Abstract
Background
Huanglian (HL), also known as Coptis chinensis Franch., was initially documented in “Sheng Nong’s Herbal Classic” as a high-quality product used to cure ailments such as fever, indigestion, bloating, nausea, dysentery, diarrhea, abdominal pain, and tuberculosis. Nowadays, it is extensively employed in clinical settings to alleviate pulmonary diseases, like tuberculosis, lobular lung disease, and pulmonary abscess, and is worth exploring further. Nevertheless, there is limited research on the precise advancements of anti-lung adenocarcinoma (LUAD).
Objectives
Using biological experiments and network pharmacology, the objective of this research was to investigate the most effective parts and chemical constituents of C. chinensis against LUAD and ascertain the probable targets and pathways.
Materials and Methods
First, an MTT experiment verified the impact of each part on A549 and BEAS-2B cells. High performance liquid chromatography (HPLC) was then used to identify the chemical components of the effective substance in C. chinensis against LUAD. Then, network pharmacology was used to screen potential pathways and binding target proteins. Finally, a subcutaneous tumor model was established in mice using LLC cells to observe the effects of the effective components of C. chinensis against LUAD in tumor-bearing mice.
Results
The MTT assay demonstrated the efficacy of oxyepiberberine (OPB) as an anti-LUAD compound with minimal toxicity and side effects. According to the results of network pharmacology and molecular docking, SRC appears to be the primary target of C. chinensis active substance against LUAD. In vivo experiments revealed that the OPB group exhibited lowered toxicity and side effects. Additionally, the density of tumor cells decreased and the nuclei were destroyed in OPB groups of tumor tissues. Finally, OPB could regulate apoptosis, migration, and protein expression significantly related to the SRC pathway in tumor cells.
Conclusion
Our research reveals the anti-LUAD properties of C. chinensis through a combination of experimentation and network analysis. These findings establish a theoretical foundation for C. chinensis as a clinical antitumor agent. Furthermore, our work provided an essential groundwork for further investigations into the medicinal properties of C. chinensis.
Introduction
In total, 85% of lung cancer which has the highest incidence rate in the world was non-small cell lung cancer (Chen et al., 2019). This cancer can be sub-divided into large cell carcinoma, squamous cell carcinoma, and adenocarcinoma subdivisions based on the histological characteristics, which highlights the heterogeneity of non-small cell lung cancer (Seguin et al., 2022). Lung adenocarcinoma (LUAD) is the principal sub-type of non-small cell lung cancer (Li et al., 2014). The World Health Organization (WHO) divides it into five types based on the histological characteristics: adherent, acinar, papillary, microemulsion head, and solid (Schneider et al., 2016). Additionally, various pathological subtypes exhibit distinct histological features that impact gene expression and prognosis, as well as serving as clinically relevant factors for the auxiliary diagnosis and prognosis of lung cancer. The development of LUAD is determined by the build-up of mutations in numerous genes, leading to the unchecked proliferation of tumors. A considerable proportion of patients receive a diagnosis in advanced stages, presenting significant problems for both patients and healthcare providers (Zhu et al., 2022).
Huanglian (HL) is a perennial plant in the Ranunculaceae family, and its dried rhizomes are used for medicinal purposes. HL is known to have therapeutic benefits such as clearing heat, drying dampness, purging fire, and detoxifying. To date, more than 100 chemical components, including alkaloids, flavones, and lignans, have been isolated from HL. Among these components, alkaloid is the most effective one. It is important to note that the use of HL for medical purposes should be consulted with a healthcare professional (Wu et al., 2019). In the research done on non-small cell lung cancer cells, HL has demonstrated the ability to impede growth and metastasis as well as initiate cell apoptosis (Li et al., 2018; Ni et al., 2017). Studies have demonstrated that the use of HL in conjunction with silver nanoparticles can restrict the proliferation of lung cancer cells by regulating apoptosis through the intrinsic pathway (Pei et al., 2019). Berberine found in HL has the capability to attain an anti-tumor effect through regulating autophagy, eliminating tumor cells, enhancing sensitivity and efficiency, reversing drug resistance, blocking tumor cell invasion and metastasis, impeding tumor angiogenesis, regulating cell cycle, inducing cell death, scavenging and inhibiting free radicals, as well as other mechanisms (Liu et al., 2019; Su et al., 2017; Wang et al., 2015). Furthermore, HL is recognized as a “Traditional Chinese Medicine (TCM) antibiotic”, attributed to its anti-bacterial and anti-inflammatory properties (Hao et al., 2020; Wang et al., 2021).
