Abstract
Background
The proportion of dementia patients in Asian countries is rapidly increasing, and because vascular dementia is more common than in the West, it is recognized as important as Alzheimer’s disease.
Purpose
The aim of this study was to verify the benefits of Pinellia ternata Breitenbach tuber aqueous extract (PTex) in improving cognitive function and reducing inflammation in brain damage resulting from bilateral common carotid artery stenosis (BCAS).
Materials and Methods
Four groups of five C57BL/6 male mice each were randomly assigned (a sham group, a BCAS surgery group, and two experimental groups that received PTex concentrations of 25 and 50 mL/kg, respectively). Measuring behavioral learning and memory, the two concentrations of the extract were given orally three times a week for six weeks following BCAS surgery. Furthermore, the inflammatory response in the cerebral cortex was observed, and RNA sequencing analysis was carried out.
Results
The novel object recognition test revealed that the PTex-50-treated group had improved cognitive function, and the anti-inflammatory effect was confirmed by immunofluorescence staining of brain tissue. Proteins such as Apob, Shh, Dscc1, Cdc45, and Cdc6 were identified through protein-protein interaction network analysis as having a beneficial effect on BCAS-induced cognitive deficit.
Conclusion
Results show that PTex-50 was found to have a beneficial effect on BCAS-induced cognitive deficit.
Introduction
Dementia is a clinical syndrome characterized by the deterioration of various cognitive functions such as memory, performance function, language function, visuospatial ability, and attention as a result of chronic progressive degenerative diseases of the brain (Cheng et al., 2022; Kalaria et al., 2008). Dementia is directly proportional to age, and the proportion of dementia patients in Korea, which is transitioning into an aging society, has rapidly increased in recent years (Choi et al., 2020; Kim, 2012; Korea National Statistical Editorial Office, 2021).
Alzheimer’s disease (AD) is the most common cause of dementia. Other possible causes include head trauma, thyroid issues, and HIV infection. Vascular dementia (VD) and Lewy body dementia are next in frequency. Among them, AD accounts for 60%–70% of all dementia cases and is the most prevalent degenerative brain disease globally when considered as a single illness. Following AD, VD is known to account for approximately 20% (Arvanitakis et al., 2019; Cheng et al., 2022; Kalaria et al., 2008; Kuang et al., 2021). VD is more common in Asian countries such as Korea, China, and Japan than in the West, and it is recognized as being as serious as AD (Choi et al., 2020; Kalaria et al., 2008; Xu et al., 2018). While AD progresses slowly, VD frequently shows up with abrupt dementia symptoms, a long-term course of improvement followed by deterioration, a history of stroke, or focal neurological abnormalities. Symptoms of nervous breakdown, such as headache, dizziness, lethargy in the upper and lower extremities, numbness, fatigue, and difficulty concentrating, are common in VD. Personality changes can be observed at a young age, and in many cases, the original personality is sharpened (Arvanitakis et al., 2019; Cheng et al., 2022; Choi et al., 2020; Kalaria et al., 2008; Kuang et al., 2021; Xu et al., 2018).
In Western medicine, there is no single drug that can treat VD. Symptomatic treatment is used instead for prevention and treatment. Aspirin, warfarin, and other drugs are used to prevent the progression and recurrence of cerebrovascular disease as well as to manage risk factors such as diabetes, hypertension, hyperlipidemia, and atrial fibrillation. Cholinesterase inhibitors such as donepezil, and memantine are used to improve cognitive function (Diniz et al., 2013; Farkas et al., 2007; Iadecola, 2010; Jorm & Jolley, 1998; Knopman, 2006; Venkat et al., 2015), so it is still necessary to develop drugs to effectively treat VD.
Clinicians of Asian traditional medicine have used a variety of herbal medicines and formulas to treat dementia, but the classification of dementia has not been systematic, and there has been a lack of research on which medicine shows the most efficacy due to a lack of scientific evidence. As a result, Pinelliae tuber (PT) was chosen as one of the medicines commonly used to treat dementia in this study to confirm its effect.
PT, or Pinellia ternata Breitenbach tuber, is a popular Korean herbal medicine. Since it was first described in Shen Nong Ben Cao Jing (Shennong’s Classic of Materia Medica), PT has been widely used in Asian traditional medicine clinics as an expectorant. It has been widely used traditionally to treat sputum, cough, vertigo, headache, and vomiting due to its ability to dry dampness and resolve phlegm, reduce adverse flow, and relieve vomiting (Peng et al., 2022; Yang et al., 2018). Recent research indicates that it possesses a range of pharmacological activities, including neuroprotective and sedative–hypnotic effects (Deng et al., 2020; Lin et al., 2019; Peng et al., 2022). With cognitive dysfunction being one of the most prevalent symptoms of VD, we hypothesized that PT could also be used to treat it.
The impact of PT aqueous extract (PTex) administration on improvements in cognitive function in mice with bilateral common carotid artery stenosis (BCAS)-induced chronic cerebral blood flow reduction was evaluated by the researchers using the Y-maze test and the novel object recognition test (NORT). Alterations in microglial cells and astrocytes, two types of brain cells involved in the inflammatory response, were also noted. Furthermore, post-mortem brain tissue observation and RNA sequencing analysis were used to identify the proteins on which PTex is expected to act, and it was discovered that PTex regulated the inflammatory response caused by BCAS.
Materials and Methods
Animals
Male C57BL/6 mice aged six weeks, weighing between 20 and 22 g, were acquired from Samtako Bio (Gyeonggi-do, Korea). The mice were kept in polypropylene cages with a 12-hour lighting and shade cycle and a temperature control of 24°C ± 4°C for a minimum of one week before the experiment.
