Abstract
Background
Andrographis paniculata (Burm. f.) Nees is a well-recognized medicinal plant known for its benefits against a multitude of ailments. The intent of the present study was to evaluate the effect of a standardized extract of A. paniculata on innate immunity.
Materials and Methods
Phagocytic activity was measured in terms of phagocytic index using carbon clearance methodology in mice. In another experiment, NK cells (effector cells) were isolated from the mouse spleen after the dosing period, and NK cells-mediated lysis of target cells (YAC-1 cells) were determined using an MTT assay.
Results
The phagocytic index and NK cell activity were found to be significantly (p < 0.05) different in the treated group compared to the control group in the tested animal models at a dose of 41 mg/kg.
Conclusion
The results suggest the potential of A. paniculata to enhance innate immunity.
Introduction
Ayurveda (an Indian traditional system of medicine) has been practiced for several thousand years and is now gaining global acceptance (Chaudhary & Singh, 2011; Mukherjee et al., 2017). The ayurvedic plant Kalmegh, Andrographis paniculata (Burm. f.) Wall. ex Nees, and its extracts have been traditionally used to treat different diseases (Kumar et al., 2004). The aerial parts region of the A. paniculata plant contains flavonoids, diterpenoids, and lactones (Akbar, 2011; Hossain et al., 2014; Rao et al., 2004). The herb is reported majorly for its broad scale of pharmacological properties viz. antiviral (Cáceres et al., 1997; Chang et al., 1991; Churiyah et al., 2015), antioxidant (Kamdem et al., 2002; Sheeja et al., 2006), antimicrobial (Deepak et al., 2014; Zhang et al., 2020), anti-cancer (Kumar et al., 2004; Rajagopal et al., 2003), hepatoprotective (Maiti et al., 2010; Nagalekshmi et al., 2011) against upper respiratory tract infections (Gabrielian et al., 2002; Saxena et al., 2010), and immunostimulatory (Puri et al., 1993; Rajagopal et al., 2003) among others. The present study explores the effects of the standardized botanical extract of Andrographis paniculata (AP-Bio®/KalmCold®) (SEAP) on innate immunity.
Innate immunity is the first level of cellular defense against invaders (non-specific). It is regulated by dendritic cells, mast cells, erythrocytes, and lymphocytes (natural killer (NK) cells, phagocytes, neutrophils, eosinophils, basophils, and monocytes). Phagocytes (e.g., macrophages, monocytes, neutrophils, etc.) are known to perform a crucial function in the innate/non-specific immune response of the human body. Phagocytes bind to the cell surface receptors present on the pathogens, engulfing, lowering its pH, and disintegrating its contents (Rosales & Uribe-Querol, 2017). Phagocyte dysfunction or reduction in their numbers is associated with an increase in clinical illnesses (Engelich et al., 2001).
NK cells possess the ability to kill tumor cells and pathogens without prior stimulus by the release of perforins. Perforins create holes in the cell membranes, increase permeability, and thereby cause the cells to swell up and burst. The function of NK cells in fighting viral and bacterial illnesses is established through several studies (Bruunsgaard et al., 1997; Haller et al., 2000). A marked reduction in NK cells is correlated to a rise in infections and tumors (Fujisaki et al., 2009; Knorr et al., 2014). The major histocompatibility complex (MHC) proteins present in normal cells help NK cells differentiate between healthy cells and infected/tumor cells (Hamerman et al., 2005).
Reported studies have concluded the relationship between diet and immune function. The activity of phagocytes and NK cells is significantly altered with diet and the consumption of certain foods (Childs et al., 2019; Ravaglia et al., 2000). Since A. paniculata has traditional claims in the context of immunity, the current research is focused on scientific validation of SEAP for its phagocytic and NK cell activity through in vivo and ex vivo experimentation, respectively.
Materials and Methods
Test materials, chemicals, and reagents. The investigational substance used was SEAP known as AP-Bio®/KalmCold®, developed by M/s Natural Remedies Pvt. Ltd., Bengaluru, India. SEAP contains ˃33% diterpene lactones with ˃30.0% w/w of andrographolide as analyzed by HPLC. As per the USP, the 14-deoxy-11,12-didehydroandrographolide content is not >15% of the total diterpene lactones (Raj et al., 2023).
