Abstract
Background
This study investigated the therapeutic effects of Rosa canina L. seed oil (RCSO) on PCOS using an animal model, focusing on evaluating biochemical factors and the Bax/Bcl-2/p53 and Caspase-3 (Cas-3) pathways.
Materials and Methods
The PCOS model was created by injecting estradiol valerate into 60 adult Wistar rats, which were assigned to six groups of 10 animals: normal, PCOS, metformin, RCSO, and co-treatment with MET and RCSO groups. After 50 days, the serum levels of testosterone, progesterone, LH, and FSH hormones were measured, along with the activity of catalase, superoxide dismutase, glutathione peroxidase, and nitric oxide. Additionally, ovarian tissues were taken for gene expression analysis of Bax/Bcl-2/p53 and Cas-3 using real-time PCR, as well as protein expression analysis with western blot. Immunohistochemistry was performed to assess the levels of p53 and Ki-67 proteins, indicating the apoptosis of ovarian granulosa cells.
Results
RCSO, in synergy with MET, demonstrated reductions in the LH/FSH ratio along with a decrease in progesterone and increased activity of antioxidant enzymes. Additionally, RCSO, along with MET, upregulated the Bax/Bcl-2/p53 and Cas-3 pathways, resulting in increased p53 expression and decreased Ki-67-positive cells, indicative of enhanced apoptosis.
Conclusions
These findings suggest the potential use of RCSO in women with ovulation disorders, particularly those with PCOS.
Introduction
45% of the reasons for infertility in couples are due to disorders related to women, and polycystic ovary syndrome (PCOS) includes 12%–15% of these disorders. PCOS is a complex condition influenced by endocrine, metabolic, and genetic factors (Azziz et al., 2019). It is characterized by various symptoms, such as hirsutism, irregular menstrual cycles, oligo/anovulation, hyperandrogenism, and the presence of polycystic ovaries. In PCOS, there is a dysregulation of the hypothalamic–pituitary–ovary (HPO) feedback axis, leading to an elevated level of luteinizing hormone (LH) compared to follicle-stimulating hormone (FSH). This imbalance suppresses steroidogenesis in both granulosa and theca cells. Furthermore, the increased LH/FSH ratio inhibits the activity of 17-hydroxylase and 17,20-lyase enzymes, resulting in elevated testosterone (T) levels instead of estrones (Ganie et al., 2019).
In PCOS, there is an elevation in reactive oxygen species (ROS) and nitrogen species (RON), leading to the suppression of endogenous antioxidant enzymes. Studies suggest that individuals with PCOS may exhibit alterations in the activity and expression of antioxidant enzymes, such as superoxide dismutase (SOD), catalase, and glutathione peroxidase. These enzymes play a crucial role in neutralizing ROS to maintain cellular homeostasis. This, in turn, negatively affects insulin signaling, inhibits the activity of enzymes in the cytochrome P450 system, and disrupts the mitochondrial apoptotic pathway in granulosa cells (Gong et al., 2020; Zhu et al., 2019). The Bax/Bcl-2/p53 and Caspase-3 (Cas-3) pathway is a significant axis involved in androgen modulation, stimulation of steroidogenesis in theca and granulosa cells, promotion of dominant follicle differentiation, and increased mitochondrial apoptosis of granulosa cells (Wang et al., 2021). In PCOS, alterations in Bax and Bcl-2 expression have been reported. Studies have shown decreased Bax expression within the ovarian tissues of women with PCOS, which suggests an enhanced pro-apoptotic influence. Also, studies have reported downregulation of Cas-3, indicating suppressing apoptosis within ovarian follicles of women with PCOS (Cui et al., 2022). Insulin resistance and hyperinsulinemia, frequently observed in PCOS, can dysregulate apoptosis-related gene expression. Elevated insulin levels may promote Bcl-2 expression and decrease Bax expression, shifting the balance toward apoptosis. Androgen excess, a hallmark of PCOS, can also impact the expression of these genes, although the specific mechanisms are not yet fully understood. Studies have shown that the utilization of certain whole extracts or plant-derived compounds not only enhances the hypothalamic–pituitary–gonadal (HPGA) axis through their aphrodisiac properties but also reduces free radicals, thereby leading to an overall increase in the ovarian parenchyma’s total antioxidant capacity (Xu & Qiao, 2022).
Rosa canina L. seed oil (RCSO) is derived from the Rosaceae family and has been traditionally used in therapeutic doses to address various conditions such as cardiovascular disorders, menopausal symptoms, urogenital tract issues, anorexia, and skin lesions. With its abundant proteins, vitamins, carbohydrates, and minerals, RCSO finds applications in food products, antimicrobial films, and the preservation of oil quality (Soltan et al., 2023). Throughout history, RCSO powder and extracts have been utilized in the preparation of cakes, soups, and decoctions to alleviate various ailments. Liquid chromatography-electrospray ionization-tandem mass spectrometry (LC-ESI-MS/MS) analysis has revealed that RCSO is rich in essential fatty acids (oleic acid, linoleic acid, and palmitic acid), isoflavonoid compounds (luteolin, biochinin A, formononetin, and daidzein), anthocyanins, carotenoids, flavonoids (caffeic acid, apigenin, quercetin, gallic acid, and kaempferol), and saponins (Charpentier et al., 2022). It has been suggested that RCSO can improve reproductive and digestive system functions and aid in the treatment of conditions such as hyperlipidemia, Alzheimer’s disease, and various endometrial, brain, and breast tumors (Khazaei et al., 2020). Key compounds found in RCSO, such as kaempferol, daidzein, quercetin, biochinin A, apigenin, and quercetin, have been shown to stimulate the HPO axis and the PI3K/AKT pathway, promoting steroidogenesis through the activation of enzymes such as 17-hydroxylase, 17,20-lyase, 17β-hydroxysteroid dehydrogenase, and 5α-reductase (Dubey et al., 2021; Rashidi et al., 2021).
