Abstract
Background
Prunella vulgaris L. (Lamiaceae) is a perennial medicinal plant commonly used to treat thyroid dysfunction, infectious hepatitis, and mastitis in north-east Asia and Europe. In China, there has been a historical change in the medicinal use of P. vulgaris from the use of whole plants to the use of the spicas.
Purpose
This study aimed to evaluate the protective effects of whole plant P. vulgaris water extracts and spicas on hyperthyroidism with liver-yang hyperactivity syndrome and to preliminarily explain the mechanism of action of these water extracts.
Materials and Methods
Rats were induced with aconite decoction and levothyroxine and treated with P. vulgaris whole plant and spicas water extract for 28 days at the same time. Body weight, food intake, and rectal temperature were monitored. The serum thyroid function indicators thyroid stimulating hormone (TSH), thyroid hormone-free triiodothyronine (FT3) and free thyroxine (FT4), liver function index alanine aminotransferase (ALT) and aspartate aminotransferase (AST), oxidative damage index malondialdehyde (MDA), and superoxide dismutase (SOD) were detected. Liver and thyroid tissues were taken for sectioning and observation. The liver inflammatory factors human tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) were detected by immunohistochemistry, and the expression levels of the apoptotic proteins caspase-3 and B-cell lymphoma-2 (Bcl-2) were detected.
Results
Both P. vulgaris whole plant and spica water extract groups exhibited markedly reduced FT3 and FT4 contents, increased the TSH content, and decreased ALT and AST activities. Furthermore, these agents improve thyroid and liver lesion tissue; reduce MDA content; increase SOD activity; reduce IL-6, TNF-α, and caspase-3 expression; and increase Bcl-2 expression.
Conclusion
The whole plant and spicas water extracts of P. vulgaris have obvious protective effects on hyperthyroidism and hyperactivity in liver yang, and the protective effect of the water extract of spicas is greater than that of the whole plant water extract.
Keywords
Introduction
Prunella vulgaris L. (Lamiaceae) is a perennial medicinal plant that is distributed mainly in the Northern Hemisphere (e.g., Asia, Europe, and North America) and is commonly used to treat thyroid dysfunction, mastitis, and infectious hepatitis in Asian and European countries (Hu et al., 2023). P. vulgaris is rich in polyphenols, triterpenes, and polysaccharides and has pharmacological activities, such as anti-inflammatory, antioxidant, and immune regulatory effects (Akkol et al., 2022; Li et al., 2015; Mir et al., 2022; Wang et al., 2019; Wu et al., 2009). Our previous research revealed that the curative effect of P. vulgaris has been consistent from ancient to modern times in China, but there has been a historical change in the medicinal component, that is, from the whole plant to mature spicas (Chen et al., 2010). Till date, it has not been clear whether historical changes in the medicinal use of P. vulgaris affect the clinical efficacy equivalence of medicinal materials.
Hyperthyroidism is a common endocrine disease, and abnormal secretion of thyroid hormone is the main clinical symptom (Łacka & Fraczek, 2014). According to Traditional Chinese Medicine (TCM) syndrome differentiation, hyperactivity of liver yang is the most common syndrome type of hyperthyroidism (Yin et al., 2008). The etiology of hyperthyroidism is mostly caused by qi stagnation (a pathological change in which the liver is depressed), excessive liver fire, phlegm coagulation, and blood stasis, leading to yin deficiency and yang hyperactivity (Mu et al., 2020; Wang et al., 2020). Liver function is related to thyroid function, and liver cell activity depends on the regulation of thyroid hormones. Moreover, the liver plays a crucial role in the metabolism of hormones such as thyroid stimulating hormone (TSH), the thyroid hormone free triiodothyronine (FT3), and free thyroxine (FT4), thereby affecting the systemic endocrine function of the thyroid gland (Anugwom & Leventhal, 2021). There is a close relationship between liver function and thyroid function, suggesting that thyroid dysfunction is highly common in patients with chronic liver diseases. Chronic liver diseases of different etiologies have unique mechanisms of influence on the thyroid and can lead to different types of thyroid dysfunction (Zhang & Tang, 2021).
Therefore, our purpose was to evaluate the equivalence of P. vulgaris whole plants and spicas for the treatment of hyperthyroidism with liver-yang hyperactivity and to preliminarily explain the protective effect of P. vulgaris on hyperthyroidism with liver-yang hyperactivity. The results not only reveal the historical changes in the medicinal parts of P. vulgaris on a modern scientific basis but also provide new ideas for the comprehensive development and utilization of P. vulgaris medicinal plant resources.