Carboxymethyl cellulose (CMC) (HiMedia Labs, India), Polyinosinic:polycytidylic acid (Poly (I:C)) (Sigma-Aldrich, USA), 1x phosphate-buffered saline (PBS) (Hyclone), complete RPMI-1640 medium (Gibco®, Thermo Fischer Scientific, Inc., USA), fetal bovine serum 10% (FBS), Penicillin (100 U/mL, Himedia), Streptomycin (100 µg/mL, HiMedia), (NH4Cl, KHCO3, Na2EDTA (HiMedia)), and (NaHCO3, C3H3NaO3 (Sigma)) were procured and stored at specified temperature.
Preparation of Test Substance
The 0.5% CMC (HiMedia Labs, India) was used as a vehicle to suspend the test substance. The test substance was mixed in the 0.5% CMC and administered orally at the rate of 10 mL/kg mouse body weight. An animal dose of 41 mg/kg of SEAP was selected by considering a human equivalent dose of 200 mg (Saxena et al., 2010).
Experimental Design and Procedure
All the experimental animals were inbred at the Central Animal Facility, Natural Remedies Pvt. Ltd., Bengaluru. The mice were maintained at 22 ± 3°C in a well-ventilated animal house between 30% and 70% relative humidity under a 12-h light/12-h dark cycle. The animals had free access to available standard food pellets and UV-treated water ad libitum. Animals were allowed to adapt to the laboratory environment for a minimum of a week prior to experiments. All the experimental procedures involving animals were approved by the Institutional Animal Ethics Committee (IAEC) of Natural Remedies Pvt. Ltd., Bengaluru, India (Approval No.: IAEC/NR-PCL-01/03/2020), and experiments were conducted in accordance with the guidelines of Committee for Control and Supervision of Experiments on Animals (CCSEA) and in compliance with the Animal Research: Reporting of in vivo Experiments (ARRIVE) guidelines.
In vivo Phagocytic Activity
The male Swiss albino mice aged about 6−8 weeks were used to measure phagocytic activity. The randomization was done based on the body weight of the mice. Animals with individual body weight within ±20% coefficient of variation of the group mean were considered. The mice were dosed by oral gavage with the help of graduated syringes. Twelve mice were divided into two groups. Group I was considered as the control and only 0.5% CMC (vehicle) was administered at 10 mL/kg mice body weight. Group II was treated with SEAP at 41 mg/kg mice body weight. These were administered daily for a period of 10 days. On day 11, all mice were injected with Pelikan Tusche A ink (0.1 mL) suspension through the tail vein (Halpern et al., 1953). After which, 25 µL of blood sample was collected from the orbital plexuses before and at 5 and 15 min after the carbon suspension injection. The blood samples were then mixed thoroughly in 2 mL of 0.1% sodium carbonate solution. The absorbance was read at 675 nm using a spectrophotometer (VersamaxTM, BN02314, Molecular Devices Corp., California) (Atal et al., 1986).
The rate of carbon clearance, expressed as the phagocytic index, was calculated using the following formula:
Phagocytic index = K = (log OD1 – log OD2)/(T2 − T1)
where OD1 and OD2 are the optical densities at 5 and 15 min, respectively.
Ex vivo NK Cell Activity
Twelve male BALB/c mice aged about 8−10 weeks were randomly divided into three groups of four each. For 10 days, group I was administered (p.o.) with 0.5% CMC at 10 mL/kg body weight and group II with SEAP at 41 mg/kg mice body weight. Group III was injected with a single dose of 150 µg Poly (I:C) intraperitoneally (i.p.) 18 h before euthanizing the mice. After 24 h of the last dose (i.e., 11th day), spleens were isolated from all the groups and processed as mentioned below. The activated spleens were aseptically removed from the BALB/c mice, and single cell suspension was prepared and allowed to go through a cell strainer for separation (BD biosciences, NC, USA). Erythrocytes in the suspension were removed by treatment with a lysis buffer (recipe −0.15 M NH4Cl + 1.0 M KHCO3 + 0.1 mM Na2EDTA) for 5 min. Cells were washed in RPMI-1640 medium and used as NK cell population (Hwang et al., 2012; Madaan et al., 2015; Moore et al., 2014).