This study aimed to examine the impact of RCSO on endocrine and biochemical parameters, specifically focusing on antioxidant effects, as well as the modulation of folliculogenesis and granulosa apoptosis. The study utilized an estradiol-induced PCOS rat model to investigate the therapeutic effects of RCSO. Furthermore, the Bax/Bcl-2/p53 and Cas-3 pathways, which play crucial roles in driving granulosa cell apoptosis, were also evaluated.
Materials and Methods
Reagents, Antibodies, and Instruments
Soil grinding was performed by a soil grinder (Humboldt Co., USA; Cat. No. H-4199.5F), and filtration was carried out using paper filters (No. 42 Cat. No. 1442-125; Millipore, USA). The rotary evaporator used was a Buchi Rotavapor (model: 9230; Buchi Rotavapor, Switzerland). LH (Cat. No. NBP2-68054), FSH (Cat. No. KA2330), T (Cat. No. KG010), and P (Cat. No. NBP2-60127) hormone levels were measured using Novus ELISA kits (Novus Biologicals, USA), and ELISA readings were obtained with an ELISA reader (Milton Roy Company, Spain; Model No. Spectronic 20). NO concentrations were determined by the ZellBio commercial kit (ZellBio GmbH, Germany; Cat. No. ZX-44107-96), and a UV-visible spectrophotometer (JASCO, USA; Model No. V-630 Bio) was used. GPx activity was assessed with the Elabscience GPx ELISA kit (Elabscience, USA; Cat. No. E-EL-R2491); SOD activity was measured using the Elabscience SOD ELISA kit (Cat. No. E-EL-R1424; Elabscience, USA); and CAT activity was determined using the Abcam CAT ELISA kit (Cat. No. ab277396; Abcam, USA). Total RNA purification was conducted using the Bio Basic column animal total RNA purification kit (Bio Basic Inc., Canada; Cat. No. EZ-10), followed by cDNA synthesis using the Takara cDNA synthesis kit (Takara Bio., Japan; Cat. No. RTK0104) and amplification in an applied biosystems thermocycler (Quantstudio 1, USA). SDS polyacrylamide gel electrophoresis was performed using the Millipore Bio Inc. SDS polyacrylamide gel (Cat. No. WBKLS0100; Millipore Bio Inc., UK). For immunoblotting, primary antibodies for Bax (1:500; Cat. No. GTX109683), Bcl-2 (1:200; Cat. No. GTX100064), p53 (1:400; Cat. No. GTX70214), and Cas-3 (1:200; Cat. No. GTX1105403) were obtained from GeneTex. Microscopic observations were made by an optical microscope (BX61TRF; Olympus, Japan). Chemiluminescence detection was performed using a chemiluminescence reagent provided by the e-BLOT company (China). Additionally, primary antibodies for p53 (1:500; Cat. No. AF1355) and Ki-67 (1:200; Cat. No. AF7617) were obtained from R&D Systems, Inc. (USA).
Preparation of Seed Extracts
Preparation of RCSO
After obtaining 1,000 g of dried RCS and grinding it into a powder by a soil grinder, the obtained powder was added to a hexane and methanol solution (30:70 v:v). This mixture then underwent incubation for 72 hours at 40°C in the dark. To remove impurities, a paper filter was used. The filtered solution was further concentrated using a rotary evaporator. Finally, the resulting oil extract, weighing 250 g and exhibiting an extraction efficiency of 20%, was kept at −4°C (Kaushik et al., 2020).
Experimental Design
Sixty adult female Wistar rats (three months old; 200 ± 20 g) were housed in propylene cages under controlled conditions. The temperature was kept at 22 ± 3°C under 12:12 hour light–dark cycle with a relative humidity of 40 ± 3%. Prior to inducing the PCOS model, adaptation to standard laboratory conditions was done for 24 hours. During this period, animals had unrestricted access to standard laboratory pellets and clean tap water. Each 100 mg pellet comprises 4 Mcal energy, with 25 g of protein, 45 g of carbohydrates, and 50 g of total fat. Additionally, it contains 0.9 g of phosphorus, 1.2 g of calcium, 0.2 g of magnesium, 0.2 g of cobalt, 0.3 g of iron, and 0.9 g of sodium, as well as 250 mg each of linoleic acid, palmitic acid, and arachidonic acid. The rats were maintained following international standard instructions and protocols and the ethics committee of the Tangdu Hospital Air Force Medical University (Li & Hong, 2020).
