Abstract
Background
Lung cancer is one of the most common cancers worldwide and a leading cause of cancer-related deaths. The study delves into melezitose, a naturally occurring compound known for its biocompatibility.
Purpose
This study aims to uncover its therapeutic potential and molecular mechanisms within lung cancer, particularly in A549 cells. Melezitose’s impact on inhibiting cell proliferation and influencing epithelial-mesenchymal transitions (EMT) was the primary focus.
Materials and Methods
In a time-dependent manner, A549 cells, representative of lung adenocarcinoma (LUAD), underwent melezitose treatment. Analysis of cytotoxicity by MTT assay, cell migration assay, and its responsible genes were analyzed by reverse transcription polymerase chain reaction (RT-PCR) and enzyme-linked immunosorbent assay (ELISA) kit method.
Results
The MTT assay indicated a significant reduction in A549 cell growth after 48 hours of melezitose treatment. Additionally, melezitose induced G1 phase cell arrest and stimulated apoptosis in A549 cells. Subsequent determination of IC50 values represents the concentration at which melezitose inhibits 50% of cell growth. The study also investigated EMT-related gene expression like claudin 1 (CLD1), E-cadherin (ECADH), SNAIL1, SLUG, and vimentin (VIM) through RT-PCR. The findings revealed strong binding associations between melezitose and these EMT targets, suggesting a potential regulatory role of melezitose in impeding EMT processes.
Conclusion
Overall, this study illuminates the significant role of melezitose in lung cancer. Its observed inhibition of lung cancer cell proliferation and its influential impact on EMT-related gene expression highlights. Melezitose has potential as a therapeutic agent, particularly in the context of NSCC. The multifaceted effects of melezitose on A549 cells open promising avenues for advancing our understanding of this disease and developing innovative therapeutic strategies.
Introduction
Lung cancer has the unfortunate distinction of being the most often diagnosed cancer worldwide, with a projected 2.3 million cases and a concerning increase in fatality rates, as emphasized by Sung et al. (2021). This disease is more prevalent than other well-known cancers such as colorectal, pancreatic, and breast cancer. Despite ongoing efforts, the prognosis for lung cancer remains grim, and the occurrence of epithelial-mesenchymal transition (EMT) in lung cancer has significant therapeutic consequences. Tumors that have undergone EMT are often linked with increased chemoresistance and resistance to targeted therapies (Cui et al., 2022). Additionally, the acquisition of mesenchymal characteristics enhances the capability of cancer cells to infiltrate blood vessels and lymphatics, facilitating metastasis to remote organs. These factors collectively create a formidable barrier to successful treatment intervention (Fontana et al., 2023). Extensive studies have been conducted to explore the complex web of signaling pathways that are pivotal in the unabated advancement of lung cancer. These findings highlight the need for focused therapy efforts aiming at blocking these pathways and preventing tumors from spreading uncontrollably. The identification and comprehension of these signaling pathways are critical in the ongoing efforts to produce more effective and precisely tailored treatments for lung cancer, addressing the complexity of the molecular landscape and increasing the chances of better patient outcomes (Quail & Joyce, 2013).
The EMT is a biological process that plays a critical role in several physiological and pathological conditions, including cancer progression (Nisticò et al., 2012). In this context of cancer, EMT is an intricate cellular program that influences the conversion of epithelial into mesenchymal cells. This transition is defined by changes in cell morphology, loss of cell–cell adhesion, increased cell motility, and enhanced invasive capabilities (Kalluri & Weinberg, 2009). The molecular mechanisms underlying EMT in cancer involve a variety of signaling pathways and transcription factors. Transforming growth factor-beta (TGF-β) is an essential cytokine that induces EMT by activating downstream signaling cascades (Moustakas & Heldin, 2016). Other signaling pathways, such as Wnt (Gonzalez & Medici, 2014) and Notch (Wang et al., 2010), also contribute to the regulation of EMT in cancer. Various transcriptional factors, namely Snail, Slug, and Twist, perform crucial responsibilities in orchestrating genetic reprogramming during EMT (Garg, 2013). These factors repress epithelial genes while activating mesenchymal genes, driving the transition process. Furthermore, alterations in the expression of cell adhesion molecules, such as N- and E-cadherin, signal the shift from an epithelial to a mesenchymal phenotype (Huang et al., 2022).
