Abstract
Background:
Shikonin, a purified naphthoquinone separated from a Traditional Chinese medicinal herb
Objectives:
To clarify the molecular mechanisms of therapeutic effects of shikonin against osteosarcoma.
Materials and Methods:
Cell Counting Kit-8 (CCK-8) assay was employed to evaluate cell viability. Annexin V-fluorescein isothiocyanate/propidium iodide (Annexin V-FITC/PI) double-staining was conducted to evaluate the apoptotic ratio of the MG-63 cells. The effects of shikonin on the invasiveness of osteosarcoma cells were implemented by a transwell cell migration assay. In the meantime, a western blot assay was employed to detect alterations in the relevant mechanism proteins within osteosarcoma cells. Molecular docking analysis was conducted to anticipate the binding interaction between shikonin and EGFR/protein kinase B (AKT)/mTOR.
Results:
We observed that shikonin suppressed proliferation and induced apoptosis in the MG-63 cells in a dose-dependent manner. Pursuing these findings, the potential molecular mechanisms were detected. Shikonin intervention blocked epidermal growth factor receptor (EGFR) phosphorylation and decreased epidermal growth factor (EGF)-induced activation of downstream signaling molecules, such as AKT and mammalian target of rapamycin (mTOR) in the MG-63 cells. However, the additional recombinant human epidermal growth factor (rHuEGF) could stimulate the activation of EGFR/AKT/mTOR signaling and reverse cell apoptosis caused by shikonin. Molecular docking analysis showed that shikonin presented the highest bonding ability with EGFR, AKT, and mTOR.
Conclusion:
Our results show that shikonin inhibits human osteosarcoma development via inactivating EGFR/AKT/mTOR signaling. It demonstrates that shikonin may act as a potential therapeutic agent in osteosarcoma treatment.
Introduction
The osteosarcoma is the most aggressive primary solid tumor of the bone in children and adolescents. The current treatment for osteosarcoma mainly includes surgical excision and systemic chemotherapy (Harrison et al., 2018; Jiang et al., 2014; Wu et al., 2016; Zhang et al., 2018). The efficacy of these treatments in improving five-year survival for patients with osteosarcoma, particularly those with metastatic or recurrent osteosarcoma, remains limited despite the presence of significant side effects. (Harrison et al., 2018; Wu et al., 2016). Therefore, a new therapeutic approach with increased efficiency and reduced toxicity is urgently needed.
Shikonin is isolated from the roots of the herb
As a receptor tyrosine kinase, the epidermal growth factor receptor (EGFR) has an important effect on physiology and tumors (Lemmon & Schlessinger, 2010). A recent study shows that higher EGFR expression exits in human osteosarcoma tissues than in adjacent normal tissues, which suggests a therapeutic role in targeting EGFR in osteosarcoma (Wang et al., 2018). EGFR inhibition, by EGFR inhibitor ZD6474 or anti-EGFR antibody cetuximab positively has some effects on osteosarcoma therapy (Liu et al., 2015; Pahl et al., 2012). Previous research has demonstrated that EGFR can regulate cell metastasis, survival, and proliferation by activating numerous signaling pathways, including PI3K/protein kinase B (AKT) (Liang et al., 2019), and Ras/Raf/MEK/ERK pathways (K. Li et al., 2017). EGFR blockade with cetuximab decreases the activity of AKT/mammalian target of rapamycin (mTOR) signaling and alleviates the resistance to antitumor immunity through increasing attraction of T cells within head and neck squamous cell carcinoma (Ma et al., 2018). In esophageal cancer, shikonin shows its antitumor effect by suppressing the EGFR/AKT/mTOR signal cascade (Tang et al., 2018). However, whether shikonin treatment for osteosarcoma may be a good therapy targeting EGFR/AKT/mTOR signaling is unclear.
Osteosarcoma cells from human subjects exposed to shikonin demonstrated a significant inhibition of cell growth. It indicates that shikonin inhibited osteosarcoma cell growth through the EGFR/AKT/mTOR signaling pathway.
