Abstract
Psoralea corylifolia (PC) seed extract (PCE) has been extensively utilized as an herbal drug due to its favorable effects on human health. It’s extracts and active components have demonstrated a variety of biological actions including estrogenic, anticancer, antioxidant, anti-bacterial, anti-depressant, anti-inflammatory and anti-ageing properties. A study of this medicinal herb has major constituents extracted using different plant parts has been depicted and among them, Bakuchiol has the highest ethyl acetate extract content. The mechanism behind PCE’s anti-lipotoxic activity against palmitate (PA)-induced lipotoxicity in PC12 cells was examined. CE therapy not only raised the quantities of mRNA for antioxidant genes such as heme-oxygenase and nuclear factor (erythroid-derived 2)-like 2 but also lowered the production of reactive oxygen species caused by PA. Prenylflavonoids play a unique role in the treatment of Alzheimer’s disease (AD) and can be used as a nootropic agent. This article comprehensively enumerated the literature on the anti-apoptotic effects of PC and its potential pathogenesis in AD.
Introduction
Psoralea corylifolia (PC) L., a member of the Fabaceae family and also known as Babchi, is an erect herb that grows annually. Height varies between 30 and 180 cm and the tiny flowers are shaped like red clover (Alam et al., 2018). It consists of a solitary seed, which is dark brown in colour and elongated in shape with a smooth surface. Some other nomenclatures of this botanical specimen include Cullen corylifolium, Lotodes corylifolia, Psoralea patersoniae and Trifolium uniflorum (Alam et al., 2018; Brands, 1989). It flourishes throughout India’s plains, particularly in Rajasthan’s semiarid regions and Punjab’s eastern districts that border Uttar Pradesh. It is also found throughout India, including in the Himalayas, Dehradun and Karnataka. Additionally, this plant is widely dispersed throughout the world’s tropical and subtropical climates, particularly in China and southern Africa (Alam et al., 2018; Khushboo et al., 2010). The seeds and oil of this herb consist of a number of medical advantages. People sometimes use this plant because it has diuretic and laxative qualities, but they also use it as a stimulant or even a diaphoretic. It is regarded as a good tonic herb by the Chinese for boosting general well-being and vigour (Dey, 1896). The fruit of this plant is thought to have aphrodisiac effects and can be used as a genital organ tonic. With a wealth of flavonoids and meroterpenes, the active fraction isolated from fruits, seeds and roots has anti-bacterial, anti-oxidative and immunomodulatory effects. It has been proven that PC seed extracts (PCEs) significantly reduce cytotoxicity and may be used as therapeutic agents to treat neurological diseases (Im et al., 2014).
A serious threat to human health is presented by neurodegenerative diseases. These age-related illnesses are becoming more common, partly due to the rise in the older population in recent years (Heemels, 2016). Parkinson’s disease (PD), frontotemporal dementia, spinocerebellar ataxias, Huntington’s disease (HD), Alzheimer’s disease (AD) and amyotrophic lateral sclerosis (ALS) are a few examples of neurodegenerative disorders. These illnesses include a variety of pathophysiologic symptoms; some impair memory and cognition while others have an impact on a person’s strength to communicate, walk and sight (Abeliovich & Gitler, 2016; Canter et al., 2016; Gitler et al., 2017; Taylor et al., 2016; Wyss-Coray, 2016). To understand the root system of each disease is the only way to develop effective treatments, which are urgently needed. As a result of their widespread usage as a model for brain development, PC12 cells have been utilized to investigate how differentiation affects exocytotic release, particularly release at varicosities (Zeliger, 2015). As the quantal size increases dexamethasone-differentiated cells have also been shown to have an increased number of releasable vesicles, opening the door to large areas of applications, including neurotoxicological and pools of vesicles, each about half the total number of releasable vesicles and one about twice the submaximal size of the other are present in PC12 cells displaying high numbers of events. It is expected that PC12 cells will continue to be used as a model to investigate exocytosis in the coming decade because they offer an advantage compared to other neurosecretion models, such as chromaffin cells (Westerink & Ewing, 2008; Westerink et al., 2000).
There are two categories for neurosecretions: To affect nearby cells, neurotransmitters are released into the synaptic cleft. They have a brief lifespan and range. When neurohormones are released into circulation, they can travel farther. Certain neurotransmitters are also neurohormones (Kostal et al., 1999).
The objective of the current article is to provide a comprehensive review of PC including its ethnophar macological properties, major constituents present in different plant parts, their phytochemistry and special emphasis on its neuroprotective effects and their mechanism. Hopefully, our analysis will serve as a solid scientific foundation for more in-depth research on PC pure chemicals, which could result in the discovery of novel medicinal uses.
PC: An Indigenous Traditional Medicinal Herb
PC has many ethnopharmacology and medicinal utilization. Most parts of the plant like roots, leaves, stems, flowers and seeds are used in treating various kinds of disease (Chishty & Bissu, 2016). The plant is frequently employed as a pigment, heart tonic, vasodilator, anti-cancer, anti-bacterial, cytotoxic and anti-helminthic in Ayurvedic and Chinese Medicinal systems (Alam et al., 2018). Oleoresin is a compound used to treat lumbago and some kidney diseases (Lim et al., 2011). The plant can be used both externally and internally. PC seeds are used orally to treat a variety of illnesses, including intestinal worm infections and can cause unpleasant side effects like headaches, nausea, diarrhoea and vomiting if consumed in excess (Alam et al., 2018). Essential oil is the seed’s active ingredient and can be used to treat psoriasis, tinea versicular, scabies, ringworm and other skin conditions such as vitiligo (Shrestha et al., 2018).
Phytoconstituents and crude extracts from diverse plant sections have been found to have a variety of biological functions, according to pharmacological and biochemical studies described below:
Seeds are brownish-black sticky oily pericarp covering seeds (Alam et al., 2018). Most of the PC active ingredients that have been isolated so far are found in the seed. Monoterpene phenol, Bakuchiol (BAK) and the two novel dimeric monoterpenoids bisbakuchiols A and B are three crucial components that were extracted from seeds of PC (Alam et al., 2018; Panda, 1999). In indigenous systems of medicine, the plant’s seeds are employed as laxatives, aphrodisiacs, anthelmintics and diaphoretics in feverish situations due to their superior therapeutic potential. For inflammatory skin problems like eczema, leprosy, psoriasis hair loss, leukoderma and other conditions utilize seed in the form of ointment or paste via both oral and external administration for treatment (Cui et al., 2015; Singh & Agrawal, 2022). The crude methanolic seed extract of this herb was evaluated in vitro and demonstrated considerable antimycobacterial activity against Mycobacterium aurum and Mycobacterium smegmatis (Husain et al., 2018; Ling et al., 2013).
Fruits are sticky oily pericarp with hard coats and bitter in taste (Khushboo et al., 2010). In one of the studies, spectroscopic techniques were used to isolate and identify six novel flavonoid compounds and a meroterpenoid from fruit. It helps to prevent vomiting, used to treat febrile disease, piles, bronchitis, anaemia and premature ejaculation (Ibrahim et al., 2013). Traditional remedies for this plant’s fruits include treatment for psoriasis, vitiligo, gynaecological, haemorrhage and frequent urination (Cui et al., 2015; Ma & Meredith, 2021).
Leaves—Simple, round and mucronate leaves at the apex. It contains raffinose, psoralen and isopsoralen. The use of leaves helps to treat diarrhoea (Singh & Agrawal, 2022).