Plant-derived compounds offer great promise for treating a variety of ailments while causing no harm in normal cells. The fascination for scientists in this field stems from phytochemicals’ unique qualities, which include the ability to alter cell cycle control, avoid apoptosis, and influence crucial processes such as angiogenesis and metastasis. The acceptance of supplementary alternative medicines has increased significantly over the last 20 years, particularly in nations such as India and the United States. Surprisingly, medicinal plants and other natural resources account for over 60% of all anti-cancer drugs (Aruchamy & Duraisamy, 2021). Melezitose is a type of sugar that belongs to the group of oligosaccharides, specifically a trisaccharide: α-
Materials and Methods
Reagents
Himedia and Abcam supplied Dulbecco’s Modified Eagle’s Medium (DMEM), fetal bovine serum (FBS), propidium iodide (PI) stain, and penicillin/streptomycin antibiotics. The primers for claudin 1 (CLD-1), E-cadherin, SNAIL1, SLUG, and vimentin (VIM) were obtained from Eurofins Genomics India Pvt. Ltd., India. Melezitose was obtained from Sigma-Aldrich, India. The ribonucleic acid (RNA) isolation kit was sourced from Invitrogen, USA.
Cell Line
The A549 cell line (human lung adenocarcinoma) was procured from the National Infrastructure of Cell Line Resource (Beijing, China). Cultivation and maintenance of A549 cells occurred at 37°C within a controlled and humidified environment using a CO2 incubator. To support cell growth, DMEM supplemented with 10% FBS and 1% penicillin-streptomycin antibiotics were employed.
Cell Viability—MTT Assay
The impact of melezitose on cytotoxicity against the A549 cell line, a model for lung cancer, was investigated using the MTT colorimetric assay (Jayaraman, Natarajan, Veeraraghavan, et al., 2023; Jayaraman, Natarajan, Ponnusamy, et al., 2023). A seeding of 1 × 104 A549 cells in serum-free media was exposed to different concentrations of melezitose ranging from 0 to 150 µM. Following the addition of diluted melezitose samples, the A549 cell-containing wells were placed in a CO2 incubator for a 48-hour incubation period. Subsequently, the MTT solution was introduced, and after removing the supernatant, the formed formazan crystals were dissolved by adding DMSO and incubating for an additional hour. The optical density was then measured at 590 nm. Morphological images at 20× magnification were captured using an inverted microscope with a Zeiss camera.
Cell Cycle Analysis by Flow Cytometry
To analyze the distribution of deoxyribonucleic acid (DNA) content across various cell cycle phases, the flow cytometric technique was employed (Jayaraman, Natarajan, Veeraraghavan, et al., 2023; Jayaraman, Natarajan, Ponnusamy, et al., 2023). A549 cells were seeded, and treatment with melezitose was carried out at its IC50 concentration, with each well containing approximately 1 × 106 cells. Following a 24-hour incubation, the cells underwent a 48-hour treatment period before being fixed overnight in 70% ice-cold ethanol. Subsequently, the fixed cells were subjected to treatment with RNase A at a concentration of 10 mg/mL, and post-treatment, they were stained with PI dye. The fluorescence released by PI-labelled nuclei was evaluated employing a BD FACS Caliber flow cytometer from Becton Dickinson, USA, and the resulting data were quantified.
Estimation of Enzymatic Antioxidant and Lipid Peroxidation Level
The cells were obtained by trypsinization. Phosphate-buffered saline (PBS) was used to suspend the pellet. The supernatant was used to test enzymatic antioxidant activity, specifically superoxide dismutase (SOD) (Kakkar et al., 1984) and catalase (CAT) (Sinha, 1972), as well as to determine the extent of lipid peroxidation by TRABS assay (Niehaus & Samuelsson, 1986).
Cell Migration Assay
A549 cell was seeded in 6-well plates and allowed to attain confluency. Then, using a micropipette, a scratch was made in the center of the plates. The cells were washed with PBS to remove the detached cells. Then, the cells were incubated with or without melezitose. An image was immediately taken for consideration as 0 hours, and the cells were allowed to grow. After 48 hours, the images of melezitose-treated and untreated cells were captured under an inverted microscope (Motic, Japan).
Analysis of the TGF-β/Smad2 Using Enzyme-Linked Immunosorbent Assay (ELISA) Assay
Melezitose-treated and untreated cells were collected. The samples were used for the analysis of the TGF-β and Smad2 signaling molecules as per protocol prescribed by the manufacturing company, Invitrogen.