TCM has a significant role in treating lung cancer. It is important to note that the effectiveness of TCM in treating lung cancer has been well-established through research studies. It not only provides effective standalone treatment but also complements modern surgery, radiotherapy, and chemotherapy to improve curative effects and minimize adverse reactions (Chen et al., 2014; Zhang et al., 2021a). Based on research findings, TCM has the ability to combat lung cancer through various mechanisms, including destruction of direct tumor cell, induction of tumor cell apoptosis, inhibition of tumor angiogenesis, regulation of immune function, and modulation of oncogene expression (Su et al., 2020; Yang et al., 2023). Studies have demonstrated that HL has the potential to considerably impede lung cancer, liver cancer, and other tumorous growths via a range of means; therefore, it holds promising prospects for anti-tumor applications in the future (Zhao et al., 2016).
In 2006, protoberberine alkaloid oxyepiberberine (OPB) was isolated from the rhizome of Coptis japonica Makino (Ranunculaceae) by natural means. We acquired OPB samples via chromatographic separation techniques. The compound’s activity and toxicity were tested, while its effects against LUAD were assessed using network pharmacology. In vivo experiments were then carried out to furnish a scientific foundation for OPB’s action against LUAD.
Materials and Methods
Reagents and Instruments
HL was acquired from Hubei Tianji TCM Decoction Co. Ltd. and verified by experts to comply with the Chinese Pharmacopoeia standards. It is conserved in the Key Laboratory of Resource Science and Chemistry of Chinese Medicine in Hubei Province, which belongs to Hubei University of Chinese Medicine, located in Wuhan, Hubei, China. Berberine hydrochloride, jatrorrhizine hydrochloride, tetrandrine hydrochloride, palmatine hydrochloride, and OPB were procured from Chengdu AIFA Biotechnology Co. Ltd. All substances had a purity exceeding 98%.
Methanol, ethanol, petroleum ether, ethyl acetate, and n-butanol were obtained from Sinopharm Chemical Reagent Co. Ltd. We purchased high performance liquid chromatography (HPLC) from Agilent Technology Co., Ltd. and the pathological slicer from Shanghai Leica Instrument Co. Ltd.
Cytotoxicity and Chemical Composition Analysis
In total, 200 g of herbal material was extracted repeatedly using 10 times its weight of 75% ethanol. The filtrate was combined and concentrated under reduced pressure until all alcohol taste was gone, resulting in a concentrated solution. The solution was then separately extracted from petroleum ether, ethyl acetate, and n-butanol. Vacuum recovery was performed and resulted in the extraction of petroleum ether, ethyl acetate, n-butanol, and water. Dry extracts of four parts of HL were obtained through vacuum drying and subsequently stored at low temperature for future use. Solutions with concentrations of 10 and 0.2 mg/mL were prepared by dissolving the active and reference substances in methanol. The above-mentioned samples were subjected to gradient elution using a TC-C18 chromatographic column. The mobile phases employed were acetonitrile (A) and 0.03 mol/L ammonium bicarbonate solution (B). The gradient elution procedure was as follows: 0−15min, A:10%−25%, B:90%−75%; 15−25 min, A:25%−27%, B:75%−73%; 25−40min, A:27%−45%, B:73%−55%; 40−45min, A:45%−45%, B:55%−55%. The flow rate was 1 mL/min, and the column temperature was at 30°C. The detection wavelength was 270 nm, and the injection volume was 10 µL. Additionally, we conducted an MTT assay on A549 and BEAS-2B cells. The experimental groups comprised a blank group with only culture medium, a control group with only cells, and administration groups with different concentrations of extraction. The microplate reader detected the optical density (OD) value of absorbance at 495 nm, and the survival rate of cells was calculated using SPSS 19.0 statistical software. The formula for cell survival rate is provided below:
Cell viability (%) = (OD experimental group − OD blank group)/(OD control − OD blank group) × 100% (Almutary & Sanderson, 2016).
