (100 µl), and then underwent incubation at 37°C for 30 minutes and centrifugation (12,000 g/5 minutes). The supernatant absorbance was then assessed at 593 nm by an ELISA reader.
To determine thiol levels in liver tissue, an important tissue antioxidant indicator, 100 µl of homogenized intestinal tissue mixture was combined with 5,5-dithiol-bis-(2-nitrobenzoic acid) (DTNB; 20 µl), followed by incubation at 37°C for 15 minutes and centrifugation (12,000 g/5 minutes). The supernatant absorbance was assessed at 412 nm by an ELISA reader (Alam et al., 2023).
Serum Concentrations of Tumor Necrosis Factor-α (TNF-α), Interleukin (IL)-1β, IL-10, and IL-6
To assess the anti-inflammatory properties of VAFE extract, levels of pro-inflammatory cytokines (IL-6 (Cat. No.: M6000B), TNF-α (Cat. No.: NBP2- DY410), and IL-1β (Cat. No.: RLB00)) and the anti-inflammatory cytokine IL-10 (Cat. No.: R1000) were quantified using sandwich-oriented rodent-specific ELISA kits obtained from Novus Biologicals (United States) as instructed (Shin et al., 2019).
HOTAIR, miR-124, Notch1, and Jagged1 Genes Expression
Liver tissue total ribonucleic acid (RNA) extraction was conducted utilizing EX6101-RNX Plus Solution (Cat. No.: EX6101-RNX; SinaClon BioScience, China). In brief, cell lysis was initiated by the addition of RNX Plus buffer, followed by the incorporation of 200 µl of chloroform. Subsequent centrifugation (13,000 g/15 minutes/4°C) facilitated the separation of phases, yielding a supernatant. This supernatant was then combined with cold isopropanol (200 µl) and left to incubate on ice for 15 minutes before undergoing another round of centrifugation at 13,000 g for 15 minutes. Following this, 1 ml of 50% ethanol was introduced to the samples, which were then subjected to centrifugation (15 minutes/13,000 g at 4°C). The resulting pellet was ultimately re-suspended in 50 µl of DW and stored at –80°C.
The Revert AidTM First Strand cDNA Synthesis Kit (Cat. No.: K1621; USA) was employed for cDNA synthesis, adhering to the manufacturer’s protocols. The sequences of HOTAIR, miR-124, Notch1, and Jagged1 genes were designed utilizing gene runner software (Hastings Software, United States). Following the design phase, the genes underwent a blast search in the NCBI database (
Primer Sequences.

The fold formula change = 2–∆∆Ct; ∆∆Ct = [(Ct sample – Ct GAPDH gene) – (Ct sample – Ct control)] (Chen et al., 2021).
Expression miR-124, HOTAIR, Notch1, and Jagged1 Proteins in Liver with Western Blotting
To assess the protein expression levels of HOTAIR, miR-124, Notch1, and Jagged1, Western blot analysis was performed. Initially, a homogenized liver tissue sample was prepared, with 100 mg of the sample combined with 50 µl of PBS and 100 µl of radioimmunoprecipitation assay buffer. Following centrifugation, 20 µl of loading buffer was added to a polyvinylidene fluoride (PVDF) membrane containing primary antibodies sourced from Abcam, UK, targeting HOTAIR (Cat. No. ab062614; 1:400), Notch1 (Cat. No. ab114178; 1:500), Jagged1 (Cat. No. ab152171; 1:500), and miR-124 (Cat. No. ab0616241; 1:500). Subsequently, the mixture was subjected to separation on an SDS polyacrylamide gel (10%) using appropriate techniques. Following a 12-hour incubation at 4°C, the membrane underwent additional incubation with horseradish peroxidase (HRP)-conjugated secondary antibody (40 minutes/37°C). Finally, the protein bands’ signals were captured and analyzed by Bio-Rad software, employing an enhanced chemiluminescence reagent (e-BLOT company/China). Further analysis was conducted using ImageJ software (Wang et al., 2020).
Immunohistochemistry (IHC) Assay
Identifying p53-positive cells in liver tissues was employed as an indicator of apoptotic differentiation within tumor cells. Liver tissues underwent PBS washing and standard tissue processing, leading to the creation of paraffin blocks. Sections measuring 5 µm were affixed to slides and subjected to an overnight incubation (in a Cole-Parmer WB-400 Digital General water bath incubator, Model No. UX-12105-90, Cole-Parmer, US) at 95°C with primary p53 antibodies (1:500; Cat. No.: GAF1355, R&D Systems, Inc., US). Following this, the slides were subjected to a 1-hour incubation at 25°C. PBS-Tween 20 (PBST) buffer 0.1% (v/v) served as the washing buffer, and any remaining antibodies were blocked using 5% bovine serum albumin. Subsequently, the slides were exposed to 3% hydrogen peroxide (H2O2) for 20 minutes at 25°C, followed by staining with 3,3′-diaminobenzidine. Hematoxylin was utilized for counterstaining on all slides. A BX61TRF optical microscope (Olympus, Japan), attached to ImageJ software, facilitated the examination of the slides at a magnification of 400×. Following counting p53-positive cells in 10 fields of view in 400× and calculating the p53-positive cells/the total cells ratio, the percentage of p53 expression was determined (Dhar et al., 2018).