Gene Expression by Reverse Transcription Polymerase Chain Reaction (RT-PCR)
Each well was seeded with A549 cells at a volume of 5 × 104 per well. Subsequent to an incubation period, the cells were treated with melezitose in serum-free media at their respective IC50 values for 48 hours. RNA extraction from A549 cells was carried out using the TRIR kit from ABgene, UK. Spectrophotometric measurement was used to determine the extracted RNA’s concentration. Specifically designed for this investigation, Table 1 provides details on the forward and reverse primers. The CFX96 Touch Real-Time PCR detection apparatus was employed for this technique.
Reverse Transcription Polymerase Chain Reaction (RT-PCR) Primers.

Statistical Analysis
GraphPad Prism 8 was used to do the statistical analysis of the data, and the findings are shown as mean ± standard deviation (SD).
Results
Melezitose Reduces Cell Growth in A549 Lung Cancer Cells
The impact of melezitose on A549 cells was investigated through the MTT assay, assessing cell viability. Melezitose was administered to A549 cell line cultures at concentrations ranging from 0 to 150 µM for 48 hours. The results indicated a notable dose-dependent reduction in cell viability, with the cytotoxic effect of melezitose becoming more evident following the 48 hours (Figure 1A). Post-treatment, the calculated IC50 was determined to be 95.18 µM after 48 hours, signifying the concentration at which half-maximal inhibition occurred. Morphological assessments, as depicted in Figure 1B, further corroborated the substantial decrease in A549 cell viability induced by melezitose, evident in cell blebbing. These findings collectively underscore the robust cytotoxicity of melezitose against A549 cells, emphasizing its potential as a new therapeutic avenue to treat lung cancer.
A. MTT Assay was Assessed to Evaluate the Efficiency of Melezitose (0–150 µm) for 48 Hours of Treatment. IC50 Value was Determined Using GraphPad Prism 8 Software. B. A549 Morphological Images (20× Magnification) Compared with Control and Melezitose Treated for 48 Hours Time Interval. C. 2D Structure of Melezitose PubChem ID: 92817.
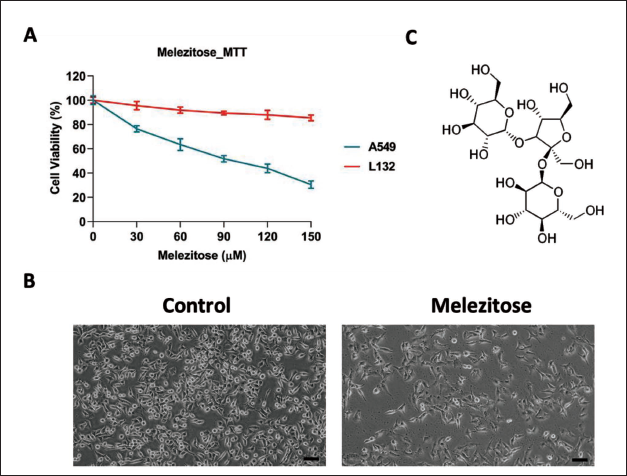
Melezitose Induces G1 Phase Cell Arrest in A549 Cells
Using flow cytometry to evaluate DNA content and cell cycle distribution produced some interesting results. After 48 hours of melezitose treatment, the proportion of A549 cells arrested in the G0/G1 phase increased significantly to 69.90%. On the other hand, 47.48% of the cells in the G0/G1 cell cycle phase were present in the untreated control group (Figure 2). These data clearly show that melezitose, especially in the G0/G1 phase, causes a substantial cell cycle halt in A549 cells. These findings highlight melezitose’s effectiveness in preventing cell division and offer the drug’s ability to slow the growth of lung cancer.
Cell Cycle Analysis by Treated Melezitose in A549 Cells.