Animals and Grouping
100 rats were subjected to intramuscular injection of estradiol valerate at a dosage of 2 mg/kg, dissolved in 0.5 ml of olive oil. The injections were administered twice, with a one-week interval between each injection. After a period of 30 days, the rats were evaluated based on clinical indicators such as permanent vaginal cornification, assessment of vaginal smears, and observation of vaginal plaques. As a result, 60 rats (n = 10 per group) were selected and grouped for further study.
In the sham group, normal rats were administered 1 ml of distilled water (DW) daily via gavage for a duration of 50 days. The PCOS group consisted of PCOS rats who were given 1 ml of DW daily via gavage for 50 days. The PCOS + MET group comprised PCOS rats who received 300 mg/kg of MET (dissolved in 1 ml of DW) daily via gavage for a period of 50 days. The 250 and 500 RCSO + PCOS groups consisted of PCOS rats receiving 250 and 500 mg/kg of AKO, respectively, daily via gavage for 50 days. In the 500 RCSO + MET + PCOS group, PCOS rats were given 500 mg/kg of RCSO along with 300 mg/kg of MET (dissolved in 1 ml of DW) daily by gavage for 50 days.
During the research, which lasted for 50 days, the rats were administered RCSO daily at 9:00 a.m. and MET at 3:00 p.m. To ascertain the suitable dosage of MET, various references and a pilot study were consulted, and in order to select the appropriate dose of RCSO, the LD50 technique was employed alongside the references and pilot study. This approach ensured the utilization of the most efficient and safe therapeutic dosage (Parandin et al., 2023; Venegas et al., 2019).
LD50 for RCSO
To assess the most suitable treatment dosage while minimizing toxicity, we applied Lork’s two-step method. Initially, we divided nine rats into three groups, with each group containing three rats, followed by administrating the doses (50–5,000 mg/kg) through oral gavage. Subsequently, we allocated another three rats to three separate groups followed by administrating the doses (10–1,000 mg/kg) by oral gavage. We closely observed these groups for a period of 24 hours. By using the provided formula, we calculated the LD50 (median lethal dose) of RCSO, which helped us determine the appropriate dosage for our study.
LD50 = (A × B)1/2
Here, A denotes the minimum dosage at which toxic manifestations (diarrhea, weight reduction, queasiness, and skin irritations) or fatality were noted, while B signifies the maximum dosage wherein neither mortality nor toxic symptoms were found within the group (Zhang et al., 2023).
Serum Concentrations of Follicle-stimulating Hormone, Testosterone, Progesterone, and Luteinizing Hormones
After the experiments (on the 51st day), rats were euthanized by a pre-anesthesia protocol of xylazine 2% (100 mg/kg) followed by anesthesia with ketamine 10% (15 mg/kg). The LH, FSH, T, and progesterone (P) serum levels were measured by commercial ELISA kits as instructed (Xu et al., 2019).
Nitric Oxide Concentrations in Serum
Serum total nitric oxide (NO) concentrations were quantified by a commercially available ELISA kit. To perform the assay as instructed, we added 50 µl of either the serum or the standard sample to all wells in a 96-well plate. Additionally, 50 µl of assay buffer was introduced as a zero standard. Subsequently, each well received 10 µl of nitric reductase solution. After an incubation period of 20 minutes at a temperature of 25°C, we added 25 µl of solutions A and B from the kit to all wells, followed by another 20-minute incubation at 25°C. Ultimately, we measured the mixture absorbance at 540–570 nm (Malfertheiner et al., 2023).
Serum SOD, CAT, and GPx Activity
The measurement of serum activity of GPx, CAT, and SOD was conducted using the commercial ELISA kit (Mohammad et al., 2022).
Real-time PCR Assay for Bcl-2, Bax, p53, and Cas-3 Genes Expression
RNA Purification
To extract total RNA from ovarian tissue, we employed the column animal total RNA purification kit (Bio Column). We initiated the procedure by grinding 50 mg of ovarian tissue into a fine powder using liquid nitrogen (N2). The powdered tissue was subsequently placed into tubes, including Buffer Rlysis-AG (350 µl). Following incubation of the mixture at 25°C for 5 minutes, we introduced 300 µl of ethanol and gently mixed it in. Then, it was transported to a new column and centrifuged (15 seconds at 14,000g). Following this, we performed a two-step process by adding 0.5 ml of NT and GT solutions to the mixture and centrifuging for 15 seconds at 14,000g. Finally, we supplemented the mixture with 50 µl of RNase-free water. After a 2-minute incubation, the obtained mixture underwent centrifugation for 15 seconds at 14,000g (Kritikos et al., 2021).
Then, the cDNA synthesis kit was used to perform cDNA synthesis from ovarian tissue. Following the instructions of the producer, the procedure commenced by preparing a mixture including oligo/random primers (1 µl for each primer), 1,000 ng of total RNA, deionized water (8 µl), and Takara Bio Mastermix (10 µl).