Materials and Methods
Reagents
Shikonin was purchased from Sigma (Cat# S7576, USA). Dulbecco’s modified eagle medium (DMEM) (Cat# 10566024) was purchased from Gibco (USA). Antibodies against EGFR (Cat# 2232), p-EGFR (Cat# 2234), AKT (Cat# 9272), p-AKT (Cat# 9271), mTOR (Cat# 2972), phosphorylated mammalian target of rapamycin [p-mTOR] (Cat# 2971), β-catenin (Cat# 8480), N-cadherin (Cat# 4061), E-cadherin (Cat# 14472), and Bcl-2 (Cat# 15071) were provided from Cell signaling Technology (USA). Cleaved-caspase-3 (Cat# ab2302) and Bax (Cat# ab53154), were purchased from Abcam (USA). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (Cat# sc-47724), rabbit, and mouse secondary antibodies were purchased from Santa Cruz (USA). Cell Counting Kit-8 (CCK-8) for cell viability analysis was purchased from Sigma (Cat# 64987, USA). FITC-conjugated Annexin-V/propidium iodide (PI) for apoptosis analysis was purchased from BD Biosciences (Cat#556547, USA). Recombinant human epidermal growth factor (rHuEGF) was purchased from R&D systems (Cat# 236-EG, USA).
Cell Cultures and Chemical
Human osteosarcoma MG63 cells and human primary osteoblastic cells (hFOBs) were offered from ATCC (USA). The cells were cultured in DMEM, which was supplemented with 10% fetal bovine serum (FBS) sourced from GIBCO (USA). Subsequently, the cells were incubated at a constant temperature of 37°C within a humidified chamber.
Cell Proliferation Assay by CCK-8
The CCK-8 assay was used to measure cell proliferation in line with the manufacturer’s instructions. The cells (5 × 104 cells/well) were seeded in 96-well plates with 100 µL DMEM medium compounded with 10% FBS. Following the observation of significant cell death in osteosarcoma cells exposed to a high dose (>5 µmol/L) (Fu et al., 2013), here we studied the effects of shikonin at low doses (0.2 and 2 µM). After incubating for 48 h, a mixture of 10 µL CCK-8 reagent and 100 µL DMEM was added to each well, followed by additional culturing for another hour in a 5% CO2 environment. The absorbance rate at 450 nm was mediated by a Microplate Reader (Bio-Rad, USA).
Apoptosis Analysis by Flow Cytometry
Cells were seeded in six-well plates with an occurrence of 1 × 106/mL and then handled with shikonin at concentrations including 0.2 and 2 µM for 24 h. After treatment with shikonin, cells were collected, rinsed twice with cold PBS, and then resuspended in 1× binding buffer. Subsequently, cells were incubated with FITC-conjugated Annexin V and PI for 15 min in the dark at room temperature, followed by a one-hour flow cytometry analysis to examine the samples.
Transwell Cell Migration Assay
Migration assays were conducted employing transwell chambers equipped with polycarbonate membrane inserts (pore size: 5 µm, BD Biosciences, USA), following the manufacturer’s guidelines. The suspension of cells was conducted in a medium devoid of serum, and 100 µL (approximately 5 × 104 cells) was placed into the upper chamber of the transwell chambers. Afterward, the lower chamber was gently supplemented with 1 mL of complete medium containing 10% FBS. After a 24 h incubation period at a temperature of 37°C, non-migratory cells located on the upper side of the membrane were carefully removed using cotton-tipped swabs. Subsequently, migratory cells that traversed the membrane were fixed with a solution containing 4% paraformaldehyde for a duration of 30 min and subsequently stained with a solution comprising 0.1% crystal violet for an additional period of 30 min. The migrated cells located on the lower side of the chamber were meticulously captured and quantified under microscopic observation. (Olympus Corporation, Japan).
Western Blot
The full total proteins were extracted from cells by typical procedures, and then separated by SDS-PAGE gel electrophoresis and wet-transferred to PVDF membranes. The membranes were clogged, rinsed thrice with PBS, then incubated with antibodies against β-Catenin, N-Cadherin, E-Cadherin, Bcl-2, cleaved-caspase-3, BAX, p-AKT, AKT, p-mTOR, mTOR, p-EGFR, and EGFR at 4°C overnight, accompanied by incubation with HRP-conjugated secondary antibodies. The blots were scanned on the X-ray film applying electrochemiluminescence (ECL).
