Abstract
Aim
This research endeavored to explore the impact and underlying mechanisms of resveratrol on the phenomenon of “metabolic memory” in cultured human retinal vascular endothelial cells (HRVECs) under high-glucose (HG) conditions.
Materials and Methods
According to the glucose level and treatment, cultured HRVECs were divided into seven groups: normal glucose (NG), HG, high glucose followed by NG (HN), mannitol (Man), resveratrol, thioredoxin-interacting protein (TXNIP)-small interfering ribonucleic acid (siRNA), and N-acetylcysteine (NAC). The expression levels of TXNIP, nucleotide oligomerization domain (NOD)-like receptor family pyrin domain-containing 3 (NLRP3) inflammasome, intercellular adhesion molecule 1 (ICAM-1), caspase-1, interleukin-1β (IL-1β), B-cell lymphoma 2 (Bcl-2), caspase-3, and Bcl-2-associated X (BAX), as well as reactive oxygen species (ROS) production, were measured. Cell apoptosis was assessed through a terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) assay.
Results
In HRVECs from the HG group, expression levels of TXNIP, NLRP3, caspase-1, ICAM-1, and IL-1β were upregulated. However, in the HN group, the above upregulations were not reversed. After the administration of resveratrol, the expression levels of TXNIP, NLRP3, and other inflammatory cytokines were significantly reduced. Resveratrol mitigated the elevated ROS production induced by HG conditions. In the NAC group, the expression of TXNIP and inflammatory cytokines was downregulated. TXNIP-siRNA treatment showed similar effects. Resveratrol inhibited apoptosis as well as reversed the downregulation of BCL-2 and the upregulation of caspase-3 and BAX induced by HG conditions.
Conclusion
Resveratrol mitigated the HG-induced phenomenon of “metabolic memory” by inhibiting inflammation and apoptosis via modulation of the ROS/TXNIP/NLRP3 signaling pathway in cultured HRVECs. Therefore, resveratrol may have therapeutic potential to treat diabetic retinopathy and related metabolic memory complications.
Keywords
Introduction
Diabetes mellitus (DM) is a systemic metabolic disorder characterized by a myriad of complications, such as diabetic retinopathy (DR), which involves retinal microvasculature and may lead to visual impairment if not promptly treated (Song et al., 2018). DM will affect 552 million people worldwide by 2030, and DR will affect at least 300 million people at that time (Song et al., 2018; Tomić et al., 2013). It has been documented that retinal microvascular complications can progress even after hyperglycemia is reversed; this phenomenon is known as “metabolic memory” (Zhao et al., 2016; Mishra & Kowluru, 2014, 2016). This “metabolic memory” has been demonstrated in animal models (Zhong & Kowluru, 2013). Clinically, diabetic patients suffer from uncontrolled DM and its complications.
Oxidative stress, persistent low-grade inflammation, and apoptotic processes are pivotal factors in the pathogenesis of DR (Singh et al., 2023; Rao & Dlouhy, 2012; Al-Shabrawey et al., 2015). In response to hyperglycemia, thioredoxin-interacting protein (TXNIP) emerges as an early response gene (Ao et al., 2021). It serves as an endogenous inhibitor of thioredoxin (TRX). Under oxidative stress, TXNIP dissociates from TRX and then associates with the nucleotide oligomerization domain (NOD)-like receptor family pyrin domain-containing 3 (NLRP3), finally activating the inflammasome to mediate inflammatory signaling. The reactive oxygen species (ROS)/TXNIP pathway was demonstrated in inflammation (Li et al., 2022; Luo et al., 2022; Yang et al., 2023). Moreover, TXNIP can directly inhibit TRX function and activate downstream apoptosis signaling (Luo et al., 2022). Furthermore, early DR can be blocked by inhibiting the expression of TXNIP (Yang et al., 2023; Tang et al., 2021).
Resveratrol, a natural polyphenolic mixture found in various plants such as grapes, peanuts, and blueberries (Hou et al., 2019), is a strong scavenger of ROS (Yang et al., 2018). It has been revealed that resveratrol provides a protective effect in the treatment of DM complications, including DR (Zeng et al., 2017; Huang et al., 2020; Popescu et al., 2018; Chen et al., 2019; Dong et al., 2019; Zeng et al., 2022; Giordo et al., 2021; Ghadiri Soufi et al., 2015).