The real-time PCR analysis was conducted using reverse and forward primers (1 µl), cDNA templates (1,000 ng), and Takara Bio Mastermix (8 µl). The PCR mixture was obtained, and the amplification was performed in a thermocycler over 42 temperature cycles. Each cycle consisted of an annealing stage (2 minutes at 60°C). Subsequently, a melting curve was built by a gradual increase in temperature (60°C to 95°C) at 1°C/second. The Bcl-2, Bax, p53, and Cas-3 expression levels were measured during this process. The threshold cycle (CT) values for the target genes [control, sample, and β-actin (as internal reference genes)] were determined and applied in the fold change method. Primer design was conducted by Oligo Primer v3.2.3. software, and the primers were approved by blasting them against the NCBI (Table 1).
Primer Sequences.

Fold change = 2−(Ct sample − Ct internal gene) − (Ct sample − Ct control) (Changizi et al., 2021).
Western Blotting Assay
The evaluation of Bcl-2, Bax, p53, and Cas-3 pathway proteins expression was conducted through the utilization of the western blotting technique. Initially, the ovarian tissue was rinsed using PBS (200 µl). Subsequently, 50 mg of ovarian homogenized tissue was used in radio-immunoprecipitation buffer, which comprised NaCl (150 mmol/l), NP-40 (0.1%.), and Tris-HCl (50 mmol/l). Following this, the resulting mixture was centrifuged (−4°C/10 minutes/12,000g). To prepare the samples, 50 µg of the aforementioned mixture was added to sodium deoxycholate (5 µl), β-mercaptoethanol (1 µl), glycerol (5 µl), and Tris-HCl (2 µl). This mixture was incubated for 10 minutes before being transferred onto a PVDF membrane. Additionally, a 10% SDS polyacrylamide gel was loaded. The PVDF membrane harbored antibodies for Bax, p53, Bcl-2, and Cas-3, which were procured from GeneTex Bio., USA. Following overnight incubation (4°C), HRP-conjugated secondary antibodies were introduced and incubated for an additional hour at 37°C. Excess antibodies were blocked using Tris-buffered saline. The signals emitted by the protein bands were captured and assessed by Bio-Rad software in conjunction with ImageJ software (Wang et al., 2019).
Immunohistochemistry Assay
The expression levels of Ki-67 and p53 proteins in ovarian follicular and parenchymal cells were examined as indicators of apoptotic differentiation. The procedure began with the washing of ovarian tissues using PBS, followed by routine tissue processing. Paraffin blocks are then created using the ovaries, and 5 µm slices of tissue were placed onto slides. Next, all slides were subjected to incubation at 95°C overnight, followed by an additional incubation at 25°C for 1 hour with primary antibodies for p53 and Ki-67. Subsequently, the slides received 3% hydrogen peroxide (H2O2) at 25°C for 20 minutes and then underwent 3,3’-diaminobenzidine (DAB) staining. Hematoxylin was applied as a counterstain to all slides. The evaluation of the slides was done using an optical microscope attached to a light microscope at a magnification of 100×. p53 and Ki-67 positive cells/ total counted cells × 100 (Pisani et al., 2020).
Ovarian Tissue Assay
The assessment of follicular differentiation and histopathological observations concerning cystic follicles, inner and outer theca layers, as well as granulosa and corona radiata cells was conducted at a magnification of 100× using an optical microscope connected to ImageJ. Consistent with the IHC assay, all ovary sections were evaluated using hematoxylin and eosin staining (Yu et al., 2020).
Statistical Analysis
GraphPad Prism and SPSS 16.0 were used in the design of graphs and statistical analysis, respectively. One-way analysis of variance (ANOVA) followed by the Newman–Keuls post hoc test was used to compare means (p < 0.05 was considered significant).
Results
LD50 of RCSO
According to the results of 24-hour monitoring of the groups, the highest safe and lowest fatal doses were 5,000 mg/kg and 1,000 mg/kg, respectively. According to Lorek’s formula, the LD50 of RCSO was 2236 mg/kg, which can be used in studies with lower doses.
Rat’s Body and Ovary Weight
PCOS led to a significant (p < 0.05) elevation in weight parameters (body weight and ovarian weight) compared to the sham group. Nonetheless, RCSO, with its synergistic effects, caused a dose-dependent decrease in both weight parameters in comparison to the PCOS group. This reduction was particularly significant (p < 0.05) in the 500 mg/kg RCSO (in the 500 RCSO + MET + PCOS group) (Figure 1).