Materials and Methods
Chemicals
P. vulgaris plants were collected in a medicinal garden (30°49’32.41” N, 104°12’7.3” E, 471 m above sea level) at Chengdu Medical College (Chengdu city, Sichuan Province, P. R. China) in early June 2022. The lateral roots of Aconitum carmichaelii Debeaux (Fuzi, or black sliced aconite) were purchased from Sichuan Qianfang Chinese Herbal Pieces (Zhong Yao Yin Pian) Co., Ltd. (Chengdu, China); levothyroxine sodium tablets were purchased from Merck KGaA (Darmstadt, Germany); detection kits, including TSH, FT3, FT4, ALT, AST, MDA, and SOD, were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China); and primary anti-rabbit TNF-α and IL-6 antibodies, primary antibodies against caspase-3 and Bcl-2, an internal reference β-actin antibody, and a secondary mouse anti-rabbit IgG antibody were purchased from Bioss Biotechnology Co., Ltd. (Beijing, China). Paraformaldehyde, TEMED, phosphate buffer, and other chemical reagents were purchased from Biosharp Biotechnology Co., Ltd. (Hefei, China).
Preparation of Extract
Fuzi decoction mixed with levothyroxine sodium (FDMWLS): Samples of Fuzi (black sliced aconite) were soaked for 30 minutes, after which the decoction was applied twice, after which the solution was combined and concentrated. Levothyroxine sodium tablets were crushed. The two were mixed into a single liquid containing 0.2 g of aconite and 75 µg of levothyroxine sodium per milliliter. The solution was stored at −4°C for later use.
P. vulgaris water extract: The dried powders of whole plants and spicas were soaked for 20 minutes, decocted twice, combined, and concentrated to afford a 1 g.mL−1 crude drug. The solution was stored at −4°C for later use.
Animal Model
Forty female rats (SPF grade), weighing 200 ± 20 g, were provided by the Animal Experimental Center of Chengdu Medical College (Chengdu City, Sichuan Province, P. R. China), and the rats were provided free and sufficient drinking water and feed. The laboratory-generated rats were divided into four groups (n = 10) at random: the normal control group (NC), the model group (MO), P. vulgaris whole plant water extract group (PvW), and P. vulgaris spicas water extract group (PvS). After adapting to feeding for 1 week, in addition to those in the NC group, the rats in the other groups were administered FDMWLS (10 mL·kg−1·d−1) at 9:00 AM every day, which was equivalent to administering the crude drugs Fuzi (black sliced aconite, 2 g·kg−1·d−1) and levothyroxine sodium (0.75 mg·kg−1·d−1) at the same time (Yin et al., 2008). The plants in the treatment group were given P. vulgaris whole plant and spicas water extract (10 mL·kg−1·d−1) at 3:00 PM, equivalent to crude drugs (5 g·kg−1·d−1). During the course of the experiment, changes in the behaviors and temperaments of the rats were observed, and the rectal temperature, body weight, and food intake of the rats were measured weekly for 28 consecutive days. After the last administration, the rats were fasted for 24 hours, weighed, and anesthetized by injecting 1 ml of 4% chloral hydrate solution into the abdominal cavity every 100 g.
Dissection
Heart apex blood was collected and centrifuged at 3,000 rpm min−1 for 10 minutes, after which the serum and aliquot were collected. The heart was removed to obtain the heart–body weight coefficient. A portion of the liver tissue was stored at −80°C. Liver tissue and thyroid glands on both sides of the rat neck were fixed with 4% paraformaldehyde.
Thyroid Function Index Detection
The levels of TSH, FT3, and FT4 in the serum samples of the rats in each group were quantified by a modified method according to the manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute, China) (Gan et al., 2021).
Liver Function Index Detection
The ALT and AST levels in the serum samples from the rats in each group were quantified using a detection kit according to the manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute, China) with an improved quantitative histopathological detection method (Gan et al., 2021).
Oxidative Index Detection
According to the manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute, China), the SOD activity and MDA content were quantified via a detection kit in the serum samples of the rats in each group.
