Abstract
Background
Hepatocellular carcinoma (HCC) is one of the most common malignancies and a leading cause of cancer-related mortality and morbidity worldwide.
Objectives
This study evaluated the enhanced efficacy of a combination of herbal medicine with sorafenib (MIX) in a transplantation tumor model induced by HepG2 cells in HCC.
Materials and Methods
The HepG2 cell transplantation tumor model was established by injecting HepG2 cells subcutaneously into BALB/c nude mice. The impact of MIX on tumor growth was observed. Granzyme B was used to measure natural killer (NK) cell activity. Prognostic markers of HCC, telomerase reverse transcriptase (TERT), and alpha-fetoprotein (AFP) were analyzed via real-time polymerase chain reaction. Additionally, inflammation and apoptosis-related markers were assessed through serum analysis and Western blotting.
Results
Among the MIX groups, MIX25 (sorafenib 10 mg/kg + Dioscoreae rhizoma and Polygalae radix mixture 25 mg/kg) demonstrated the most significant effect. MIX25 effectively suppressed tumor volume and weight, enhanced apoptosis-promoting proteins (Bax, Cytochrome c, and Caspase-3), and reduced apoptosis-inhibiting proteins like survivin. Furthermore, MIX25 inhibited the mitogen-activated protein kinase inflammatory response and inflammatory markers, including tumor necrosis factor-alpha and cyclooxygenase-2. The MIX25 group exhibited superior effects on NK cell activity and cancer marker genes (AFP and TERT) compared to other drug-treated groups.
Conclusion
MIX25 effectively suppressed the proliferation of HepG2 cell transplantation tumors in BALB/c nude mice. It not only inhibited inflammatory markers but also enhanced cell apoptosis. Additionally, MIX25 significantly increased NK cell activity and reduced mRNA expressions of AFP and TERT. Overall, the combination of MIX25 with sorafenib shows promise as a potential treatment for patients with HCC.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is reported as the third cause of tumor-related mortality worldwide (Dat et al., 2022) and few drugs are available for its clinical treatment. However, recently, hopeful breakthroughs have been made in the development of new drugs in the field like traditional therapy or immunotherapy (Luo et al., 2021; Zhang et al., 2021). If HCC is diagnosed at early stages, it is treatable with locoregional treatments including surgical resection (SR), liver transplantation, transarterial chemoembolization (TACE), and radiofrequency ablation (RFA). However, due to the lack of techniques and high mortality for diagnosis, most of the patients diagnosed at an advanced stage cannot be resected tumors. Moreover, it shows the low efficiency of treatment when considering the high recurrence rate and low survival rate (Xing et al., 2021). Hence, it is imminently required to seek new treatment strategies for HCC (Zhang et al., 2021).
Sorafenib is prescribed currently as an effective first-line drug in HCC. Sorafenib is called a multikinase inhibitor and is responsible for facilitating apoptosis, suppressing tumor cell proliferation, and mitigating angiogenesis (Tang et al., 2020). Peak plasma concentrations reached about 3 hours after oral intake. Subsequently, sorafenib is metabolized in the liver by oxidation. However, sorafenib reduced bioavailability by about 29% in high-fat meals (Kane et al., 2006). The most commonly reported side effects are fatigue, weight loss, hand-foot skin reaction, rash, alopecia, pruritus, and diarrhea (Keating, 2017; Zhang et al., 2011). Besides, pulmonary toxicity has been reported during sorafenib use, with diffuse pulmonary opacities, cough, dyspnea, interstitial pneumonia, and fever. Sometimes, its use was fatal in some patients (Yamaguchi et al., 2015).