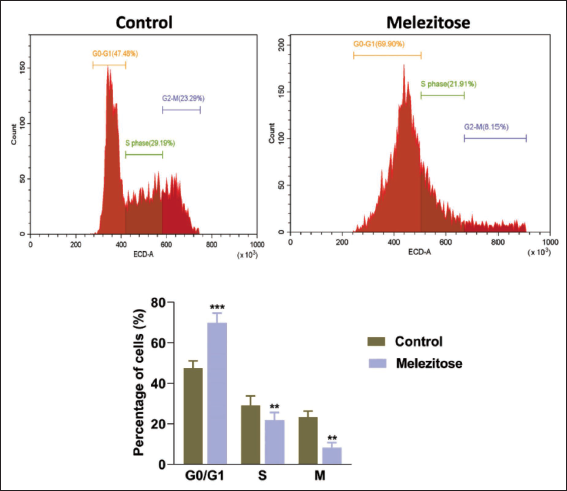
Melezitose Induce the Reactive Oxygen Species (ROS) in Lung Cancer Cells
As seen in Figure 3, we examined the concentrations of antioxidant enzymes like SOD and CAT assays as well as the production of ROS using the lipid peroxidation assay in the groups that received melezitose treatment. When compared to the control (untreated groups), melezitose exposure led to significantly reduced levels of antioxidant enzymes like SOD and CAT as well as increased lipid peroxidation in the lung cancer A549 cell line.
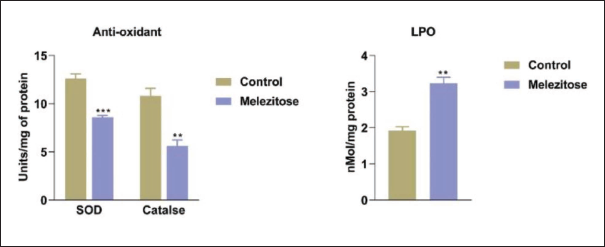
Melezitose Inhibits Cell Migration
We evaluate the cancer cell migration inhibitory effect of melezitose in A549 cells compared with the control group depicted in Figure 4. The results indicate a significant inhibition of cell migration by melezitose. The treated cells exhibited a notably reduced ability to close the scratch area compared with the control group. This observation suggests that melezitose effectively hinders the migratory capacity of the cells, indicating a potential anti-metastatic effect.
Cell Migration Assay. The Cells were Treated with Melezitose for 48 Hours. Images were Captured at 20× Magnification.
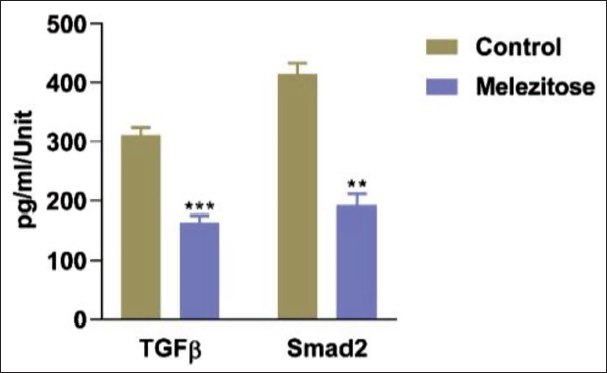
Melezitose Inhibits the TGF-β/Smad2 Signaling in A549 Cells
The results of our study indicate that melezitose effectively reduces TGF-β/Smad2 signaling, as analyzed through ELISA as shown in Figure 5. The ELISA analysis revealed a significant decrease in the levels of TGF-β/Smad2 signaling components in melezitose-treated group compared with the control (untreated group).
Transforming Growth Factor-Beta (TGF-β)/Smad2 Signaling was Analyzed by Enzyme-Linked Immunosorbent Assay (ELISA) Method. The Data are Represented as Mean ± Standard Deviation (SD) of Three Independent Experiments in Each Group. Statically Different as Mention **p < 0.01, ***p < 0.0001.
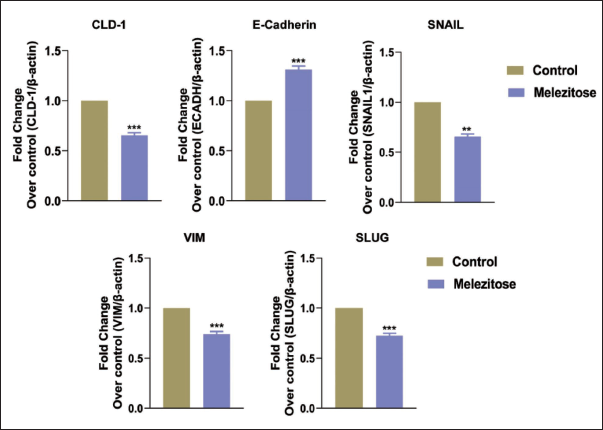
Melezitose Regulates the EMT Pathway in Lung Cancer Cells
Furthermore, our study aimed to unravel the underlying mechanisms behind melezitose’s ability to impede migration and invasion, coupled with its capacity to induce cell cycle arrest in A549 cells. The investigation involved employing RT-PCR to quantify messenger ribonucleic acid (mRNA) expression levels of genes associated with metastasis in A549 cells. The results yielded significant insights, shedding light on melezitose’s role in inhibiting metastatic properties in A549 cells. Administration of melezitose led to a notable reduction in the expression of genes crucial to the EMT regulation in A549 cells, including CLD1, SNAI1, SNAI2, and VIM mRNA levels (Figure 6). These results strongly imply that melezitose may be essential for stopping metastasis. The observed inhibitory effects on EMT-related genes provide compelling evidence for the potential of melezitose as an effective anti-cancer drug, particularly in impeding the EMT in lung cancer cells.