Pathological Tissue Test
The liver and thyroid tissues of the experimental rats were cut into small pieces. Then, the tissue samples were fixed in paraformaldehyde for 24 h. Subsequently, the paraffin-treated tissue samples were cut into 4–6 µm thick sections with an embedding machine (B7126-A, Leica, Wetzlar, Germany) and a microtome (TK-218, Leica, Wetzlar, Germany). Hematoxylin and eosin (H&E)-stained tissue was observed under a positive fluorescence microscope (BX63, Olympus, Tokyo, Japan) (Gan et al., 2021).
Detection of Inflammatory Factors
Paraffin sections of liver tissue were added with BSA (5%) serum-blocking solution and incubated at 37°C for 15 minutes. Primary anti-rabbit, anti-IL-6, and TNF-α antibody (1:300; for negative controls, PBS was used instead of primary antibody) were added dropwise and incubated at −4°C overnight (more than 12 hours); then the secondary mouse anti-rabbit IgG antibody (1:300) was added dropwise and the solution was incubated at 37°C for 40 minutes and then washed with PBS. The expression level was detected by a DAB chromogenic kit (Boster, China) (Yu et al., 2020); sections were observed under a positive fluorescence microscope (BX63, Olympus, Tokyo, Japan); and the optical density values were obtained by using micrographs analyzed by an ImageJ simulation tool.
Apoptotic Protein Detection
Following the procedure of the mammalian total protein extraction kit (Biosharp, China), the total protein from each group of livers was extracted with the protein concentration of the BCA protein quantification kit. The proteins were electrophoretically separated via SDS-PAGE (Bio-Rad, CA, USA) and transferred to PVDF membranes. The cells were blocked in skim milk powder (5%) for 90 minutes, primary antibodies against caspase-3 and Bcl-2 (1:500) were added, and the internal reference β-actin (1:1,000) was incubated overnight. The secondary antibody (1:2,000) was added, and the samples were incubated at 37°C for 1 h (Yu et al., 2020). An appropriate amount of ECL chemiluminescent solution was added (Biosharp, China), protein bands were collected with a ChemiDocTM Touch Imaging System (Bio-Rad, CA, USA), and the bands were analyzed with the ImageJ simulation tool to obtain gray density values.
Statistical Analysis
All the experimental data were statistically analyzed with GraphPad Prism 7.0 (GraphPad Software, San Diego, CA, USA) and Origin 9.0 (OriginLab, Hampton, MA, USA). Comparisons among the groups were performed by one-way ANOVA, and there was a significant difference (p < 0.05).
Results
Growth Status, Food Intake, Body Weight, and Heart–Body Weight Coefficient
The activities, fur quality, and respiration of the rats were observed every day. Irritation, yellow fur, excessive eating, frequent defecation, and extreme resistance during gavage caused some rats to become emaciated. The rats in each cage were given 200 g of feed every day; the remaining feed was weighed the next day; the daily food intake was calculated for each cage; and the average daily food intake of the rats in each group was calculated for one week (Table 1). Compared with those in the NC group, the food intake in the other three groups significantly increased, and the food intake in the MO group increased the most rapidly. The body weights of the rats were measured every week (Table 2). The results showed that the NC group exhibited the most rapid weight increase. The body weight of the MO group was markedly lower than that of the NC group at 28 days after administration. The results showed that the rectal temperature of the rats in the MO group was markedly greater than that of the rats in the NC group, and the body temperatures of both the PvW group and PvS group were lower than that of the MO group. The heart-to-body weight ratio was significantly greater in the MO group than in the NC group (Table 3).
Effects of P. vulgaris Whole Plant and Spicas Water Extracts on the Food Intake (g) of Hyperthyroidism Patients with Liver-yang Hyperactivity Syndrome–Induced Rats.
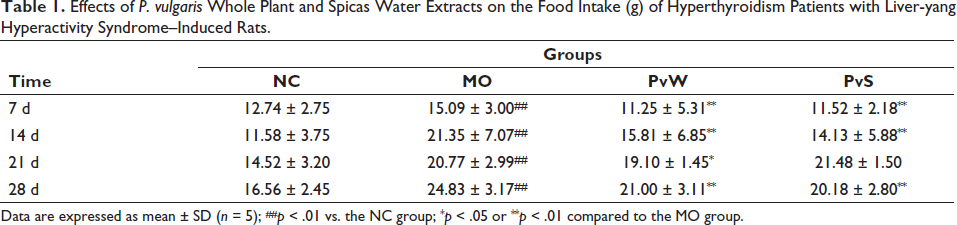
Data are expressed as mean ± SD (n = 5); ##p < 0.01 vs. the NC group; *p < 0.05 or **p < 0.01 compared to the MO group.