Dioscoreae rhizoma is used medically when dried and stripped of periderm of Dioscorea plants like Dioscorea batatas Decaisne or Dioscorea japonica Thunberg. Dioscorea has the effect of “promoting blood circulation, eliminating phlegm, relaxing tendons, promoting digestion, and inhibiting malaria” (Lian et al., 2020). Recent studies have confirmed that diosgenin extracted from Dioscorea plants exhibits antitumor activities (Li et al., 2010). The most common secondary metabolites in Dioscorea roots are saponins, with more than 100 steroidal saponins identified. Diosgenin, primarily found among these saponins, along with volatile compounds, is prevalent in Dioscorea roots and holds commercial significance in the pharmaceutical industry. However, due to its low content and underdeveloped biotransformation, various methods are being explored to efficiently produce it (Salehi et al., 2019; Yang et al., 2023). Additionally, films utilizing starch isolated from Dioscorea demonstrate various applications, not only maintaining fresh quality but also extending shelf life (Shen et al., 2022; Wang et al., 2023).
Another herb, Polygalae radix, the dried root of Polygala tenuifolia, has possessed wide-ranging pharmacological activities, such as anti-inflammatory, antioxidant, antiaging, antiviral, neuroprotective, antidepressant, and antitumor. Moreover, it has been applied in the treatment of insomnia, forgetfulness, cough, and other diseases. However, long-term use or excessive doses of Polygalae radix exhibit slight toxicity to the gastrointestinal tract (Zhao et al., 2020). Meanwhile, Dioscoreae rhizoma extracts improved digestive capability and also influenced the conversion of the intestinal microflora to helpful bacteria (Jeon et al., 2006). Overall, when these two herbal medicines are used in combination, it will be possible to remove the toxicity of Polygalae radix through the gastrointestinal protective effect of Dioscoreae rhizoma and may alleviate the side effects of sorafenib.
Until now, the antitumor effect of the mixture of Dioscoreae rhizoma and Polygalae radix (DP) has little been known and the effects related to co-administration with sorafenib also have not been studied. Therefore, the present study was performed to elucidate the pharmacological effects and the underlying mechanism of sorafenib alone and in combination with Dioscoreae rhizoma and Polygalae radix mixture (MIX) in the HepG2 cell transplantation tumor model.
Materials and Methods
Preparation of the Plant Material
Dioscoreae rhizoma and Polygalae radix were supplied by Bonchowon (Yeongcheon-si, Korea). Each dried Dioscoreae rhizoma (100 g) and Polygala radix (100 g) was boiled at 100℃ with water (1,000 mL) at room temperature (RT) for 2 hours and each solvent was evaporated in vacuo. Then, the powders of Dioscoreae rhizoma or Polygalae radix mixture extract were obtained as a yield of 14.4% and 21.0%, respectively.
Cell Culture
The human HCC cell line, HepG2 cell (American Type Culture Collection, Rockville, MD, USA) was cultured in DMEM high glucose (4.5 g/L) medium provided by Corning Life Sciences (Massachusetts, USA) supplemented with 10% fetal bovine serum (FBS) (Gibco, Grand Island, NY, USA) and 1% penicillin/streptomycin (PS). Cells were maintained in a humidified atmosphere containing 5% CO2 at 37℃.
BALB/c Nude Mice and Tumor Model Treament
Five-week-old male BALB/c nude mice were purchased from OrientBio (Seongnam, Korea). The mice were maintained under a 12-hour light/12-hour dark cycle, controlled humidity (50 ± 10%), temperature (22 ± 1℃), and illuminance (150–300 lux). To generate the xenograft tumor model, we modified the method of Seyhoun et al. (2019). Nude mice were subcutaneously injected with a 100 µL cell suspension containing 1 × 106 HepG2 cells under the right axillary region. When tumors reached a volume of more than 100 mm3, tumor mice except for normal mice were randomly divided into five groups of eight mice as follows:
Normal group, mice administered with distilled water (DW); Control group, DW administered and HepG2 cell-transplanted nude mice; S10 group, sorafenib 10 mg/kg body weight administered and HepG2 cell-transplanted nude mice; MIX25 group, sorafenib 10 mg/kg plus DP 25 mg/kg body weight administered and HepG2 cell-transplanted nude mice; MIX50 group, sorafenib 10 mg/kg plus DP 50 mg/kg body weight administered and HepG2 cell-transplanted nude mice; and MIX100 group, sorafenib 10 mg/kg plus DP 100 mg/kg body weight administered and HepG2 cell-transplanted nude mice.