Reagent
Choongwae Pharmaceutical (Seoul, Korea) provided the physiological saline, and SK Chemicals provided the ethyl alcohol (Ulsan, Korea). Bio Basic (Ontario, Canada) provided the phosphate buffer saline (PBS), and Thermo Fisher Scientific provided the tissue fixative (OCT compound) for frozen sections (Massachusetts, USA).
PTex Preparation
Gwangmyung Natural Pharmaceuticals (Ulsan, Korea) provided dried PT, and the residue was removed using a non-woven fabric after 50 g of PT was heated in 500 mL of boiling water for 2 hours. Adults weighing 60 kg typically take 1 bag of PTex solution once, three times per day, and because each pack contains 90 mL, the daily dose is 270 mL. The daily dose was administered only once to the experimental animals since it was found that giving them three times a day would cause a high-stress load. The ideal dosage for mice in this study is roughly 50 mL/kg, and half of this concentration, or two concentrations of 50 and 25 mL/kg, were used for comparison. This is because rodent metabolism is roughly 10–13 times that of humans (Nair et al., 2018). At the time of administration, 25 mL/kg was used by diluting 50 mL/kg twice so that the dosage of both concentrations was the same.
BCAS-induced VD Model Construction
VD was caused by BCAS. Isoflurane was used to sedate laboratory mice in N2O/O2 (70%/30%) gas until they ceased reacting to mechanical stimuli on the tail. A midline incision was made in the neck region, and in order to induce BCAS, the carotid artery was carefully isolated from the surrounding tissues and wrapped with an inner diameter micro-coil (Sawane Spring, Hamamatsu, Japan) measuring 0.18 mm. Throughout the surgical procedure, a thermostat was employed to maintain the mice’s body temperature at 37°C ± 0.5°C, which was connected to a temperature sensor placed in the rectum.
The PTex Treatment
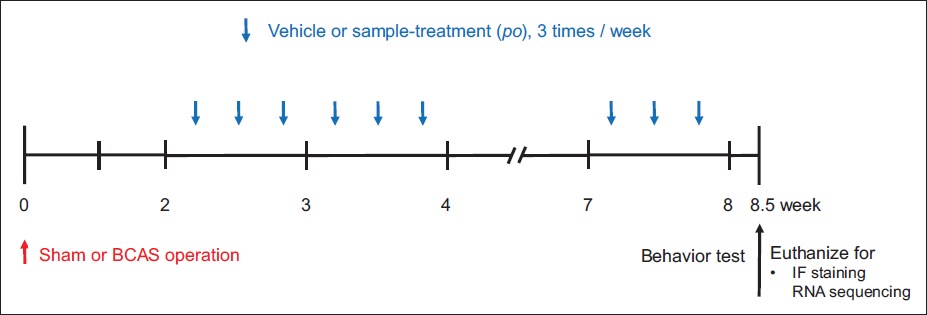
The experimental rats were divided into four groups (n = 5) at random. (a) Sham group, (b) BCAS control group, (c) BCAS + PTex 50 mL/kg dose (PTex-50), and (d) BCAS + PTex 25 mL/kg dose (PTex-25). During the BCAS surgical procedure, only non-BCAS surgical procedures were used in the sham group, and the same dose of distilled water was administered instead of the drug during the drug administration period. The BCAS control group received distilled water as well, while the PTex-50 and PTex-25 groups received PTex at doses of 50 and 25 mL/kg, respectively, three times a week during the drug administration period (Figure 1).
An Overview of the Experimental Procedure. Vehicle, PTex-25, or PTex-50 was Post-treated Three Times a Week for Six Weeks After Surgery. Each Group had Five Mice. We Experimented with Behavior Tests Such as the Y-maze Test and NORT After 8.5 Weeks of Surgery. The Subjects were Also Sacrificed on the Same Day for Brain Harvesting for IF Staining and RNA Sequencing.
NORT and the Y-maze Test
Maze experiments with arms, such as the Y-maze, cause less stress in rodents than other methods (Vorhees & Williams, 2014). Because stress impairs working memory significantly, maze experiments using pathways are accepted as an appropriate method for measuring working memory ability in rodents (Olver et al., 2015). The experimental animals are housed in a Y-shaped maze (Figure 2A) with three identically shaped arms (each measuring 35 × 7 × 40 cm). The animals are given a predetermined amount of time to investigate the maze before the data is examined. After being put in the center of the maze and given 2 minutes to move around freely, each experimental mouse was given three arms of the Y-maze to investigate for 8 minutes before the blocked passage was opened. The mouse’s motion was recorded and analyzed on top of the maze using a digital camcorder, as was the degree of sequential exploration of the three arms. The recorded video was examined for the path and time the animal moved utilizing the behavior analysis software SMART ver. 3.0.06 (Panlab, Barcelona, Spain). That is, the degree of sequential search of these passages was expressed as a percentile of the number of times the three arms were explored, (e.g., C–A–B, B–C–A, and A–B–C). The spontaneous alternation behavior was calculated as % Alternation = {(number of alternations)/(total number of arm entries − 2)} × 100.
Aerial View of the Y-maze for the Spontaneous Alternation Test (A) and the Open Field Box for the NORT Test (B). The Y-maze was Made Up of Three 35 cm (Length), 7 cm (Width), and 40 cm Sections (Height). The Field Arena Used for NORT is 40 cm (Length), 40 cm (Width), and 40 cm (Height).