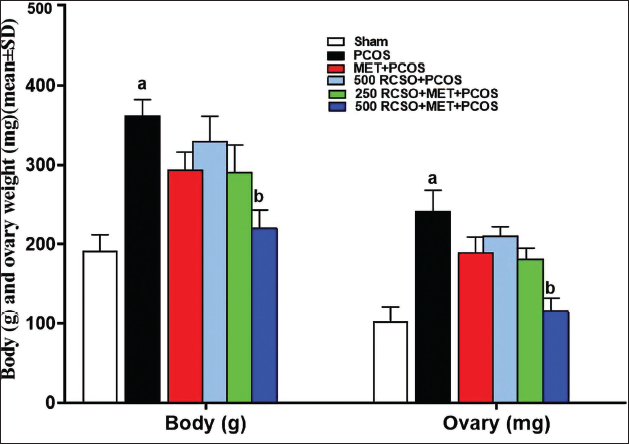
Serum FSH, P, T, and LH Levels
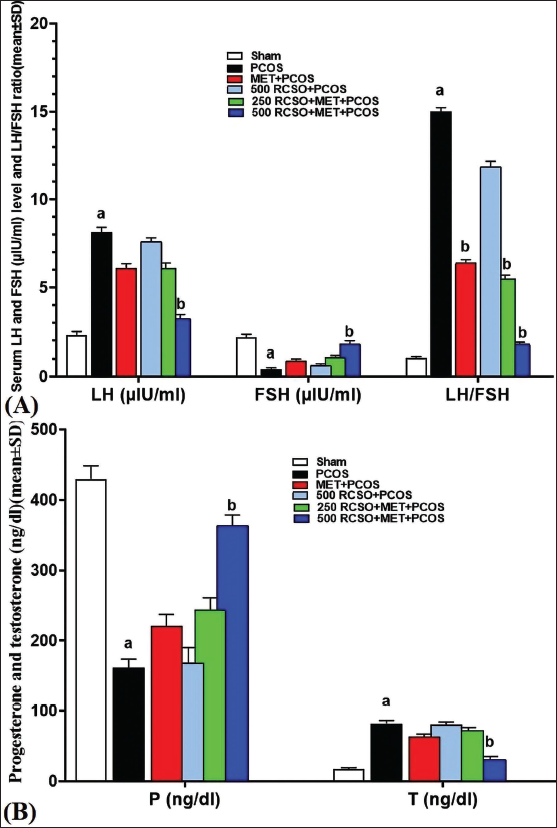
After evaluating the serum levels of hormones related to PCOS, it was found that in comparison to the sham group, the LH level was significantly (p < 0.05) higher and the FSH level was lower in PCOS rats. This ultimately resulted in a significant (p < 0.05) elevation in the LH/FSH ratio. Additionally, in PCOS rats, the serum T level was significantly (p < 0.05) higher, while the serum P level was significantly (p < 0.05) lower compared to the sham group. RCSO, along with MET, exhibited a dose-dependent regulation of the HPG axis and effectively modulated the hormones associated with PCOS compared to the PCOS group. Specifically, in the 500 mg/kg RCSO (in the 500 RCSO + MET + PCOS group), there was a significant (p < 0.05) reduction in serum LH concentrations and a notable increase (p < 0.05) in FSH concentrations. Consequently, this shift led to a significant (p < 0.05) reduction in the LH/FSH ratio compared to the PCOS group. Also, in the 500 mg/kg RCSO group (in the 500 RCSO + MET + PCOS group), there was a significant (p < 0.05) increase in serum P concentrations and a significant (p < 0.05) decrease in T concentrations in comparison to the PCOS group (Figure 2).
Serum NO Levels Along with GPx, SOD, and CAT Activity
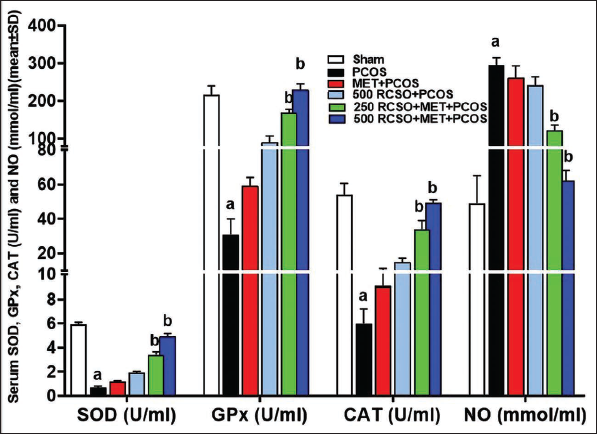
PCOS triggered a notable (p < 0.05) increase in NO levels compared to the sham group, primarily through the generation of harmful free radicals. Moreover, this surge in free radicals substantially hindered and significantly (p < 0.05) diminished the activities of endogenous antioxidant enzymes such as GPx, SOD, and CAT in comparison to the sham group. On the other hand, RCSO, being abundant in polyphenolic compounds, exhibited a dose-dependent reduction in NO levels relative to the PCOS group. This reduction reached significance (p < 0.05) in the 250 and 500 mg/kg RCSO treatment groups (i.e., the 250/500 RCSO + MET + PCOS group). Additionally, in the 250/500 RCSO + MET + PCOS group, RCSO at 250 and 500 mg/kg caused a significant (p < 0.05) elevation in the serum activity of all three antioxidant enzymes—GPx, SOD, and CAT—in comparison to the PCOS group (Figure 3).
