Abstract
Background
Poor sleep quality and even insomnia are global problems, especially after the COVID-19 pandemic, and bioactive substances in natural herbal medicines may improve subjective sleep for people with insomnia. However, their active components still need to be isolated, determined, and verified by pharmacological experiments.
Objectives
To verify the efficacy of emodin-8-O-β-D-glucoside (EG), derived from Caulis Polygoni Multiflori in improving sleep in mice.
Materials and Methods
Hypnotic effect test, hypnotic experiment of sodium pentobarbital at a subthreshold dose, measurement of sleep latency induced by sodium barbital, and extending sodium pentobarbital sleep time experiment were performed in mice after different concentrations of EG were given intragastric administration for 30 days. The concentrations of 5-hydroxytryptamine (5-HT) and gamma-aminobutyric acid (GABA) in mouse brain tissue were determined by enzyme-linked immunosorbent assay. Levels of tryptophan hydroxylase 2 (TPH2) were detected by western blotting analysis and immunofluorescence labeling.
Results
After 30 days of continuous administration, behavioral experiments showed EG could shorten the time to fall asleep and prolong sleep duration. Tests revealed EG dose-dependently increased the levels of 5-HT in the brain tissue and the cause might be it could increase the expression of TPH2, the rate-limiting enzyme of 5-HT, in nerve cells in the hypothalamus.
Conclusion
Experimental data confirmed that EG could shorten sleep latency and prolong sleep time to improve sleep, and its mechanism of sleep meliorating is related to the upregulation of TPH2 and 5-HT levels in brain tissue.
Introduction
Sleep is necessary for normal physiological function. Seven hours of sleep is associated with the longest lifespan, while sleep less than 7 hours per 24 hours has poorer health in adults (Siegel, 2022). Due to the quickening pace of life in modern society, the increase in psychological pressure, and the influence of inappropriate sleeping environments, the incidence of insomnia in the population is growing. The COVID-19 pandemic is associated with a noticeable growth in insomnia and psychological symptoms compared to prepandemic levels. Large-scale public sleep intervention programs should be prioritized during and after pandemics such as COVID-19 (Morin et al., 2022). The occurrence of sleep disorders involves many systems, such as the central nervous and endocrine systems, and medication is the most effective way (Perlis et al., 2022). Drugs commonly used for insomnia mainly include barbiturates and benzodiazepines, but long-term use has many side effects (Dujardin et al., 2022). The bioactive substances in natural herbal medicines may have effective sleep meliorating, and accumulated research results (Ni et al., 2015) show that natural oral herbal medicines, such as traditional Chinese medicine, are used as monotherapies or adjuncts to conventional therapies.
The monoamine neurotransmitter 5-hydroxytryptamine (5-HT), also known as serotonin, is important in promoting sleep. Serotonin depletion in the brain will cause insomnia (Murray et al., 2015), and the release of serotonin creates a more relaxed state, especially after accumulating sleep debt, resulting in a transition to sleep; this is one of the mechanisms by which increasing 5-HT in the brain may improve insomnia (Jones, 2011). Tryptophan hydroxylase 2 (TPH2) is the rate-limiting enzyme of 5-HT in the brain tissue of mice. The electroencephalographic recording showed decreased sleep, especially rapid eye movement sleep in TPH2-deficient mice (Zhang et al., 2018). Based on behavioral and electroencephalogram analysis of genetic mutants, these results provide the strongest evidence yet that 5-HT in the brain is very important in sleep. Gamma-aminobutyric acid (GABA) is a nonproteinogenic amino acid and functions as an inhibitory neurotransmitter in the central nervous system. It is another neurotransmitter involved in sleep (Nuss, 2015). Low levels or impaired functioning of GABA may be associated with the cause and maintenance of sleep disorders such as insomnia (Bruni et al., 2021; Hepsomali et al., 2020). The active components of some natural products have been shown to improve sleep by reducing sleep latency and increasing subjective and objective indicators of sleep quality, and in-depth studies have shown that their mechanisms of action rely on neurotransmitter systems in the brain involved in promoting sleep (Feizi et al., 2019).