Effects of P. vulgaris Whole Plant and Spica Water Extracts on the Body Weight (g) of Hyperthyroidism Patients with Liver-yang Hyperactivity Syndrome–Induced Rats.
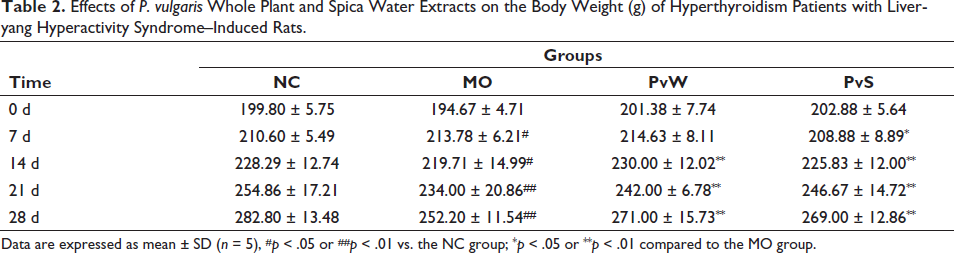
Data are expressed as mean ± SD (n = 5), #p < 0.05 or ##p < 0.01 vs. the NC group; *p < 0.05 or **p < 0.01 compared to the MO group.
Effects of P. Vulgaris Whole Plant and Spicas Water Extracts on the Rectal Temperature and Heart–Body Mass Ratio of Hyperthyroidism with Liver-yang Hyperactivity Syndrome–Induced Rats.
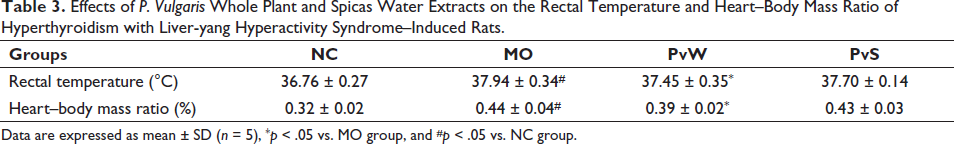
Data are expressed as mean ± SD (n = 5), *p < 0.05 vs. MO group, and #p < 0.05 vs. NC group.
FT3, FT4, and TSH Contents
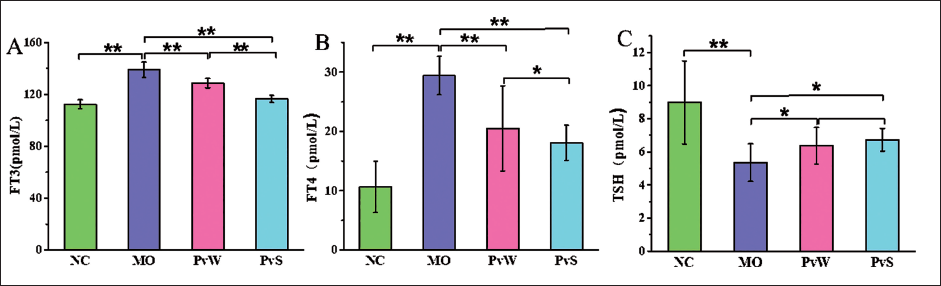
Compared with those in the NC group, the FT3 and FT4 levels in the MO group were markedly greater. However, the contents of FT3 and FT4 in both the PvW and PvS groups were significantly lower than those in the MO group. In addition, compared with that in the NC group, the TSH content in the MO group was lower, and the levels in both the PvW and PvS groups were significantly greater than those in the MO group (Figure 1).
ALT and AST Contents
Compared with that in the NC group, the alanine aminotransferase (ALT) content in the MO group was significantly greater, and the ALT content in both the PvW and PvS groups was markedly lower than that in the MO group. Compared with that in the NC group, the aspartate aminotransferase (AST) content in the MO group was markedly greater; however, the AST content in both the PvW and PvS groups was markedly lower than that in the MO group. The AST content in the PvS group was dramatically lower than that in the PvW group (Figure 2).
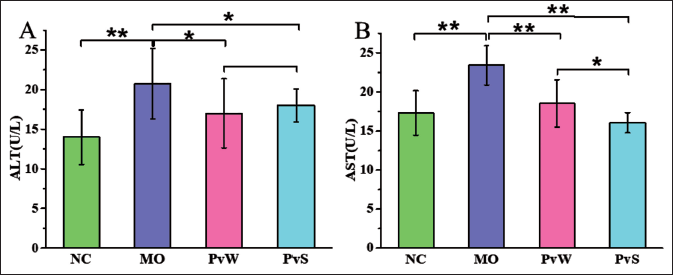
(A) ALT and (B) AST levels in the different groups.