Nevertheless, the impact and mechanism of resveratrol on diabetic “metabolic memory” were not clearly known. Therefore, we established an in vitro model to investigate the effects and underlying signaling pathways of resveratrol utilizing human retinal vascular endothelial cells (HRVECs) under high-glucose (HG) conditions.
Materials and Methods
Cell Culture and Treatment
Human endothelial cell medium (Cell Biologics, Chicago, IL, USA) was used to culture HRVECs (Cell Biologics) and refreshed every 24 h. The culture conditions were 5% carbon dioxide (CO2) at 37°C, and passages 3 or 4 were used for the experiments. According to the treatments, cells were grouped as follows: normal glucose (NG), 5 mM
Reverse Transcription-quantitative Polymerase Chain Reaction (RT-qPCR)
RT-qPCR was employed to assess the messenger RNA (mRNA) expression levels of TXNIP, NLRP3, intercellular adhesion molecule 1 (ICAM-1), caspase-1, interleukin-1β (IL-1β), caspase-3, Bcl-2-associated X (BAX), and B-cell lymphoma 2 (Bcl-2). Briefly, in accordance with the manufacturer’s guidelines and following the method mentioned in our prior investigation (Jiang et al., 2019), cellular total RNA was extracted. After RNA was transcribed into complementary deoxyribonucleic acid (cDNA), qPCR was conducted for these cDNA. The primer sequences are shown in Table 1. The values of each group were compared using the relative level. The experiments were repeated three times.
Primer Sequences for RT-qPCR.

Western Blotting
Briefly, according to the manufacturer’s protocols and the method mentioned in our prior study (Jiang et al., 2019), radioimmunoprecipitation assay buffer and bicinchoninic acid protein assay were used to extract and quantify the entirety of cellular protein derived from HRVECs. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (Thermo Fisher Scientific, Waltham, MA, USA) was conducted using 50 g of protein samples from each group. Briefly, the protein was transferred onto polyvinylidene fluoride membranes. Then, the membranes were blocked and subjected to overnight incubation at 4°C with primary antibodies (purchased from Cell Signaling Technology, Beverly, MA, USA) to the following antigens: TXNIP (1:2,000; Abcam, Cambridge, MA, USA), caspase-3, NLRP3, ICAM-1, BAX, IL-1β, caspase-1, Bcl-2 (all were 1:1,000), and actin (1:2,000). On the second day, the membranes were washed and incubated with secondary antibodies for 1 h. Finally, the immunoreactive bands were imaged and analyzed for protein expression. The experiments were repeated three times.
Terminal Deoxynucleotidyl Transferase-mediated dUTP Nick-end Labeling (TUNEL) Assay
A TUNEL assay kit (Beyotime Biotechnology, Shanghai, China) was used. The staining procedures were conducted in accordance with the instructions provided by the manufacturer.
HRVECs in each experimental group were cultured on coverslips, subjected to triple 10-min washes in phosphate-buffered saline (PBS), and permeabilized with 0.5% Triton X-100 in PBS for 5 min, followed by two additional PBS washes. Then, cells were incubated in a TUNEL-detection solution for 1 h at 37°C in darkness and then underwent three subsequent washes in PBS. Ultimately, the specimens were treated with 4′,6-diamidino-2-phenylindole for a duration of 20 min at room temperature. Cells positive for TUNEL were labeled with green fluorescence and examined using a confocal microscope (FV1200, Olympus, Tokyo, Japan).
Measurement of ROS
Intracellular ROS production was quantified through the utilization of 2’,7’-dichlorodihydrofluorescein diacetate (DCFH2DA) (Beyotime Biotechnology). Briefly, the methods were implemented in conformity with the guidelines provided by the manufacturer. Cultivated cellular specimens were incubated with DCFH2DA for a duration of 30 min at 37°C, followed by triple washes with PBS. Subsequently, the fluorescence intensity was quantified using a fluorescence microplate reader (Thermo Fisher Scientific Inc., Grand Island, NY, USA) at excitation and emission wavelengths of 488 nm and 525 nm, respectively.