Molecular Docking
We first obtained the crystal structure of AKT (PDBID: 6npz), EGFR (PDBID: 1m17), and mTOR (PDBID: 5gpg) in the PDB bank. Then, we used the CB-Dock Server (Liu et al., 2020) to predict the binding modes between the 3 targets and Shikonin. Finally, Discovery Studio software elucidated the 2 best docking results between the compound and the 3 target proteins.
Statistical Analysis
All information was examined by utilizing the SPSS 19.0 software system (IBM Corporation, USA). The statistical differences were evaluated using the Student’s
Results
The Influence of Shikonin on Proliferation in Osteosarcoma Cells
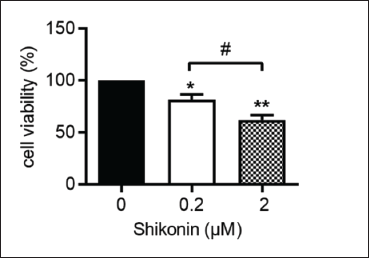
To investigate the effects of shikonin on osteosarcoma cells, the MG63 cells were treated with various concentrations of shikonin (0, 0.2, and 2 µM) for 24 h. Then, a CCK-8 assay was carried out. As shown in Figure 1, shikonin visibly decreased the proliferation of MG63 cells in a dose-dependent manner, as compared with that in the control group.
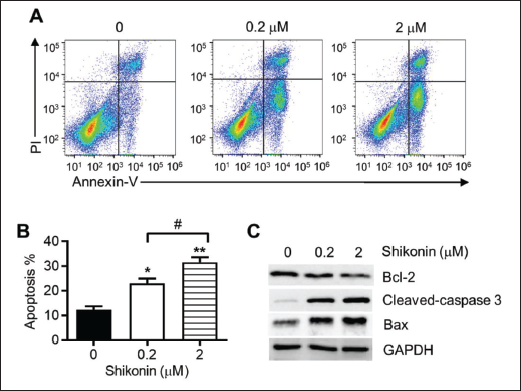
The Effect of Shikonin on Apoptosis in Osteosarcoma Cells
Then, we detected the apoptosis of the MG63 cells after shikonin treatment. As expected, shikonin significantly stimulated the apoptosis of the MG63 cells (Figure 2A and B). To explain the molecular mechanisms that shikonin inhibited osteosarcoma cell growth, we measured proteins related to apoptosis through the implementation of a western blot assay. The findings revealed that shikonin decreased the levels of anti-apoptotic protein Bcl-2, and increased the expressions of pro-apoptotic proteins cleaved-caspase-3 and Bax when compared to the control groups (Figure 2C). All these results showed that shikonin could have a positive therapeutic effect on human osteosarcoma cells by promoting cell apoptosis.
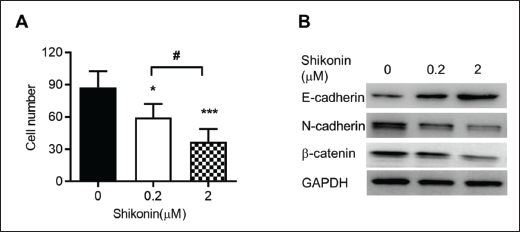
The Impact of the Shikonin on Migration of Osteosarcoma Cells
The osteosarcoma cell migration was executed using a transwell cell migration assay. Figure 3A demonstrates a significant dose-dependent reduction in the number of migrated cells following shikonin treatment when compared to the control groups. Given that epithelial-mesenchymal transition (EMT) is regarded as a key delegate in tumor metastasis and invasion and has proven to promote tumor development (Liu et al., 2018). Then, we examined proteins closely related to the EMT event, such as the epithelial marker E-cadherin and, correspondingly, the mesenchymal markers N-cadherin and β-catenin utilizing western blot assay. Our findings displayed that E-cadherin was upregulated obviously while N-cadherin and β-Catenin were downregulated evidently in shikonin groups comparable to control groups (Figure 3B).