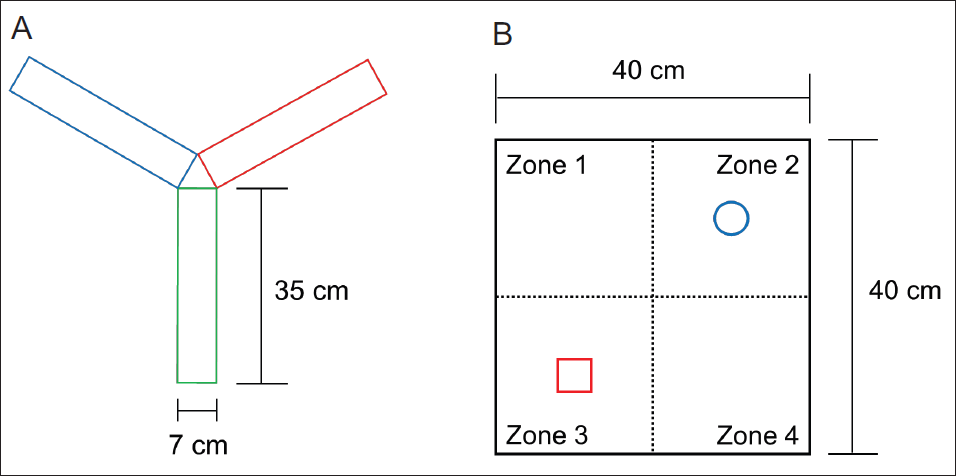
NORT is used to assess changes in cognitive function by utilizing rodent’s traits that are more curious about novel objects than familiar ones (Denninger et al., 2018; Tahamtan et al., 2013). One week before the experiment, there were three pre-adaptation periods. To acclimate to the field box, each mouse explored a gray open field box (40 × 40 × 40 cm) for 10 minutes without an object during the acclimatization phase (Figure 2B). On the day of measurement, a period of object adaptation was given to confirm cognitive function. That is, the experimental mice were given 10 minutes to get to know two identical objects that were placed in the field box’s two opposite corners. In the second trial, the experimental mouse is returned to the field box after at least 20 minutes, and just before that, one of the same objects {familiar (F)} provided in the first trial is replaced with a novel object {novel (N)} (replaced objects in zone 3). The mice in this condition were given 10 minutes to freely explore objects before a digital camera was used to record their actions. The experimenter was prepared to begin after the mice had been acclimated for 10 minutes, and the mice’s behavior was recorded. We calculated the total amount of time the mice spent examining each object by subtracting the start time from the stop time for each object investigation path instance and the duration of the mice’s movement from the recorded video. SMART software was utilized to analyze the behavior. The formula of total N time/(N time + F time) × 100 was used to analyze the discriminant ratio and object contact time for each experimental group. Between measurements, 70% ethanol was used to thoroughly clean the two objects, as well as the field box, to eliminate any remaining smells.
Physiological Parameters are Measured
The mice’s body weight was measured once every two weeks during the experiment to confirm whether the decrease in blood flow had an effect on body weight. At the conclusion of the experiment, blood was drawn while under deep anesthesia, and serum was separated from the blood using a centrifuge set to 1,500 × g for 15 minutes at 4°C. Because severe changes in electrolytes in the body can influence behavior, blood electrolytes were also examined. An electrolyte analyzer (Fuji Dri-Chem 3500i, Tokyo, Japan) was used to measure the serum concentrations of electrolytes such as sodium (Na+), potassium (K+), and chloride (Cl−) in order to monitor and rule out any potential electrolyte imbalances. During the experiment, mice’s blood pressure (BP) and heart rate (HR) were measured once every two weeks with a dedicated device (Visitech System, BP-2000, NC, USA) to see if BCAS and PTex administration affected BP and HR. BP and HR were measured non-invasively from the mice’s tails using this technique.
Euthanize of Experimental Mice, Cardiac Perfusion, and Brain Tissue Frozen Section Preparation
An incision was made in the center of the experimental mice’s abdomen, and PBS was used to perform cardiac perfusion. To sum up, the procedure involved occluding the pulmonary artery, inserting a 21-gauge needle into the left ventricle, and securing the needle to the ascending aorta. The right atrium was cut with scissors immediately after perfusion began to drain the perfused blood from the body. Fixation was carried out after sufficient perfusion with PBS with 4% paraformaldehyde (PFA). The brain was immersed in 4% PFA for post-fixation before being placed in 10%, 20%, and 30% sucrose solutions for cryoprotection. The cryoprotected frozen mouse brains were immersed in OCT compound and stored at −70°C. Using a cryostat (Leica, Wetzlar, Germany), before being used, 20 µm-thick brain slices from each mouse were put on a glass slide and kept in a refrigerator at a temperature of −70°C.
Brain Tissue Immunofluorescence (IF) Staining
The brain tissue slices were dried and reacted with blocking buffer (5% BSA) at 25°C for 1 hour, then washed and a cluster of differentiation 68 (CD68), neuronal nuclei, glial fibrillary acidic protein (GFAP) antibodies (#97778, #94403 and #12389, Cell Signaling Technology, MA, USA), etc. were added and reacted at 4°C for 12 hours. After washing the primary antibody three times in PBS for 5 minutes, the tissue sections were reacted with diluted secondary antibodies (ab150077, ab150078, ab150113, and 150114, Abcam, Cambridge, UK) at 25°C for 2 hours for fluorescence development. After that, the secondary antibody was washed three times in PBS for 5 minutes each, and a slide glass fixing solution containing DAPI dilution (ab104139, Abcam, Cambridge, UK) was dropped on the tissue, covered with a cover slide, and sealed with nail polish. Tissue observation was performed using a fluorescence microscope (Ni-U, Nikon, Tokyo, Japan), and tissue picture merging was performed using ImageJ (NIH, MD, USA). The cell marker protein expression level per unit area (1 mm2) of the brain section was determined by calculating the fluorescence intensity following antibody staining, and this was done using Digimizer version 4.6.1 (MedCalc Software Ltd, Ostend, Belgium).