YAC-1 Mus musculus lymphoma cell (target cells) suspension containing 1 × 104 cell per well was plated onto the flat bottom of a 96-well plate as target cells and incubated for 1 h. The cells were counted in a hemocytometer using trypan blue. YAC-1 cells were co-incubated with NK cells (Effector:Target ratio of 100:1) in RPMI-1640 containing 10% FBS and 1% penicillin-streptomycin. NK cells alone (Effector) and YAC-1 cells alone (Target) were also incubated as controls. After 4 h of incubation in a CO2 incubator at 37°C, the NK cells-mediated lysis of target cells was measured using MTT assay. The percentage of specific cytotoxicity was calculated as follows (Hwang et al., 2012; Madaan et al., 2015):
% specific cytotoxicity = [1 − (OD(effector + target) – OD(effector)/OD (target)] × 100
Statistical Analysis
In vivo data analysis was done by independent t-test, and the values were reported as mean ± standard deviation (SD), whereas the statistical analysis of the ex vivo study was done by using a one-way analysis of variance (Dunnet’s test) using the GraphPad Prism 5 software. The statistical significance was set at p < 0.05 for both studies.
Results
Phagocytic Activity
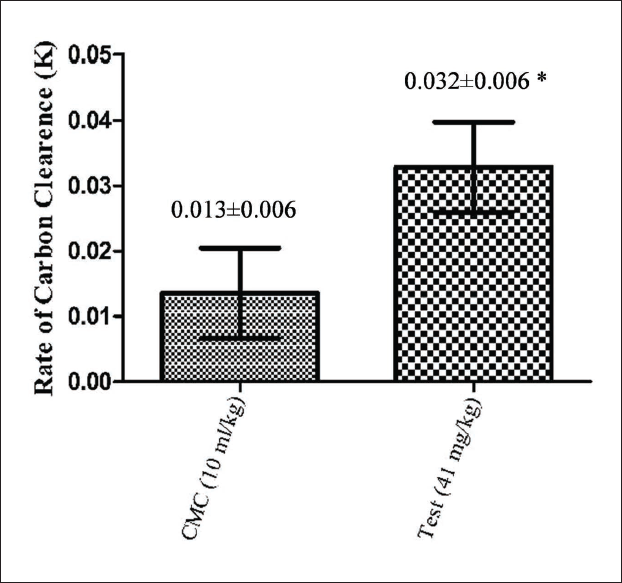
The SEAP at 41 mg/kg showed a significant increase in the phagocytic index when compared to the control (Figure 1). The group administered with SEAP showed a 2.46-fold increase compared to the control group.
NK Cell Activity
We evaluated the NK cell activity and the cytotoxic effect on target cells by co-culture with NK-sensitive YAC-I cells. The cytotoxicity was assessed using a microplate reader and calculated. The percentage of specific lysis i.e., the difference in the percentage of lysis of YAC-1 target cells by NK cells was represented as mean ± SD from four replicates per treatment group (Figure 2).