Target Prediction
The structures of seven alkaloids from HL were sourced from PubChem before the target was predicted via the SwissTargetPrediction and PharmMapper server databases. Genes related to LUAD were obtained from the GeneCards database (Pan et al., 2019; Zhang et al., 2022). The obtained compound targets and disease-related targets were imported into the Venny 2.1.0 database for screening candidate core target proteins of HL (Liang et al., 2019).
Bioinformatics Annotation
The identified core targets were imported into DAVID databases for the gene ontology (GO) analysis as well as the Kyoto Encyclopedia of Genes and Genomes (KEGG). Visual representation of the data obtained from both sources was presented using column and bubble charts (Wang et al., 2019; Y. Zhang et al., 2019; Y. F. Zhang et al., 2019). The active constituents of HL, potential primary target proteins, and LUAD-associated pathways were inputted into the Cytoscape 3.7.1 application, resulting in the production of the compounds-targets-pathways (C-T-P) network, which facilitated the exploration of the core targets. Additionally, the protein-protein interactions (PPI) network was established using the String database (C. Wang et al., 2022).
Molecular Docking
Seven compounds were introduced into AutoDock for the purposes of energy minimization, hydrogenation, and charge treatment. The first four target proteins from the RSCB PDB database were screened and imported into PyMOL for the removal of non-protein ligands. The processed receptors and ligands were subsequently added into AutoDock for docking, and a result indicating a binding bond energy of less than −4 kcal/mol was selected (W. X. Wang et al., 2022; Yuan et al., 2021).
Establishment of Lung Cancer Model
Five BALB/c mice were chosen and injected with LLC cells at a density of 1 × 106 cells/mL. They were then fed for 7 days, and their ascites were extracted and diluted to 2 × 107 cells/mL. Another 50 mice were injected with 0.2 mL in their right forelimb; 40 mice were chosen for subsequent experiments when the tumor reached 50 m3 (Berceanu et al., 2016).
Experimental Grouping and Administration
The 40 mice were grouped and administered as follows: the Normal group received 0.2 mL/10 g of normal saline via stomach irrigation, the Model group received 0.2 mL/10 g of normal saline, the OPB low-dose group (Low) was given 20 mg/kg of a drug and 0.2 mL/10 g of irrigation in the stomach, the OPB high-dose group (High) received 40 mg/kg of the drug and 0.2 mL/10g of irrigation in the stomach, and the Dasatinib group received 20 mg/kg via intraperitoneal injection. The drug was administered once a day for 18 days. Mice consumed food and water ad libitum. The weight and volume of tumors in mice were measured daily. After 24 h of last administration, the mice were weighed and their blood was collected. Tumor tissues and liver of mice were dissected, photographed, and weighed. Additionally, a few tumor tissues were preserved in wax blocks.
Hematoxylin & Eosin Staining
The paraffin sections of mice in each group underwent dewaxing, hydration, hematoxylin staining, differentiation, blue washing, eosin staining, dehydration, transparency, and sealing to produce Hematoxylin & Eosin (H&E) staining sections (Nadarajan & Doyle, 2020).
Immunohistochemistry
The paraffin-embedded tissue was sliced and then dewaxed in xylene, washed in PBS, dewaxed in absolute ethanol, and washed again in PBS. After pretreatment, the samples underwent 3% hydrogen peroxide washing, followed by incubation in primary and secondary antibodies, and chromogenic agent application. Hematoxylin re-staining was performed, and then, a gradient alcohol dehydration step was taken before the sections were clarified in xylene and sealed in resin. Lastly, the sections were examined under a microscope (Cleary et al., 2020).
Results
Cytotoxicity of Active Fractions
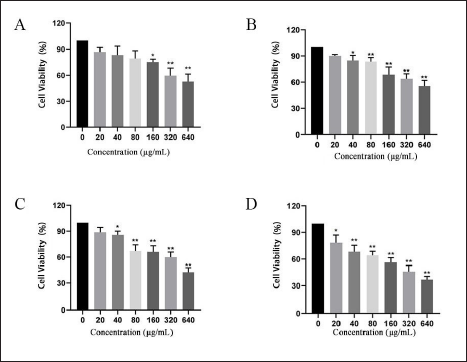
The following are the weights of HL extracts: Petroleum ether: 8.16 g, ethyl acetate: 5.89 g, n-butanol: 16.1 g, and water: 16.98 g. The MTT results demonstrated that the activity of A549 cells was considerably affected at 160 µg/mL (water section), 40 µg/mL (petroleum ether and ethyl acetate sections), and 20 µg/mL (n-butanol section) concentrations. These results are depicted in Figure 1. Therefore, the n-butanol extraction of HL was chosen as the active fraction against LUAD to investigate its active constituents further.