RNA Sequencing Examination
After the behavioral analysis was finished, the animals were euthanized with CO2, the brain tissue was removed in a cold experimental setting, and the cerebral cortical region that contained the white matter was divided and quickly frozen. Trizol was used to isolate total RNA, and RNA with verified purity and ratio was used in the investigation. Capillary electrophoresis (Agilent 2100 Bioanalyzer, CA, USA) was used to determine the quality of total RNA before the experiment began. mRNA was isolated from total RNA, cDNA was synthesized from it, and mass sequencing was carried out. Sequencing raw data was mapped to the reference genome to derive a raw count, and the level of expression was verified after normalization using Edge R, an analysis platform. To determine the genes that needed to be identified, expression data was sorted using the Excel-based analysis program ExDEGA.
To determine the RNA expression level, expression levels in the control and experimental groups were compared to the sham group when its expression level was set to 1. In this research, if the expression level increased or decreased by less than fourfold compared to the sham group, it was to be within the normal range. Genes were chosen based on whether their expression level in the sham group was higher or lower than four times in the BCAS control group. Among the genes, genes whose expression was controlled to normal levels in the experimental group administered PTex-50 were chosen. These genes-related proteins were chosen, and they underwent additional examination. Following the selection of proteins for analysis, the STRING database (
Statistical Analysis
A one-way analysis of variance test was used to determine the statistical significance between groups, and the experimental data are presented as mean ± standard deviation. Holm-Sidak post hoc analysis was utilized when the sample values were from a normal distribution, and Tukey’s post hoc analysis was used when they were not. Sigmaplot v12.0 (Systat Software Inc., IL, USA) was utilized for the statistical analysis mentioned above, and statistical significance was established when the p value was less than 0.05.
Results
Physiological Parameters Affected by BCAS Operation and PTex Administration in the Experimental Mice
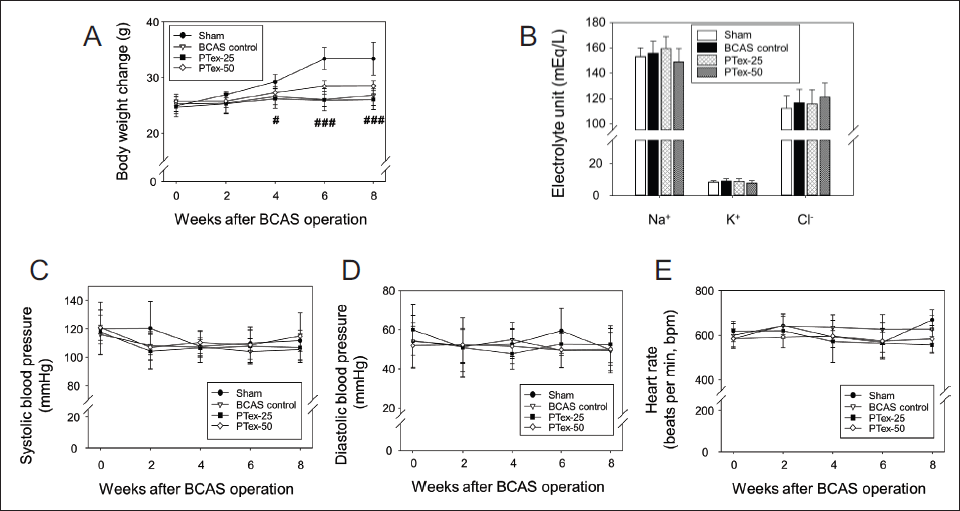
BCAS reduced CBF, which resulted in a significant decrease in weight gain beginning in the fourth week. PTex administration resulted in no significant change in body weight when compared to the BCAS control group (Figure 3A). Other physiological changes were not observed because BCAS or PTex administration had no effect on blood electrolyte content (Figure 3B), BP (Figure 3C and D), or HR (Figure 3E).
Body Weight (A), Serum Electrolytes (B), Blood Pressure (C and D), and Heart Rate (E) in All Experimental Groups were Evaluated. Mice were Weighed Every Two Weeks During the Eight-week Experimental Period. The Results are Given as Mean ± SD. #p < 0.05 Versus Sham Group, ###p < 0.001 Versus Sham Group; n = 5 in Each Group.
Changes in Behavioral in the Y-maze Test and NORT
The BCAS-induced decrease in blood flow or PTex administration had no effect on behavioral changes in the Y-maze test experiment. In the Y-maze test, did not differ significantly among the four groups. The latency time for the first arm entry, the probability of sequentially entering three arms, and the total number of entries in three arm entries (Figure 4A).
Spontaneous Triplet Arm Alternation Test Using Y-maze (A) and NORT Using Open Field Box (B). In the Y-maze Test, the Time to Get to the First Arm Entry, the Total Number of Times it Went into Three Arm Entries, and the Probability of Sequentially Entering Three Arms were Measured by Filming with a Digital Camcorder at an Illuminance of 15 LUX. NORT was Captured with a Digital Camcorder at 200 LUX Illumination. The Results are Given as Mean ± SD. #p < 0.05 Versus Sham Group, ###p < 0.001 Versus Sham Group, *p < 0.05 Versus BCAS Control Group; n = 5 in Each Group.