Liver Histopathology
Liver tissues were subjected to fixation in 10% formalin for 72 hours and then received a gentle rinse with PBS. Subsequent to fixation, the specimens underwent dehydration in progressively increasing levels of ethanol, clarification in xylene, and embedding in paraffin wax. Sections measuring 5 µm were obtained from the paraffin blocks utilizing a microtome (Model No. SM2010RV1.2 microtomes, LEICA, Germany) followed by drying in an incubator at 37°C. Slides were prepared for hematoxylin and eosin (H&E) staining. A light microscope performed histological analysis of the slides at 400× magnification. The images were captured employing a BX61TRF calibrated light microscopic system (Olympus, Japan) and processed using ImageJ software (Chen et al., 2020).
Statistical Analysis
To compare the quantitative results among the studied groups, we performed statistical analysis by one-way analysis of variance (ANOVA) followed by the Newman–Keuls test. A significance threshold of p < 0.05 indicated statistical significance. The Kolmogorov–Smirnov test assessed normality and homogeneity of the data, with a p value above 0.05 indicating normal distribution and homogeneity. All results are expressed as mean ± standard deviation (SD). Data analysis was conducted by SPSS 16, while GraphPad Prism software 9 was utilized for graphical representation of the results.
Results
LD50 of VAFE
Following a 24-hour observation of the groups subjected to VAFE treatment, the outcomes indicated the identification of a safe dose (D safe) at 2,000 mg/kg, with the toxic dose (D toxic) established at 5,000 mg/kg of VAFE. Employing Lork’s formula, the calculated LD50 for VAFE was determined to be 3,162 mg/kg. This implies that animal studies can utilize doses lower than the LD50 of VAFE.
Body Weight (BW) and Liver Weight (LW)
Upon analyzing the outcomes related to total LW and BW, DEN in the HCC group caused a significant (p < 0.05) reduction in BW and a notable (p < 0.05) increase in LW when compared with the healthy group. In contrast, VAFE showed a dose-dependent impact when given at doses of 200 and 400 mg/kg in the HCC + 200 and 400 mg/kg VAFE groups. This caused a significant elevation in the rats’ weight (p < 0.05) and a significant reduction in LW in comparison to the HCC group (Figure 1a).
(a) Rats Body Weight (BW) and Liver Weight (LW) (g); (b) Serum Activity of Alanine Aminotransferase (ALT), Aspartate Aminotransferase (AST), and Alkaline Phosphatase (ALP) Enzymes (IU/l); and (c) Serum Levels of Albumin (ALB) (mg/dl), Total Protein (TP) (g/l), and Bilirubin (BIL) (µmol/l), C-reactive Protein (CRP) (mg/l) in Experimental Groups (Means ± SD; n = 10/group). (a) (p < 0.05) Hepatocellular Carcinoma (HCC) Versus Healthy Groups; (b) (p < 0.05) HCC + 200 and 400 Vaccinium arctostaphylos L. Fruit Extract (VAFE) Treated Versus HCC Groups.
Effects of DEN and VAFE on Serum Liver Biochemical Parameters
Following the assessment of serum liver enzyme activities (AST, ALP, and ALT), DEN notably (p < 0.05) raised the activity levels of these liver enzymes in comparison with the controls. In the groups receiving treatment (HCC + 200 and 400 mg/kg VAFE), both showed a significant reduction (p < 0.05) in the activity of all three enzymes in comparison with the HCC group (Figure 1b).
The examination of serum CRP and BIL data unveiled a notable increase (p < 0.05) in both indicators within the HCC group when contrasted with the controls. Notably, VAFE exhibited a dose-dependent impact in reducing (p < 0.05) the concentration of these serum indicators in comparison to the HCC group. The assessment of serum TP and ALB levels across the groups indicated that DEN significantly (p < 0.05) decreased the concentrations of both markers in comparison to the controls. Conversely, in the HCC + 400 VAFE treatment group, a significant increase (p < 0.05) was detected in the concentrations of both markers in comparison to the HCC group (Figure 1c).