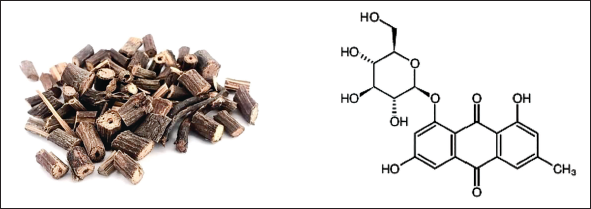
The stem of Caulis Polygoni Multiflori is consumed and has many pharmacological effects such as sleep-enhancing in China, and emodin-8-O-β-D-glucoside (EG, Figure 1) is one of the active ingredients in this herbal extract. Some natural chemicals have been reported to improve sleep and were evaluated by a pentobarbital-mediated sleeping model in mice (Jeon et al., 2015). This study aimed to verify through animal experiments whether EG improves sleep and provides a pharmacological experimental basis for further development of anti-insomnia drugs.
The Stem of Caulis Polygoni Multiflori and the Molecular Formula of Emodin-8-O-β-D-Glucoside (EG).
Materials and Methods
Sample Preparation of EG
EG was produced through the following steps. Dried medicinal tuber fleece flower stems (25 kg) were weighed and crushed, and flux extraction was performed 3 times with 75% ethanol for 2 hours each time. The extraction solution was reduced in pressure and concentrated until no alcohol flavor remained. Alcohol extract was suspended in an appropriate amount of water and extracted with petroleum ether to obtain the petroleum ether extract and the water extract. After petroleum ether extraction, a macroporous resin column was wetly eluted with an ethanol gradient to obtain macroporous resin eluates in distilled water and 30%, 60%, and 90% ethanol. The 60% ethanol-eluted portion (250 g) of the macroporous resin was eluted by silica gel column chromatography using a gradient elution of chloroform-methanol to obtain four parts. The third part was eluted with chloroform-methanol (20:1) by repeated silica gel column chromatography, and EG was obtained as yellow needle crystals.
Experimental Animals
One hundred and sixty SPF male BALB/c mice (18–22 g, 6-8 weeks) were provided by the Hubei Experimental Animal Research Center. Considering the experiment period was long and there were obvious hormone fluctuations in female mice, male mice were selected to avoid interference caused by intrinsic hormones. Animal experiments fully met ethical requirements.
Hypnotic Effect Test
Forty mice were randomly divided into five groups according to body weight: normal control group (distilled water), positive control group (diazepam 1 mg/kg), and EG low, middle, and high dose groups (10, 30, and 100 mg/kg). Mice were given free access to food and water during the experiment to avoid the stress effects of fasting, and test substances were given by oral gavage once a day for 30 days. When the mice were lying on their back, the disappearance of the righting reflex indicated that they had entered sleep, and the time between the disappearance and recovery of the righting reflex was recorded as the sleep time (Jeon et al., 2015). The observation was continued for 2 hours after each administration, and the number of mice that fell asleep in each group and the corresponding sleep times were recorded. The animals were eventually used to detect 5-HT, GABA, and TPH2 in brain tissue.
Hypnotic Experiment of Sodium Pentobarbital at a Subthreshold Dose
The animals were divided into the same groups and the treatment was the same as above, with daily administration for 30 days. Fifteen minutes after the last intragastric administration, pentobarbital sodium (38 mg/kg) was injected intraperitoneally to determine the time required for the disappearance of the dorsal righting reflex. The number of mice falling asleep in each group was recorded, and the falling asleep rates were calculated.
Measurement of Sleep Latency Induced by Sodium Barbital
Animals were treated as above, and 15 minutes after the last intragastric administration, the righting reflex was induced to disappear due to hypnosis by intraperitoneal injection of sodium barbital (360 mg/kg). The judgment index was the supine position of mice, and the time of sleep latency in each group was recorded.
Extending Sodium Pentobarbital Sleep Time Experiment
The animals were divided into the same group and the administration method was the same as above. On the 30th day, 15 minutes after the last treatment of EG and control substances, pentobarbital sodium (53 mg/kg) was injected intraperitoneally, and the dorsal righting reflex disappeared. The sleep times of the mice were recorded according to the recovery condition.
Detection of 5-HT and GABA in Brain Tissue
After the experimental observations, mice brain tissues in each group were separated, the cerebellum was removed on a frozen petri dish and the brain (including the cerebral cortex, basal ganglia, thalamus, and brainstem) was obtained and weighed, homogenized with nine times the volume of cold phosphate-buffered saline (PBS) buffer solution (pH 7.2), and centrifuged at 4°C at 1,000 r/min for 15 minutes to obtain the supernatant. The levels of 5-HT and GABA in the supernatant were determined by enzyme-linked immunosorbent assay (ELISA) kits.