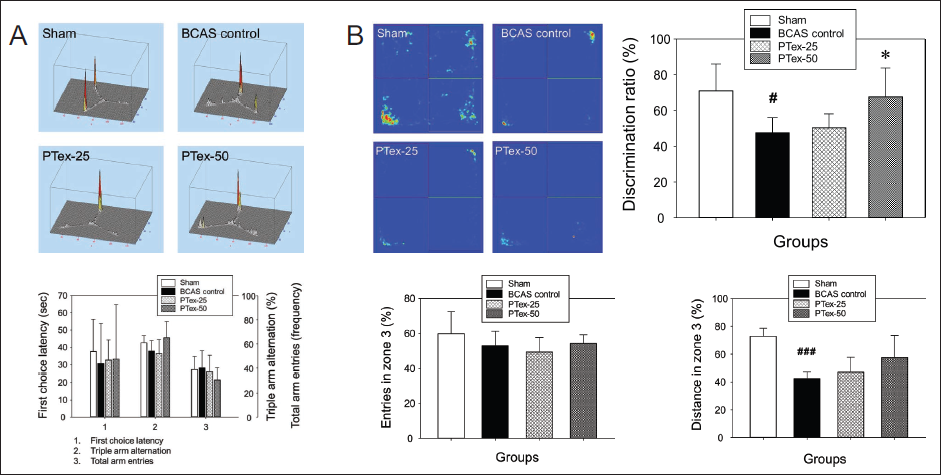
There was no change in the number of searches as a result of NORT analyzing the behavior in zone 2 and zone 3 with distance moved, number of searches, and time spent. As shown in Figure 4B, there was no significant difference between groups of entries in zone 3 (%), but the explored distance of mouse in zone 3 (%) was 73.00% ± 5.67% in the sham group versus 42.51% ± 4.68% in the BCAS control group, indicating that the BCAS operation affected spontaneous moving activities, but PTex experimental groups showed no significant difference compared to the BCAS control group. The time spent in zone 3 was significantly reduced in the BCAS control group compared to the sham group, while it was significantly increased in the PTex-50 group compared to the BCAS control group. The retention time corresponds to the discrimination ratio (DR), and the DR (%) in the sham group was 71.09% ± 14.87%, 47.61% ± 8.40% in the BCAS control group, 50.40% ± 7.73% in the PTex-25 group, and 67.75% ± 16.04% in PTex-50 group, respectively (Figure 4B). It was discovered that decreased CBF causes negative changes in object recognition function, and it was confirmed that PTex administration could improve these pathological symptoms.
In the Brain Tissue, Cellular Expression is Involved in the Inflammatory Response
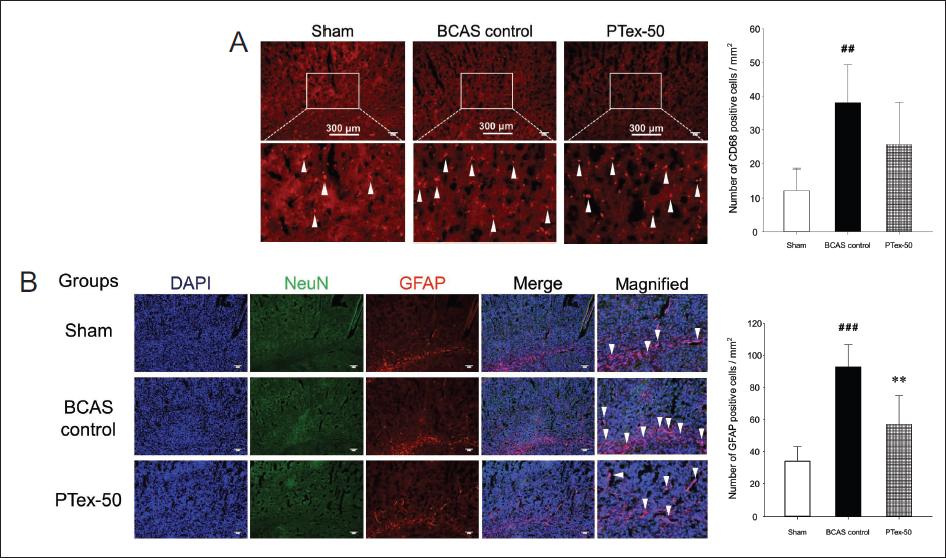
CD68 has been identified as a common marker for activated phagocytic microglia/macrophages, a key inflammatory cell in the central nervous system, in a number of studies using mouse models of ischemic stroke (Doust et al., 2021; Perego et al., 2011). BCAS increased the expression of activated microglia in the cerebral cortex, as shown in Figure 5A, and PTex-50 administration had no effect on this response (Figure 5A). GFAP is a cytoskeletal protein that is the main component of the glial filament of astrocytes (Guo et al., 2018; Ludwin et al., 1976). When there is central nervous system damage such as stroke, dementia, or brain damage, astrocytes change into reactive astrocytes such as hypertrophy and hyperplasia, and GFAP is the most characteristic to mark these reactive astrocytes (Doust et al., 2021; Fukuyama et al., 2001; Zhang et al., 2000). The expression of activated astrocytes was found to be increased in the cerebral cortex, particularly in the white matter region, and this expression was suppressed by PTex-50 administration (Figure 5B).
BCAS Causes Changes in the Expression of CD68-positive Microglia (A) and GFAP-positive Astrocytes (B) in the Cerebral Cortex. The Arrowheads Represent Activated Microglia (A) and Astrocytes (B). BCAS Increased the Activation of Microglia and Astrocytes, but This Increase was Reduced by PTex-50 Administration, and Such Suppression in Astrocytes was More Evident. The Results are Given as Mean ± SD. ##p < 0.01 Versus Sham Group, ###p < 0.001 Versus Sham Group, **p < 0.01 Versus BCAS Control Group; n = 5 in Each Group.
