Abstract
Background:
Petasites japonicus (PJ), also known as Butterbur, has a rich history as a traditional healing remedy across numerous countries.
Objectives:
This study was designed to evaluate the potential anti-cancer properties of the methanol extract derived from PJ (PJE).
Materials and Methods:
Cell viability was measured with 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide. Cell cycle analysis, caspase activity assays, western blotting, and reactive oxygen species (ROS) assays were also used to investigate the anticancer effects of PJE on cancer cells.
Results:
It was shown that PJE inhibited the cell viability of the colon carcinoma cell line Caco-2 (half-maximal inhibitory concentration [IC50]: 268.4 µg/mL), of the hepatocellular carcinoma cell line Hep3B (IC50: 420.2 µg/mL), and of the bladder carcinoma cell line 5637 (IC50: 99.43 µg/mL). Analysis of DNA content indicated an increase in the sub-G1 population of 5637 cells as a result of PJE treatment. Furthermore, PJE caused a reduction in mitochondrial membrane potential and the ratio of Bcl-2 to Bax. Moreover, PJE enhanced the levels of various components involved in the proapoptotic cascade, such as caspase-3, caspase-9, and poly-adenosine diphosphate-ribose polymerase. Moreover, it was observed that PJE modulated mitogen-activated protein kinases (MAPKs) activation and induced an elevation in intracellular production of ROS.
Conclusion:
These combined results strongly suggest that PJE possesses significant proapoptotic properties as an herbal medicine, acting through ROS-dependent MAPK signaling pathways in bladder cancer cells.
Introduction
Bladder cancer ranks among the most prevalent malignant cancers affecting women and the fourth most common malignant cancer in men (Lenis et al., 2020; Richters et al., 2020). Bladder cancer comes in a variety of forms, with non-invasive cancers recoverable and quite aggressive cancers leading to death (Golijanin et al., 2006; Lenis et al., 2020; Richters et al., 2020). In particular, smoking has been known to be the most common cause of bladder cancer (Cumberbatch et al., 2016). Recently, many studies have been conducted on the causes and treatments of bladder cancer. Although cisplatin-based chemotherapy is still selected as the most basic perioperative treatment, various treatment methods, and novel therapeutic targets are being developed (Patel et al., 2020; Plimack et al., 2014).
Plant compounds are generally known to have the function of preventing cancer with chemical preventive drugs and also have the effect of apoptosis, which inhibits cell growth in cancer cells (Samandari-Bahraseman et al., 2023). This is because plant compounds have both pro-oxidant and antioxidant effects and thus show a very important therapeutic role in treating cancer (Bahraseman et al., 2022). The genus Petasites Mill (Asteraceae) is widely found in Europe, Asia, and North America and has long been used as a traditional remedy (Tzoneva et al., 2021). Among the numerous species in the genus, Petasites japonicus (PJ), also known as Butterbur, has attracted the attention of many researchers owing to its long use as a traditional medicine in many countries (Aydin et al., 2013). PJ has many properties, such as antioxidant, anti-inflammatory, analgesic, diuretic, antispasmodic, and antitumor, especially its roots, which are used as anodynia and antidotes for food in Korea (Aydin et al., 2013; Kim et al., 2015; Lee et al., 2011; Lee et al., 2013; Tys et al., 2015). Nonetheless, a systematic study on the anticancer effect of PJ has not been carried out. In this study, our hypothesis was that the methanol extract of Petasites japonicus (PJE) has the potential to modulate molecular signaling pathways and inhibit the proliferation of cancer cells. To examine this conjecture, we examined the impacts of PJE on apoptosis and the growth of 5637 human bladder cancer cells.