Roots—PC roots have been examined for bioactive compounds such as Psoralen and Isopsoralen. Roots are used to treat various dental problems and to encourage the calcification of the bone, making them effective in the treatment of osteoporosis and bone fractures (Singh & Agrawal, 2022).
Pharmacologically Bioactive Ingredients of PC
PC comprises a variety of phytochemicals notably lipids, chalcones, coumarins, meroterpenes, flavonoids, resins, stigmasteroids and coumarins (Alam et al., 2018). Their nature and quantity vary depending on the climatic conditions. Researchers discovered that in comparison to other plant parts, seeds had the highest concentration of active ingredients (Table 1) (Tang et al., 2004). Major constituents extracted from various parts of the plant are Bavachinin (BCN), Bavachin, Corylifol A, Isobavachalone, Corylifol B, BAK and Psoralen which are mentioned in (Table 1) along with their structure, content, method of analysis, parts of the plant they are extracted, the solvent used and chemical nature of compounds (Ruan et al., 2007). The method of extraction is the most important parameter to obtain a pure compound from the crude extract and different plant parts. Researchers reported that the concentration of isolated compounds varies with the different solvent systems (Stéphane et al., 2021). This observation was made to analyse from which part of the plant, a particular compound was found in its highest concentration and which solvent system is suitable for targeted compound isolation (Liu et al., 2004). Taking, for instance, BCN obtained from PC seeds through high-performance liquid chromatography (HPLC) using a solvent system, 70% ethanol has a higher yield ratio when compared to ethyl acetate extract (Chopra et al., 2013; Wang et al., 2013). Among the estimated compounds, BAK has the highest ethyl acetate extract content (Alam et al., 2018). BCN, which is biogenetically produced from bavachin, has a substantially higher level. Corylifol B and Isobavachalcone (IBC) both had substantial concentrations in the extract (Cui et al., 2015).
Major Phytochemicals Present in Psoralea corylifolia Plant and Their Method of Extraction.
Neuronal Apoptosis
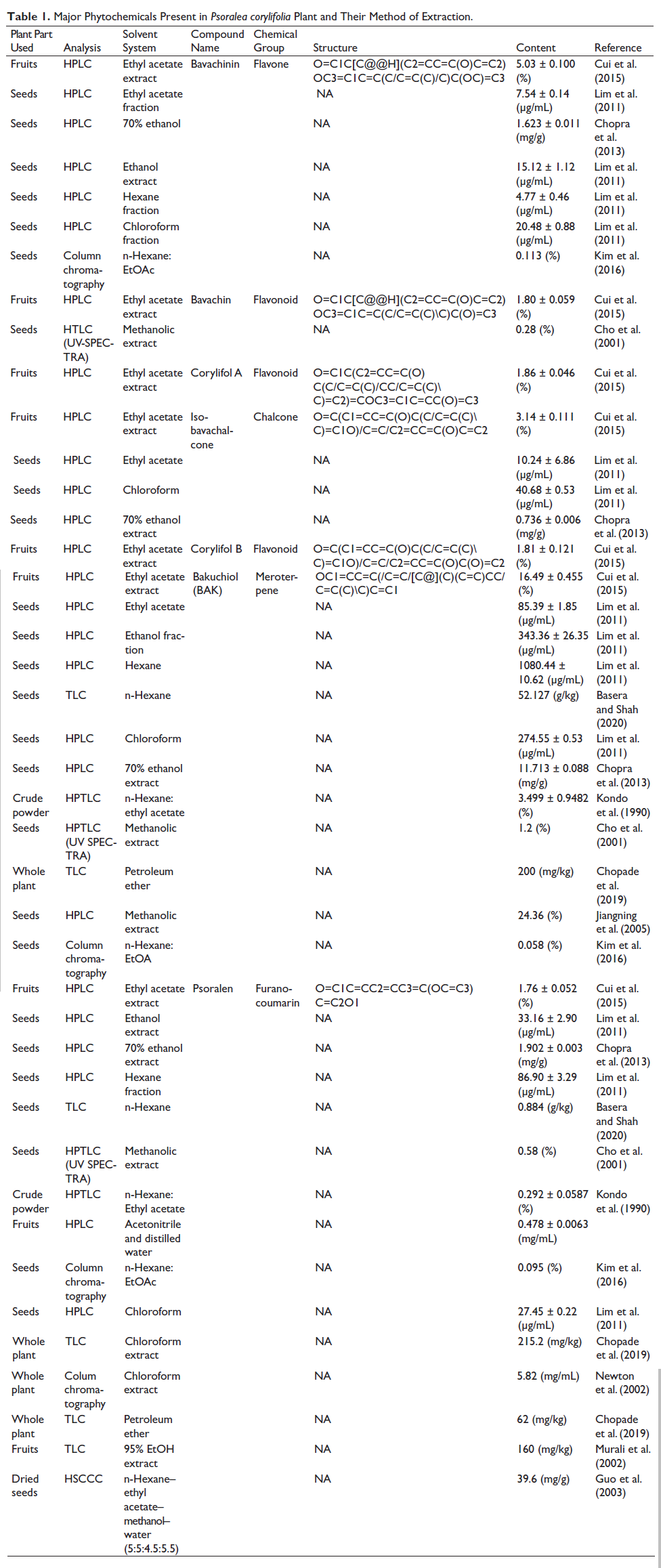
Unlocking the enigma of neuronal demise and delving cell death. A neuron orchestrates its demise through a process called neuronal apoptosis, which is considered an inbuilt suicide program. Physiologically appropriate neuronal loss leads to a sculpting process that eliminates around half of all neurons created during neurogenesis during normal nervous system development. Following this developmental window, neuronal loss is physiologically inappropriate and can have a role in neurodegenerative diseases like Alzheimer’s (Figure 1) and Parkinson’s (Karvandi et al., 2023). Therefore, illuminating the molecular processes causing neuronal apoptosis may aid in clarifying the underlying causes of human neuropathology and aid in the discovery of new therapeutic and preventative approaches (Kawahata & Fukunaga, 2023).
The Neuronal Demise in Neurodegenerative Disorders Such as Alzheimer’s Disease Cerebral Aβ Pathological Interaction with Transition Metals Causes Oxidative Stress, Disturbed Cellular Ca2+ Balance and Altered Energy Metabolism, All of Which Increase the Production of ROS and Cause the Death of Neurons (Benilova et al., 2012; Nakano et al., 2020).
Physiological process, neuronal apoptosis occurs during neurogenesis in the central nervous system (CNS) and abnormal apoptosis has been linked to the aetiology of neurodegeneration (Ekshyyan & Aw, 2004). A lot of research has been done on the regulation and/or activation of apoptosis as a result of the realization that lack of apoptotic control may be the basis for disease aetiology.
Different Forms of Cell Death Pathways
During typical neuronal development, tightly controlled programmed cell death (PCD) signalling events take place in a geographically and temporally constrained way. Neurodegenerative illnesses like ALS, AD, HD and PD are frequently characterized by aberrant activation of PCD pathways, which leads to an unintended loss of neuronal cells and function (Garofalo et al., 2020; Moujalled et al., 2021). On the other hand, the inactivation of PCD is believed to influence brain cancer growth and therapy response (Bialik et al., 2018) (Table 2).
Morphological and Biochemical Signatures Across Diverse Cell Death Pathways.

Apoptosis
Cell shrinkage is a hallmark of the active, tightly controlled form of cell death known as apoptosis. When the CNS is developing, it arbitrates the accurate and predetermined neuron’s natural death and is a crucial physiological mechanism in neurogenesis. Although neurodegeneration is a complex phenomenon due to premature apoptosis that results in several chronic disease conditions, including spinal muscular atrophy, HD, ALS, PD (diabetic encephalopathy) and AD. A cell must reach an adenosine triphosphate (ATP) threshold in order to undergo apoptosis. Necrosis replaces apoptotic cell death when ATP depletion is severe (Yuan & Yankner, 2000).