PPI Network Analysis Based on RNA Sequencing
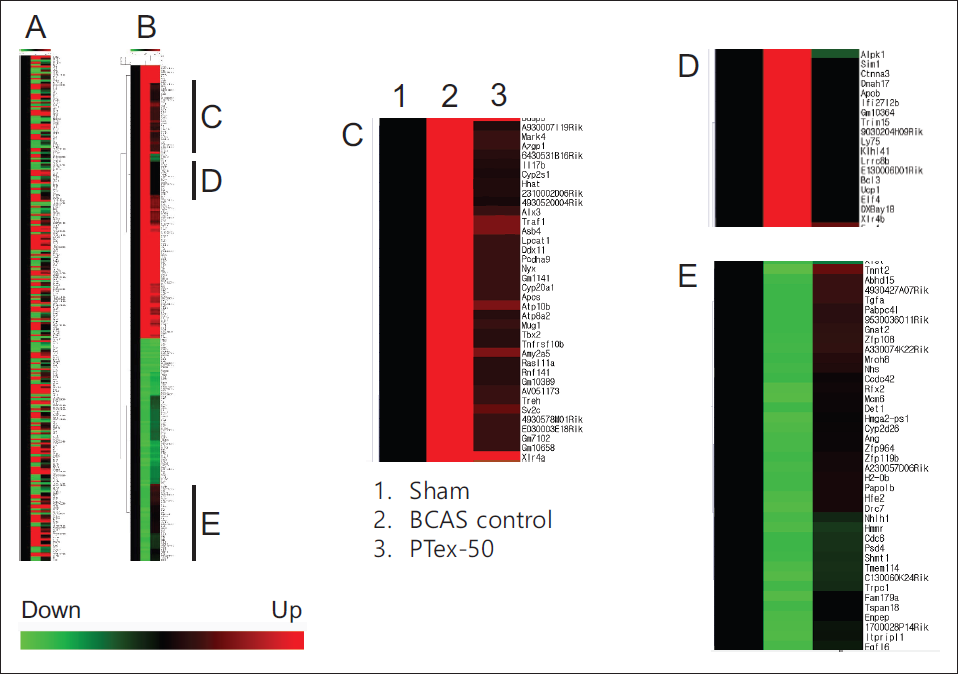
RNA sequencing analysis was used to determine the level of gene expression, and the amount of change in expression is shown in Figure 6. When the gene expression in the BCAS control group decreased by 1/5 times or less compared to the sham group or increased by five times or more, it was chosen for further investigation. The horizontal bar at the bottom of Figure 6 indicates the gene whose expression is down-regulated in comparison to the sham group, and the red bar indicates the expression is up-regulated. The leftmost black line in Figure 6A corresponds to the sham group, and because the value corresponds to the standard of gene expression, all appear in black. The second line represents the BCAS control group, while the rightmost line represents the PTex-50 group. Then, depending on the degree of expression, it was visualized as green or red. Figure 6A was reconstructed into Figure 6B to make it easier to identify the genes that increased or decreased in the control group, and Figure 6C and D represents genes whose expression was down-regulated by PTex-50 administration, and Figure 6E represents genes whose expression was up-regulated by PTex-50 administration (Figure 6).
Clustering Analysis of Expressional Profiling. Normalized Data were Hierarchically Clustered According to Gene Expression Ratio. Red, Green, and Black Colors Indicate Up-regulation, Down-regulation, and No Change in Gene Expression, Respectively. Color Intensity is Related to the Expression Level of a Gene. Genes Whose Expression was Down-regulated by PTex-50 Administration were Clustered in (C and D), and Genes Whose Expression was Up-regulated by PTex-50 Administration were Clustered in (E).
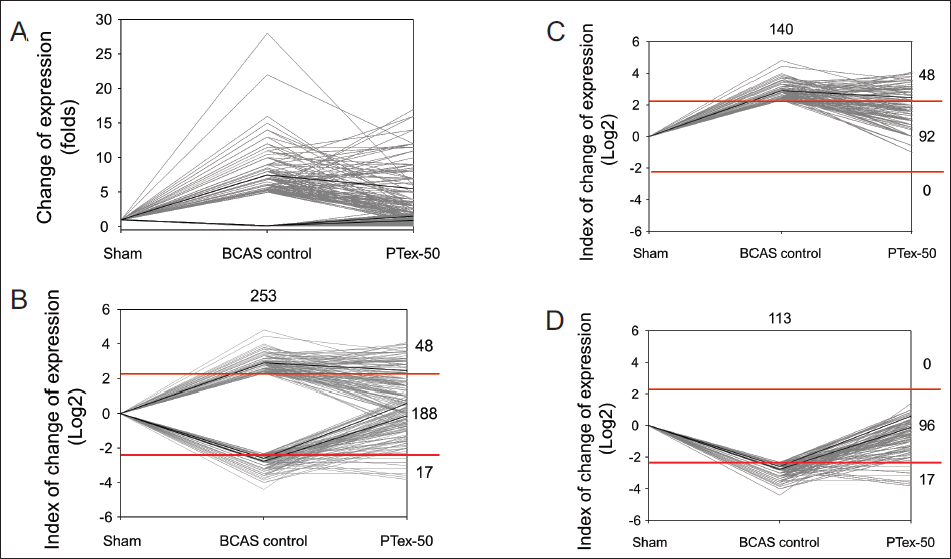
To make it easier to identify the change in the expression of the target gene whose expression was regulated by PTex-50 administration, it was visualized as a line graph (Figure 7). Figure 7A depicts the fold change in gene expression, and the fold change in gene expression change was expressed in log2 form to understand the trend of up- or down-regulation of gene expression better (Figure 7B). To clearly identify the number of up- or down-regulated genes, Figure 7C and D shows them separately. The number of genes whose expression changed as a result of BCAS-induced chronic CBF reduction was 253 (Figure 7B), with 140 genes having increased expression (Figure 7C) and 113 genes having decreased expression (Figure 7D). PTex-50 administration down-regulated 92 of 140 (65.71%) genes and up-regulated 96 of 113 (84.96%) genes, as shown in Figure 7C. Normal levels have been restored. PTex-50 administration had no effect on the expression of any of the 253 genes, so it is assumed that PTex-50 administration had a little negative effect on the VD model in mice (Figure 7).