HE Dye
The thyroid follicular structure of the rats in the NC group was complete, their shape was regular, they were round or polygonal, they were neatly arranged, and the follicular cavity was filled with colloids. Compared with those in the MO group, the follicular epithelial cells in the MO group were flat, and most of the follicular structures were destroyed and atrophied. Additionally, the follicular atrophy in both the PvW and PvS groups was alleviated compared with that in the MO group (Figure 3). The liver tissue structure for the rats in the NC group was classical and complete, and the hepatocytes were arranged radially. Compared with those in the MO group, the arrangements in both the PvW and PvS groups were more regular, and the pathological damage was reduced (Figure 4).
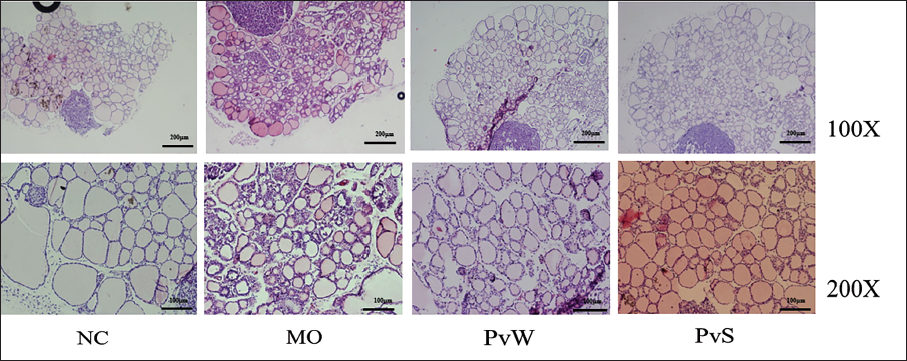
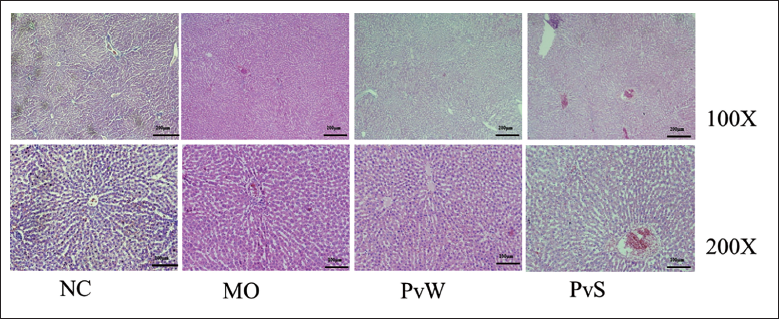
MDA Content and SOD Activity
Compared with that in the NC group, the malondialdehyde (MDA) content in the MO group was markedly greater, and compared with that in the MO group, the MDA content in both the PvW and PvS groups was markedly reduced among the PvS groups. In contrast, the superoxide dismutase (SOD) activity in the MO group was significantly lower than that in the NC group, and the SOD activity in both the PvW and PvS groups was significantly greater than that in the MO group. The SOD activity in the PvS group was markedly greater than that in the PvW group (Figure 5).
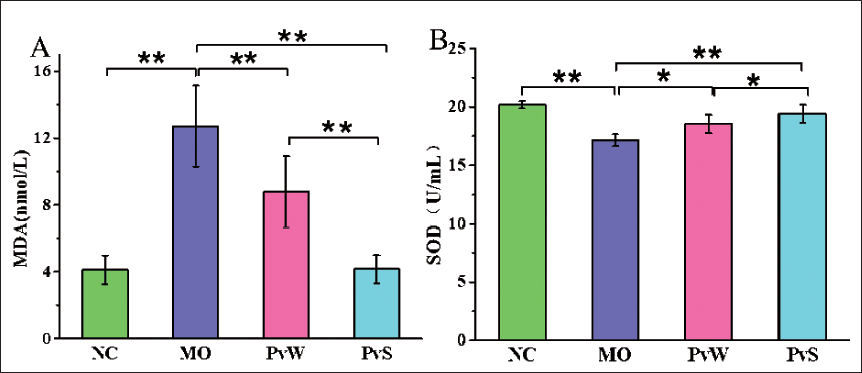
(A) MDA and (B) SOD levels in the different groups.