Autophagy
Unlike necrosis and apoptosis, autophagy is a type of caspase-independent cell death where lysosomal enzymes break down the cytoplasm. Lysosomes catabolize broad protein complexes or intracellular organelles as part of the evolutionarily conserved system known as autophagy, which helps the cells remove damaged organelles and remodel cells during development and differentiation (Roth & D’Sa, 2001). When nutrients are limited, mitochondrial ATP generation uses recycled membrane lipids and proteins, which led to the initial description of autophagy in yeast as a pro-survival mechanism. Autophagy can play a role in cell death after oxidative stress or famine (Buja et al., 1993; Chaabane et al., 2013). Autophagy exhibits the capacity for trichotomous differentiation, delineated as macro-autophagy, micro-autophagy and chaperone-mediated autophagy (Yang & Klionsky, 2020). Each of these physiological processes remains discrete in their operational mechanisms; nevertheless, they collectively culminate at lysosomal compartments for the purpose of cargo degradation and the recycling of intracellular constituents (Doherty & Baehrecke, 2018).
Necroptosis
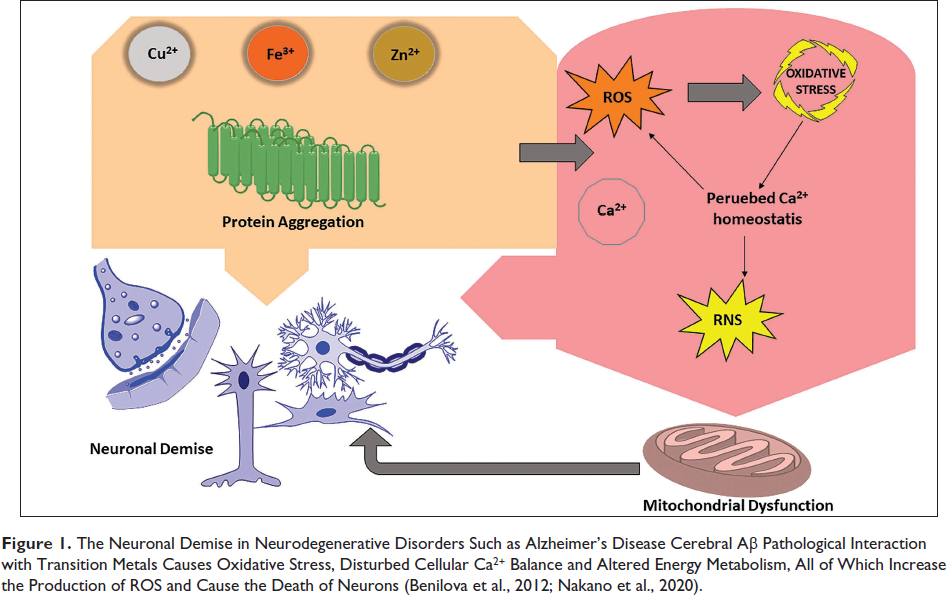
Necroptosis is a kind of necrosis that triggers innate immune responses by rupturing dead cells and releasing intracellular components; it can be caused by Toll-like receptor (TLR)-3 and TLR-4 agonists, tumour necrosis factor (TNF), certain microbial infections and T cell receptors (Lee et al., 2016). The following characteristics of necroptosis: (a) It mimics necrosis when dead cells are grouped together and have membranes ruptured, their cell bodies and organelles enlarged and their chromatin fractured; (b) an abundance of inflammasomes; (c) necroptotic cells passively flow through the damaged membrane into the extracellular matrix during autophagy as opposed to apoptosis (Das et al., 2018). Through its potent pro-inflammatory qualities, necroptosis acts as a host defence mechanism, having antipathogenic and anti-cancer effects (Lee et al., 2016). According to recent investigations, necroptosis may contribute to the aetiology of neuroinflammatory disorders. Necroptosis inhibitors, such as necrostatin-1 (Nec-1) and stable Nec variants (Nec-1s), have demonstrated efficacy in treating a variety of neurological conditions. RIPK1-dependent transcription increases disease-associated microglia and lysosomal abnormalities to drive the buildup of amyloid plaques in AD. Microglia dysfunction is important to the aetiology of AD. After being phosphorylated by the kinase RIPK3, which is necessary for necroptosis, the pseudo kinase MLKL is transferred from the cytosol to the plasma membrane (Figure 2) (Lee et al., 2016).
Necroptosis is Affected by Different Receptors and Other Factors.
Necrosis, an alternative cell demise mechanism, manifests as cellular swelling, leading to membrane disruption and intracellular content release. While DNA damage plays a role in degradation, necrosis does not entail chromosome condensation (Chan et al., 2015; Kraupp et al., 1995). Traditionally, necrosis has been regarded as an inadvertent demise stemming from an excessive cytotoxic insult, devoid of requisite molecular orchestrations. The pathological attributes of necrosis can be discerned from those of apoptosis (Krysko et al., 2008). The presence of TNF, capable of provoking necrosis, challenges this notion. The triggering of specific death receptors or TLRs initiates the process of necroptosis (Feoktistova & Leverkus, 2015).
Ferroptosis
Ferroptosis is an iron-dependent necrotic PCD. While it shares traits with traditional oxidative stress-induced cell death, it possesses distinct morphological and functional differences from generic oxidative stress, like hydrogen peroxide-induced necrosis (Dixon et al., 2012). Ferroptosis inducers comprise GPX4 inhibitors (e.g., RSL3, ML210, ML162, FIN56, FINO2), disruption of glutathione synthesis (buthionine sulfoximine), inhibition of system xCT to disrupt cysteine supply (erastin, sorafenib, sulfasalazine, glutamate) and iron-related agents. Endogenous ferroptosis inhibitors encompass glutathione, ubiquinone, vitamin E and selenium (Moujalled et al., 2021). Morphologically, ferroptosis is characterized by conspicuous mitochondrial shrinkage, augmented membrane density, and the diminishment or disappearance of mitochondrial cristae, setting it apart from other cellular demise pathways (Li et al., 2020; Yang & Stockwell, 2008). Excessive H2O2 reacts with cellular Fe2+, leading to Fenton reaction-derived reactive oxygen species (ROS) that induce intracellular LPOs and Initiate ferroptosis (Lučiūnaitė et al., 2020).
Pyroptosis
Nerve cell demise is no longer limited to apoptosis, autophagy and necrosis, as previously established. In the past few decades, the revelations regarding the gasdermin (GSDM) protein family and pivotal caspase molecules have introduced pyroptosis as an emerging form of PCD (Yue et al., 2022). Caspase-1 activation can be induced by inflammasomes, including NOD-like receptor proteins such as NLRP1, NLRP3, NLRC4, AIM2 (absent in melanoma 2) and pyrin, leading to the initiation of pyroptosis (Man et al., 2017). Aβ aggregation may stimulate NLRP1 inflammasome, triggering pyroptosis via caspase-1 and IL-1β release through the P2X7-purinoceptor/pannexin-1 pathway (Yang et al., 2019). During AD progression, microglial pyroptosis fosters the assembly of the apoptosis-associated speck-like protein containing a CARD (ASC) binding and cross-seeding extracellular Aβ (ASC-Aβ) complex. This complex not only amplifies Aβ oligomerization and aggregation but also triggers NLRP3 inflammasome formation, caspase-1 activation, IL-1β maturation, GSDMD cleavage, and induces pyroptosis in neighbouring microglia (Heneka et al., 2018; Liao et al., 2023).