Serum GPx, CAT, and SOD Activity Alongside Serum NO Levels
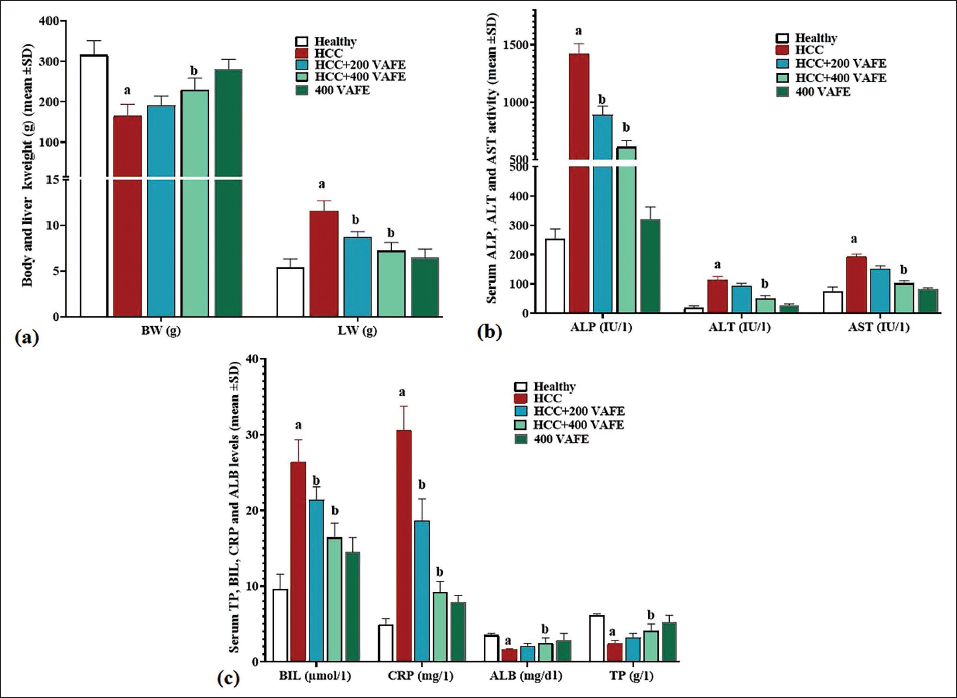
DEN, by triggering the generation of free radicals, led to a significant rise in the serum NO concentrations when compared to the control group. In contrast, VAFE exhibited a dose-dependent reduction in its concentrations in comparison with the HCC group. This decrease was statistically significant (p < 0.05) in the 400 mg/kg dosage (within the HCC + 400 VAFE group). Furthermore, DEN markedly lowered the serum activity of all three antioxidant enzymes in comparison with the controls. Conversely, VAFE boosted the serum concentrations of all three enzymes dose-dependently in comparison to the controls, with a significant increase (p < 0.05) observed at the 400 mg/kg dosage (within the HCC + 400 VAFE group) compared to the HCC group (Figure 2a).
(a) Serum Levels of Nitric Oxide (NO) (mmol/ml), Alongside the Mean Serum Activity of Superoxide Dismutase (SOD), Catalase (CAT), and Glutathione Peroxidase (GPx) (U/ml) and (b) Intestine Tissue Levels of Thiobarbituric Acid Reactive Substances (TBARS) (nmol/mg) and Ferric-reducing Antioxidant Power (FRAP) (µmol/mg) (means ± SD; n = 10/group) in Experimental Groups. a(p < 0.05) Hepatocellular Carcinoma (HCC) Versus Healthy Groups; b(p < 0.05) HCC + 200 and 400 Vaccinium arctostaphylos L. Fruit Extract (VAFE) Treated Versus HCC Groups.
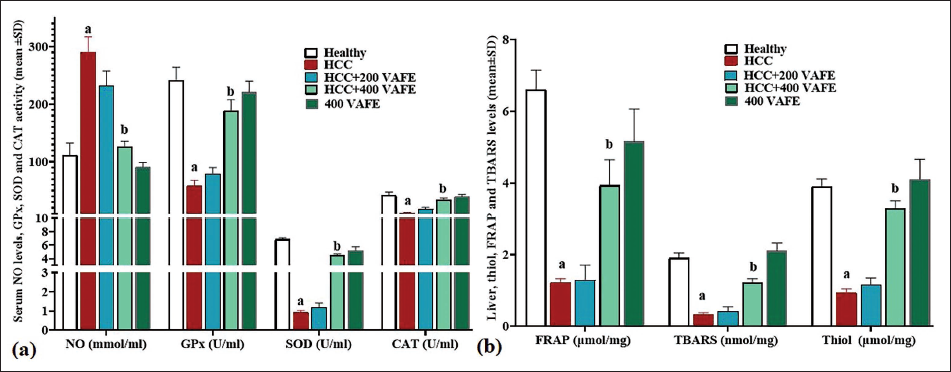
Thiol, FRAP, and TBARS Levels in Liver
Thiol, FRAP, and TBARS levels served as key markers of overall antioxidant capacity and LPO. The findings indicated that DEN notably (p < 0.05) diminished the levels of all three factors in the tissues compared to the control group. In contrast, VAFE, attributed to its rich antioxidant properties, demonstrated a dose-dependent elevation in the levels of these factors in comparison to the HCC group. This elevation was significant (p < 0.05) at the 400 mg/kg dosage (within the HCC + 400 VAFE group) in comparison with the HCC group (Figure 2b).