Effects of PTex-50 Administration on Genes Whose Expression Changes Due to BCAS-induced CBF Reduction. Fold Change of Gene Expression (A) was Transformed Using log2 (B) to Easily Overview the Fold Change, and Divided into Two Parts of Increased (C) or Decreased (D) Expression by BCAS-induced Chronic CBF Reduction.
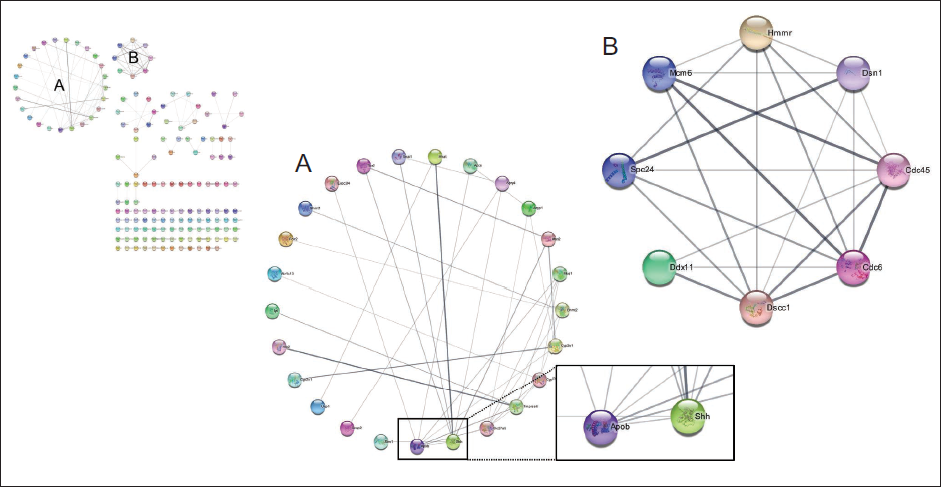
A PPI network was built using the STRING database for 188 genes whose expression was up- or down-regulated by PTex-50 administration, and proteins with major functions were identified, as shown in Figure 8. Apob (ApoB) and Shh proteins were important in the A network (Figure 8A), while Dscc1, Cdc45, and Cdc6 proteins were important in the B network (Figure 8B).
PPI Network Constructed Using the STRING Database. Two (A and B) Major Clusters were Identified from the Network. The Edge Between Proteins Means that the Two Sides are Known or Predicted to have an Interaction.
To study gene functions, gene ontology (GO) analysis classifies biological processes, cellular functions, and molecular functions involved in individual genes. In this study, it was confirmed that PTex acts most significantly on DNA duplex unwinding in biological processes, on motile cilium in cellular function, and on DNA replication origin binding in molecular function (Supplementary Tables S1–S3).
Discussion
The syndrome known as dementia is defined by a decline in cognitive function that is greater than what would be predicted from biological aging. Supporting and caring for individuals with dementia has significant effects on families, the healthcare system, and society at large. Dementia is a major problem in South Korea, where the number of cases is predicted to double every 20 years (Doust et al., 2021; Jang et al., 2021; Kalaria et al., 2008; Srikanth et al., 2005).
VD is defined as cognitive dysfunction brought on by ischemic or hemorrhagic brain lesions brought on by cerebrovascular lesions or cardiovascular disease. It is the second most common type of dementia after AD (Kalaria et al., 2008; Sarti et al., 2002; Srikanth et al., 2005; Wolters & Ikram, 2019). As a result, the pattern and prognosis differ depending on the type of underlying cerebrovascular disease. The mechanism is explained as follows: Similar to general cerebrovascular disease, VD is caused by large or small blood vessel occlusions in the brain that happen to people who have risk factors for vascular disease, such as smoking, diabetes, hypertension, and old age. This destroys the association of nerve fibers in the cerebral cortex or subcortex. Patients with cortical lesions may exhibit aphasia, apraxia, epileptic symptoms, and other symptoms, whereas patients with subcortical lesions may exhibit ataxia, executive dysfunction, gait disturbance, urinary incontinence, Parkinsonian symptoms, and other symptoms (Honig et al., 2003; Maclin et al., 2019; Sengupta et al., 2019; Wolters & Ikram, 2019).
Because the majority of the drugs used to treat VD aggravate cerebrovascular diseases (Diniz et al., 2013; Farkas et al., 2007; Iadecola, 2010; Jorm & Jolley, 1998; Knopman, 2006; Venkat et al., 2015), it is still necessary to develop effective drugs that act directly on VD.
Natural substances have long been used in Asian traditional medicine, especially in Korean and Chinese medicine, to treat diseases related to dementia. However, research on the efficacy of these substances and their precise mechanisms of action is still lacking. As a result, in this study, PT, which is commonly used in Korean traditional medicine and has recently been shown to improve brain function (Deng et al., 2020; Lin et al., 2019; Peng et al., 2022), was used as a research material and applied to a mouse model of VD.