Adult human brain mature neurons, in contrast to many somatic cells, are resistant to a variety of stressors and pro-apoptotic triggers, such as the absence of neurotrophic chemicals. Only a small number of neurons is lost as we grow old and numerous classical apoptotic molecules have been associated with the pathophysiology of neurodegenerative illnesses as mature neurons must somehow be able to avoid cell death pathways. One can hypothesize that cell death and ageing, which is frequently accompanied by a decline in motion- and cognition-based activities similar to the clinical signs of neurodegenerative illness may be causally related.
Several key factors drive these cell death pathways are as follows:
Excitotoxicity: Excitotoxicity results from neuronal overstimulation by neurotransmitters like glutamate, causing excessive calcium influx and cell death. It plays a role in ALS and ischemic stroke, as overactive glutamate receptors, particularly N-methyl-
Oxidative stress: Oxidative stress arises from an imbalance between ROS production and cellular detoxification capacity, causing harm to cellular elements like proteins, lipids and DNA. It is a prevalent factor in neurodegenerative diseases, including Alzheimer’s, and can impair neuronal function and viability.
Protein misfolding and aggregation: Neurodegenerative diseases involve the misfolding and aggregation of particular proteins, leading to cellular disruption and activation of cell death mechanisms. Substantial evidence strongly supports the theory that the buildup of misfolded proteins instigates synaptic dysfunction, neuronal apoptosis, brain injury and disease pathogenesis (Dong et al., 2009; Soto & Estrada, 2008).
Endoplasmic reticulum (ER) stress: ER stress can arise from the buildup of misfolded proteins within the ER, triggering the unfolded protein response (UPR). Prolonged or severe UPR activation can culminate in cell death. Caspase-4, located in the ER, undergoes proteolytic cleavage and activation. Numerous studies have shown that ER stress elevates both the expression and cleavage of caspase-4 across various experimental models (Hitomi et al., 2004). Activation of caspase-4 can initiate apoptosis, an inflammatory response, or a combination of both processes (Kim et al., 2006).
Mitochondrial dysfunction: Mitochondria play a vital role in energy generation within neurons. Their malfunction can result in energy depletion and the release of pro-apoptotic factors, ultimately contributing to neuronal demise (Wang et al., 2020).
Inflammation: Persistent neuroinflammation is a prevalent characteristic in numerous neurodegenerative disorders. Inflammatory mechanisms can contribute to neuronal demise by releasing inflammatory mediators and activating immune cells. TNF binding to TNFR1, which contains a death domain, and the subsequent assembly of complex 1 can result in either the initiation of NFκB signalling or cytotoxic outcomes, including apoptosis or necroptosis, upon the formation of complex 2. Apoptosis is initiated when receptor-interacting kinase 1 (RIPK1) undergoes deubiquitination facilitated by the CYLD protein, a deubiquitinating enzyme. Subsequently, RIPK1 is cleaved by activated caspase 8 (Moquin et al., 2013).
Aberrant protein aggregates are a common feature that appears in many neurodegenerative diseases. It is well acknowledged that the production and accumulation of these proteinaceous materials serve as a readout for numerically evaluating the severity of various neurodegenerative disorders, despite the fact that the composition and placement of aggregates vary (Melo et al., 2011).
Palmitate (PA)-induced Neuronal Apoptosis
The primary fatty acid present in palm oil is palmitic acid, which is present in PA (more technically known as retinyl PA). Saturated fatty acid intake in excess can cause neuroinflammation, ER stress and resistance to peripheral signals, which can ultimately result in hyperphagia, obesity, decreased reproductive function and disrupted circadian rhythms (Yu et al., 2021).
From PA de novo ceramide production occurs and induces Schwann cell death. In the cultured rat, primary Schwann cells and immortalized mouse Schwann cells (IMS), PA, C2-ceramide, a ceramide analogue and de novo ceramide synthesis inhibitors (myriocin and fumonisin B1) were used (Suzuki et al., 2011). PA in a dose-dependent manner was incubated for 48 hours, and this dramatically increased the amount of IMS apoptosis that could be seen by cell membrane inversion and nuclear staining. Myriocin and fumonisin B1 greatly but partially inhibited this increased apoptosis (Burhans, 2013; Moujalled et al., 2021). PA unmistakably activated caspase-3 in IMS, according to immunostaining and Western blot analyses. Despite a complete restoration in ceramide accumulation, reduction in PA-induced caspase-3 activation unexpectedly failed by the ceramide synthesis inhibitors (Listenberger et al., 2001).
In Schwann cells apoptosis is induced via a ceramide-mediated, caspase-3-independent method by PA as well as a ceramide-independent, caspase-3-dependent pathway because PA’s activation of the ER stress pathways eIF2 and X-box binding protein-1 was inhibited by a specific c-Jun N-terminal kinase (JNK) inhibitor, ER stress was brought on by PA administration via a JNK-dependent pathway (Listenberger et al., 2001).
Suitability of PC12 Cells for Neurosecretion
Rat phaeochromocytoma (PC12) cells were created concurrently as a substitute model to study exocytosis using amperometry. The extraordinary adaptability of PC12 cells for pharmacological manipulation, their simplicity in culture and the wealth of knowledge on their proliferation and differentiation are the primary reasons for their popularity. Additionally, they resemble neurons more closely than chromaffin cells, having smaller vesicles and quanta (Suzuki et al., 2011; Uchida et al., 2003; Westerink & Ewing, 2008).
It is common practice to utilize PC12 cells to examine a drug’s potential for neurotoxicity, for instance by evaluating the impact on the viability of cells, protein expression levels or DNA damage. The use of this cell line as a model for neurodegenerative illnesses is, nevertheless, very common. They are helpful in the study of physiology and pharmacology nerves since they have certain properties of neurons and are quite simple to passage and culture (Wiatrak et al., 2020).
There are two types of PC12 cells: One type is grown in suspension, and the other is derived from selected easily adherent cells (Oprea et al., 2022). Dopamine and norepinephrine (NE) are produced, stored and released by catecholamine cells also known as PC12 cells (Alemà et al., 1985; Das et al., 2004; Greene & Tischler, 1982).
These cells originate from a rare adrenal medulla tumour called pheochromocytoma, which produces neurotransmitters like dopamine, NE and epinephrine. They have been crucial in exploring the control of neurotransmitter production and release in nerve and hormone systems. The level of catecholamine synthesis and storage in PC12 cells can vary depending on several factors, including the stage of cell differentiation, culture conditions and experimental manipulations. The specific levels of catecholamine synthesis and storage in PC12 cells can be quantified through various biochemical assays, including enzyme activity assays, HPLC and radioimmunoassay.
A summary of PC12 cell catecholamine synthesis and storage is listed here.
Catecholamine synthesis: Catecholamines, including within PC12 cells, are produced via a sequence of enzymatic reactions. The synthesis pathway comprises these stages:
Tyrosine uptake: PC12 cells acquire tyrosine from their external surroundings, with tyrosine being an amino acid serving as the precursor for catecholamine production (Paravati et al., 2023).
Tyrosine hydroxylation: Within PC12 cells, tyrosine undergoes conversion into 1-dihydroxyphenylalanine (L-DOPA) through the action of the enzyme tyrosine hydroxylase. This step is critical and serves as a rate-limiting factor in catecholamine synthesis (Fernstrom & Fernstrom, 2007; Kuhn & Billingsley, 1987).