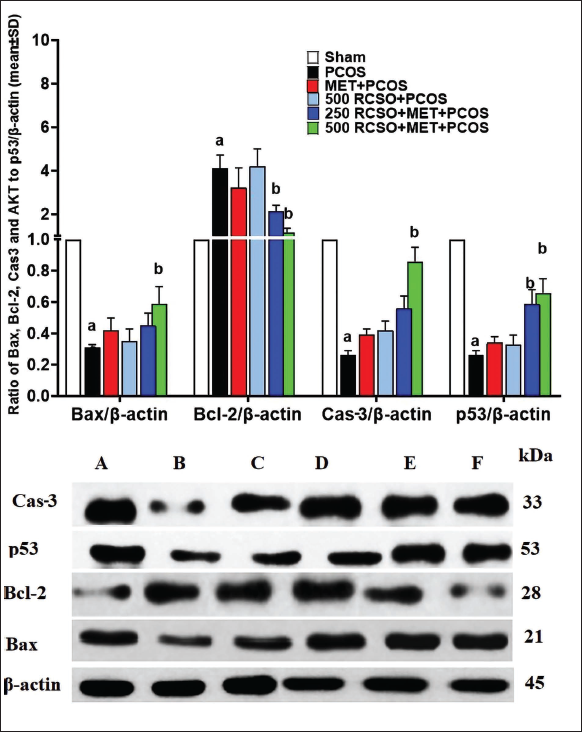
Expression of Ovarian Bax, Bcl-2, p53, and Cas-3 Genes
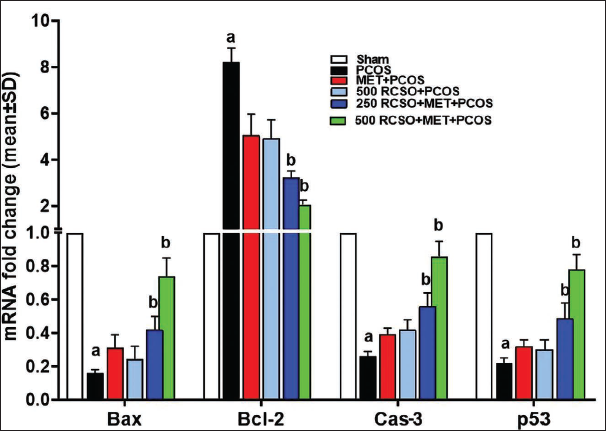
PCOS disrupts the natural apoptosis process of ovarian cells, particularly granulosa and theca parenchymal cells. The findings of this study revealed that PCOS notably (p < 0.05) impeded the expression of pro-apoptotic genes such as Bax/p53 and Cas-3 compared to the sham group. Conversely, the anti-apoptotic gene Bcl-2 expression displayed a significant (p < 0.05) decrease. RCSO, along with MET, dose-dependently exerted a synergistic effect by promoting the expression of pro-apoptotic genes while inhibiting the expression of anti-apoptotic genes in comparison to the PCOS group. Intriguingly, both doses of RCSO at 250 and 500 mg/kg, along with MET, significantly (p < 0.05) elevated the expression of pro-apoptotic genes Bax/p53 and Cas-3 relative to the PCOS group. Moreover, the anti-apoptotic gene Bcl-2 expression experienced a remarkable (p < 0.05) reduction in apoptosis (Figure 4).
Expression of Ovarian Bax, Bcl-2, p53, and Cas-3 Proteins
After analyzing the results pertaining to the Bax, Bcl-2, p53, and Cas-3 expression levels, it was observed that PCOS caused a significant (p < 0.05) reduction in Bax/p53 and Cas-3 proteins, along with a notable (p < 0.05) elevation in Bcl-2 protein expression, compared to the sham group. These findings indicated that PCOS inhibits apoptosis in ovarian parenchymal cells. However, it is noteworthy that RCSO at 250 and 500 mg/kg, in conjunction with MET, enhanced apoptotic pathways. This enhancement was evident through a significant (p < 0.05) increase in Bax/p53 and Cas-3 protein expression, coupled with a significant (p < 0.05) reduction in Bcl-2 protein expression, as compared to the PCOS group (Figure 5).
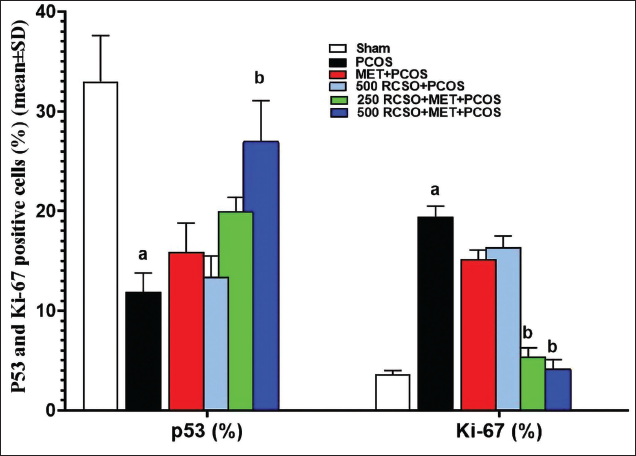
Ki-67 and P53-positive Cells (%) in Ovarian Tissue
After evaluating the results of ovarian tissue immunohistochemistry to evaluate Ki-67 protein and p53 apoptotic indices, it was found that PCOS caused a significant (p < 0.05) decrease in percentage of p53 positive cells and a significant (p < 0.05) increase in percentage of Ki-67 positive cells compared to the sham group. RCSO at 250 and 500 mg/kg significantly (p < 0.05) reduced the percentage of Ki-67 positive cells compared to the PCOS group. In addition, RCSO at 250 and 500 mg/kg significantly (p < 0.05) increased the percentage of p53 positive cells in comparison to the PCOS group (Figures 6 and 7).