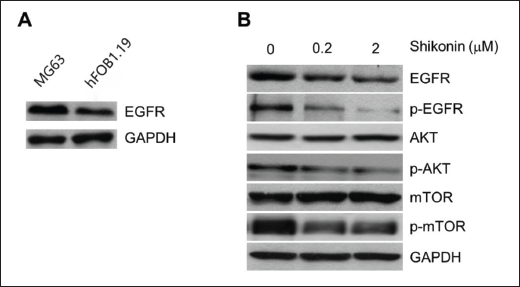
The Impact of Shikonin on EGFR/AKT/mTOR Signaling
The levels of EGFR in both osteosarcoma tissues and cells are higher, which is positively correlated with the GTM grade of osteosarcoma (Wang et al., 2018). As shown in Figure 4A, the expression of EGFR in the MG63 cells was higher than that of the hFOB1.19 cells. Given that EGFR/AKT/mTOR plays a crucial role in tumorigenesis and progression (Ma et al., 2018; Tang et al., 2018). And the EGFR/mTOR signaling pathway is considered to be associated with osteosarcoma growth (Geng et al., 2014). Thereby, we conducted further investigations to determine whether the key signaling pathways are involved in the potent inhibitory effects of shikonin on osteosarcoma cell proliferation and apoptosis. The results expectedly demonstrated a significant reduction in phosphorylation levels of EGFR, AKT, and mTOR upon administration of shikonin to MG63 cells. Nevertheless, there were no discernible alterations detected in the overall levels of AKT and mTOR. (Figure 4B).
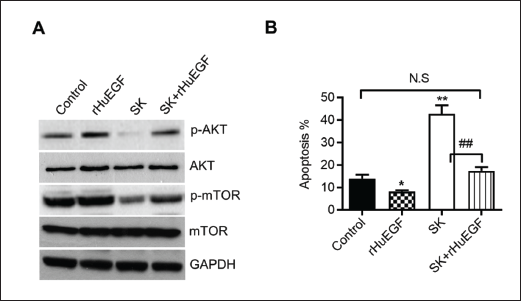
The Inbitory Potency of rHuEGF on the Apoptosis of Osteosarcoma Cells Induced by Shikonin
To validate the suppressive effect of shikonin on osteosarcoma cells through the EGFR/AKT/mTOR signaling pathway, the MG63 cells were pretreated with 100 ng/mL rHuEGF for 30 min, then followed by a common incubation with 2 µM of shikonin for 24 h. The findings indicated that rHuEGF obviously alleviated the impact of shikonin on the phosphorylation of AKT and mTOR (Figure 5A). Corresponding to the findings, rHuEGF remarkably reversed the increase of cell apoptosis induced by shikonin (Figure 5B).
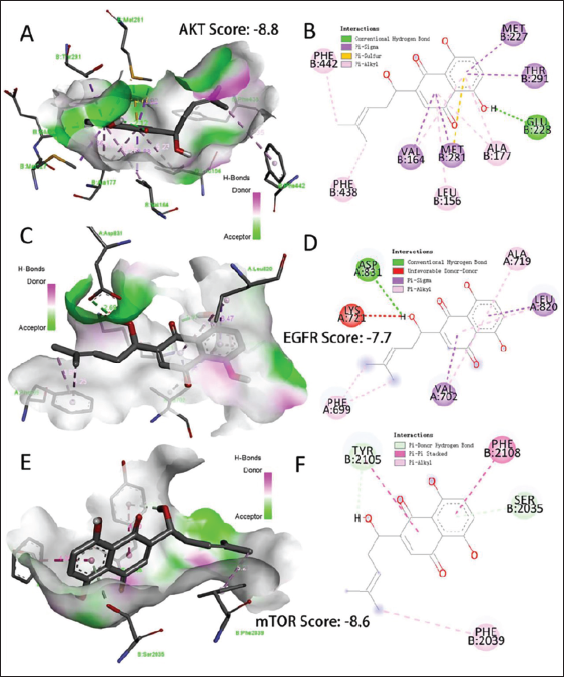
Molecular Docking
The interactions between shikonin and the three structural proteins (AKT, EGFR, and mTOR) were elucidated using the CB-Dock server and Discovery Studio software. A lower docking score indicates a stronger binding between the compounds and their targets due to reduced energy requirements. For AKT (Figure 6A and B), shikonin presented the highest bonding ability (Score: −8.8) than other targets and showed 12 interactions including hydrogen bond, pi-alkyl, and pi-sulfur, which were connected with MET227, GLU228, PHE442 and LEU156, and so on. For EGFR (Figure 6C and D), shikonin presented the higher bonding ability (Score: −7.7) and showed 9 interactions including hydrogen bond, unfavorable donor-donor, pi-alkyl, and pi-sigma, which were connected with ASP831, ALA719, PHE699, and LEU820, and so on. For mTOR (Figure 6E and F), shikonin presented the higher bonding ability (Score: −8.6) and showed 5 interactions including hydrogen bond, pi-alkyl, and pi-pi stacked, which were connected with TYR2105, PHE2039, PHE2108, and SER2035, and so on.