Analysis of Chemical Composition
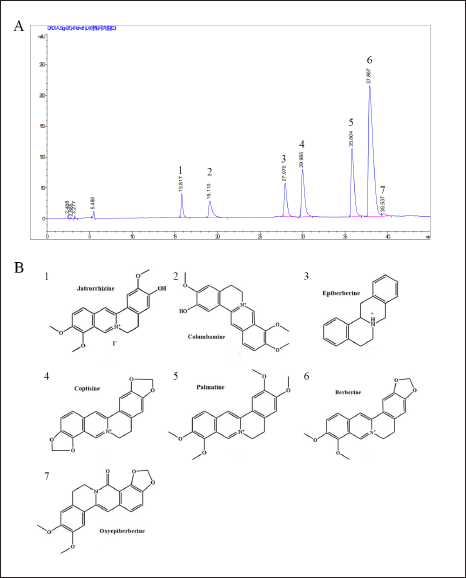
The n-butanol sample underwent the HPLC analysis, and upon comparing it with the reference substance and previous research, it was found that HL’s n-butanol extract comprises of seven vital compounds. The liquid phase is illustrated in Figure 2A, and these seven compounds, namely: jatrorrhizine, African tetrandrine, epiberberine, coptisine, palmatine, berberine, and OPB are identified by their retention times. Their molecular structures are illustrated in Figure 2B.
Active Compounds Inhibited Growth in A549 Cells
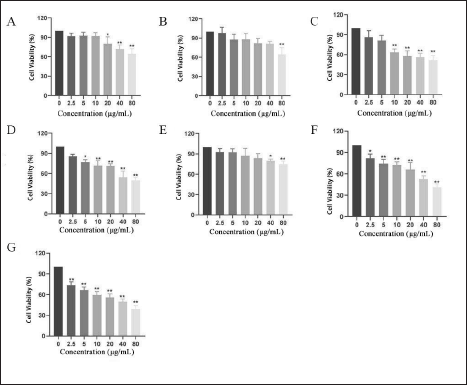
The MTT assay was used to investigate the effect of seven compounds on the proliferation of A549 cells. The results are shown in Figure 3. According to the results, berberine, coptisine, and OPB had the most obvious inhibitory effect on the proliferation of A549 cells after 24 h of administration, among which OPB had the strongest inhibitory effect and showed obvious concentration dependence. When the administration concentration was 2.5 µg/mL, which significantly inhibited A549 cells (p < 0.01), and the concentration reached 80 µg/mL, the cell survival rate decreased to less than 50% (p < 0.01).
Low Toxicity of OPB in BEAS-2B Cells
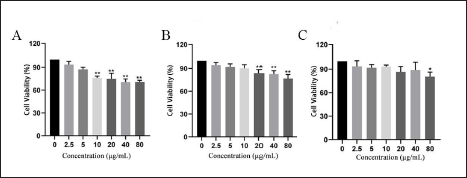
According to the experimental results of the effects of seven compounds on the proliferation of A549 cells, we selected berberine, coptisine, and OPB and detected their toxic effects on human lung epithelial cell BEAS-2B by the MTT method. As shown in Figure 4, the concentrations of coptisine, berberine, and OPB at which the cell viability reached a significant difference after 24 h were 10 µg/mL (p < 0.01), 20 µg/mL (p < 0.01), and 80 µg/mL (p < 0.05), respectively. This result suggests that among the seven active compounds of the n-butanol fraction, OPB has the least toxic effect on human normal lung epithelial cells.
Screening of Potential Targets
The seven active alkaloids of the n-butanol fraction of HL were imported into the SwissTargetPrediction database for analysis. The top 100 targets corresponding to each component were intercepted. After removing duplicates, a total of 272 active component targets of HL were obtained. The related targets of LUAD were searched from the GeneCards database and 1999 up- and downregulated disease genes were obtained. The disease genes were then analyzed using the Venny2.1.0 database. The intersection of 272 targets of the screened compounds and 1999 disease targets was obtained, and 126 potential targets of HL compounds for the treatment of LUAD were obtained. The most important compounds are shown in Table 1.