Statistical Analysis
Data processing and analysis were conducted utilizing SPSS 17.0 (IBM Corp., Armonk, NY, USA). The data were expressed as the mean ± SD. Student’s t-test was employed for intergroup value comparisons, while one-way analysis of variance followed by Tukey’s multiple comparison tests was utilized for comparing data across multiple groups. Statistical significance was established at a p-value less than 0.05.
Results
Resveratrol Decreased ROS Production Under HG Conditions
Under conditions of HG, there was a notable elevation in ROS production. Nevertheless, in the HN group, the transition from HG conditions to NG conditions over a span of 4 days failed to reduce ROS production. The introduction of resveratrol to the HN group effectively attenuated the elevated ROS production, as illustrated in Figure 1.
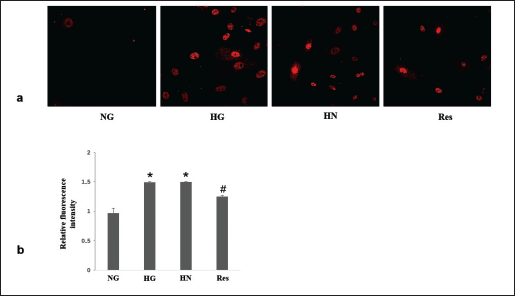
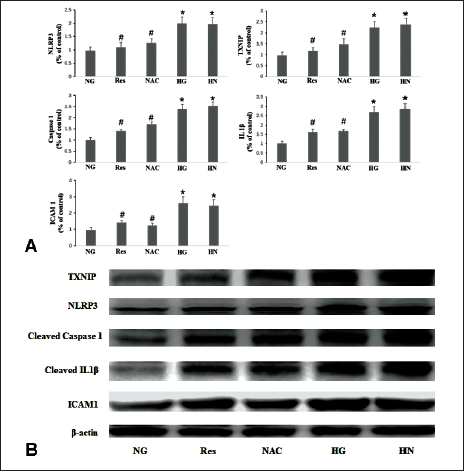
Resveratrol Reduces Inflammatory Factors Under Hyperglycemic Conditions
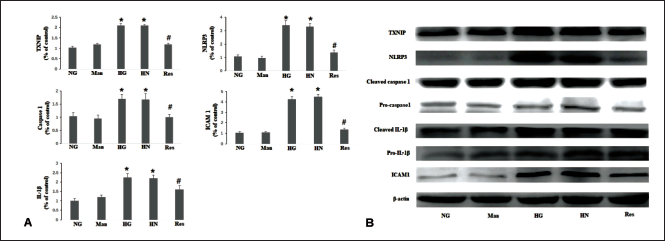
In the current study, HG conditions lead to the upregulation of TXNIP, NLRP3, caspase-1, IL-1β, and ICAM-1. In the HN group, the transition from HG conditions to NG conditions over a span of 4 days failed to reduce the expression levels of the aforementioned mRNAs and proteins. Conversely, supplementation with resveratrol in the HN group resulted in a downregulation of the expression levels of TXNIP, NLRP3, and the aforementioned inflammatory cytokines (Figure 2).
Resveratrol Attenuated HG-induced Cell Apoptosis
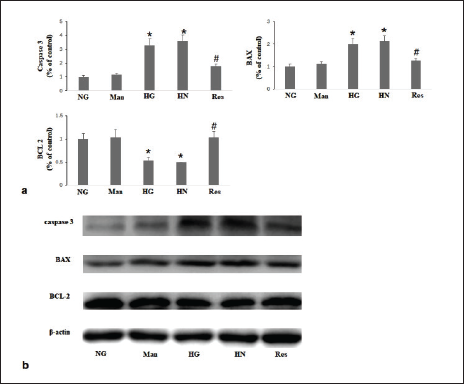
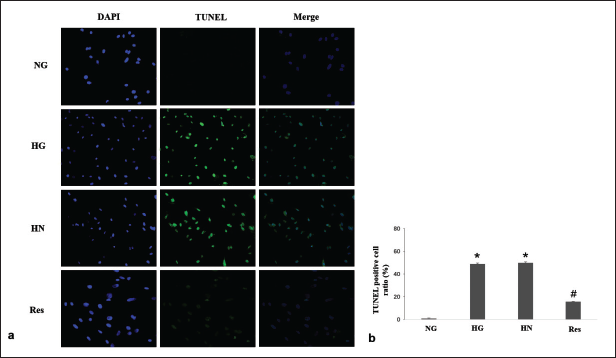
In the present study, HG conditions significantly downregulated Bcl-2 expression and upregulated BAX and caspase-3 expression. In the resveratrol group, the above changes were reversed (Figure 3). The number of TUNEL-positive cells was significantly increased under HG conditions and did not improve after continued culture in NG conditions in the HN group. In the resveratrol group, a reduction in the quantity of TUNEL-positive cells was observed (Figure 4).