Discussion
New findings have indicated that shikonin, a bioactive compound derived from a traditional Chinese herb, displays promising anticancer properties across different types of malignancies. (Feng et al., 2021; Gong & Li, 2011; Hsu et al., 2004; Huang & Hu, 2018; X. Li et al., 2017; Tang et al., 2018; Wang et al., 2014; Yang et al., 2013; Zhao et al., 2015), including osteosarcoma (Chang et al., 2010; Deng et al., 2015; Deng & Qiu, 2015; Fu et al., 2013; Li et al., 2018; Yang et al., 2017). Nevertheless, a complete understanding of the molecular mechanisms involved remains elusive. In the present study, we investigated the biological efficacy of different concentrations of shikonin on the human MG63 osteosarcoma
Previous studies have indicated the therapeutic functions of shikonin on many kinds of tumors. Shikonin-inhibited tumor cell growth through increasing caspase activity-dependent apoptosis and preventing proliferation has been found in colorectal cancer cells (Hsu et al., 2004), endometrioid endometrial cancer (Huang & Hu, 2018), hepatocellular carcinoma (Feng et al., 2021), non-small cell lung cancer (X. Li et al., 2017). Few reports have suggested the anti-tumor efficacy of shikonin on osteosarcoma. Shikonin could decrease cell proliferation and promote apoptosis in 143B osteosarcoma cells through enhanced cell cycle arrest and DNA condensation (Chang et al., 2010). Shikonin-prevented osteosarcoma cell growth and invasiveness were evidenced by its dose-dependently diminished MMP13 levels (Deng et al., 2015), and increased RIP1 and RIP3-dependent necroptosis (Fu et al., 2013; Li et al., 2018). In the MG63 cells, we detected the cell viability and survival rate and found an obviously inhibitory proliferation and elevated apoptosis rates by both 0.2 and 2 µM shikonin, and the effects might be in a dose-dependent manner, which is similar to a previous study (Fu et al., 2013). However, the results displayed non-obvious cell toxicity while treating hFOB1.19 cells and other human normal cells, such as normal liver cells (L02), normal lung epithelial cells (BEAS-2B), and normal skin cells (HF) (Supplementary Figure S1), suggesting shikonin could be safe for clinical application. Further study showed that shikonin dose-dependently decreased the migration cell number using a tumor cell migration assay. Given that EMT is regarded as a key delegate in tumor metastasis and invasion and has proven to promote tumor development (Liu et al., 2018). Then, the proteins closely related to EMT occurrence were examined, and the results revealed E-cadherin was upregulated obviously while N-cadherin and β-catenin were downregulated evidently in shikonin groups comparable to the control groups. As a result, shikonin might function as a therapeutic effect on human osteosarcoma cells.