Main Active Compounds in HL.
Biological Function Analysis of Screened Potential Targets
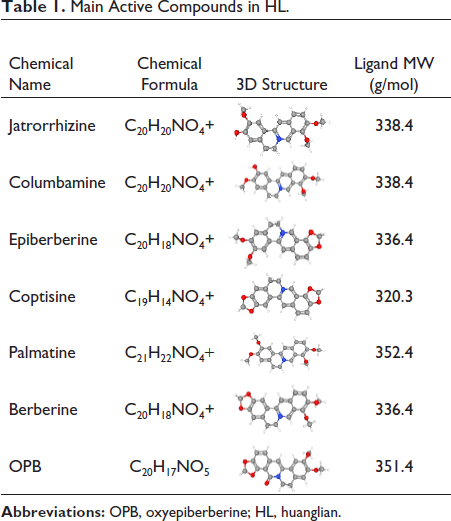
A total of 126 potential targets were screened using the SwissTargetPrediction and GeneCards databases. Potential targets were imported into the DAVID database for the GO and KEGG enrichment analysis. Using a p-value of less than 0.05 as a standard, the top 10 were selected for visual analysis. The visualization results of GO and KEGG enrichment analysis were shown in Figure 5.
C-T-P Network and PPI Construction Analysis
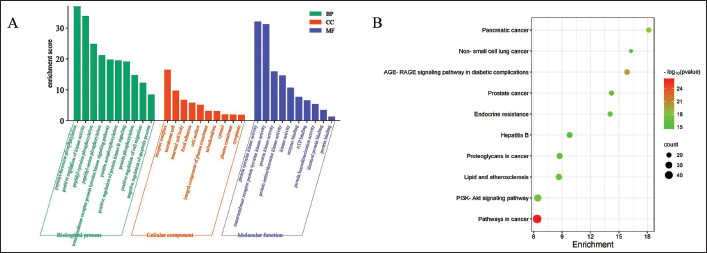
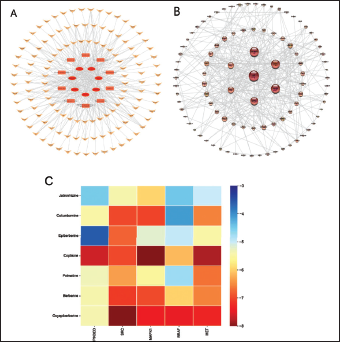
The network analysis of active compounds, potential targets, and enrichment pathways of HL for the treatment of LUAD was constructed by Cytoscape3.7.1 software, as shown in Figure 6A. In order to further explore the interaction between the targets, the potential targets were put into STING database; we got the PPI network, as shown in Figure 6B. As shown in Table 2, after analyzing the data results of Cytoscape3.7.1, we got the top five potential targets: PIK3CD, SRC, MAPK1, BRAF, and MET. We regard these five targets as key targets. According to the PPI results, we further found that SRC interacts most closely with other proteins. We preliminarily believe that SRC gene is the core target of these five genes.
Top Five Codes in the Order of Degree of C-T-P.
Molecular Docking Display
Based on the analysis of the effective parts of HL and the results of compounds targets pathways (C-T-P) and PPI, seven alkaloids and the above five key targets were selected for molecular docking by AutoDockTools. The docking fractions and amino acid residues of the compounds and targets are shown in Table S1 in the supplementary material. PyMOL software was then used for visualization. The visualized matrix thermogram is shown in Figure 6C. The results of the matrix thermogram showed that the binding energies of coptisine, berberine, and OPB to the five receptors were the Top 3. It is speculated that these three compounds may be the key compounds in the n-butanol fraction of HL against LUAD. Furthermore, we found that the docking binding energies of SRC and MAPK1 to the seven compounds were in the top 2, indicating that they are closely related to the mechanism of the active components of HL against LUAD. Figure 7 shows the molecular docking results of OPB, and the other six compounds presented in the supplementary material.