Gene Expression for Epithelial-Mesenchymal Transition (EMT) Targets (CLD1, ECADH, SNAIL, SLUG, and VIM) by Reverse Transcription Polymerase Chain Reaction (RT-PCR). The Data are Represented as Mean ± Standard Deviation (SD) of Three Independent Experiments in Each Group. Statically Different as Mention **p < 0.01, ***p < 0.0001.
Discussion
As targeted therapy has grown in popularity, chemotherapy remains the most effective treatment for advanced lung cancer despite the substantial side effects that frequently limit its clinical use. As a result, doctors are increasingly turning to herbal medicine as an adjuvant therapy due to its perceived safety and efficacy (Yin et al., 2013). Melezitose, a trisaccharide-based molecule (Figure 1C), has a variety of biological actions, including anticancer properties. The purpose of this study is to evaluate the possible therapeutic effect of melezitose against lung cancer, utilizing the A549 cell line as a model.
Initially, we examined melezitose cytotoxicity against the lung cancer A549 cell line and the human normal lung epithelial cell line L132. Interestingly, we found that melezitose has no toxicity for the L132 cell line, although it does display cytotoxicity depending on the dosage of melezitose. The study indicated that phytocompound concentrations significantly limit cancer cell proliferation, with an IC50 value of 95.18 µM. Morphological examination of the lung cancer A549 cell line shows that the control cells’ morphology and population remain unchanged. In the melezitose-exposed group, the A549 cell population was drastically reduced with changed cell architecture like cell blebbing, cell shrinkage, and nuclear condensation, which is connected to the apoptotic cell death pattern (Brauchle et al., 2014).
Moreover, our findings demonstrate that melezitose exhibits potential anticancer effects against lung cancer through its ability to induce a substantial amount of ROS within cancer cells. Previous studies of fucoidan, brown algae, have shown the ability to induce ROS mediated by suppressing the antioxidant enzyme level with increased free radicals in cancer to cause cell death in several cancers, namely, breast cancer (MCF-7) (Banafa et al., 2013), bladder (5637) (Han et al., 2017), liver (SMMC-7721) (Yang et al., 2013), and colon (Caco-2) (Narayani et al., 2019). Similarly, we observed mechanisms involving the inhibition of two key antioxidant enzymes, SOD and CAT, leading to an increase in intracellular ROS levels. One of the notable outcomes of melezitose-induced ROS is the initiation of lipid peroxidation, a chain reaction that damages cellular membranes and disrupts their structural integrity. The elevated ROS level can result in damage to cellular components by free radicals, potentially causing binding and configuration changes or damaging biomolecules, such as DNA fragmentation, and protein dysregulation (Checa & Aran, 2020). In our study, melezitose induces ROS, leading to DNA fragmentation to inhibiting lung cancer progression.
Furthermore, the cell cycle is a tightly regulated process that governs the orderly progression of a cell from its formation to division, ensuring the faithful replication of genetic material and the maintenance of cellular integrity (Ding et al., 2020). In cancer cells, alternations in the key regulatory proteins and signaling pathways disrupt the delicate balance between cell growth and cell cycle arrest. Mutation in tumor suppressor genes, such as p53, as well as overexpression of oncogenes, can result in aberrant cell cycle progression (Chen, 2016). The G1 checkpoint, which monitors DNA integrity and cell size before committing to DNA replication, is frequently in cancer cells, allowing them to enter the S phase even in the presence of DNA damage (Barnum & O’Connell, 2014). A previous report by Hamadou et al. (2023) revealed that melezitose has high binding energy with P53 protein, which stimulates the cell cycle arrest in the MCF-7 breast cancer cell line. Similarly, our flow cytometry data reveal that melezitose can induce cell cycle arrest at the G0/G1 phase. This observation suggests that melezitose possesses significant potential to inhibit the progression of lung cancer by triggering cell death mediated by ROS and inducing cell cycle arrest.