Decarboxylation: L-DOPA is subsequently transformed into dopamine through the enzyme aromatic 1-amino acid decarboxylase (Gnegy, 2012).
NE synthesis: Not all PC12 cells produce abundant DBH, leading to variable NE synthesis in different cell lines. Neurons utilizing NE as a signal carrier possess an extra enzyme, dopamine-β-hydroxylase, which transforms dopamine into NE. Neurons using epinephrine as a signal carrier feature an additional enzyme, phenylethanolamine-N-methyl transferase, responsible for converting NE to epinephrine (Gnegy, 2012; Kaufman, 1995; Nagatsu et al., 1964).
Catecholamine storage: Catecholamines are stored in specific compartments within PC12 cells called secretory vesicles or chromaffin granules (Rebois et al., 1980). The storage and release of catecholamines are highly controlled processes.
Uptake into chromaffin granules: Catecholamines are moved into chromaffin granules after their synthesis, a process facilitated by vesicular monoamine transporters (Montero-Hadjadje et al., 2013; Wimalasena, 2011).
Storage as complexes: Within chromaffin granules, catecholamines are housed in conjunction with a variety of proteins, including chromogranins and secretogranins. These partnerships shield catecholamines from breakdown and oversee their release (Montero-Hadjadje et al., 2013; Zhang et al., 2011).
Storage dependent on pH: The pH inside chromaffin granules is acidic, a condition that preserves catecholamines in their stable, storable states (Montero-Hadjadje et al., 2013).
Catecholamine release: Catecholamines stored in chromaffin granules can be discharged in reaction to different triggers, like nerve signals or stress (Burgoyne, 1995). The release process includes these stages:
Depolarization: Upon stimulation, the PC12 cell membrane depolarizes, causing the activation of voltage-gated calcium channels.
Calcium influx: Calcium ions flow into the cell through these channels.
Fusion of chromaffin granules: The rise in intracellular calcium prompts the fusion of chromaffin granules with the cell membrane, resulting in the discharge of catecholamines into the extracellular space. This mechanism is referred to as exocytosis.
Catecholamine release: Catecholamines, mainly dopamine and NE, are liberated from PC12 cells and can subsequently influence neighbouring cells or tissues.
Effect of 3-nitropropionic Acid (3-NP)
Consumption of 3-NP, a particular inhibitor of mitochondrial respiratory complex II, by animals, may lead to experiencing symptoms similar to HD. In previous studies, 3-NP inhibition of complex II resulted in neuronal cell death and mitochondrial fragmentation via N-methyl-
Brain neuron cells are more sensitive to oxidative stress than other types of cells in apoptosis and neural degeneration because of their dependence on oxidative phosphorylation as their main energy source (Bhute et al., 2020). Mitochondria are crucial because they are constantly exposed to high energy and oxygen levels and damage from oxidative stress. This stress brought on by mitochondrial electron leakage might result in cell death. Uncertainty surrounds the mechanisms driving oxidative damage-induced neuronal death, including whether mitochondrial abnormalities are the key contributor to the toxicity or merely a side effect of the damage (Liot et al., 2009; Ludolph et al., 1991).
Mitochondria carry out vital cellular tasks, integrate extracellular signals, and control the life and demise of neurons (Park & Hayakawa, 2021). Since mitochondria are known to be the prime source of ROS, it is hypothesized that mitochondrial biogenesis will boost oxidative stress in neurons (Cobley et al., 2018; Im et al., 2014).
Pharmacological approaches that make use of this respiratory reserve may be helpful in treating neurodegenerative diseases. A respiratory reserve may provide an effective therapeutic target for the therapy of mitochondrial diseases, according to previous findings (Singh et al., 2021).
Anti-apoptotic Effect of PCE Against Lipotoxicity
An analysis of apoptosis-related proteins using a western blot revealed that pre-treatment of PCE drastically reduced the expression level of pro-apoptotic proteins such as Bax, cleaved-caspase-3 and cleaved-PARP, while PA treatment raised these protein expression levels (Sharma et al., 2021). Lipotoxicity causes apoptosis in neuronal cells (PC12 cells) (Padilla et al., 2011). The protective effect of PCE against PA-induced lipotoxicity in these cells and the mechanism involved in the anti-lipotoxic effect of PCE were investigated. Cells were treated with 0.4 mM PA for 6–48 hours to see if it had any impact on PC12 cell viability using MTT assay. The most effective amount was found at 50 µg/mL PCE. Oxidative stress brought on by mitochondrial electron leakage might result in cell death. We do not fully understand how oxidative damage leads to neuronal death, and it is unclear if mitochondrial deficiencies are the key factor causing the toxicity or whether they are just a side effect of the damage. Age-related factors decrease in cell respiratory capacity are exacerbated in neurodegenerative mitochondrial disorders (Wu et al., 2019). Mitochondria carry out vital cellular tasks, integrate extracellular signals, and control the life and demise of neurons. Since it is known that mitochondrial metabolism is the main generator of ROS, it is hypothesized that mitochondrial biogenesis will increase oxidative stress in neurons and enhance superoxide dismutase (SOD) activity (Gorman et al., 2000; Jovanović & Jovanović, 2011).
According to the previous investigation, autophagy is regulated by oxidative stress and its correlation to lipotoxic neuronal apoptosis. PA treatment induced high dichlorofluorescein fluorescence, which was reduced by NAC (N-acetyl-l-cysteine), a ROS scavenger. The exact mechanism of lipotoxicity is unknown and the increased production of ROS has been seen as one of the major causes of PA-induced apoptosis (Lipke et al., 2022).
Hepatocytes exposed to high concentrations of lipids and carbohydrates suffer hepatic injury, which is referred to as lipotoxicity or glucotoxicity. Both conditions result in neuronal death (Li & Hu, 2015; Okouchi et al., 2007).
Role of PCE in Protein Kinase Inhibition
Over 500 members make up the protein kinase gene family, which makes up about 2% of all human genes. ATP’s terminal phosphate group is transferred onto an amino acid residue in a polypeptide chain by protein kinases, usually serine, threonine, or tyrosine. In AD, the activation of the protein kinase signalling cascade results in the development of amyloid precursor protein and beta-promoted neurodegeneration. Protein kinase-related signalling pathways regulate neuronal death in AD (Yu et al., 2021).
According to research, PC can cause apoptosis in human tumour cell lines, which blocks the mitogen-activated protein kinases (MAPK) pathway and has anti-chemotherapy characteristics. The isolated chemicals showed substantial toxicity at very low concentrations, the protein kinase was inhibited by a substance known as corylifol C (Almansa-Ordonez et al., 2020). It demonstrated a strong ability to affect cellular processes, such as via inhibiting protein kinases, which might be useful for pharmaceutical uses. For this reason alone, kinases constitute a large target class for creating new therapies, as the deregulation of kinases and their substrates is linked to a variety of illnesses. Blocking or decreasing the kinase activity of a kinase-like CK1 that may target several sites both directly and indirectly through kinase priming might be very helpful in the case of AD (Zhang, 2020).
JNK3 is an enzyme that belongs to the MAPK family that is mostly expressed in the brain and is involved in a number of pathological processes, such as neuronal development, synaptic plasticity and neuronal death (Sunkari et al., 2022). Although many kinase inhibitors are still in various pre-clinical phases, failure rates are still substantial. To develop these inhibitors from the laboratory to the bedside, much research is needed.