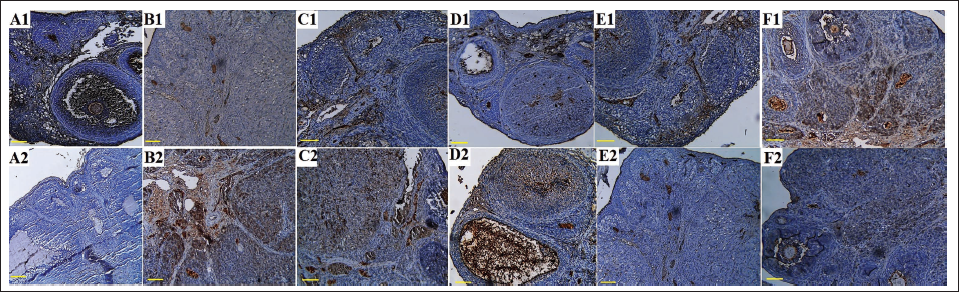
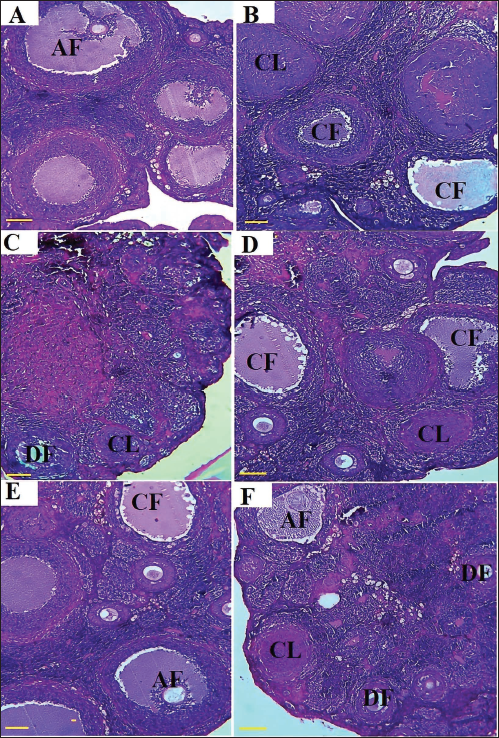
Histopathology of Ovarian Tissue
PCOS leads to an increase in cystic follicles (CF) on the cortical surface of the ovaries. Moreover, PCOS is associated with edema in CF, collagen fiber synthesis, and lymphocyte infiltration into the ovarian germinal parenchyma. This process results in a reduction in the number of differentiating follicles (DF) and graafian (antral) follicles (AF), with fibrotic tissue replacing the developing follicles. The administration of RCSO, either alone or in combination with MET, effectively prevented the formation of CF and increased the number of DF. Additionally, RCSO mitigated the formation of fibrotic tissue and collagen fibers, thereby preserving the normal architecture of the germinal parenchyma (Figure 8).
Discussion
In the current study, it was observed that RCSO effectively reduced CF and improved the normal functioning of ovaries. This was achieved through the strengthening of the HPG axis, along with antioxidant effects and the regulation of apoptosis in ovarian parenchymal cells.
In PCOS, the LH/FSH ratio, which increases during the condition, inhibits the function of granulosa and theca cells, leading to suppressed steroidogenesis. Consequently, the levels of androgen metabolites, such as testosterone and dehydroepiandrosterone, rise. Additionally, PCOS disrupts the HPO axis, resulting in increased LH levels and decreased FSH levels (Saadia, 2020). In this study, PCOS inhibited the HPO axis, ultimately leading to an increased LH/FSH ratio. Consequently, the androgen (T) level increased, while the serum level of P decreased due to suppressed steroidogenesis. Plants containing polyphenols, particularly isoflavonoids, have the ability to regulate the HPO axis by reducing the LH/FSH ratio. These compounds can lower the LH rate compared to FSH by modulating the levels of gonadotropin-releasing hormone (GnRH) (Ma et al., 2021). Studies have demonstrated that isoflavonoids, in addition to reducing the LH/FSH ratio, enhance steroidogenesis in granulosa and theca cells, leading to increased estrone levels and inhibition of androgen production (Darabi et al., 2020). In the present study, RCSO, rich in polyphenols, including isoflavonoids, increased steroidogenesis by reducing the LH/FSH ratio. Consequently, the level of P increased, while the production of androgen (T) decreased. This can be attributed to the high content of polyphenols, especially isoflavonoids, in RCSO.