Drugs were orally administered for 24 days. The reactivity and behavior of mice in the cages were meticulously observed. After group separation, the length and width of the tumors were newly measured on days 1, 4, 7, 10, and final days using a digital caliper (Mitutoyo series 500, Kanagawa, Japan), and the weight of the mice was recorded every day.
The tumor volume (mm3) was calculated using the formula:
Tumor volume = 0.5 × long distance (L) × short distance (W)2.
Measurement of Aspartate Aminotransferase (AST) and Alanine Aminotransferase (ALT) Levels in Serum
Serum evaluation for the liver function was performed using the commercial kit as used in the previous study (Shin et al., 2022).
Measurement of Relevant Factors in Serum
Both levels of inflammatory markers including tumor necrosis factor-alpha (TNF-α) and cyclooxygenase-2 (COX-2) in serum and granzyme B level for natural killer (NK) cell activity were analyzed using commercial enzyme-linked immunosorbent assay (ELISA) kits according to Thermos Fisher Scientific (Waltham, MA, USA)’s instructions. Also, serum biochemical analysis was measured using an automatic biochemical analyzer (TBA-120FR, Toshiba Ltd., Tokyo, Japan).
Western Blot Analysis
Tumor samples were cut into small pieces and homogenized. Tissues were lysed at 4°C using protein lysate buffer as used in the previous study (Shin et al., 2021). Protein concentration was quantified using the BCA method and proteins were separated by 10% SDS-PAGE and transferred onto a nitrocellulose membrane. Membranes were incubated with primary antibodies against p-c-Jun, p-c-Fos, p-p38, p-ERK, p-JNK, c-Jun, c-Fos, p38, ERK, JNK, Bax, Cytochrome c, survivin, Caspase-3, β-actin, and histone diluted in 1:1,000 and overnight at 4°C. Then, membranes were soaked in horseradish peroxidase (HRP)-conjugated secondary antibody for 1 hour at RT. Bands were visualized using the previous method (Shin et al., 2021). The antibody lists for Western blot analysis are shown in Supplementary Table 1.
The Effect of Dioscoreae Rhizoma and Polygalae Radix Mixture (DP) in Combination with Sorafenib (MIX) on Body Weight Change.
Bars are expressed as the mean ± SEM, n = 8. Significance: ##p < 0.01, ###p < 0.001 versus the normal group and *p < 0.05, **p < 0.01, ***p < 0.001 versus the control group. DP, Dioscoreae rhizoma and Polygalae radix mixture; MIX, Dioscoreae rhizoma and Polygalae radix mixture in combination with sorafenib.
Real-time Polymerase Chain Reaction (PCR) Analysis
The tumor tissue was synthesized using a cDNA synthesis kit (Thermo Fisher Scientific, MA, USA) after RNA extraction using an RNA extraction kit (Qiagen, Hilden, Germany), and then the expression of hepatic carcinoma index (alpha-fetoprotein (AFP), telomerase reverse transcriptase (TERT)) was analyzed using real-time PCR (Roche, Basel, Swiss). Each primer sequence is shown in Supplementary Table 2.
Statistical Analysis
Data were analyzed using SPSS (version 26.0, IBM Co., Armonk, NY, USA) and presented as the mean ± SEM. Statistical comparisons were performed by one-way analysis of variance (ANOVA) with the least significant difference (LSD) test. Values of p < 0.05 were considered significant.