PT, the dried tuber of P. ternata is known to possess antitussive, antiemetic, antitumor, antibacterial, and sedative–hypnotic activities; however, few research results associated with dementia have been reported till date (Bai et al., 2022). According to Kim et al. (2017), a study finding indicated that PT-infused herbal formula can mitigate cognitive function loss by reducing inflammatory response in the brain. The Y-maze test and NORT were used in this study to evaluate learning and working memory behavior in mice that had chronic CBF reduction via BCAS in order to determine the impact of PTex administration on improvements in cognitive function. Furthermore, post-mortem brain tissue observation and RNA sequencing analysis were used in this study. Vehicle, PTex-25 or PTex-50 were orally administered three times a week for six weeks to rats whose chronic CBF reduction was induced by the BCAS procedure two weeks after the sham or BCAS operation. No significant differences were found between the groups in the Y-maze test (Figure 4A). However, in the NORT, the BCAS control group’s duration time in zone 3 was significantly shorter than that of the sham group, and the BCAS control group’s duration time was longer than that of the sham group because of the effect of PTex-50 administration (Figure 4B).
IF staining was used to assess the expression of cells involved in the inflammatory response in brain tissue; BCAS increased the expression of activated of microglia in the cerebral cortex, while PTex administration had no effect (Figure 5A). However, it was discovered that BCAS-induced CBF hypoperfusion increases astrocyte expression and that this tendency was suppressed by the administration of PTex-50 (Figure 5B), indicating that PTex has an anti-inflammatory effect because an increase in GFAP means activation of astrocytes and because activated astrocytes promote gliosis, it is an indicator to distinguish the degree of central nervous system damage (Guo et al., 2018; Sticozzi et al., 2013; Zhang et al., 2000).
RNA sequencing was used to investigate the extent and change of gene expression. As a result, 253 genes whose expression was altered by BCAS-induced chronic CBF reduction were discovered, and PTex-50 administration restored normal levels to 65.71% of 140 genes with increased expression and 84.96% of 113 genes with decreased expression (Figure 7). It was discovered that a total of 5 proteins, ApoB, Shh, Dscc1, Cdc45, and Cdc6, each have 7 connections and are thought to play a major role after constructing a PPI network with 188 genes whose expression was regulated to a normal level by PTex-50 administration. ApoB is found in chylomicron and represents the amount of triglyceride-rich ultra-low-density lipoproteins and low-density lipoproteins in the body (Barter & Rye, 2006; Elovson et al., 1988). As a result, studies have shown that the ratio can be used to evaluate insulin resistance and abnormal fat metabolism, and is more useful than cholesterol in predicting the occurrence of cardiovascular disease (Holme et al., 2008; Lind et al., 2006; Pitsavos et al., 2008). Furthermore, the Cdc45 protein is required for both the initiation and elongation stages of DNA replication and thus plays an important role in genome stability. Additionally, the Cdc6 protein is required for the initiation of chromosomal replication and functions as a licensing factor to maintain chromosome integrity (Broderick & Nasheuer, 2009; Kim et al., 2017). Although it is not yet known whether the ApoB protein plays a role in the onset and progression of VD, a recent study has found that the protein regulates blood LDL levels and that VD is associated with elevated LDL expression (Picard et al., 2022). Furthermore, a study has recently reported that ApoB may be a new target for Alzheimer’s research. Moreover, Yurov et al. (2011) hypothesized that DNA replication stress could be the cause of AD, therefore the proteins chosen for this investigation may be linked to cognitive decline in VD.
Because PTex-50 administration worked to keep these genes at a normal level, it is thought that more research into these proteins is needed. Furthermore, GO analysis revealed that the genes whose expression was normalized by PTex-50 administration had the greatest impact on DNA duplex unwinding in the biological process, motile cilium in cellular function, and DNA replication origin binding in molecular function (Tables S1 and S2). According to the previous description, these findings are believed to be associated with the actions of proteins such as ApoB and Cdc45, and their actions will need to be clarified in further research. In this study, it was found that such an integrated analysis would be useful in verifying the drug’s mechanism. Because PT, the material used in this study, contains various substances, it is assumed that it acts comprehensively through several mechanisms rather than showing effects through one or two mechanisms. PTex-50 was found to be involved in several cell survival mechanisms, in addition to the ones mentioned above (Table S3).
Many studies are reporting a connection between changes in the differential gut microbiota and abnormalities in brain function (Açar et al., 2023), and our research team’s previous report also indirectly verified the “brain–gut axis” hypothesis by revealing changes in isoflavone metabolites due to ischemic cerebral infarction (Kim et al., 2021). As a result, in follow-up studies, it is believed that it will be necessary to conduct more research on the relationship between VD and the administration of PTex and the differential of gut microbiota.
Conclusion
To assess the efficacy of PTex oral administration on the VD mouse model, we used Y-maze tests and NORT to assess learning and working memory. Furthermore, the expression of CD68 and GFAP in the cerebral cortex was confirmed, and RNA sequencing was performed. In comparison to the BCAS control group, the PTex-50 group significantly increased DR, which is the ability to recognize a novel object in NORT. Additionally, BCAS-induced chronic CBF reduction increased the expression of microglia and astrocytes in the cerebral cortex and white matter, while PTex-50 administration inhibited astrocyte activation. Proteins such as ApoB, Shh, Dscc1, Cdc45, and Cdc6 were identified through RNA sequencing-based PPI network analysis, and PTex-50 was found to have a beneficial effect on BCAS-induced cognitive deficit.
Authors’ Contributions
Conceptualization: K.-L. Kim, S. Cho, and K.-M. Kim. Methodology: C. Lim and S. Cho. Investigation: K.-L Kim and S. Cho. Data analysis: C. Lim and S. Lim. Writing—original draft: K.-L. Kim and S. Cho. Writing—review and editing: S. Cho and K.-M. Kim. Supervision: K.-M. Kim. All authors read and approved the final version of the manuscript.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All procedures, including surgical operations, were approved by the Pusan University Animal Experimental Ethics Committee (Approval No. PNU 2017-1759), and each experimental procedure was conducted in compliance with related provisions.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