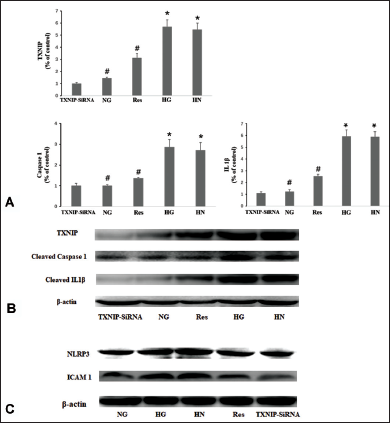
Inhibition of ROS and TXNIP Blocked Inflammasome Activation
In the NAC group, the expression levels of TXNIP, caspase-1, IL-1β, and ICAM-1 were markedly attenuated by NAC, a ROS inhibitor (Figure 5). In the TXNIP-siRNA group, the upregulation of the above-mentioned inflammatory cytokines was also inhibited by the silencing of TXNIP (Figure 6).
Discussion
In DM patients who have recent glycemic control, the development of DR does not stop immediately (Diabetes Control and Complications Trial Research Group et al., 1993; Nathan & DCCT/EDIC Research Group, 2014). This “metabolic memory” phenomenon indicates that the effects induced by previous HG levels can continue to lead to retinal microvascular complications. Therefore, inhibiting metabolic memory is crucial in the therapeutic approach to DR. Our investigation found that resveratrol effectively mitigated the “metabolic memory” in cultured HRVECs induced by HG conditions. This effect was associated with the suppression of inflammation via the ROS/TXNIP/NLRP3 signaling pathway, concomitant with a decrease in cellular apoptosis.
Oxidative stress, inflammation, and apoptosis are essential components of DR (Barber et al., 2011; Kang & Yang, 2020; Forrester et al., 2020). The NLRP3 inflammasome, a protein complex composed of NLRP3, procaspase-1, and apoptosis-associated speck-like adaptor protein containing a caspase recruitment domain (ASC), assumes a pivotal role in inflammatory responses. When the NLRP3 inflammasome is activated, NLRP3 is ligated with ASC, which combines with procaspase-1, causes cleavage of caspase-1, and then regulates the maturation of IL-1β (Raman & Matsubara, 2022). The overexpression of the NLRP3 inflammasome is involved in the pathogenesis of DM and DR (Yang et al., 2023; Kuo et al., 2022). In the current study, elevated expression levels of caspase-1, IL-1β, and ICAM-1 were observed concomitant with the activation of the NLRP3 inflammasome, which was induced by HG stimulation. The upregulation of these proinflammatory cytokines persisted even after the culture medium was changed to NG conditions for 4 days. This finding, known as “metabolic memory,” has been reported in previous investigations (Zhao et al., 2016; Kowluru et al., 2024; Gao et al., 2021).
TXNIP, recognized for its involvement in the inflammatory cascade associated with DR, assumes a pivotal role in the activation of NLRP3 inflammasomes (Yang et al., 2021). In the current study, persistent elevation of TXNIP expression was noted under HG conditions. The inhibition of TXNIP reduced the expression levels of NLRP3 and the measured inflammatory cytokines. Furthermore, ROS is a key factor in various signaling pathways associated with hyperglycemia (Singh et al., 2023). TXNIP is a redox-sensitive component that can be induced by ROS. ROS can cause TXNIP to bind to NLRP3, leading to inflammasome activation (Yang et al., 2021; Chen et al., 2017; ElSayed et al., 2023). ROS–TXNIP signaling is also involved in the development of DR (Yang et al., 2023). In the present study, NAC, a ROS scavenger, reduced the upregulation of TXNIP, NLRP3, and inflammatory cytokines. This result illustrated the involvement of the ROS/TXNIP signaling pathway in the activation of the NLRP3 inflammasome induced by HG.