Moreover, targeting TGF-β mediate cell migration is a crucial aspect of cancer therapy, as it directly impacts the metastatic potential of tumor cells. In our study, we observed a significant inhibition of cell migration, and this effect can be attributed to the modulation of the TGF-β pathways by melezitose.
TGF-β is a multifunctional cytokine known to play a dual role in cancer progression, acting as both a tumor suppressor and a promotor of tumor metastasis, depending on the context and cellular milieu. In many cancer types, including lung cancer, TGF-β signaling is often dysregulated, leading to increased cell migration, invasion, and metastasis. Our finding indicates that melezitose intervenes in the TGF-β signaling pathway, resulting in the inhibition of cell migration. The inhibition of TGF-β signals through the Smad family of proteins, and disruptions in this signaling cascade, can impede the induction of genes associated with cell migration and invasion. Our result results indicate that targeting TGF-β signaling can be an effective strategy to hold the cancer cell motility. EMT plays a critical role in the progression of lung cancer, contributing to increased invasiveness and metastatic potential of tumor cells. Several key molecular players have been identified in the intricate orchestration of EMT, including vimentin, Slug, Clauidn-1, E-cadherin, and Snail. Vimentin, an intermediate filament protein, is often upregulated during EMT and is associated with the acquisition of a mesenchymal phenotype in cancer cells. Slug, a zinc finger transcription factor, acts as a potent repressor of E-cadherin, a key component of epithelial cell–cell adhesion. The downregulated of E-cadherin is a hallmark of EMT and is closely linked to increased motility and invasiveness of cancer cells. Additionally, CLD1, a member of the claudin family of tight junction protein, has been implicated in EMT-associated changes in cell–cell interactions and permeability. Snail, another transcription factor, has an influenced role in repressing E-cadherin expression, facilitating the transition of cells from an epithelial to a mesenchymal state (Ikenouchi et al., 2003; Xiao & He, 2010; Zhuo et al., 2008).
The dysregulation of these EMT-associated molecules in lung cancer promotes a phenotypic shift toward a more invasive and migratory state, contributing significantly to disease progression and metastasis. According to Karacosta et al. (2019), it was revealed that the induction of the EMT process stimulates cancer progression in three non-small-cell lung carcinoma (NSCLC) cell lines (HCC827, A549, H3255) via the addition of TGF-β. Another study conducted by Guo et al. (2021) suggests that the EMT process can enhance tumor invasion and confer immunotherapy resistance via Zeb1 transcriptional factors. Targeting the EMT process emerges as a promising option to inhibit lung cancer progression. In our study, we investigate how melezitose modulates EMT-associated genes through RT-PCR analysis. Melezitose was found to suppress Snail, leading to the upregulate of E-cadherin and a reduction in the vimentin expression, thereby inhibiting lung cancer progression. A study by Hsu et al. (2014) demonstrated that fucoidan, a polysaccharide found in brown seaweed, can inhibit lung cancer progression via the Smurf2-dependent ubiquitin degradation of TGF-β receptors, thereby repressing the EMT process. Consequently, melezitose, a trisaccharide, demonstrates anticancer efficacy. Recently, the focus of researchers on carbohydrate and amino acid-based anticancer drug development has increased, underscoring the potential of these compounds in cancer therapy (Hossain & Andreana, 2019).
Conclusion
When combined, melezitose causes lung cancer A549 cell line cell proliferation, antioxidant state, lipid peroxidation, and cell cycle arrest to all decrease, which starts the cancer cell death process. The current study’s findings unequivocally show that melezitose has a therapeutic impact on lung cancer in an
Abbreviations
EMT: Epithelial-mesenchymal transitions; LUAD: Lung adenocar cinoma; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; CLD1: Claudin 1; ECADH: E-Cadherin; VIM: Vimentin; TGF-β: Transforming growth factor-beta; LPO: Lipid peroxidation; SOD: Superoxide dismutase; CAT: Catalase; ROS: Reactive oxygen species.
Author Contributions
QZ and ZW—manuscript drafting and experimental design; XC and ZZ—experimental analysis; CF—review the manuscript.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not Applicable.
Funding
Regulation and molecular mechanism of acute lung injury induced by Tollip (EZ01-002-20210137).