IL-6 and TNF-α Expression
Compared with that in the NC group, the IL-6 expression in the MO group was markedly greater, and the expression of IL-6 in both the PvW and PvS groups was obviously lower than that in the MO group. Similarly, the expression of the inflammatory factor antitumor necrosis factor-α (TNF-α) in the MO group was significantly greater than that in the NC group, and the TNF-α expression in both the PvW and PvS groups was markedly lower than that in the MO group; moreover, the TNF-α expression in the PvS group was markedly lower than that in the PvW group (Figure 6).
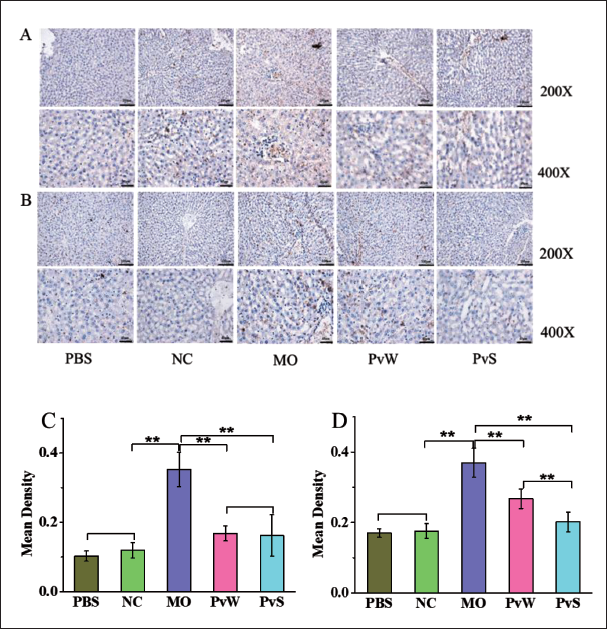
(A) Immunohistochemical Morphology of the Liver Inflammatory Factor IL-6. (B) Immunohistochemical Morphology of the Liver Inflammatory Factor TNF-α. (C) The Optical Density Value of the Inflammatory Factor IL-6. (D) The Optical Density Value of the Inflammatory Factor TNF-α.
Caspase-3 and Bcl-2 Expression
Compared with that in the NC group, the expression of the apoptosis protein caspase-3 in the MO group was markedly increased. The expression of caspase-3 in the PvW group was significantly reduced compared with that in the MO group, but there was no significant difference between the PvS and MO groups. Compared with that in the NC group, the expression of the apoptotic protein B-cell lymphoma-2 (Bcl-2) in the MO group was decreased. Compared with that in the MO group, Bcl-2 expression in both the PvW and PvS groups was greater (Figure 7).
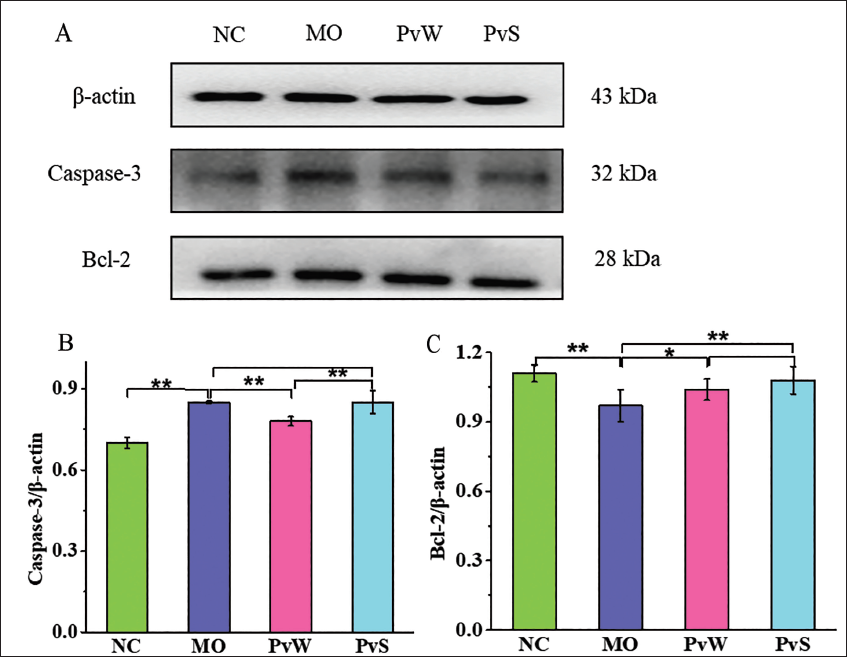
(A) The Protein Bands Corresponding to Caspase-3 and Bcl-2 in Liver Tissue. (B) The Relative Expression Levels of Caspase-3 Proteins in Liver Tissue. (C) The Relative Expression Levels of the Bcl-2 Protein in Liver Tissue.