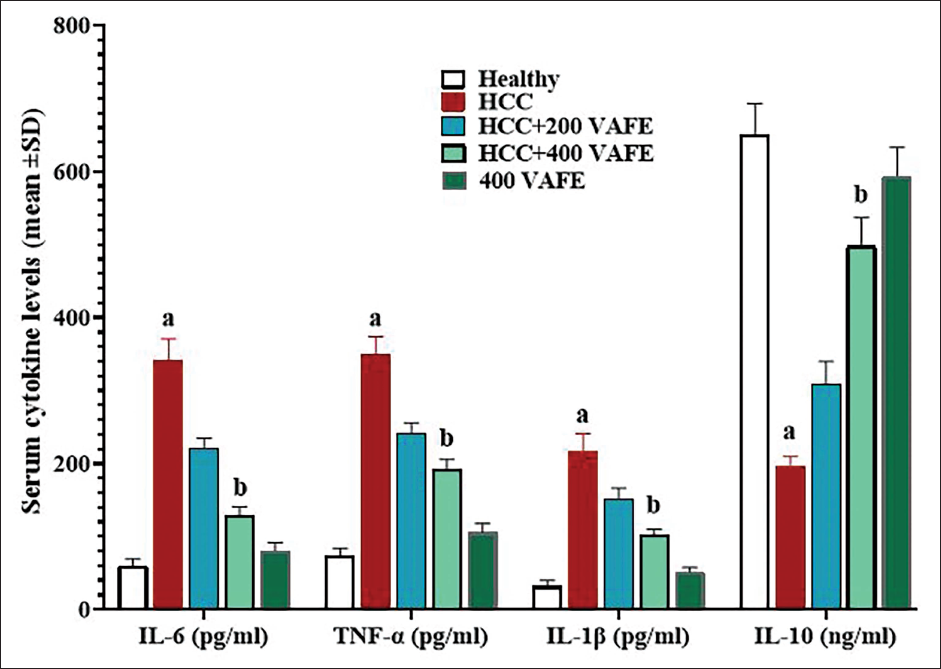
Serum Concentrations of TNF-α, IL-6, IL-10, and IL-1β
By stimulating inflammatory responses, DEN increased the concentrations of pro-inflammatory cytokines and suppressed the activity of systemic anti-inflammatory cytokines. This resulted in a significant elevation (p < 0.05) in the serum concentrations of all three pro-inflammatory cytokines (TNF-α, IL-1β, and IL-6) in comparison to the controls, while the level of IL-10 notably decreased. However, the findings of the study indicated that VAFE possesses strong anti-inflammatory properties. It progressively raised the IL-10 serum concentrations and reduced the levels of all three pro-inflammatory cytokines (IL-1β, TNF-α, and IL-6) compared to the HCC group. These changes were significant (p < 0.05) at 400 mg/kg (within the HCC + 400 VAFE group) of this herbal remedy in comparison to the controls (Figure 3).
Serum Levels of Interleukin (IL)-1β, Tumor Necrosis Factor-α (TNF-α), IL-6 (pg/ml), and IL-10 (ng/ml) (Means ± SD; n = 10/group) in Experimental Groups. a(p < 0.05) Hepatocellular Carcinoma (HCC) Versus Healthy Groups; b(p < 0.05) HCC + 200 and 400 Vaccinium arctostaphylos L. Fruit Extract (VAFE) Treated Versus HCC Groups.
Expression of Liver miR-124, HOTAIR, Notch1, and Jagged1 Genes
Analysis of gene expression related to proliferation, differentiation, and apoptosis pathways revealed that DEN treatment significantly decreased HOTAIR, Notch1, and Jagged1 expression while increasing miR-124 expression in hepatocytes compared to the controls (p < 0.05). In the HCC + 200 VAFE group, there was an elevation in Notch1, HOTAIR, and Jagged1 expression and a reduction in miR-124 expression compared with the HCC group; however, these alterations were not significant (p > 0.05). The most pronounced changes were detected in the HCC + 400 VAFE group where HOTAIR, Notch1, and Jagged1 expression significantly increased while miR-124 expression significantly reduced compared to the HCC group (p < 0.05) (Figure 4a).
(a) HOTAIR, miR-124, Notch1, and Jagged1 Genes Expression and (b) and (c) HOTAIR, miR-124, Notch1, and Jagged1 Proteins Expression in Liver (Means ± SD; n = 10/group) in Healthy (a), Hepatocellular Carcinoma (HCC) (b), HCC + 200 Vaccinium arctostaphylos L. Fruit Extract (VAFE) (c), HCC + 400 VAFE (d), and 400 VAFE (e) Groups. a(p < 0.05) HCC Versus Healthy Groups; b(p < 0.05) HCC + 200 and 400 VAFE Treated Versus HCC Groups.
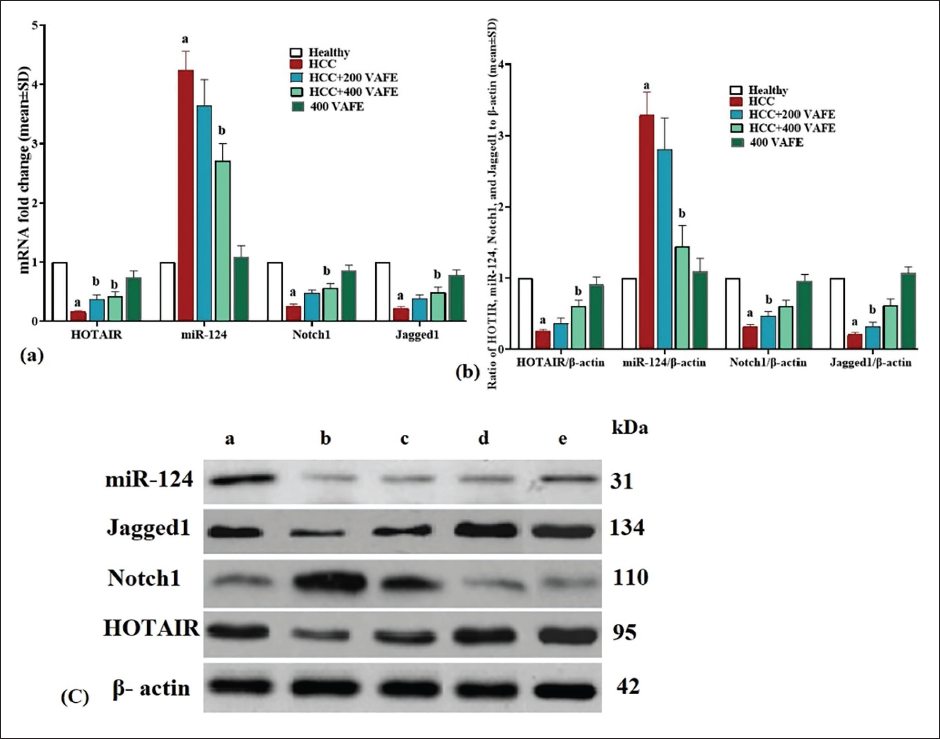
Expression of Liver HOTAIR, miR-124, Notch1, and Jagged1 Proteins
In order to assess the impact of DEN on liver pathways related to proliferation, differentiation, and apoptosis comprehensively, we investigated the protein expression levels of Notch1, miR-124, HOTAIR, and Jagged1. The findings from this analysis showed a significant reduction (p < 0.05) in the HOTAIR, Notch1, and Jagged1 expression, along with a significant elevation (p < 0.05) in the protein expression of miR-124 compared to the healthy group. In contrast, in the HCC + 200 VAFE group, there was an increase in the protein expression of HOTAIR, Notch1, and Jagged1, accompanied by a decrease in the protein expression of miR-124 in comparison to the HCC group. However, these alterations were not significant (p > 0.05). The most notable alterations were observed in the HCC + 400 VAFE group, where there was a significant elevation (p < 0.05) in the HOTAIR, Notch1, and Jagged1 expression, while a significant reduction (p < 0.05) was seen in the protein expression of miR-124 in comparison with the HCC group (Figure 4b and c).