Materials And Methods
Materials
The PJ sample was obtained from the Korea Plant Extract Bank (Cheongju, Republic of Korea). Chlorogenic acid (CGA), 3,4-dicaffeoylquinic acid (3,4-DCQA), 3,5-dicaffeoylquinic acid (3,5-DCQA), SB203580, SP600125, or PD98059, tetramethylrhodamine methyl ester (TMRM), 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) assay, N-acetyl-
HPLC Analysis
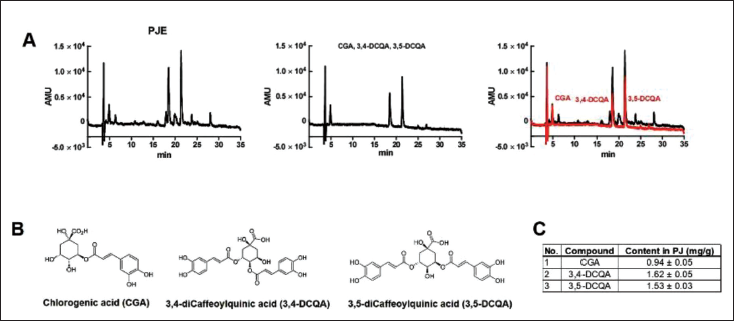
We sought to identify the major components of PJE. According to previous studies, PJ has CGA, 3,4-DCQA, and 3,5-DCQA as its major components (Choi et al., 2017; Hiemori-Kondo & Nii, 2020). Similarly, the HPLC results of the present study revealed that PJE contains CGA, 3,4-DCQA, and 3,5-DCQA, with concentrations of 0.94 ± 0.05 mg/g, 1.62 ± 0.05 mg/g, and 1.53 ± 0.03 mg/g, respectively (Figure 1). To create stock solutions, the PJE and each individual compound were dissolved in 50% methanol. Standard calibration solutions spanning from 6.25 to 100 µg/mL were also prepared. All solutions underwent filtration using a 0.45-µm filter (Sartorius Stedim, Goettingen, Germany). For the analysis, a JASCO HPLC system was utilized, which comprised an AS-950-10 auto-sampler, a column oven, a binary pump, and an MD-2010 Plus multiwavelength detector. The direct injection of 10 µL of standard or PJE sample solutions was made into the Symmetry300TM C18 column (4.6 × 250 mm × 5 µm). The mobile phase utilized in the experiment comprised two components: (A) 0.1% phosphoric acid in distilled water and (B) ACN. The following gradient conditions were applied: 85%–85% A for 0–10 min, 85%–75% A for 10–20 min, 75%–65% A for 20–30 min, and 65%–0% A for 30–35 min. The total runtime for the assay determination was 35 min. For the experiment, a flow rate of 1 mL/min was chosen, and the column temperature was maintained at 30℃. The UV detection wavelength was specifically configured at 350 nm.
Cell Culture
Colon cancer Caco-2 cells, hepatocellular carcinoma Hep3B cells, and bladder cancer 5637 cells were acquired from the Korean Cell Line Bank located in Seoul, Korea. These cells were cultured in Roswell Park Memorial Institute-1640 medium supplemented with 10% heat-inactivated fetal bovine serum and 1% penicillin/streptomycin, both supplied by Fisher Scientific (Hampton, New Hampshire, USA). Human bladder epithelial cells (HBlEpC) were purchased from Cell Applications (San Diego, CA, USA) and cultured in bladder epithelial cell growth medium (Cell Applications). The extract was dissolved in DMSO and the final concentration of DMSO in the medium was always <0.1%.
MTT Assay
Cell viability was determined using an MTT assay. The absorbance at 570 nm was measured.
Cell Cycle Assay
After centrifugation of the 5637 cells for 5 min, the resulting cell pellets were resuspended in a propidium iodide staining solution supplemented with 2 µL of RNase. Following that, the cells underwent another centrifugation at 20,000×g for 10 s. Analysis of the samples was conducted using a fluorescence-activated cell sorter (FACScan; BD, Franklin Lakes, NJ, USA) with a wavelength of 488 nm.
Mitochondrial Membrane Potential Assay
The 5637 cells were exposed to a concentration of 50 nM TMRM for a duration of 30 min. Subsequently, the fluorescence intensities of the samples were assessed using a FACSCanto II (BD, Franklin Lakes, NJ, USA) with excitation and emission wavelengths set at 510 and 580 nm, respectively.
Western Blot Assay
To extract lysates from 5637 cells, we utilized RIPA buffer supplemented with a mixture of protease and phosphatase inhibitors. The total protein content was quantified using the Bradford method (Bio-Rad Laboratories, Hercules, CA, USA). Equal quantities of protein (20 µg/lane) from the samples were separated on sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gels with either 8% or 10% concentration and then probed with the designated antibodies. Specific protein bands detected by western blots were quantified with the Image J program and normalized against β-actin or GAPDH.