Results
Effects of MIX on Animal Changes in Body Weight, Tumor Volume, and Tumor Weight
As shown in Table 1 and Figure 1, body weight, tumor volume, and tumor weight were measured in all groups. Symptomatic index of the tumor model, such as body weight increase according to the increase in tumor weight, was observed during experimental periods. Compared with the normal group, the control group considerably increased in both final body weight and body weight change (p < 0.01, p < 0.001, respectively). However, S10 and MIX25 treatment significantly reduced such elevated levels on final body weight (p < 0.05). While, there was no significant change in the DP25, MIX50, and MIX100 groups. Furthermore, body weight change in all drug-treated groups dramatically decreased compared with the control group. When the tumor inhibitory potential of MIX was measured between groups, drug treatment led to a significant decrease in both tumor volume change and tumor weight.
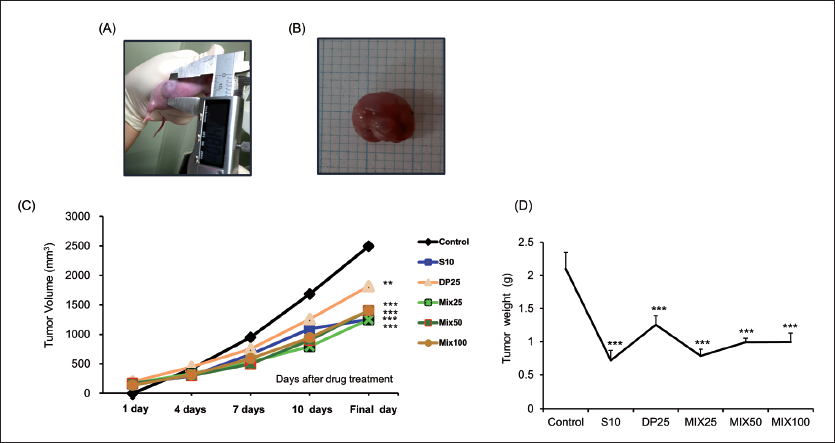
In particular, S10 and MIX25 showed similar inhibitory effects on tumor volume and tumor weight.
Effect of MIX on Serum Liver and Kidney Function
As shown in Figure 2, the serum levels of AST and ALT in the HepG2 cells transplantation tumor model were significantly increased. Whereas, all drug treatments effectively reduced two factors. In particular, MIX25 treatment decreased powerfully ALT as well as AST compared with the control group (AST, 45.8% reduction; ALT, 20% reduction). Moreover, the creatinine level in the MIX25 group was reduced by 19.1% when compared with the control group.
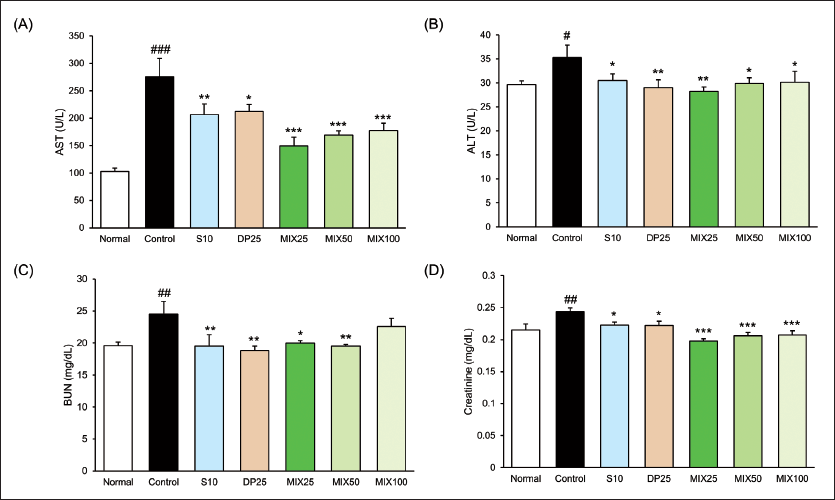
Compared to the liver and kidney function between S10 and MIX25, there was a tendency for MIX25 to seem better in serum AST, ALT, and creatinine levels than S10, but serum blood urea nitrogen (BUN) level showed a similar effect.