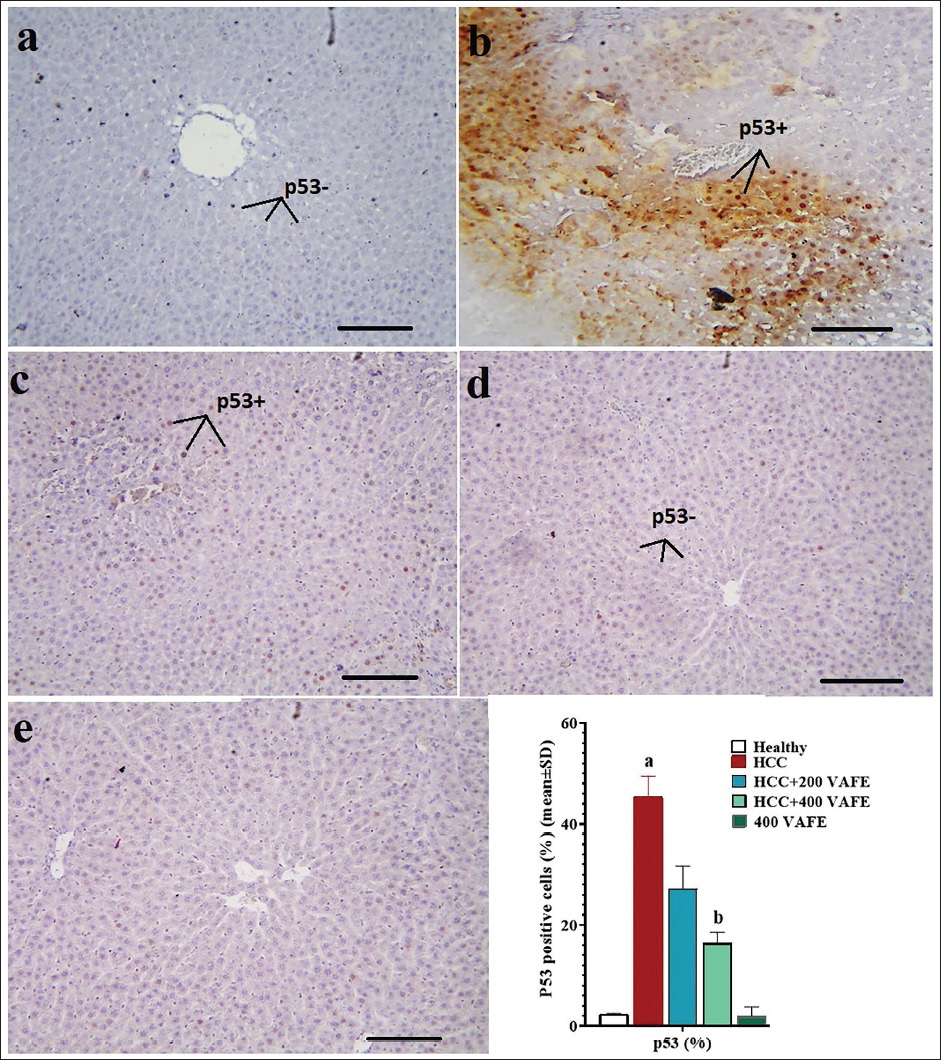
Liver p53-positive Hepatocytes
An analysis of the outcomes concerning the percentage of p53-positive cells indicated that DEN led to a notable (p < 0.05) rise in the proportion of p53-positive cells in comparison to the healthy group. Furthermore, the groups receiving solely 200 and 400 mg/kg of VAFE extract also exhibited enhancements in the apoptotic indices (decrease in p53-positive cells) when contrasted with the HCC group. Particularly, the decrease in the percentage of p53-positive cells in the HCC + 400 VAFE group was statistically significant in comparison with the HCC group (Figure 5).
p53 Positive Cells (%) in Liver Tissue by Immunohistochemistry (n = 10 rat/group, Data are Means ± SD) (Means ± SD; n = 10/group) in Healthy (a), Hepatocellular Carcinoma (HCC) (b), HCC + 200 Vaccinium arctostaphylos L. Fruit Extract (VAFE) (c), HCC + 400 VAFE (d), and 400 VAFE (×100 with Scale Bar = 200 µm). a(p < 0.05) HCC Versus Healthy Groups; b(p < 0.05) HCC + 200 and 400 VAFE Treated Versus HCC Groups. p53 Positive Cell (p53+) and p53 Negative Cell (p53–).