Caspase Assay
Cellular activity assay kits plus for caspase-3 and caspase-9 were utilized. Following the resuspension of 5637 cells, the resulting supernatants were subjected to incubation with a caspase substrate (400 µM Ac DEVD pNA; 50 µL) at a temperature of 37°C.
Reactive Oxygen Species (ROS) Production Assay
Fluorescence emitted by DCF-DA was detected using FACS (BD, Franklin Lakes, NJ, USA) at excitation/emission wavelengths of 488/525 nm, respectively.
Statistics
For conducting multiple comparisons, a one-way analysis of variance (ANOVA) with Dunnett’s post hoc test was employed. The analysis was performed using Origin 8.0 software (OriginLab, Northampton, MA, USA) and Prism 6.0 software (GraphPad Software Inc., La Jolla, CA, USA). The results are expressed as mean ±SEM, and p-values <0.05 were regarded as statistically significant.
Results
PJE Induces Apoptotic Cell Death in 5637 Cells
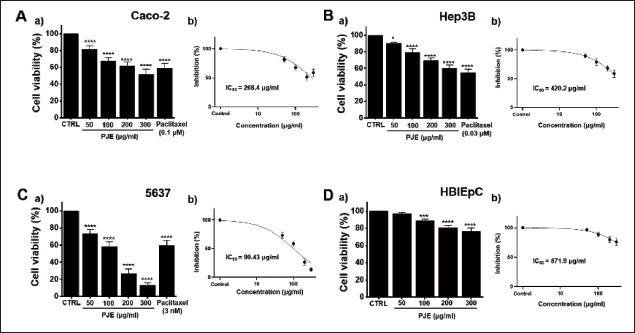
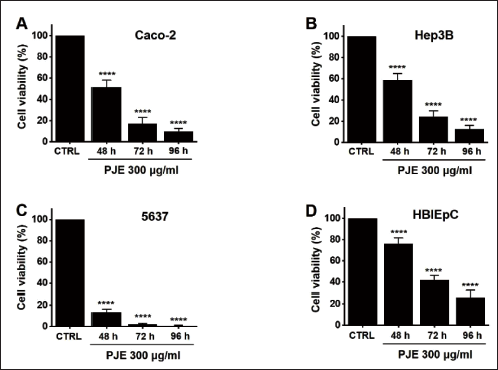
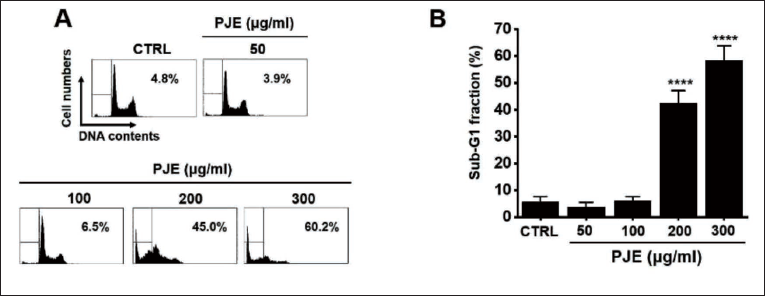
Initially, the cytotoxic effect of PJE was examined in three different cancer cell lines (Caco-2, Hep3B, and 5637). The results showed that PJE treatment produced dose-dependent cytotoxicity in all three cell lines (Figure 2A–C). To confirm cell viability, we used a positive control, paclitaxel, which is an anticancer drug. The half-maximal inhibitory concentration (IC50) values for PJE in the Caco-2, Hep3B, and 5637 cells were 268.4 (Figure 2Ab, p < 0.0001), 420.2 (Figure 2Bb, p < 0.0001), and 99.43 µg/mL (Figure 2Cb, p < 0.0001), respectively. As the cytotoxic effect of PJE was most potent in the bladder cancer 5637 cells and PJE exhibited much lower cytotoxicity toward a normal HBlEpC (Figure 2Db, IC50 = 871.9 µg/mL (p < 0.0001)), at the same concentrations as were used on 5637 cells (Figure 2D), the 5637 cell line was selected for subsequent experiments. In addition, the efficacy of PJE was also confirmed over a long period of time to show the best effects on 5637 cells (Figure 3). In order to investigate whether the cytotoxic effects of PJE were associated with apoptotic cell death, an analysis of the cell cycle was performed on PJE-treated 5637 cells. In a dose-dependent manner, the sub-G1 phase ratios were augmented at concentrations of 50, 100, 200, and 300 µg/mL of PJE (Figure 4). These findings indicate that apoptosis contributes to the cell death induced by PJE.