Discussion
At present, the commonly used methods for the clinical treatment of hyperthyroidism include the use of anti-thyroid drugs, radioactive iodine, and thyroidectomy, which are often accompanied by side effects (Lin et al., 2021). The combination of TCM and Western medicine can improve the autoimmune status and clinical symptoms of patients. For example, P. vulgaris oral liquid is combined with other drugs to treat thyroiditis, thyroid nodules, and goiter (Graves disease) and can effectively shorten the treatment time, relieve symptoms, and improve the treatment efficacy (Experts of Clinical Application of Xiakucao Oral Liquid, 2020). According to the basic theory of TCM, the lateral root of A. carmichaelii (Fuzi) is a hot Chinese medicine (National Pharmacopoeia Commission, 2020), and the pharmacological effects of Fuzi are bidirectional; moreover, when long-term use of Fuzi can cause damage to essence and blood, it has toxic effects on the nervous, digestive, and reproductive systems as well as the heart, kidneys, liver, etc. (Shi et al., 2023). Additionally, P. vulgaris is a cold Chinese medicine, and cold and hot Chinese medicines can change the lipid metabolism of rats at the gene and protein levels (Yang et al., 2019). Interestingly, the results showed that whole plant and spicas of P. vulgaris water extracts could significantly improve the phenomenon of overeating and wasting in rats, reduce the FT3 and FT4 contents, increase the TSH content, and alleviate the damage to pathological thyroid tissue.
Thyroid hormones have a major impact on cardiac function and structure. Excessive thyroid hormone levels affect cardiovascular hemodynamics, leading to high-output heart failure and dilated cardiomyopathy in the late stage (Osuna et al., 2017). Our results showed that the whole plant and spicas of P. vulgaris water extracts have cardioprotective effects.
The liver plays a key role in the metabolism of T3, T4, and TSH, thereby affecting the systemic endocrine function of the thyroid (Chen et al., 2010; Łacka & Fraczek, 2014). Thyroid dysfunction is very common in chronic liver disease patients, and different types of thyroid dysfunction may manifest according to different causes. ALT and AST levels can predict diseases and the cause of diseases and can reveal body liver injury, fatty liver, and/or oxidative stress (Van Beek et al., 2013). The clinical diagnosis of liver diseases and structural integrity damage is usually based on the detection of blood AST and ALT activity levels (Zhou et al., 2018). Taken together, the results indicated that the whole plant and spicas of P. vulgaris water extracts significantly improve the liver functions of rats with hyperthyroidism and liver-yang hyperactivity syndrome.
In the rapid metabolic state of the body, activated mitochondria in liver cells can overproduce free radicals involved in oxidative stress, and oxidative damage is the fundamental cause of most liver diseases (Cichoż-Lach & Michalak, 2014). A comparison of rat body mass and food intake revealed that the food intake of the model group was remarkably greater than that of the NC group, while the body weight was significantly lower than that of the NC group. This may be due to the accelerated metabolism of rats with hyperthyroidism liver-yang hyperactivity syndrome, causing thyroid and liver damage (Sadasivam et al., 2022). Thyroid injury cascade events include excessive accumulation of reactive oxygen species (ROS), followed by a significant reduction in glutathione (GSH) levels and SOD activity; a noteworthy increase in malondialdehyde (MDA) content; myeloperoxidase (MPO) activity; and inducible nitric oxide synthase (iNOS) gene expression (Mohammed et al., 2020; Venditti & Meo, 2006), which can protect against hyperthyroid liver injury by increasing endogenous antioxidant (GSH) levels (Abliz et al., 2016). Taken together, our results indicated that whole plants and spicas of P. vulgaris water extracts can protect model rats with hyperthyroidism and liver-yang hyperthyroidism by enhancing antioxidant activity.