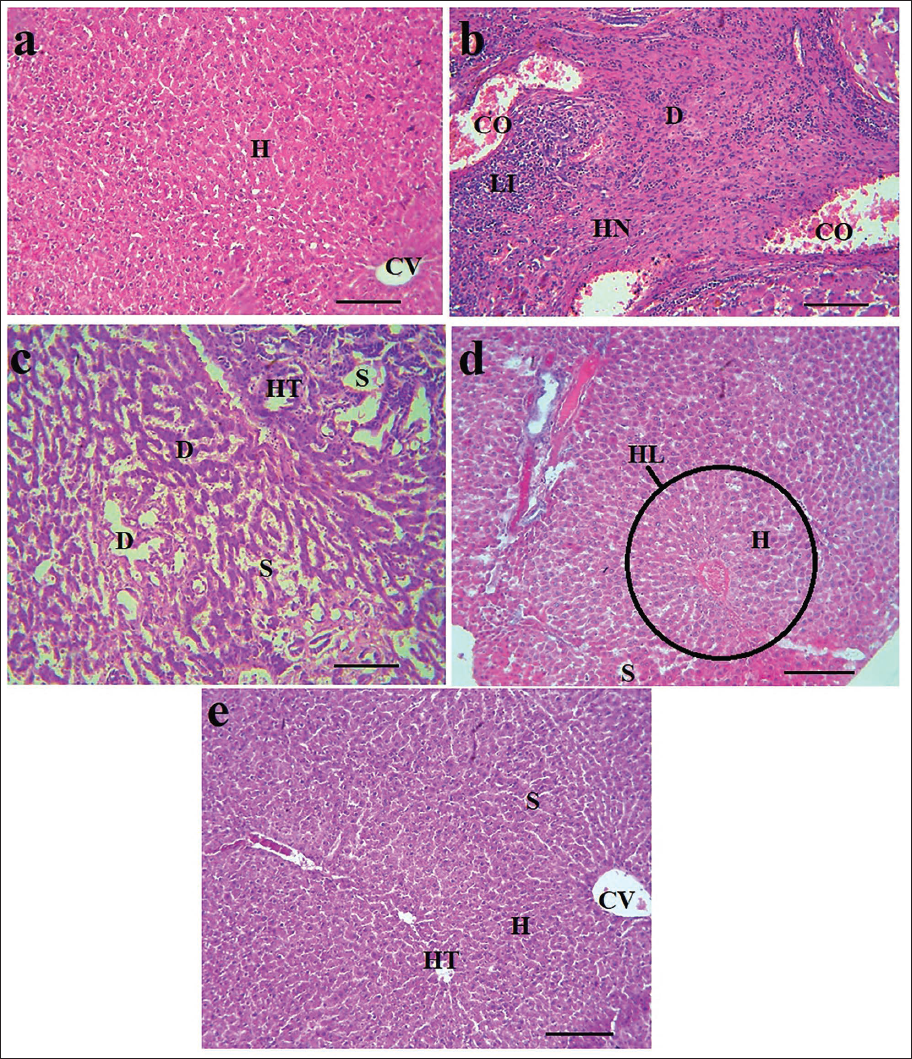
Liver Histopathological Evaluations
The histopathological analysis of the liver tissue indicated that DEN led to the development of lymphocytic infiltration (LI) with hepatic nodules (HN) and degenerated hepatocytes (D) near necrotic tissue. The typical hepatic triad (HT) and hepatic lobule (HL) were missing in this specific group, while central venule (CV) dilatation and congestion (CO) were visible. In the HCC + 200 and 400 VAFE groups, the HL formation returned to normal, and LI decreased significantly. Furthermore, HN presence was no longer observed in the liver, and the alignment of hepatocytes (H) alongside liver sinusoids (S) was visible. These changes seem to be associated with the dosage given, with the most noticeable improvement in tissue structure seen in the HCC + 400 VAFE group (Figure 6).
Histopathological Changes in Liver Tissue in Healthy (a), HCC (b), HCC + 200 VAFE (c), HCC + 400 VAFE (d), and 400 VAFE (×100 with Scale Bar = 200 µm). Histopathological Examinations of the Liver Tissue in the Studied Groups Revealed that DEN Induced the Development of Hepatic Nodules (HN) Consisting of Lymphocytic Infiltration (LI) and Degenerated Hepatocytes (D) Near the Necrotic Tissue. In this Specific Group, the Typical Hepatic Triad (HT) and Hepatic Lobule (HL) were Not Present, While Congestion (CO) and Dilatation of the Central Venule (CV) were Visible. In the HCC + 200 and 400 VAFE Groups, the Restoration of HL Formation to a Normal State and a Significant Reduction in LI were Observed. Furthermore, the Presence of HN was No Longer Identifiable in the Liver, and the Alignment of Hepatocytes (H) Alongside the Liver Sinusoids (S) was Notably Clear. These Changes Seem to be Associated with the Dosage Administered, with the Most Significant Improvement in Tissue Structure Seen in the HCC + 400 VAFE Group.