Neuroprotective Effect of PCE
Around the world, millions of people battle many neurological disorders and the primary independent factor for neurological diseases is ageing. PC is an upcoming anti-ageing herb that is introduced for neurological disorders therapy (Gupta et al., 2012). A neurological condition associated with ageing is AD. Crucial AD-related protein targets, like acetylcholinesterase (AChE), glycogen synthase kinase-3 (GSK-3), amyloid-peptide 42 and -secretase, were discovered to be variably suppressed in vivo by four PC compounds namely Bavachin, BCN, bavachalcone and IBC. The researchers also discovered that BCN and IBC regulate the aggregation of Aβ42 through a variety of pathways. Both oligomerization and fibrillization of Aβ42 were greatly reduced by IBC, whereas fibrillization was reduced by BCN, which resulted in off-pathway aggregation (Jeong et al., 2015).
The neuroprotective effect is against cytotoxic insults caused by the 3-NP (neurotoxic agent) and glucotoxicity or lipotoxicity causing neuronal apoptosis. PA-induced PC-12 cells express enhanced proapoptotic proteins namely cleaved PARP, cleaved-caspase 3 and Bax which results in neuronal apoptosis and PCE decreasing the expression of these proapoptotic genes (Hou et al., 2019).
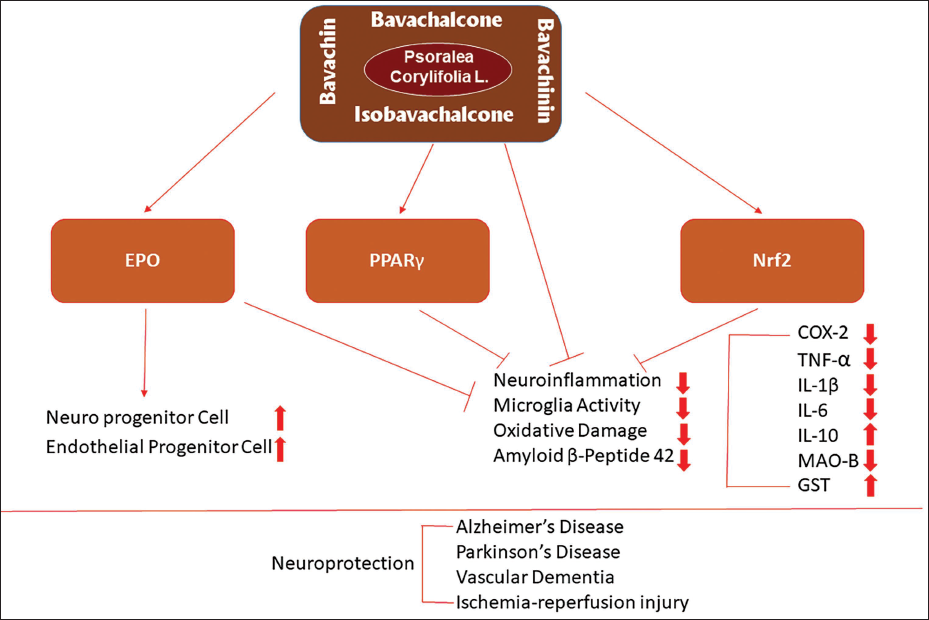
Endogenous factors are the principal regulator of the antioxidant response and a marker of oxidative stress. It mediates the neuroprotective impact of Chinese herbal supplement active components on AD. By enhancing Nrf2 activity and expression IBC may function as a possible neuroprotective drug, according to recent in vivo and in vitro investigations. Erythropoietin’s (EPO) therapeutic potential in the brain helps to prevent this disease (Gao et al., 2019). The expression and circulation concentration of EPO improved on brain diseases increased by bavachalcone and other flavonoids from Chinese herbal medicine as the therapeutic benefits. Figure 3 summarises the neuroprotective properties of Psoralea’s prenylflavonoids (Ramachandran & Thangarajan, 2016; Xu et al., 2018).
Schematic Representation of Various Prenylflavonoids on Neuroprotective Effects Through Direct Anti-inflammatory Actions, Activation of PPAR, Nrf2 and EPO (Hybertson et al., 2011).
Impact on AD
Globally, AD affects about 47 million individuals, the most prevalent cause of dementia and a deadly neurodegenerative brain illness. AD prevalence is mostly observed in the senile stage and is linked to ageing. There is an urgent need for the discovery of AD drugs because there are currently no medicines that can stop the development of AD. The aggregation and deposition of amyloid peptides result in extracellular amyloid plaques (α). This is among the two major pathogenic alterations found in AD patient’s brains and the neurofibrillary tangles, which are clumps of the abnormally hyperphosphorylated microtubule-associated protein Tau that gather inside neuronal cells (Aguilar et al., 2016; Alam et al., 2018; Rahman & Lendel, 2021; Zhou et al., 2020). It has been proposed that irreversible neuronal death and loss are primarily caused by lethal oligomers formed as an aggregate of proteins such as Aβ and Tau (Masters et al., 1985; Tetz & Tetz, 2021).
Two compounds IBC and BCN are isolated from the PC plant used in traditional Chinese medicine clinical settings, modulate amyloid peptides (peptides with 40 [Aβ40] or 42 [Aβ42] residues) and are thought to be culpable for amyloid plaque formation in AD (Sabapathy, 2012). DMSO was used in the formation of peptides in dry form in the lab; Aβ42 was employed at a concentration of 5 mg/mL, which was then diluted to 50 M in PBS. Both substances had distinct effects. IBC greatly reduces Aβ-42’s ability to oligomerize and fibrillarize, whereas BCN causes Aβ-42 to form a massive, unregulated assemblage in neuroblastoma cells. Both substances had good anti-Alzheimer effects (Alam et al., 2018; Benilova et al., 2012). As an AChE enzyme inhibitor, PC fruits psoralen was isolated and examined to determine its potential for treating AD. The concentration of psoralen administered ranged from 25 to 400 g/mL. In studies on animals, it suppressed the AchE in a dose-dependent mechanism. The study employed adult male Wistar rats weighing 180–250 g (Alam et al., 2018; Andreyev et al., 2015). Additionally, molecular docking studies showed that psoralen exhibits interactions including stacking and hydrogen bonding and binds efficiently within the binding domain of the enzyme (Andreyev et al., 2015). Despite the fact that the study’s activity assessment was modest relative to the standard chemical utilized, the compound could be used as a lead for the creation of synthetic analogues to increase the inhibitory activity.
Studies on neuroprotective strategies aimed at lowering neuronal oxidative stress and apoptosis as well as maintaining neuronal mitochondrial integrity at the cellular, animal and human levels offer hope for the future development of therapy methods against neurodegenerative diseases (Somani et al., 2015).