PJE Induces Apoptosis Through the Mitochondrial Pathway
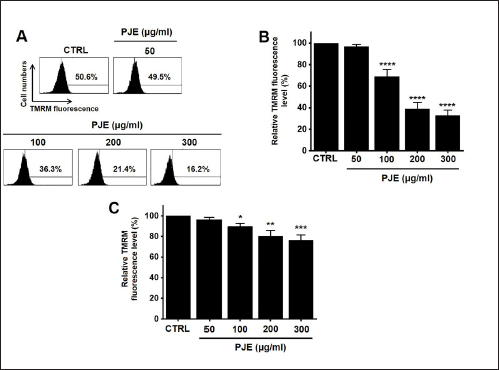
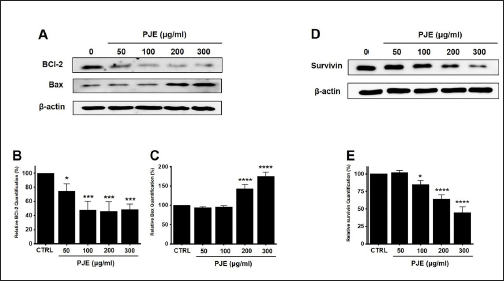
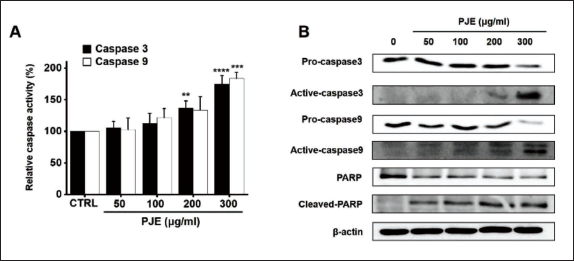
To delve into the mechanism of PJE-induced apoptosis, an analysis was conducted to assess the expression levels of specific proteins associated with the apoptosis pathway. Furthermore, PJE was found to decrease the mitochondrial membrane potential, as indicated by TMRM staining (Figure 5A). Relative fluorescence levels of TMRM were reduced by 96.9 ± 1.7%, 69.1 ± 6.4% (p < 0.0001), 38.9 ± 6.0% (p < 0.0001), and 32.9 ± 4.9% (p < 0.0001) at concentrations of 50, 100, 200, and 300 µg/mL of PJE, respectively (Figure 5B). However, in HBlEpC, only a slight decrease in TMRM was confirmed (Figure 5C). Furthermore, PJE treatment resulted in a decrease in Bcl-2 levels and an increase in Bax levels (Figure 6A). Treatment with different concentrations of PJE resulted in a significant decrease in relative Bcl-2 quantification levels (Figure 6B). Conversely, relative Bax quantification levels exhibited a notable increase upon treatment with PJE, respectively (Figure 6C). In addition, survivin was reduced by PJE (Figure 6D and E). In a dose-dependent manner, PJE was observed to increase the activities of caspase-3 and caspase-9 (Figure 7A). Western blot analysis revealed that PJE reduced the levels of preforms of caspase-3, caspase-9, and PARP while increasing the levels of active forms of caspase-3, caspase-9, and cleaved PARP (Figure 7B). These findings suggest that PJE-induced cell death involves the mitochondrial pathway.