Effect of MIX on Mitogen-activated Protein Kinase (MAPK)-associated Factors on the HepG2 Cells Transplantation Tumor Model
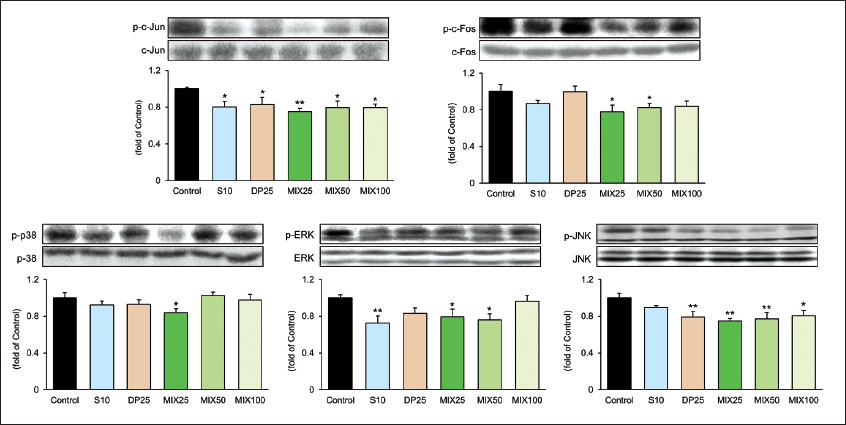
To elucidate the pharmacological effects of MIX focusing on the possible mechanism of hepatic inflammation, we performed Western blotting against their phosphorylated forms of MAPK-associated factors including c-Jun, c-Fos, p38, ERK, and JNK. As shown in Figure 3, all targeted proteins in the MIX25 group were drastically altered in the control group (p-c-Jun and p-JNK, p < 0.01; p-c-Fos, p-p38, and p-ERK, p < 0.05) according to our expectations. Especially, the MIX25 group exhibited prominently the expressions of p-c-Fos, p-p38, and p-JNK, unlike the S10 group. These data showed that MIX25 alleviated hepatic inflammation by regulating MAPK-associated factors.
Effect of MIX on Serum Inflammatory Factors and NK Cell Activity
As shown in Figure 4, mice in the control group dramatically increased serum TNF-α and COX-2 levels compared with the normal group (TNF-α, 868.98 ± 47.08 vs. 507.18 ± 38.59 pg/mL, p < 0.001; COX-2, 565.17 ± 81.57 vs. 151.69 ± 18.26 pg/mL, p < 0.001). NK cell activity was dose-dependently decreased with MIX treatment. In particular, NK cell activity in the MIX25 group was the best among the drug treatment groups.
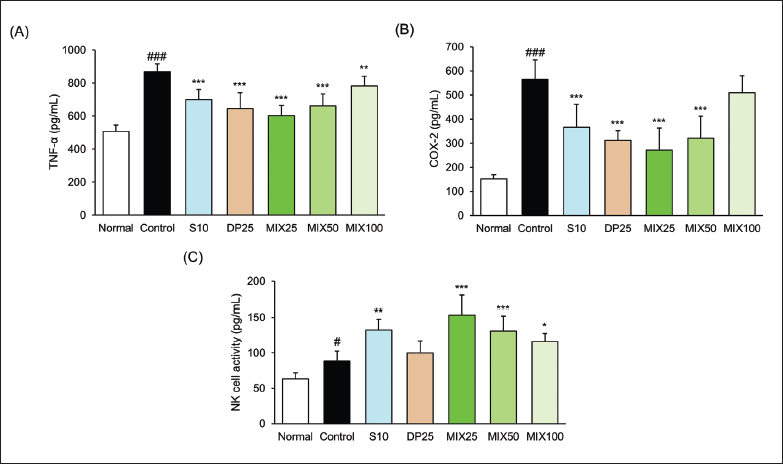
Compared to NK cell activity between the S10 and MIX25 groups, the MIX25 group is 1.73-fold and the S10 group is 1.50-fold compared with the control group. As a result, the MIX25 group was superior to the activity of the S10 group against the NK cell activity.