Mechanism of Antagonistic Effect of PC in AD
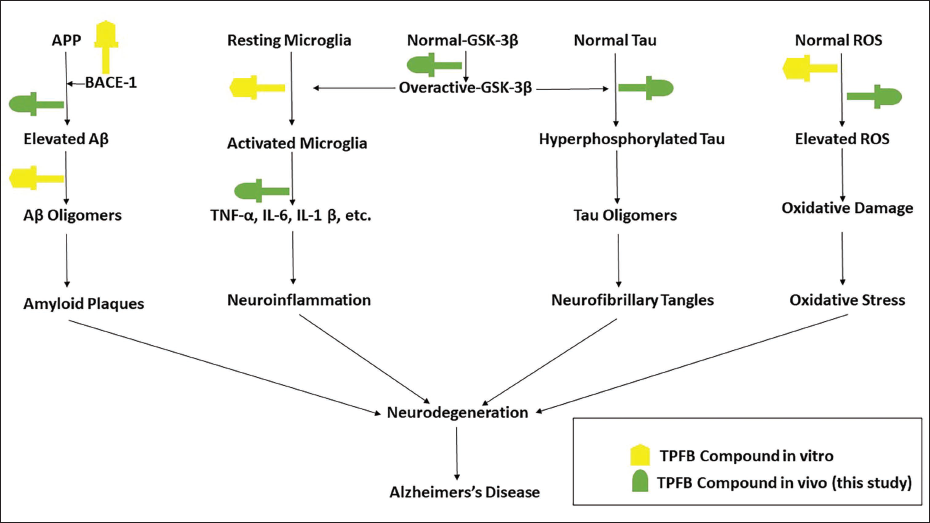
PC exhibits therapeutic benefits in AD through diverse mechanisms such as anti-inflammatory and antioxidant actions, cholinergic effects, reduction of beta-amyloid and modulation of tau proteins. Additionally, total prenylflavonoids (TPFB) exhibit significant anti-inflammatory effects (Zhou et al., 2020) and reduced Tau hyperphosphorylation, a hallmark of AD, most likely by inactivating the crucial enzyme GSK-3. All study groups clearly demonstrated the negative association between GSK-3 (Ser9) phosphorylation and Tau (Thr231) phosphorylation, indicating the crucial function of GSK-3 in Tau hyperphosphorylation in the brain of SAMP8 mice (Benilova et al., 2012; Chen et al., 2018). According to a recent study, employing antisense oligonucleotides to directly block GSK-3 activity, 12-month-old SAMP8 mice’s learning and memory abilities, as well as Tau phosphorylation levels, might be dramatically enhanced (Benilova et al., 2012). Additionally, restricted overexpression of GSK-3 in normal mice brain-induced tau hyperphosphorylation and apoptotic neuronal death and impairment in spatial learning, a pattern similar to AD. These changes could be completely reversed if normal GSK-3 levels were restored by gene shutdown (Benilova et al., 2012). Cognitive abnormalities shown in SAMP8 mice models of Tau disease may also be due to GSK-3. Indeed, new discoveries significantly support the idea that GSK-3, which connects the pathways for Aβ42, tau and neuroinflammation, performs a crucial role in the etiopathogenesis of AD (Sayas & Ávila, 2021). However, it is yet to be determined if the GSK-3 inhibitory activity of TPFB is predominantly responsible for the positive effects seen in SAMP8 mice. Additional research is required to determine the precise chemicals involved and how TPFB regulates GSK-3 (Benilova et al., 2012).
Neurodegeneration following injury to the developing rat brain has the participation of intrinsic and extrinsic apoptotic pathways. Increased neurotrophin expression could be an internal mechanism controlling this apoptotic process (Lin & Beal, 2006). Nerve growth factor, brain-derived neurotrophic factor and neurotrophins 3, 4 and 5 (NT3, NT4/5 and NT6), are the neurotrophins that inhibit the death of neurons. These elements interact with the high-affinity receptors such as Trk A, B and C and dissociate less specific apoptotic pathways. In addition to restoring trophic-depleted neurons, neurotrophins help strengthen nerve cell’s resistance to ischemia, excitotoxicity and other neurodegenerative risks (Uttara et al., 2009). Current AD medication only alleviates symptoms and only modestly improves cognitive abilities. Numerous compounds have been studied that exhibit potential results in the treatment of AD and can control cellular viability through various mechanisms (Felderhoff-Mueser et al., 2002).
Significance of Prenyflavonoids
The amount of peptide Aβ42 was significantly reduced by the overall prenylflavonoids (TPFB) extracted from PC-dried fruits (Chen et al., 2018). Additionally, Ser9 of GSK-3 was phosphorylated, Tau hyperphosphorylation was blocked, and the expression of the pro-inflammatory cytokines TNF, IL-6 and IL-1 was reduced (Figure 3) (Benilova et al., 2012). As compared to their flavonoid equivalents, prenylflavonoids are more hydrophobic and more affinitive to target proteins, which may provide them with higher bioactivities and bioavailabilities. Few investigations have focused on the neuroprotective consequence of prenylflavonoids, despite the fact that many bioactivities have been identified for them including anti-microbial, estrogenic, antioxidative, immunomodulatory and anti-cancer activity (Beeg et al., 2021; Benilova et al., 2012). In vivo, oxidative stress biomarker is the concentration of serum derivatives of reactive oxygen metabolites, which was likewise significantly decreased by prenylflavonoids (Figure 4). It can effectively stave against neurobiochemical changes resembling AD and age-related cognitive loss, and it might play a part in the illness’s prevention (Benilova et al., 2012; Chen et al., 2018).
The Schematic Diagram Depicting the Main Pathogenic Pathways Involved in Alzheimer’s Disease (AD) and the Probable TPFB Targets and Components from Earlier Studies (Benilova et al., 2012; Hybertson et al., 2011; Jiangning et al., 2005; Nishikawa et al., 2010; Panche et al., 2016; Rajpal, 2002; Xiao et al., 2010).
Charting the JNK Pathway: Unravelling Its Intricacies in Neurodegenerative Disorders
The JNK family of protein kinases controls brain plasticity, expression of genes, cell death, regeneration and cellular senescence through the stress signalling pathways (Hwang et al., 2021). JNK pathway activation has been demonstrated to occur in return to various stressors, including oxidative stress, growth hormones, cytokines, UPR signals or Aβ peptides (Figure 5). Since JNK activation is known to be a crucial component in the modulation of apoptotic signals, it is essential for pathological cell death linked to neurological illnesses like AD (Porter et al., 2015).
Activation of the JNK Signalling Pathway for c-Jun N-terminal Kinase. JNKKKs Like ASK1 Phosphorylate JNKKs, Specifically MKK4 and MKK7, Which Then Activate JNK and the Transcription Factor Activator Protein-1 (AP1) Family Proteins in Response to Environmental Stressors, Growth Factors and Cytokines (Hwang et al., 2021).
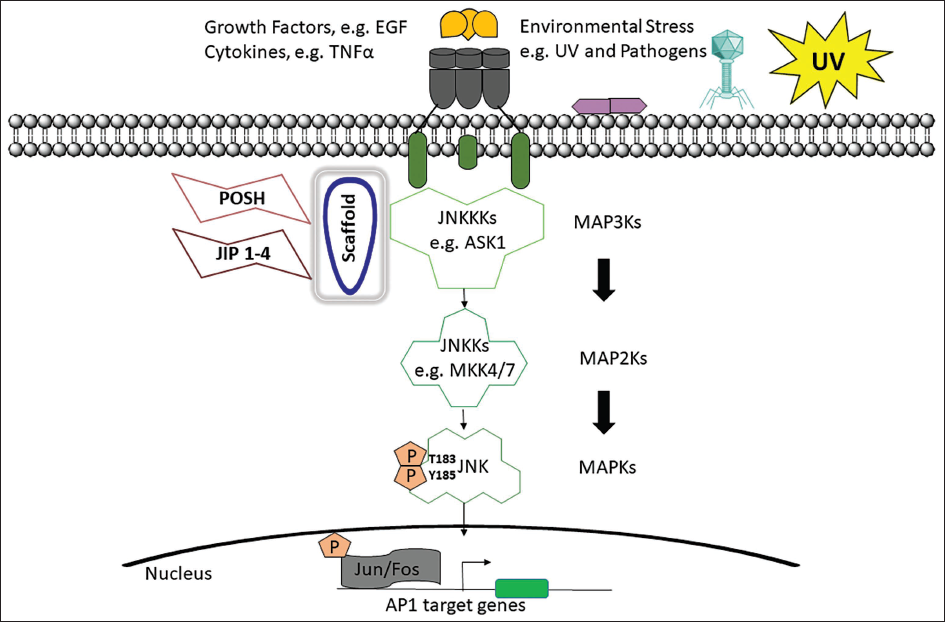
Blocking JNK3 could be a plausible potential treatment for the neurodegenerative processes associated with AD because JNK3 is activated by stresses and largely expressed in the brain. JNK signalling pathway mediates a broad range of biological processes involving cell migration, survival and proliferation along with cell death, senescence and stress responses. Premature senescence and a growth stop in G2/M in mouse embryonic fibroblast cells are brought on by the genetic inactivation of MKK. The advancement of the G1 cell cycle and the G2/M transition is controlled by JNK and activation in Jurkat cells at the G2/M transition. Additionally, it was found that from early S-phase to late anaphase, activated JNK was localised in the centromeres of human HeLa cervical cancer cells, peaking at metaphase. c-Jun is consistently phosphorylated during mitosis and the initial stages of the G1 phase (Hwang et al., 2021; Porter et al., 2015).