Detection of Levels of TPH2 in Brain Tissue
Western blotting analysis and immunofluorescence labeling were used to detect the level of TPH2, the rate-limiting enzyme in the synthesis of neuronal 5-HT. Sections of mice brains were incubated overnight at 4°C with anti-TPH2 antibody (1:40, Invitrogen PA5-27584) and fluorescently tagged with a secondary antibody for 1 hour. Images were captured using a laser scanning confocal microscope (Nikon Corporation, Japan).
Statistical Analysis
Animal weights, time of sleep latency, and duration were tested for homogeneity of variance during the experiment, and then analysis of variance (F-test) was performed. Final data were expressed as mean ± standard deviation, and p < 0.05 was considered significant.
Results
The Effect of EG on the Mice Body Weight
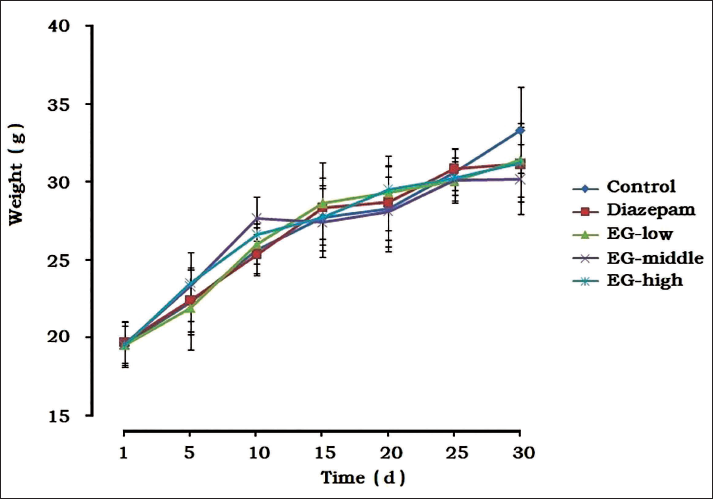
The observed results of feeding the animals for 30 days (Figure 2) manifested no significant difference in body weight among the groups during the experiment, and no abnormal effects were observed in the EG groups, indicating that EG had no toxicity in normal mice.
The Influence of Emodin-8-O-β-D-Glucoside (EG) on the Weight and Growth of Mice (
± SD, n = 8).
Hypnotic Effect Test
The behavior and activity of each group of mice were normal after the corresponding test substances were given, and no direct sleep phenomenon or behavioral abnormality was observed in EG groups, indicating that EG had no direct hypnosis action.
Hypnotic Experiment with Subthreshold Dose of Sodium Pentobarbital
After different doses of EG were given for long-term feeding, the hypnotic rate of falling asleep under sodium pentobarbital was similar to the normal control and diazepam groups (p > 0.05). Results of sleep rate also showed that EG had no direct hypnotic effect (Table 1).
The Hypnotic Effect of Emodin-8-O-β-D-Glucoside (EG) at the Subthreshold Dose of Sodium Pentobarbital.

Measurement of Sleep Latency Induced by Sodium Barbital
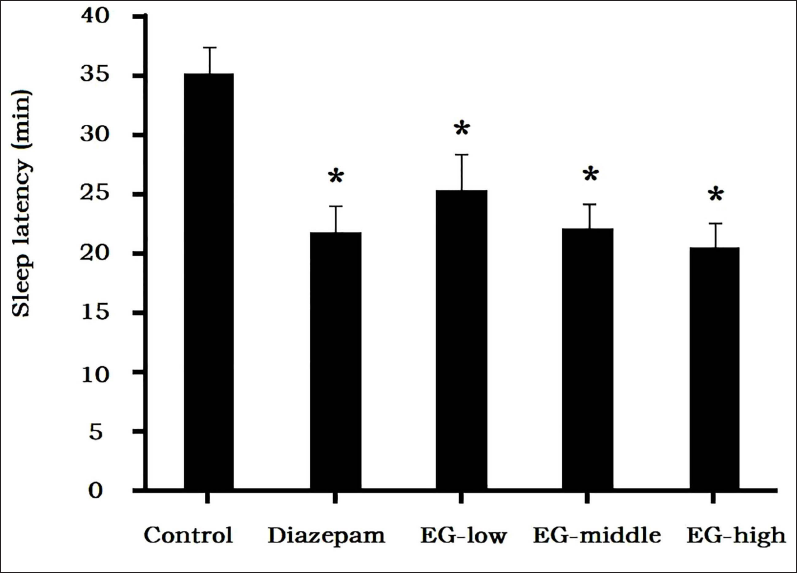
The sleep latency of the control group was 35.2 ± 2.3 minutes, while administration of EG shortened the sleep latency. Compared with the control group, sleep latencies of the middle and high-dose groups were shortened to 22.1 ± 2.2 minutes and 20.6 ± 2.1 minutes, respectively, and the difference was significant, which indicated that EG had the effect of promoting sleep (Figure 3).