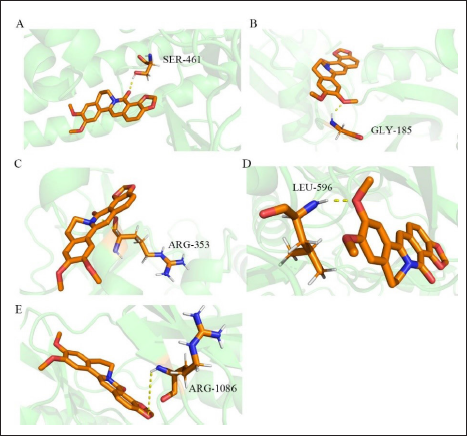
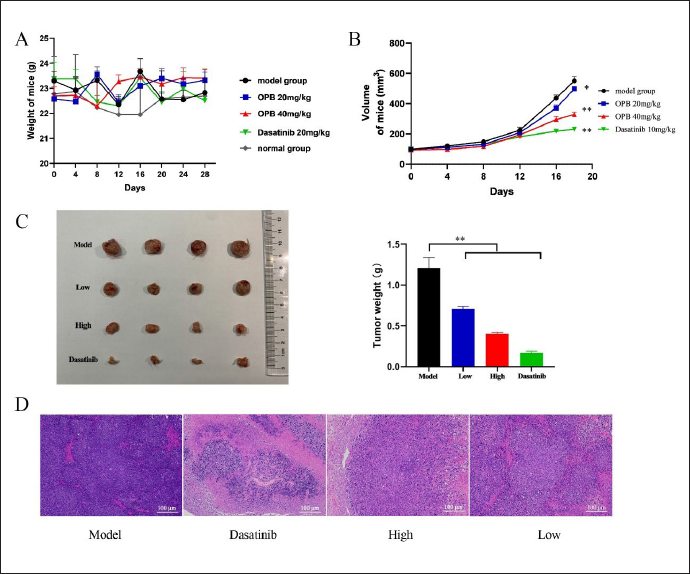
In vivo Anti-tumor Activity of OPB
After the inoculation of LLC cells, there was no significant change in food intake, water consumption, coat shine, and spontaneous activity of the mice in each group. As showed in Figure 8A, apart from the mice in the dasatinib group who experienced weight loss during the experimental period, the other five groups of mice exhibited weight gain to varying degrees, with no significant difference in weight change compared to the model group.All treatment groups, including the dasatinib group, exhibited a deceleration in tumor growth compared to the model group. The effect was more pronounced in the dasatinib group. Figure 8B displayed the changes in tumor volume in tumor-bearing mice during the experimental period. The size and weight of the final tumors in each group of mice were presented in Figure 8C. The results showed that both OPB and dasatinib inhibit tumor growth, as demonstrated by the reduction in tumor volume. Additionally, the inhibitory effect of OPB on tumor weight was dependent on the dosage.
H&E Staining
As shown in Figure 8D, it was observed under 200× microscope that the cells in the model group had good morphology and complete structure, and no obvious dead cells were found; dead cells were observed in the low-dose group, high-dose group, and dasatinib group. Compared to the model group, the tumor cells in the high-dose group were fragmented, the nuclei were solidified, and the chromatin was dense and concentrated. It could be seen from this result that the inhibitory effect of OPB on the tumor is obvious, but it does not reach the level of the positive drug.
OPB Significantly Upregulated the Expression of Bax and Downregulated the Expression of Bcl-2
Under normal circumstances, when cells are stimulated by apoptotic signals, Bax oligomerizes to form the Bax/Bax homodimer, which releases pro-apoptotic factors into the cytoplasm, leading to apoptosis (Moldoveanu & Czabotar, 2020).
Bcl-2 can compete with Bax to form the Bax/Bcl-2 heterodimer. The structure of the latter is more stable than the former, which can prevent the release of pro-apoptotic factors. Bcl-2 can therefore inhibit Bax-induced apoptosis.
As shown in Figure 9A, the expression of Bcl-2 in the dasatinib group, high-dose group, and low-dose group was significantly lower than that in the model group (p < 0.01). The expression of Bax in the dasatinib group, high-dose group, and low-dose group was significantly higher than that in the model group (p < 0.01). The above experimental results showed that dasatinib, high-dose, and low-dose OPB significantly upregulated the expression of Bax, downregulated the expression of Bcl-2, promoted the apoptosis of tumor cells, and effectively inhibited the proliferation of tumor cells.