Effect of MIX on Apoptosis-associated Factors on the HepG2 Cells Transplantation Tumor Model
Figure 5 shows the reduced expressions of Bax, Cyto C, and Caspase-3 and increased survivin expression in the control group. Unlike these results, both the S10 and MIX25 groups observed pro-apoptotic factors such as Bax, Cyto C, and Caspase-3 increased significantly whereas, the MIX25 group only reduced dramatically in the survivin level. The levels of the MIX25 group on the expressions of Bax and Caspase-3 were superior compared with the S10 group (Bax, 1.10-fold; Caspase-3, 1.10-fold) while Cyto C was shown similar effects in the two groups. Moreover, the level of the MIX25 group on the expressions of survivin was reduced by 20.42% compared with the S10 group.
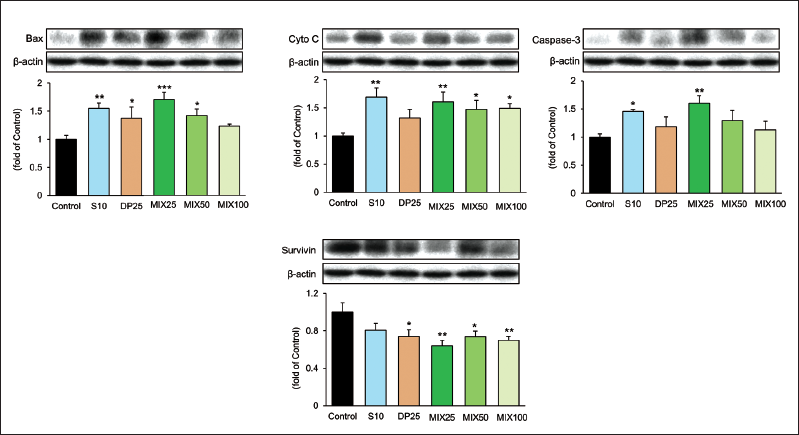
As a result, the MIX25 group was more excellent than that of the S10 group against the apoptosis-associated factors.
Effect of MIX on AFP and TERT mRNA Expressions
To verify HCC diagnostic biomarkers such as AFP and TERT, we measured their mRNA expressions as shown in Figure 6. AFP relative expression compared to the control group was drastically reduced in drug-treated groups. Moreover, the MIX25 group showed a potent inhibitory effect. These results meant that the excellent inhibition of both AFP mRNA and TERT mRNA could lead to enhanced immunity against HCC in mice.
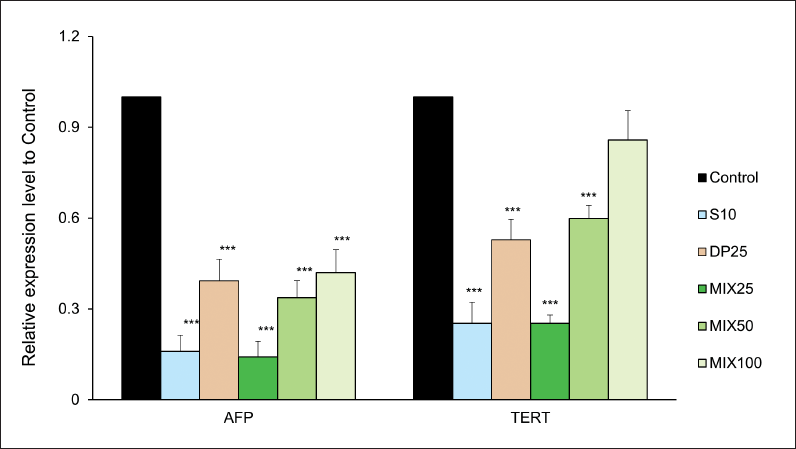
In particular, the inhibitory effects in the S10 and MIX25 groups were shown at similar levels.