Extending Sodium Pentobarbital Sleep Time Experiment
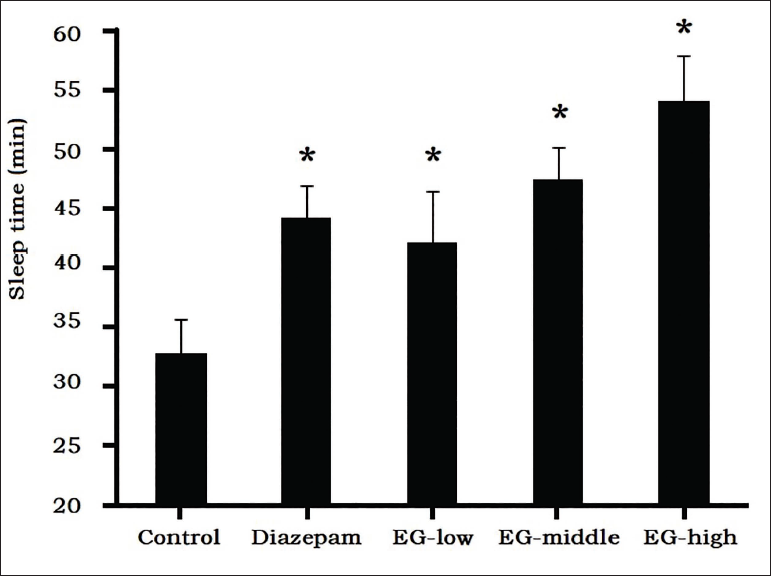
Pentobarbital-induced sleep in mice is a classic pharmacological method for screening sedative-hypnotic drugs, and results (Figure 4) showed that after the effective dose of sodium pentobarbital was given to normal mice in the control group, the duration of sleep time was 32.9 ± 2.8 minutes. EG prolonged the sleep time induced by sodium pentobarbital, which in the middle-dose group (47.5 ± 2.8 minutes) and the high-dose group (54.2 ± 3.8 minutes) were significantly longer than control group (p < 0.05).
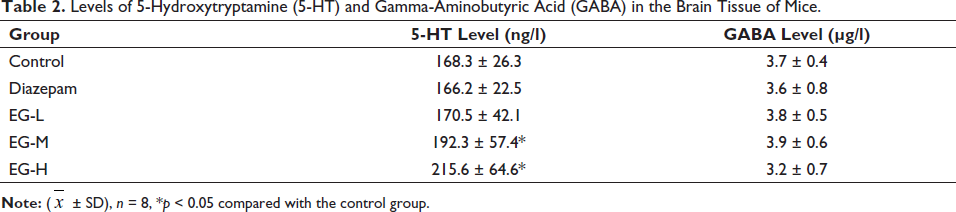
Levels of 5-HT and GABA in Brain Tissue
After continuous administration, EG dose-dependently increased the levels of 5-HT in the brain tissue of mice, which were statistically significant compared with the control group (Table 2). The levels of GABA did not change in the brain tissue.
Levels of 5-Hydroxytryptamine (5-HT) and Gamma-Aminobutyric Acid (GABA) in the Brain Tissue of Mice.
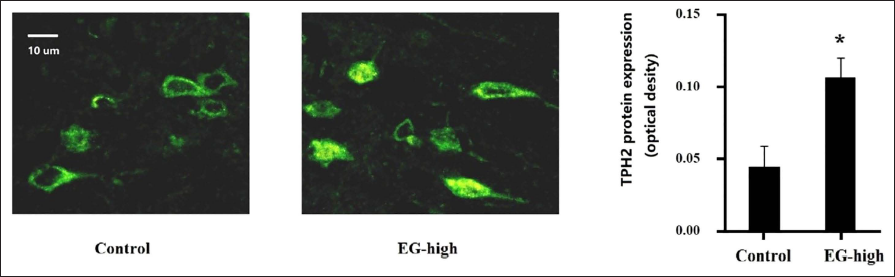
Levels of TPH2 in Brain Tissue
The western blotting results showed no significant increase in overall TPH2 content in brain tissue, but we observed significant enhancement in the fluorescence of some local nerve cells in the hypothalamus (shown in Figure 5). It was suggested that EG could increase the expression of TPH2 in nerve cells in some specific areas, so the potential mechanism and regulatory pathway need to be further studied.