PJE Induces Apoptosis Through the MAPK Pathway
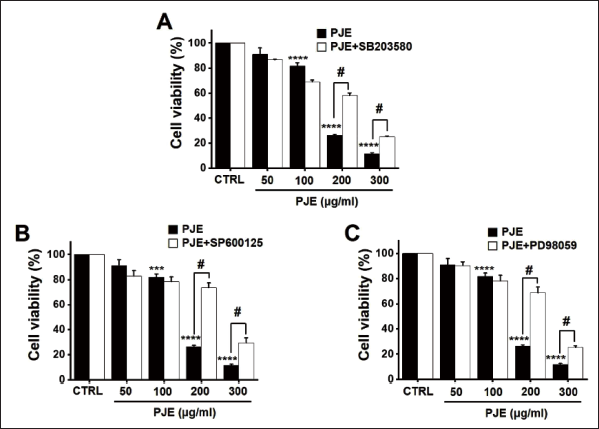
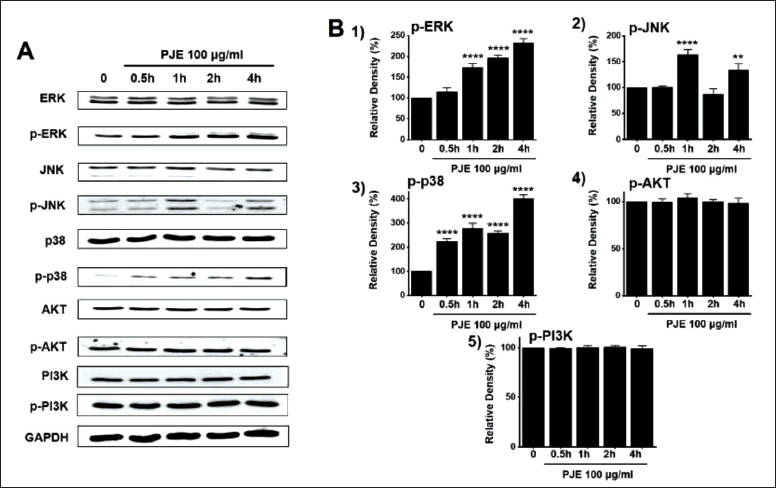
To assess the mitogen-activated protein kinase (MAPK) pathway mechanism underlying PJE-induced apoptosis, cell viabilities were measured after the co-administration of PJE and p38 MAPK inhibitor (SB203580), c-jun NH2-terminal kinase (JNK) II inhibitor (SP600125), or p42/44 MAPK inhibitor (PD98059). The co-administration of PJE and 10 µM SB203580 increased cell viabilities when PJE was employed (Figure 8A). Similarly, co-administering PJE and 10 µM SP600125 increased cell viability when PJE was used (Figure 8B). Cell viabilities were also found to increase when PJE were co-administered with 10 µM PD98059, respectively (Figure 8C). Furthermore, in order to assess the role of MAPK signaling pathways, the phosphorylation of MAPK proteins was examined through western blot analysis following treatment with PJE. It was observed that the phosphorylation of MAPKs increased over a time period of 0.5–4 h upon treatment with PJE (200 µg/mL) (Figure 9A). At 0.5, 1, 2, and 4, the relative p-ERK, p-JNK, and p-p38 density levels were increased by PJE (Figure 9B1, 9B2 and 9B3). Finally, relative p-AKT and p-PI3K density levels remained relatively unchanged after treatment with PJE (Figures 9B4 and 9B5). These results suggest that the MAPK signaling pathway is implicated in the cell death induced by PJE.
PJE Induces Apoptosis Through ROS Generation
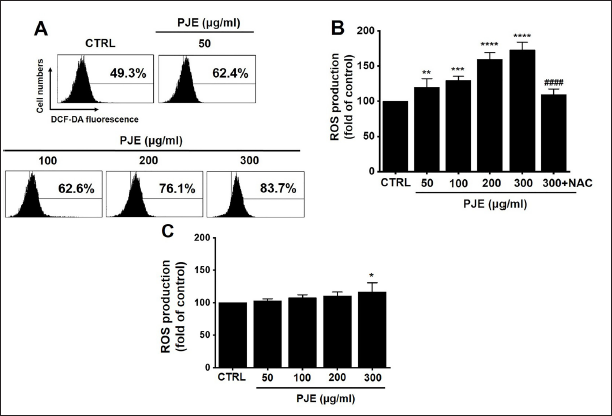
Whether treatment with PJE increased ROS production was determined herein. Based on our results, PJE dose-dependently increased ROS production (Figure 10A). ROS production levels were increased by 119.9 ± 12.3% (p < 0.01), 129.4 ± 6.0% (p < 0.001), 159.2 ± 9.9% (p < 0.0001), and 172.9 ± 11.1% (p < 0.0001) under treatment with PJE (50, 100, 200, and 300 µg/mL, respectively). However, ROS production level was reversed by 5 mM NAC with 300 µg/mL PJE (109.3 ± 8.2%) (Figure 10B). In addition, in HBlEpC, ROS production was observed only slightly at 300 µg/mL PJE (Figure 10C). The results imply that the generation of ROS contributes to the cell death induced by PJE.