Discussion
HepG2 cells have a high similarity to the human liver in terms of the expression of cellular proteins (Choi et al., 2015; Rodriguez-Antona et al., 2002). HepG2 cells have an unlimited life span and they comprise a more stable phenotype than primary hepatocytes (Ruoß et al., 2019). Accordingly, HepG2 cells are broadly utilized for screening tests of drug metabolism and toxicity as representative cells of hepatic cancers (Han et al., 2020; Lu et al., 2014; Wang et al., 2016). Regulating the proliferation of tumor cells is being accepted as an essential way to prevent tumors.
The current study focused on the pharmacological effects of sorafenib alone or in combination with DP (Dioscoreae rhizoma and Polygalae radix mixture) on the HepG2 cell transplantation tumor model. We obtained that both sorafenib and MIX (Dioscoreae rhizoma and Polygalae radix mixture in combination with sorafenib) could significantly inhibit tumor proliferation on the HepG2 cells transplantation tumor model and could be strengthened by their combination treatment. Paradoxically, such effects of MIX treatment with different concentrations were attenuated dose-dependently. In other words, the lowest concentration of MIX (MIX25 treatment) showed the most excellent effect.
To establish the HepG2 cells transplantation tumor model, we injected subcutaneously HepG2 cells in BALB/c nude mice. As shown in Table 1, the control group significantly increased body weight change (p < 0.01) in comparison with the normal group. This increase was significantly reduced only in the S10 and MIX25 groups. Additionally, we measured both tumor volume and weight (Figure 1). The previous report showed tumor growth associated with body weight (Cui et al., 2022). As we expected, administration with MIX during entire experimental periods excellently attenuated not only tumor volume but also tumor weight. Among the MIX-treated groups, MIX25, the lowest concentration, showed a powerful inhibitory effect. In particular, MIX25 and the S10 groups showed similar inhibitory effects on tumor volume and tumor weight (S10, 1250.3 ± 222.7 mm3, 0.72 ± 0.15 g; MIX25, 1250.0 ± 229.6 mm3, 0.79 ± 0.10 g, p < 0.001, respectively). Additionally, liver enzymes such as serum AST and ALT levels and kidney enzymes such as BUN and creatinine in the control group were significantly increased as compared with the normal group. Whereas drug-treated groups significantly decreased all enzymes except for MIX100 on serum BUN level (Figure 2).
A mitogen-activated protein (MAP) kinases are composed of at least three subfamilies like ERK, p38, and c-Jun N-terminal kinase (JNK). Activation of these MAP kinases is related to cell proliferation, differentiation, development, inflammation, stress response, and apoptosis (Dong et al., 2002; Shneider et al., 2021). Herein, the extracellular signal-regulated kinase (ERK) is closely associated with cell growth and survival, and the p38 not only regulates both stress and inflammation response but also induces apoptosis. p38 in recent studies emerged as an important regulator in cancer progression (Bradham & McClay, 2006). JNK is also involved in the regulation of apoptotic pathways (Wang et al., 2017). Subsequently, MAP kinases regulate AP-1, which is a transcriptional activator including c-Jun and c-Fos subunits, in response to extracellular various stimuli. Here, AP-1 induces inflammatory mediators and cytokines (Agron et al., 2017). As shown in Figure 3, three factors in the MIX25 group were significantly reduced whereas S10 treatment inhibited only the level of p-ERK. Moreover, p-c-Fos significantly reduced only the MIX25 and the MIX50 groups unlike the significant inhibition of p-c-Jun in all drug-treated groups. The expressions of TNF-α and COX-2 by the activation of MAP kinase showed the best inhibitory effects in the MIX25 group (Figure 4). NK cells, which are innate lymphoid cells with cytolytic activity, have been known to play a fundamental role in antitumor therapies (Habif et al., 2019; Vivier et al., 2011). In this study, NK cell activities in the drug-treated groups except for the DP25 group effectively increased. In particular, MIX treatment decreased in a dose-dependent manner. Through this, it was found that MIX25 showed the best NK cell activity.