Mutation in JNK Pathway
The JNK pathway’s signalling cascade can be hampered by mutations. The pathway normally becomes active in response to a variety of stimuli, including oxidative damage, inflammation, or growth factor signalling.
The route may be inadvertently activated or inactivated as a result of mutations, upsetting the balance of signalling in cells. AD is primarily characterized by abnormal brain protein accumulation (beta-amyloid plaques, tau tangles) causing neuronal dysfunction and cell death. While mutations in the JNK pathway are not linked to AD development, the pathway activates in response to cerebral stressors, like beta-amyloid and tau. This JNK activation, not pathway mutations, contributes to AD-related neuroinflammation and neuronal damage, signifying pathway dysregulation in response to AD pathologies.
Certain type of mutations that causes dysregulation in the JNK pathway are:
Genetic mutations: Mutations can occur in genes encoding components of the JNK pathway, such as JNK itself (MAPK8 and MAPK9), encode for the JNK1 and JNK2 proteins respectively, which are key components of the JNK pathway, upstream kinases like MEKK1 (MAP3K1), or downstream transcription factors like c-Jun (JUN). These mutations may cause the route to be constitutively active or inactive, which would result in aberrant cell signalling (Davies & Tournier, 2012).
MKK4, alias MAP2K4, orchestrates the synthesis of MAPK (kinase-4) serving as a pivotal upstream catalyst for JNK. Mutations in MKK4 disrupt the JNK pathway by impeding its appropriate activation, thereby influencing cellular reactions to stress and inflammation. Numerous suppressors of the JNK pathway exist, including dual-specificity phosphatases and JNK-inactivating kinases. Mutations in these regulators can lead to persistent activation of the JNK pathway (Girnius et al., 2018).
Spontaneous mutations: Due to mistakes in DNA replication or exposure to mutagenic substances like radiation or certain chemicals, simply as a result of the natural variability in DNA. Several mutations in the JNK pathway might develop spontaneously. These mutations may activate the JNK pathway uncontrollably, which can cause illnesses like cancer. Mutations impacting mRNA transcript splicing can induce modified protein isoforms with distinct functional attributes. Splicing mutations may arise in genes encoding JNK pathway constituents, potentially disrupting their function and regulation. Epigenetic modifications, encompassing DNA methylation and histone alterations, can modulate gene activity within the JNK pathway. Spontaneous epigenetic alterations can engender enduring changes in pathway activity (Papa et al., 2019).
Inflammatory diseases: Chronic inflammation is associated with dysregulation of the JNK pathway. JNK mutations that make them more active can make inflammatory reactions worse. For instance, mutations in the TLR gene, which is linked to inflammatory signalling, might change the activation of the JNK pathway and result in inflammatory disorders.
Oxidative stress: Oxidative stress can activate the JNK pathway. Increased oxidative stress can result from mutations in genes involved in antioxidant defences, such as SOD or glutathione peroxidase (GPx), which in turn stimulates JNK signalling. Mutations in the JNK pathway can contribute to neurodegenerative diseases such as AD, PD and ALS. The buildup of beta-amyloid plaques and tau protein tangles in the brain in the case of AD can cause this pathway to become active. JNK activation has been linked to cognitive impairment and neuronal cell death.
According to studies, BAK suppresses cell growth via the JNK pathway and induces S-phase arrest in MCF-7 cells via the p38-ROS-p53 pathway (Engin & Engin, 2021). One of the primary pathogenic characteristics of AD is hyperphosphorylation and the buildup of tau in neurons (and glial cells). In all types of tauopathies, brain homogenates exhibit elevated amounts of active JNK. Strong active JNK immunoreactivity has only been seen in senile plaques in AD, hyperphosphorylated tau-containing neurons, glial cells, as well as in dystrophic neurites (Sabapathy, 2012).
Significance of JNK Pathway
The JNK pathway is essential for controlling cellular stress responses, inflammatory responses and apoptosis (cell death), which are all pertinent to the development of neurodegenerative diseases. In hippocampus neurons, JNK3 activation is a mechanism that promotes apoptosis. In line with JNK3’s involvement in excitotoxic neuronal cell death (Liu & Lin, 2005). The involvement of JNK3/SAPK pathways in additional neuronal death responses and neurodegenerative illnesses was predicted from the role of JNK3 in excitotoxicity (Bogoyevitch et al., 2010; Yarza et al., 2016). In mice, ischemia-induced cell death has been shown to be clearly mediated by JNK/SAPKs. JNK3 may serve as a therapeutic target for the neurodegenerative diseases PD and AD (Choi et al., 2011).
PA therapy decreased the expression of beclin-1 and p62, two autophagy-related genes; PCE therapy restored their levels and may be a useful pharmaceutical drug in the treatment of neuronal cell damage brought on by oxidative stress or lipotoxicity (Bruckner et al., 2001).
PCE emerges as the formidable ally in the battle against AD, wielding its potent antioxidant feature to quell oxidative stress triggered by various assailants. As it decisively inhibits stress and deactivates the JNK pathway, PCE becomes the light of hope in the quest to inhibit neuronal apoptosis and combat this neurological disorder.
Conclusion
For the current review, an effort has been made to gather the morphology, phytochemistry and pharmacological profiles of PC, as a medicinal plant. It has been demonstrated that PCE can reduce the cell damage caused by PA in PC12 cells. By acting as an antioxidant, PCE therapy reduced the lipotoxic effect, and its antiapoptotic effects required activating autophagy. Leukoderma, psoriasis, vitiligo, asthma, ulcers and renal diseases have all been treated with plant parts. It contains a number of crucial chemicals for pharmacology. The seed of this plant can be utilised as a nootropic agent against neurodegenerative diseases and prenylflavonoids have a specific role in AD treatment. PCE might be promoted for its therapeutic benefits or be used as a daily novel food. Based on its pharmacological qualities, more research domains could be examined. It is a significant source of many different kinds of chemicals with a variety of chemical structures and phytochemical characteristics.
Abbreviations
AD: Alzheimer’s disease; BAK: Bakuchiol; PA: Palmitate; PC: Psoralea corylifolia; PCE: Psoralea corylifolia seed extract; PD: Parkinson’s disease.
Footnotes
Acknowledgments
The authors acknowledge the Research and Development Office of Integral University, Lucknow, for facilitating the communication of the manuscript through the provision of the manuscript communication number (IU/R & D/2024-MCN0002529). Additionally, the authors would like to express their heartfelt gratitude to the Director, CSIR-CIMAP Lucknow. The authors would like to express sincere gratitude and thanks to DST-SERB.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