Discussion
Behavioral therapy, cognitive therapy, and pharmacological interventions can effectively treat insomnia, and drug use is more frequent worldwide because it is simple and effective. For example, chemical synthesis of sedative-hypnotic drugs, after continuous use, may cause dizziness, hangover, lethargy, and other symptoms; long-term use could lead to rebound and withdrawal symptoms. On the other hand, many licensed drugs (including benzodiazepines) can effectively treat insomnia but are poorly tolerated (De Crescenzo et al., 2022; Hirschtritt et al., 2021). Hence, treatment should focus on the regulation of neurotransmitters in the part of the brain associated with normal sleep, such as 5-HT and GABA (Holst & Landolt, 2022; Luppi & Fort, 2019). 5-HT can synthesize melatonin in the body, which plays a role in regulating sleep, and p-chlorophenylalanine with tryptophan hydroxylase inhibitors can consume 5-HT and cause insomnia, so 5-HT neurons play an important role in improving sleep. The role of 5-HT neurons is to trigger downstream effects to promote and induce sleep when needed; otherwise, serotonin will be released due to accumulated sleep debt, thus transitioning to sleep, and in some cases, serotonin may induce sleep (Monti, 2011). Experiments also confirmed that animals deficient in 5-HT synthesis would sleep less (Zhang et al., 2018), and inhibition of 5-HT in the brain increases anxiety (Tu et al., 2020). Human studies also suggest symptoms such as insomnia and depression were related to the expression and regulation of 5-HT (Zeng et al., 2023). Our study showed that EG could shorten the time of sleep latency and prolong sleep duration in mice induced by a hypnotic dose of sodium pentobarbital, and increase the level of 5-HT in the brain tissue of mice, indicating that the central 5-HT nervous system is involved in the sleep-promoting effect. EG increased the content of TPH2 in nerve cells in some special parts of the hypothalamus, which may be one of the reasons. The effects of natural products on sleep improvement by increasing 5-HT and TPH2 in brain tissue may also be accompanied by an enhancement of the tryptophan and melatonin systems, as well as a decrease in serum corticosterone levels, suggesting a multi-target mechanism (Xia et al., 2024).
Poor sleep quality and even insomnia are global problems, and many factors are responsible for poor sleep quality, especially after the COVID-19 pandemic. Therefore, finding drugs that can safely and effectively improve sleep has broad application prospects. Bioactive substances in natural herbal medicines may improve subjective sleep for people with insomnia. However, their active components still need to be isolated, determined, and verified by pharmacological experiments. Our study focused on the efficacy of EG, which is derived from Caulis Polygoni Multiflori in improving sleep in mice. Results showed that EG could increase 5-HT levels in the brain, suggesting that its sleep-improving effect was related to the central nervous system, but the clinical effect needs to be confirmed by human clinical trials.
Insomnia disorders are often persistent, so the use of medications must consider their long-term effects (Morin & Jarrin, 2022). Drugs of natural origin may be better for long-term use than chemicals. In recent years, natural drug chemistry and screening of active substances have become the focus of drug development. This study provides a pharmacological experimental basis for EG in further developing anti-insomnia drugs.
Conclusion
After EG feeding for 30 days, the behavior and body weight in EG groups were not affected. Sleep tests showed that the sleep latency was significantly shortened in medium- and high-dose groups, while sleep duration was significantly prolonged, verifying that EG had the effect of sleep meliorating, and the mechanism was related to the upregulation of TPH2 of neurons in local brain tissue to increase the level of 5-HT.
Abbreviations
EG: Emodin-8-O-β-D-glucoside; GABA: Gamma-aminobutyric acid; TPH2: Tryptophan hydroxylase 2; 5-HT: 5-Hydroxytryptamine.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study has been approved by the Experimental Animal Ethics Committee of Huazhong University of Science and Technology, approval number: 20230306/459/TJ.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Foundation of Hubei Provincial Health Commission 2023-2024 Research Project (WJ2023M023).