Apoptosis, which is an intracellular death program occurs normally for a homeostatic mechanism that regulates cell numbers and removes abnormal cells. Mitochondria mediate apoptotic signalings like Bax activation the release of Cytochrome c, and the cleaves of Caspase-3 (Elmore, 2007). During the process of apoptosis, caspase-3 plays a key function and acts as the major executor of apoptosis. Dysregulation of cell apoptosis breaks down the balance between the progression and deterioration of HCC (Pistritto et al., 2016). In this study, the decreased pro-apoptotic factors including Bax, Cytochrome c, and Caspase-3 in the control group showed a significant increase by both S10 and MIX25 treatments. While the antiapoptotic factor, survivin, was significantly reduced in all groups except the S10 group (Figure 5). Moreover, AFP relative expression compared to the control group was drastically reduced in drug-treated groups. Moreover, the MIX25 group showed a potent inhibitory effect. This excellent inhibition of both AFP mRNA and TERT mRNA can lead to enhanced immunity against HCC in mice (Figure 6).
Indeed, limitations remain in this study. The exact molecular mechanism of the combined sorafenib and DP on the nuclear factor-kappa B (NF-κB) signaling pathway has been not suggested. As we know, NF-κB is a transcription factor critical for tumor progression (Xia et al., 2014; Yang et al., 2019). Currently, there is little evidence for the effect of sorafenib and DP on NF-κB in this study. Accordingly, further studies will have to follow to endorse a clearer and more enough explanation.
The combination therapy in the proper concentration can enhance the pharmacological effect. This study focused on the effects of combined sorafenib and DP on the HepG2 cell transplantation tumor model. We found that MIX25 treatment drastically suppressed cell inflammation while MIX25 treatment effectively promoted cell apoptosis and NK cell activity compared with S10 treatment. These findings provided the theoretical basis for the efficacy of combination therapy on HCC treatment.
Conclusion
MIX25 treatment inhibited HepG2 cell transplantation tumor proliferation in BALB/c nude mice. In particular, MIX is involved in not only the inhibition of inflammatory markers but also the enhancement of cell apoptosis. Moreover, the NK cell activity effectively increased in the MIX25 group, and the mRNA expressions of AFP and TERT were significantly reduced.
Summary
MIX25 treatment resulted in increased NK cell activity and the inhibition of both inflammatory markers and apoptosis.
Abbreviations
HCC: Hepatocellular carcinoma; NK cell: Natural killer cell; TERT: Telomerase reverse transcriptase; AFP: Alpha-fetoprotein; DP: Dioscoreae rhizoma and Polygalae radix mixture; MIX: Dioscoreae rhizoma and Polygalae radix mixture in combination with sorafenib; MAPK: Mitogen-activated protein kinase; ERK: Extracellular signal-regulated kinase; JNK: c-Jun N-terminal kinase; AP-1: Activator protein-1; TNF-α: Tumor necrosis factor-alpha; COX-2: Cyclooxygenase-2; Bax: Bcl-2-associated X protein; Cyto C: Cytochrome c; NF-κB: Nuclear factor-kappa B; SR: Surgical resection; TACE: Transarterial chemoembolization; RFA: Radiofrequency ablation; AST: Aspartate aminotransferase; ALT: Alanine aminotransferase; BUN: Blood urea nitrogen.
Footnotes
Acknowledgments
None.
Authors’ Contributions
M.-R. S. and J. W. C. contributed equally to this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
All animal experiment protocols were conducted following the “Guidelines for Animal Experiment” approved by DGMIF IACUC (No. 21041405).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No. 2018R1A5A2025272).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