Discussion
Cancer is one of the most global diseases, with a significantly high number of affected individuals (Rayan et al., 2017). Natural herbal medicines and their constituents hold significant importance and value in the advancement of anticancer therapeutics. The current investigation demonstrated that the cytotoxic effect of PJE was examined in three different cancer cell lines (Caco-2, Hep3B, and 5637), and PJE treatment produced dose-dependent cytotoxicity in all three cell lines. However, the cytotoxic effect of PJE was most potent in the 5637 bladder cancer cells, attributing it to the induction of apoptotic cell death (Figures 2–4). Moreover, the findings emphasize that PJE-induced apoptosis is mediated through the decrease in mitochondrial membrane potential (Figure 5) and the alteration of the BCl-2 to Bax ratio (Figure 6). PJE was also found to increase caspase-3 and caspase-9 activities (Figure 7) and the MAPK and ROS signaling pathways were identified as being involved in PJE-induced cell death (Figures 8−10). Thus, it was postulated that PJE induced the apoptosis of cancer cells through ROS-mediated JNK/p38 MAPK activation of the intrinsic mitochondrial pathway (Figure 11).
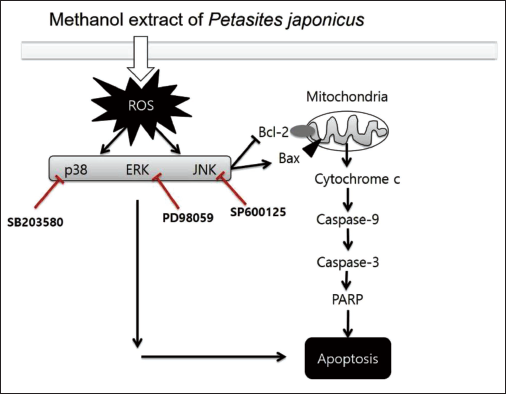
The various cellular signaling pathways include various mechanisms such as MAPKs and PI3K/Akt and are known to control cell proliferation and apoptosis (Luo et al., 2015; Sarkar et al., 2009). Controlling cell cycles and apoptosis with various cellular signaling mechanisms is important for managing cancer cell growth and cancer tissue formation (Hanahan & Weinberg, 2011). Among these diverse cellular signaling mechanisms, the MAPK cascade is emerging in the development of new anticancer drugs that are less toxic than previously known cancer-related chemotherapy drugs (Sebolt-Leopold, 2000). In the present study, cell viability was measured after the co-administration of PJE and SB203580, SP600125, or PD98059. Co-administering PJE with SB203580, SP600125, or PD98059 increases cell viability (Figure 8). Furthermore, western blot analysis demonstrated that PJE treatment led to an increase in MAPK phosphorylation in the cells (Figure 9). These findings substantiate the essential involvement of MAPK signaling pathways in the induction of cell death by PJE.
Cancer cells are generally subjected to markedly more oxidative stress than normal cells (Pelicano et al., 2004). Therefore, the levels of ROS or oxidative products are high in cancer tissues. Antioxidants act as ROS scavengers and activate the antioxidant enzymes in cells of the biological system (Moussaid et al., 2011). Naturally produced antioxidants are mainly produced in bioactive compounds, such as flavonoids, polyphenols, and carotenoids, which have received much attention as safe and powerful bioactive compounds (Miliauskas et al., 2004; Mohsen & Ammar, 2009). Therefore, antioxidants, which are abundant in medicinal plants, fruits, and vegetables, protect against various diseases, such as cancer, caused by oxidative stress (Willett, 2002). The present study shows that ROS production is increased by PJE treatment (Figure 9). Hence, the generation of ROS plays a critical role in the cell death triggered by PJE.
Many studies on the relevance of ion channels in relation to cancer pathophysiology are currently being performed (Stokłosa et al., 2020), and TRP ion channels have been receiving particular attention among ion channels (Mochizuki et al., 2009). The TRP ion channels are characterized as non-selective cation channels, enabling the passage of both monovalent and divalent cations (Nilius & Owsianik, 2011). Alterations in these channels are frequently observed in bladder cancers (Mizuno et al., 2014). Bladder cancer pathophysiology has been linked to multiple TRP channels (Gao et al., 2017; Mizuno et al., 2014; Mochizuki et al., 2009; Yamada et al., 2010). The identification of these TRP channels as potential targets for cancer treatment will be carried out using PJE. Subsequent studies will delve deeper into exploring the potential roles of these TRP channels as targets for cancer treatment with PJE.