In PCOS, the excessive production of free radicals inhibits enzymes such as 5α-reductase and 17,20-lyase/17-hydroxylase, leading to the suppression of steroidogenesis in ovarian theca and granulosa cells. Research indicates that PCOS also hampers endogenous antioxidant enzymes and the signaling pathways reliant on free radicals, such as the PI3K/AKT pathway, resulting in a decrease in overall systemic and tissue antioxidant capacity (Jiang & Li, 2022). In this study, PCOS was associated with elevated levels of NO and reduced activity of CAT, GPx, and SOD. Studies have demonstrated the strong antioxidant properties of RCSO. Parandin et al. (2023) showed that RCSO, administered at doses of 500 and 750 mg/kg, exhibited significant antioxidant effects by increasing the activity of GPx, CAT, and SOD, thus protecting against cyclophosphamide-induced testicular toxicity in mice (Parandin et al., 2023). Furthermore, Kayahan et al. (2023) found that RCSO possessed high antioxidant properties and a substantial content of polyphenolic compounds, as evidenced by its ability to scavenge various free radicals, including NO, hydrogen peroxide, and DPPH. These findings highlight RCSO’s potential as an effective radical scavenger (Kayahan et al., 2023). In the present study, it was observed that RCSO increased the activity of GPx, CAT, and SOD in a dose-dependent manner while reducing the level of NO free radicals. This further supports the notion that RCSO possesses antioxidant properties and can effectively modulate the oxidative status associated with PCOS.
During normal ovarian parenchymal apoptosis, specifically in granulosa and theca cells, an increase in the Bax/Bcl-2 ratio leads to the upregulation of p53 expression, subsequently activating caspases, including Cas-3 (Refaie et al., 2022). In PCOS, this pathway is disrupted in the inhibited cells, resulting in the formation of CF. Inhibition of apoptosis in granulosa and theca cells halts the differentiation of primary and secondary follicles into antral follicles, leading to the presence of numerous CF on the ovarian cortex surface (Hashem et al., 2020). Additionally, pathways related to cell cycle regulation, such as NF-κB/p53/Cas-3 and WNT/β-catenin, are disturbed in these CF, preventing them from reaching the Graafian stage and undergoing ovulation. Furthermore, studies indicate that PCOS affects other signaling pathways, such as the AKT/IRS-1 pathways (Gao et al., 2021). Plant polyphenols have shown the ability to enhance the endogenous apoptotic cascade associated with the mitochondrial apoptotic pathway in theca cells. These compounds, including formononetin, genistein, daidzein, luteolin, and quercetin, stimulate apoptosis by activating the WNT/β-catenin and NF-κB/p53/Cas-3 pathways in follicular cells (Cui et al., 2022). By promoting mitochondrial apoptosis, these compounds inhibit the formation of follicular cysts. Additionally, they strengthen the AKT/IRS-1 pathway, leading to increased differentiation follicles (Althari et al., 2020). RCSO demonstrated the ability to enhance the endogenous apoptotic pathway (Bax/Bcl-2/p53). It increased apoptosis in follicular parenchymal cells, as evidenced by the presence of p53 positive cells. The Ki-67 index serves as an indicator of follicle differentiation in evaluating ovarian follicles. An increase in this index (Ki-67 cells) indicates a decrease in follicle differentiation, while its reduction signifies the progression of folliculogenesis and follicle differentiation. Studies have shown that PCOS increases the Ki-67 index, ultimately leading to an elevated number of Ki-67 positive follicular cells. In contrast, in the present study, RCSO reduced the Ki-67 cells, promoting the acceleration of follicle differentiation.
Conclusion
Due to its abundant polyphenolic compounds, RCSO has varied effects and acts as an aphrodisiac while also regulating the HPG axis. It possesses antioxidant properties and influences pathways linked to mitochondrial apoptosis, thereby aiding in the prevention of follicular cyst formation. Consequently, RCSO not only maintains the normal structure of ovarian tissue but also sustains its physiological functions. Although the precise mechanism of RCSO’s impact on PCOS treatment is not confined to a specific pathway, it displays potential in alleviating the clinical symptoms associated with PCOS. Therefore, RCSO holds promise as a treatment option for individuals with PCOS.
Abbreviations
FRAP: Ferric-reducing antioxidant power; SDS: Sodium dodecyl sulfate polyacrylamide; FBS: Fetal bovine serum; TBARS: Thiobarbituric acid reactive substances; MDA: Malondialdehyde; NCBI: National center for biotechnology information; CAT: Catalase; NF-κB: Nuclear factor- kappa B; LC-ESI-MS: Liquid chromatography-electrospray ionization-mass spectrometry.
Footnotes
Acknowledgments
We would like to acknowledge the following articles for their valuable contributions to the development of our research: “Bee Pollen Stimulated BMP2 and RANK Signaling Pathway in Osteoprogenitors and Improved Bone Mineralization in Rat Model of Femur Fracture” by Xiaowan Zeng, Yajing Lin, and Chaonan Wang, 2023 (sagepub.com) and “Bee Pollen and Doxorubicin by Synergistic Effects Inhibit the Proliferation of Breast Tumors in 4T1 Tumor-bearing BALB/c Mice: A Biochemical, Immunohistochemical, and Molecular Approach” by Jinwen Li, 2023 (
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This research adhered to ethical guidelines for the use of animal subjects. Animal welfare considerations were taken into account, minimizing pain, distress, and suffering. Ethical approval was obtained from the Tangdu Hospital Air Force Medical University for animal research.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