Discussion
Our findings highlight the efficacy of VAFE in preserving the normal function of hepatocytes by facilitating antioxidant, anti-inflammatory, and antiapoptotic mechanisms. Moreover, VAFE demonstrates its potential in modulating the HOTAIR/miR-124/Notch1 axis linked to DEN-induced HCC, thereby safeguarding against tumorigenesis pathways in hepatocytes.
DEN-induced HCC
DEN impacts various pathways in the proliferation, autophagy, apoptosis, and differentiation of liver parenchymal cells. Among these, the PKB/AKT pathway, crucial for hepatocyte cell proliferation and differentiation, is suppressed by DEN, as indicated by Arboatti et al. (2019). Simultaneously, DEN activates tumorigenic pathways such as Wnt/β-catenin and TGFβ-1/Smads, expediting the hepatocellular tumorigenesis process, as reported by He et al. (2023). Another carcinogenic pathway affected by DEN involves the activation of cyclin D1/MEK1, 2 signaling pathways, and the Akt/PI3K/mTOR signaling pathways in hepatocytes. DEN plays a dual role in inhibiting the proliferation of both normal and injured hepatocytes by inducing the Bcl-2/Bax/p53 mitochondrial apoptosis pathway. Additionally, DEN initiates the ROS- and RNS-related p53-associated apoptotic pathway, mediated by cytochrome P450 in hepatocytes, which stimulates the apoptotic cascade. This cascade includes the activation of caspases 9 and 8, followed by the activation of effector caspases like caspases 3 and 6, prompted by the cytochrome c release from mitochondria (Kamel & Lamsabhi, 2021). Research indicates that DEN induces hepatocyte necrosis and apoptosis through STAT3, P38, JNK, and JAK signaling pathways, along with factors like cyclooxygenase-2 (COX-2), IGF-2, cyclin D1, and mutations. This leads to the secretion of growth factors such as epidermal growth factor (EGF) and transforming growth factor-β (TGF-β), resulting in tissue neoangiogenesis and fibrosis (Shang et al., 2018). The study findings also suggest that DEN decreases antioxidant capacity (FRAP) while increasing LPO and liver protein levels (thiol and TBARS) by suppressing antioxidant enzymes (SOD, GPx, and CAT). Elevated levels of RNS and ROS are associated with changes in liver function markers (ALT, CRP, ALB, BIL, TP, AST, and ALP). In a separate investigation, Morsy et al. illustrated DEN’s suppression of GPx, SOD, LPO, CAT, and enzyme activities, leading to increased levels of serum NO and hepatic malondialdehyde (MDA), indicating the development of OS (Morsy et al., 2023). Moreover, DEN elevated serum levels of liver enzymes (AST, ALT, and ALP), as well as BIL and CRP levels, disrupting hepatocyte physiological function. CRP, a notable inflammatory marker, rises in response to liver tissue damage, serving as a critical indicator in animals. In human/animal models, CRP induces inflammatory and pro-inflammatory cytokines, particularly TNF-α and IL-6, by the p38 mitogen-activated protein kinase (MAPK) pathway, highlighting the inflammatory response phase (Zaahkouk et al., 2019).
DEN stimulates the release of pro-inflammatory (TNF-α, IL-22, and IL-6) cytokines while inhibiting anti-inflammatory (IL-4 and IL-10) cytokines, reinforcing many inflammatory pathways involving Kupffer cells, liver lymphocytes, and damaged cells (Lyngdoh et al., 2023). These cytokines enhance apoptotic and necrotic cascades in many tissues, such as the liver. The HOTAIR/miR-124/Notch1 axis, a pivotal intracellular pathway, is influenced by metabolism, regulating growth, DEN, motility, and cell proliferation of tumor cells. DEN strengthens the nuclear factor kappa B (NF-κB)/COX-2 pathway by the HOTAIR/miR-124/Notch1 and Nrf-2/HO-1 axis, impacting hepatocyte function and liver indices after tumorigenesis and nodule formation (Sahin et al., 2014). The study also reveals DEN’s involvement in inflammatory pathways (IL-6, IL-1β, and TNF-α elevation) and stimulation of the HOTAIR/miR-124/Notch1 axis.
VAFE Ameliorates DEN-induced HCC
VAFE demonstrated dose-dependent improvements in the liver cell function while maintaining their normal structure by antioxidant, anti-inflammatory, and antitumor mechanisms. VAFE effectively reduced concentrations of liver enzymes (ALP, ALT, and AST), BIL, and CRP, while increasing ALB and TP concentrations, both individually and when combined with SIL, in a synergistic and dose-dependent pattern, particularly notable in the DEN + 500 ALOF + SIL group. Additionally, VAFE extract, particularly at a dosage of 400 mg/kg (in the HCC + 400 VAFE treatment group), suppressed inflammatory pathways (as indicated by decreased concentrations of TNF-α, IL-1β, and IL-6, alongside an increase in IL-10) and OS (demonstrated by increased activity of SOD, GPx, and CAT, and decreased NO concentrations) caused by DEN.