Radix Sophorae flavescentis caused apoptosis in gastric cancer and bladder cancer, and it could be seen that ROS and MAPK mechanisms were involved (Jo et al., 2020; Kim et al., 2019). Hemistepsin A, the main component of Hemistepta lyrata Bunge, inhibited the binding of pyruvate dehydrogenase (PDH) complex and pyruvate dehydrogenase kinase 1 and increased the ROS level and damage to mitochondria, thereby inhibiting colon cancer growth (Jin et al., 2020). Isoliquiritigenin, a component of various licorice plants, killed bladder cancer cells with caspase and ROS mechanisms in bladder cancer (Hwang et al., 2020), and Alisma canaliculatum extract and its component, alisol B 23-acetate, have been reported to cause apoptosis of AGS, a gastric cancer cell line, with a small concentration (Kwon et al., 2021a, b). In addition, the double-negative expression of E-cadherin and N-cadherin was involved in the mesenchymal transition processes of bladder urothelial carcinoma (Luo et al., 2016), and in TSGH83, one of the types of bladder cancer cells, administration of paclitaxel and Mulberry water extract together promoted mitotic catastrophe by activating of PTEN to create synergy in anticancer effects (Chen et al., 2016). In this paper, bladder cancer cell death by PJE is similar to the general cancer cell death mechanism and we will study the relevance of other mechanisms through deeper research in the future, such as the relevance of PDH and cadherin and the synergy effects with anticancer drugs such as paclitaxel.
Consequently, the objective of this present study is to unveil the cytotoxic impact of the methanol extract of PJE on 5637 bladder cancer cells. Further comprehensive investigations are necessary to discern the specific compounds responsible for inducing apoptosis.
Conclusion
This study offers compelling evidence of the apoptotic effects of PJE on human bladder adenocarcinoma 5637 cells. The administration of PJE led to heightened levels of proapoptotic cascade elements such as caspase-3, caspase-9, and poly ADP-ribose polymerase. Furthermore, PJE demonstrated the ability to regulate MAPK activation and enhance the production of ROS. These findings indicate that PJE is a potent herbal medicine with proapoptotic properties, exerting its effects through MAPK-dependent ROS signaling pathways. Significantly, this study represents the initial confirmation of PJE’s ability to induce cell death in bladder cancer cells, emphasizing its potential as a valuable resource for the future development of anticancer medications.
Summary
PJE treatment produced dose-dependent cytotoxicity in all three Caco-2, Hep3B, and 5637 cell lines. As the cytotoxic effect of PJE was most potent in bladder cancer 5637 cells, the 5637 cell line was selected for subsequent experiments.
The sub-G1 phase ratios were increased and the mitochondrial membrane potential was reduced in a dose-dependent manner by PJE in 5637 cells.
Bcl-2 survival was reduced by PJE, whereas Bax was increased.
PJE was found to dose-dependently increase the activities of caspase-3 and caspase-9 and the co-administration of PJE with SB203580, SP600125, or PD98059 increased cell viability. Also, the phosphorylation of MAPKs increased by PJE from 0.5 h to 4 h.
PJE increased ROS production.
Footnotes
Abbreviations
PJ: Petasites japonicus; PJE: Methanol extract of Petasites japonicus; ERK: Extracellular signal-regulated kinase; JNK: C-Jun N-terminal kinase; ROS: Reactive oxygen species; DCF-DA: 2′,7′-Dichlorodihydrofluorescein diacetate; CCK-8: Counting kit-8; MAPK: Mitogen-activated protein kinase; NAC: N-Acetyl-L-cysteine; HBlEpC: Human bladder epithelial cell; DMSO: Dimethyl sulfoxide.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education (2021R1I1A3042479 and RS-2023-00247033) and by the Main Research Program (E0210201-03) of the Korea Food Research Institute (KFRI), funded by the Korean Ministry of Science and ICT.
Statement of Informed Consent and Ethical Approval
Not applicable.