Hyperthyroidism can promote the release of inflammatory factors and exacerbate liver damage in patients (Xie et al., 2020). Abnormal levels of inflammatory factors are often associated with the pathogenesis of hyperthyroidism and can be used as diagnostic indicators for hyperthyroidism (Tang et al., 2020). By studying cytokine levels in the tears of patients with hyperthyroidism ophthalmopathy, the authors showed that the concentrations of the proinflammatory cytokines IL-6, interleukin-8 (IL-8), TNF-α, and interleukin-1β (IL-1β) significantly increase in the tears of patients with hyperthyroidism (Huang et al., 2012; Yoon et al., 2010); therefore, the increased expression of inflammatory factors may be caused by thyroid and liver damage. Rao’s study presented the novel finding that P. vulgaris can inhibit the TLR4/MyD88 signaling pathway to mediate hepatic inflammation (Rao et al., 2024). The results showed that the whole plant and spicas of P. vulgaris water extracts could regulate the expression of the inflammatory cytokines IL-6 and TNF-α to protect rats from hyperthyroidism and liver-yang hyperactivity.
The PvW and PvS groups exhibited decreased expression of caspase-3 and significantly increased expression of Bcl-2. The Bcl-2 protein family members are key components involved in regulating the intrinsic apoptotic pathway and have antiapoptotic effects, while caspase-3 has a promoting effect on cell apoptosis, with increased expression indicating a significant liver injury (Patel et al., 2019). A previous study demonstrated that thyroid hormone–induced liver injury in rats can be achieved by regulating mitochondria-mediated apoptotic signals (Yang et al., 2020). One study showed that the total flavonoids in P. vulgaris may be related to activation of the PI3K/Akt/mTOR pathway (Song et al., 2021). Our results indicated that whole plant water extracts and spicas from P. vulgaris protect against hyperthyroidism in rats with liver-yang hyperactivity by adjusting and controlling the expression of apoptosis-related proteins.
P. vulgaris has been found to be effective in treating a variety of thyroid-related human disorders (Ahmad et al., 2020; Zhang et al., 2022). Moreover, the bioactive ingredients in the water extract of P. vulgaris spicas are mainly polysaccharides and flavonoids, both of which have pharmacological activities, such as antioxidant, immunoregulatory, and anti-inflammatory effects, and have synergistic protective effects on hyperthyroidism with liver-yang hyperactivity (Akkol et al., 2022; Li et al., 2015; Mir et al., 2022; Wang et al., 2019; Wu et al., 2009). According to our study’s results, the water extracts of whole plants and spicas of P. vulgaris had a protective effect on hyperthyroidism caused by the liver-yang syndrome and hyperthyroidism. Hence, the water extract of P. vulgaris spicas had a greater protective effect on hyperthyroidism and liver-yang hyperthyroidism than the whole plant water extract.
Conclusion
This study provides evidence that both the whole plant and spicas of P. vulgaris significantly reduce FT3 and FT4 levels, increase TSH levels, and decrease ALT and AST activity. P. vulgaris water extract can prevent oxidative damage, reduce the expression of inflammatory factors and apoptotic proteins, and thereby protect against thyroid and liver damage caused by hyperthyroidism-related liver-yang hyperactivity syndrome. Moreover, the protective effect of the spica water extract was greater than that of the whole plant water extract. P. vulgaris is a homologous bulk medicinal material used in Chinese medicine and food, and its effects were the same in ancient and modern times; however, there have been historical changes in its medicinal uses. These findings can enrich the understanding of the expression of the medicinal parts of P. vulgaris, promote the comprehensive development and utilization of P. vulgaris resources, and offer new insights for medicinal plant resource research.
Footnotes
Acknowledgments
The authors thank the College of Pharmaceutical Sciences, Chengdu Medical College, Chengdu, Sichuan, for providing the facilities to support this study.
Authors’ Contributions
Yuhang Chen and Shuang Li designed the experiments. Chao Yan prepared the samples. Shuang Li, Zhiguo Gong, Xiangling Hu, and Yufan Li performed the experiments and analyzed the data. Shuang Li, Liping Cao, and Yuhang Chen wrote and revised the manuscript. All authors read and approved the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of Chengdu Medical College.
Funding
This study was funded by programs of the National Natural Science Foundation of China (No. 31500263), the Sichuan Science and Technology Program (No. 19YJ0368; No. 2020ZHFP0060), the Sichuan Province Innovation and Entrepreneurship Training Program for College Students (No. S202013705033; No. S202113705056; No. 202313705025; No. 202313705033), and the special project of Liyan workshop aesthetic medicine research center of Chengdu Medical College (No. 21YM003).