The protective effects of resveratrol against DR, including antiapoptotic, anti-inflammatory, and antioxidative properties, have been reported in many studies (Zeng et al., 2017; Huang et al., 2020; Popescu et al., 2018; Chen et al., 2019; Dong et al., 2019; Zeng et al., 2022; Giordo et al., 2021; Ghadiri Soufi et al., 2015). Resveratrol has demonstrated the capacity to mitigate retinal cell apoptosis provoked by hyperglycemic conditions in vitro or induced by streptozotocin (STZ) in vivo (Zeng et al., 2017; Huang et al., 2020). Resveratrol can also reduce intracellular ROS production under HG conditions (Giordo et al., 2021). Moreover, the intravitreal administration of resveratrol has been documented to downregulate the upregulation of inflammatory factors induced by STZ (Ghadiri Soufi et al., 2015). In the present study, resveratrol inhibited ROS production and TXNIP expression, suppressed NLRP3 activation, and reduced inflammatory cytokine expression in HRVECs under HG-induced “metabolic memory” conditions. Furthermore, hyperglycemia can activate proapoptotic proteins (Sankaramoorthy & Roy, 2021; Kim et al., 2020). In the current study, elevated expression levels of Bax and caspase-3, downregulated expression of BCL-2, and increased TUNEL-positive cells were observed in the cultured HRVECs under HG conditions. When resveratrol was administered, the above phenomena were reversed. Resveratrol significantly attenuated HRVEC apoptosis.
Conclusion
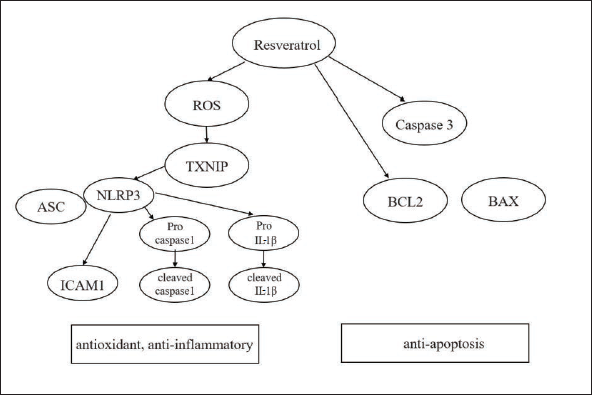
Resveratrol demonstrated its antioxidant, antiapoptosis, and anti-inflammatory effects in cultured HRVECs exposed to HG conditions (Figure 7). Our findings elucidate that resveratrol exerted a significant reduction in the expression of inflammatory genes through the inhibition of the ROS/TXNIP/NLRP3 pathway. More importantly, these effects were revealed in experimental conditions mimicking “metabolic memory” phenomena. Thus, resveratrol may have therapeutic benefits for the treatment of progressive retinal injuries of DR and other metabolic memory-related complications.
Abbreviations
HRVECs: Human retinal vascular endothelial cells; NG: Normal glucose; HG: High glucose; HN: High glucose followed by normal glucose; Man: Mannitol; TXNIP: Thioredoxin-interacting protein; NLRP3: NOD-like receptor family pyrin domain containing 3; ICAM-1: Intercellular adhesion molecule 1; BAX: Bcl-2-associated X; ROS: Reactive oxygen species; DR: Diabetic retinopathy; PBS: Phosphate-buffered saline.
Footnotes
Declaration of Conflicting Interests
The authors declare no conflict of interest.
Funding
This research was supported by the National Natural Science Foundation of China (Grant Nos. 81500734, 81870670, and 82000907). The funder had no role in study design, data collection and analysis, or preparation of the manuscript.
Statement of Informed Consent and Ethical Approval
The study is in cells and does not require ethical approval.