Molecular target therapy is an effective approach to address the issue of cancer recurrence. EGFR is triggered by peptide growth factors of the EGF family (Cataldo et al., 2011), which are commonly overexpressed in many kinds of cancer, such as hepatocellular carcinoma (Huang et al., 2020), lung cancer (X. Li et al., 2017), neuroendocrine carcinoma (Bago-Horvath et al., 2012)and glioblastoma (Wierzbicki et al., 2017). As a protooncogene, high expression of EGFR relates to progressive and metastatic tumors, indicating that there would be a common mechanism for proliferation relying on EGFR stimulation (Grandis & Sok, 2004). EGFR blockages are currently permitted for medical use or in advanced clinical trials in individuals with cancer. A recent study showed a higher expression level of EGFR in osteosarcoma areas than in nearby tissues, and miR-141-3p could hinder the development and migration of osteosarcoma by deregulating EGFR expression (Wang et al., 2018). Our results showed over-expression of EGFR in the human MG63 osteosarcoma cells when compared with the normal human osteoblastic cells
Several reports have proved that regulation of EGFR destruction and its downstream signaling cascades by shikonin results in its anti-tumor attributes in several cancer diseases (Geng et al., 2014; X. Li et al., 2017; Tang et al., 2018; Wu et al., 2013). However, the therapeutic effect of shikonin on osteosarcoma, which operates through targeting EGFR and its downstream signaling pathways has not been reported. In the present study, we revealed that shikonin at 0.2 µM or 2 µM concentration could significantly decrease p-EGFR expression levels in the MG63 cells, indicating shikonin might function on osteosarcoma cells via regulating the phosphorylation of EGFR. This is because EGFR degradation resulted in the inhibition of its downstream proliferation and anti-apoptotic signaling, which contribute to the anti-tumor function of shikonin (X. Li et al., 2017). Therapy for malignant tumors targeting EGFR/AKT/mTOR axis has been proven as a clinically important method (Bago-Horvath et al., 2012; Huang et al., 2020; X. Li et al., 2017; Wierzbicki et al., 2017). However, whether the EGFR/AKT/mTOR pathway would be a potential target for shikonin-induced osteosarcoma cell death is unclear. Indeed, shikonin treatment led to reduced p-EGFR levels, accompanied by lower phosphorylations of both AKT and mTOR in the MG63 cells. A further study showed that the addition of rHuEGF could conspicuously elevate expression levels of p-AKT and p-mTOR in the MG63 cells, which benefits cell fate by reducing apoptosis. We also proved that shikonin-induced osteosarcoma cell apoptosis could be retarded by rHuEGF. Besides, a number of researchers have applied molecular docking to predict the interaction between natural compounds and targets (Chen et al., 2020, 2021). Our molecular docking results further demonstrated that shikonin may directly target AKT, EGFR, and mTOR. Taken together we figured that EGFR/AKT/mTOR may be engaged in shikonin-regulated MG63 cell growth and migration.
Conclusion
In summary, the findings of this study indicate that the EGFR/AKT/mTOR signaling pathway in MG63 cells could be effectively suppressed by shikonin, suggesting its potential as a promising therapeutic agent for treating osteosarcoma.
Footnotes
Abbreviations
EGFR: Epidermal growth factor receptor; AKT: Protein kinase B; mTOR: Mammalian target of rapamycin; p-EGFR: Phosphorylated epidermal growth factor receptor; p-AKT: Phosphorylated protein kinase B; p-mTOR: Phosphorylated mammalian target of rapamycin; rHuEGF: Recombinant human epidermal growth factor; OS: Osteosarcoma; hFOBs: Human primary osteoblastic cells; RTK: ErbB family of receptor tyrosine kinases; DAPI: 4,6-diamino-2-phenyl indole; ECL: Electrochemiluminescence; BAX: Bcl-2-associated X; Bcl-2: B-cell lymphoma-2; GAPDH: Glyceraldehyde-3-phosphate dehydrogenase; ROS: Reactive oxygen species; ERK: Extracellular signal-regulated kinase; CCK-8: Cell counting kit-8; SD: Standard Deviation.
Acknowledgments
We are grateful to the Affiliated Cancer Hospital of Zhengzhou University and Henan Cancer Hospital for generously providing us with the invaluable experimental platform.
Authors’ Contributions
Xiao-Bin Yao, and Zhang-Jiao Wang, performed the experiments and analyzed the data. Qing-Xia Xu, Rui-Dan Zhu, and Jia-Jia Zhang drafted the manuscript. Zhi-Ling Du assisted with the data analysis. Rui-Dan Zhu was responsible for the study design and final approval of the manuscript. All authors have meticulously reviewed and consented to the final version of the manuscript.
Data Availability
The data underpinning the results of this study are accessible upon request from the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by grants from the Health Commission of Henan Province (SBGJ202102070).
Statement of Ethics and Informed Consent
Not applicable.