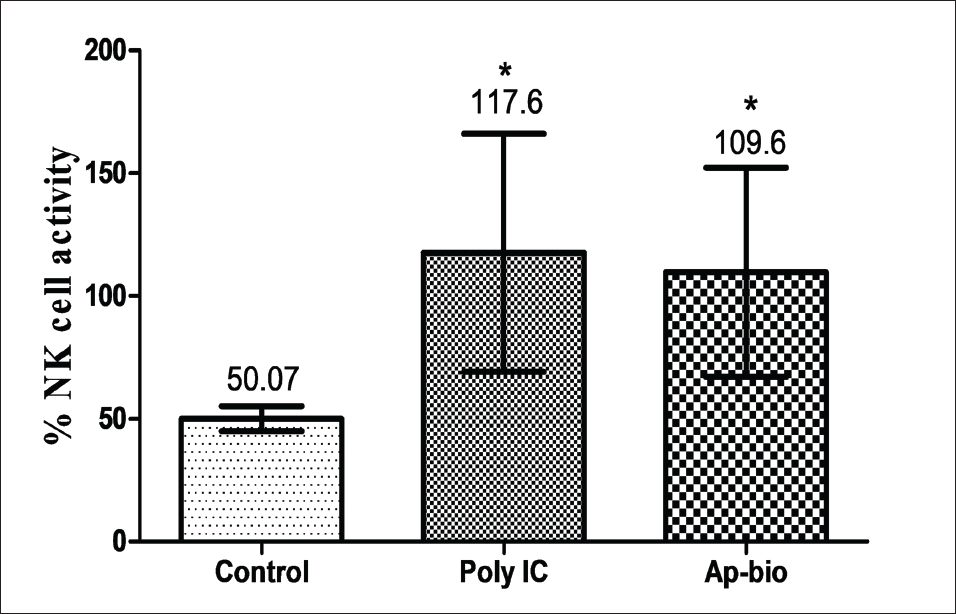
As represented in Figure 2, the NK cells pre-treated with SEAP showed a significant increase in NK cell activity when compared to the control against YAC-I cells at effectors with a target ratio of 100:1.
Discussion
The battle against viral infections is ongoing; the investigation of compounds that stimulate the body’s own defense mechanism with minimal side effects is encouraged (Grudzien & Rapak, 2018; Kumar et al., 2004). Andrographis paniculata has been extensively studied for its immunomodulatory effects (Wang et al., 2010). From the current study, it was evident that the administration of the SEAP for 10 days significantly increases both phagocytic and NK cell activities in the selected animal models. This supports the extract’s potential to favorably modulate the body’s innate immune system. The mice dose of 41 mg/kg was selected for both tests considering the human equivalent dose of 200 mg. The carbon clearance test was conducted to study the effects of the extract on the reticuloendothelial system (RES). The RES is a diffuse system that consists of phagocytic cells that are involved in the clearance of foreign particles from the bloodstream. Since a 2.46-fold increase was observed in the phagocytic index, it can be concluded that SEAP increases the activity of the RES, thereby strengthening the body’s innate immunity. The conclusion drawn from this experiment on A. paniculata substantiates the existing literature. The research work by Palanikani et al. (2020) supports the results of their phagocytic index experiment on Labeo rohita using a methanolic extract of A. paniculata.
NK cells are an integral agent of the immune system and act as the first level of immune-oriented defense cells. A significant increase was observed in the NK cell activity of BALB/c mice after the administration of SEAP for 10 days. The effect of SEAP is comparable to standard Poly (I:C) which is known to activate NK cells of mice through Toll-like receptor 3 (TLR3) (McCartney et al., 2009; Ribes et al., 2020). The experimental observations are in line with a similar study conducted with a crude extract of A. paniculata (Sheeja & Kuttan, 2010). Here, Sheeja et al. demonstrated the ability of andrographolide, a major constituent of A. paniculata to activate NK cells and support the immunomodulatory activity of SEAP.
Conclusion
The SEAP significantly increased the phagocytic and NK cell activity in the selected animal model when compared to the control. The current study provides the basis for planning a clinical study to establish the effects of SEAP on innate immunity.
Abbreviations
SEAP: Standardized extract of Andrographis paniculata; NK cells: Natural killer cells; MHC: Major histocompatibility complex; CMC: Carboxymethyl cellulose; Poly (I:C): Polyinosinic: polycytidylic acid; PBS: Phosphate-buffered saline; FBS: Fetal bovine serum; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; IAEC: Institutional Animal Ethics Committee; CCSEA: Committee for Control and Supervision of Experiments on Animals; ARRIVE: Animal research: reporting of in vivo experiments; SD: Standard deviation; RES: Reticuloendothelial system; TLR3: Toll-like receptor 3.
Footnotes
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Statement of Ethical Approval and Informed Consent
This article does not contain any studies with human participants performed by any of the authors.