Research indicates that VAFE, as an antioxidant and apoptotic inducer, boosts glutathione-S-transferase (GST) gene expression by mitigating DNA hypermethylation, consequently inhibiting prostate cancer cell line (PC-3) proliferation (Gorbanzadeh & Zaefizadeh, 2017). Matrix metalloproteinases (MMPs) are associated with in tumor cell metastasis and invasion by breaking down extracellular matrix (ECM) proteins such as collagen, laminin, proteoglycan, fibronectin, and elastin. Endogenous tissue inhibitor of metalloproteinase (TIMP) counteracts MMP activity, thus impeding metastasis and invasion (Holmberg et al., 2013). Notably, VAFE extract enhances TIMP-1 and 2 expressions, leading to MMP-2 and 9 inhibitions, thereby restraining survival and metastasis of gastric cancer cell lines (AGS and MKN-45) (Matsushima et al., 2013). Multidrug resistance (MDR) remains a significant challenge in chemotherapy, attributed to various mechanisms including activation of detoxifying systems, reduced drug uptake, activation of DNA repair mechanisms, and increased drug efflux regulated by adenosine triphosphate-binding cassette (ABC) transporters, notably ABCCs or multidrug resistance proteins (MRPs) (Cole, 2014; Ruan et al., 2020). VAFE demonstrates potential in overcoming MDR by enhancing the efficacy of chemotherapy drugs on gastric cancer cell lines (AGS and MKN-45), suppressing their growth and viability through downregulation of MRP-1 expression (Esmaeili et al., 2021). Comparative analysis of VAFE and Vaccinium myrtillus extracts on human HL60 and НСТ116 tumor cell lines reveals their antitumor effects, attributed to anthocyanin content, particularly delphinidin, which induces tumor cell suppression and apoptosis (Karcheva-Bahchevanska et al., 2017). In an investigation by Muceniece et al. (2019) on the protective effects of VAFE against diazinon-related hepatotoxicity, the plant extract exhibited a reduction in hepatic enzyme levels (AST, ALT, and ALP) while safeguarding liver function and structure against oxidative damage by stimulating SOD, GPx, and CAT enzymes (Muceniece et al., 2019). Akbari Bazm et al. (2019) investigated the protective effects of VAFE extract against oxymetholone-induced testicular toxicity in rats, revealing its ability to diminish tissue MDA, thiol, and NO levels while enhancing SOD and GPx activity (Akbari Bazm et al., 2020).
The HOTAIR/miR-124/Notch1 axis in hepatocytes, the insulin pathway, and liver cancer are interconnected elements in hepatic physiology and pathology. HOTAIR, a lncRNA, and miR-124, a microRNA, along with Notch1, a transmembrane receptor, influence cellular processes in hepatocytes, including proliferation and differentiation, with dysregulation implicated in liver cancer progression (Price et al., 2021). The intricate interactions within this axis involve HOTAIR potentially acting as a sponge for miR-124, increasing Notch1 expression, and contributing to HCC suppression. The insulin pathway regulates glucose and lipid metabolism, with insulin resistance and hyperinsulinemia linked to hepatocarcinogenesis by promoting cell proliferation and inhibiting apoptosis (Chen & Kong, 2022). VAFE effectively downregulated miR-124 by enhancing the HOTAIR/Notch1/Jagged1 axis, thereby inhibiting tumor cell proliferation and differentiation. Additionally, by reinforcing the HOTAIR/Notch1/Jagged1 antitumor pathway, VAFE induced apoptosis in hepatocytes.
Conclusion
In conclusion, our study underscores the potential of VAFE as a protective agent against DEN-induced HCC. Through a comprehensive investigation of biochemical, molecular, and antioxidant pathways, alongside its role in modulating the HOTAIR/miR-124/Notch1 axis pathway, we elucidated the significant therapeutic effects of VAFE. Our findings reveal that VAFE administration effectively regulated liver function indices, improved antioxidant status, and mitigated inflammatory cytokine levels in HCC rats. Moreover, VAFE demonstrated an ability to modulate gene and protein expressions associated with HCC progression, particularly by downregulating HOTAIR and Notch1 while upregulating miR-124. This modulation effectively countered the activation of the Notch1/Jagged1 signaling pathway induced by HOTAIR, highlighting the potential of VAFE as an anticancer agent in DEN-induced HCC. Nevertheless, further in vivo and in vitro investigations are imperative to fully elucidate the anticancer properties of VAFE.
Abbreviations
MDA: Malondialdehyde; VAFE: Vaccinium arctostaphylos L. fruit extract; DNA: Deoxyribonucleic acid; HRP: Horseradish peroxidase, DTNB: 5,5-dithio-bis-2-nitrobenzoic acid, DEN: N-diethylnitrosamine; CRP: C-reactive protein; TP: Total protein; ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; HCC: Hepatocellular carcinoma; ALB: Albumin; CAT: Catalase, RNA: Ribonucleic acid, FRAP: Ferric-reducing antioxidant power, PBST: PBS-Tween 20; COX-2: Cyclooxygenase-2; BIL: Bilirubin; MAPK: Mitogen-activated protein kinase.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The experimental protocols of this study were approved by the Jinan City People’s Hospital ethics committee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