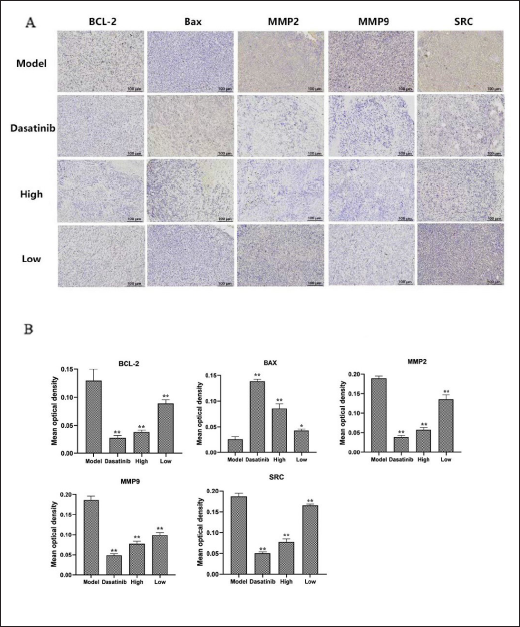
OPB Downregulated the Expression of MMP2/9
The MMP family plays an important role in inhibiting tumor migration. The positive expression rates of MMP2 and MMP9 in lung cancer tissues are higher than those in adjacent tissues and benign lung lesions, and the same is true in patients with metastatic lung cancer (T. Zhang et al., 2021).
As shown in Figure 9B, the positive expression of MMP2/9 was very high in the tumor tissue of the model group, but the positive expression was low in the dasatinib group and in the high- and low-dose groups. The expression of MMP2/9 was significantly decreased in the dasatinib group (p < 0.01). The expression of MMP2/9 in the high-dose group was significantly lower than that in the low-dose group (p < 0.01). The above experimental results showed that dasatinib, high, and low doses of OPB downregulated the expression of MMP2/9 and inhibited the migration of tumor cells, thereby preventing the growth process of the tumor.
OPB Inhibited the Expression of SRC Protein
Based on the above results, we further examined the effect of OPB on the expression of SRC in tumor cells by immunohistochemistry. As shown in Figure 9B, the positive expression of SRC was very high in the tumor tissues of mice in the model group, but the positive expression was low in the dasatinib group, the high-dose group, and the low-dose group. Among them, the expression of SRC was significantly decreased in the dasatinib group (p < 0.01). The expression of SRC in the high-dose group was significantly lower than that in the low-dose group (p < 0.01). The above experimental results showed that dasatinib, high, and low doses of OPB downregulated the expression of SRC, which was consistent with the results of network pharmacology and cell experiment in vitro.
This result suggests that in the process of HL effective substance in the treatment of LUAD, the most important compound, OPB will bind to SRC protein in cells, inhibit the expression of SRC protein, and further inhibit the activation and expression of downstream proteins, so as to inhibit the proliferation and migration of tumor cells, promote the apoptosis of tumor cells, and slow down the process of tumorigenesis.
Discussion
Many active ingredients in TCM have been found to have anti-tumor effects, and OPB is one of them. Liu et al. (2020) found that OPB can inhibit TGF-β1-induced epithelial mesenchymal transformation by interfering with the Smad3 protein, thereby inhibiting tumor cell migration. Ning et al. (2021) found that OPB could inhibit the growth of colon cancer cells by inhibiting the polymerization of tubulin. However, there is no report on the efficacy and mechanism of OPB in the treatment of LUAD. In this study, the n-butanol extract from HL exhibits the most effective anti-lung cancer activity according to MTT screening, and seven major alkaloids in the n-butanol part of HL were identified by HPLC. Berberine, coptisine, and OPB had the best inhibitory effect on A549 cell proliferation, and OPB had the least toxicity to BEAS-2B cells. This suggests that OPB may play an important role in anti-LUAD.
Network pharmacology reveals the interaction of “drug-disease-target” through systems biology, high-throughput screening, network analysis, network visualization, and other technologies and analyzes drug action from the perspective of the whole system. Based on the similarity of drugs in structure and action, combined with the complex interaction relationship and biological effects of target molecules, the drug function or drugs corresponding to specific functions can be effectively predicted by constructing networks such as “drug-target-pathway” and “PPI” (Bing et al., 2022). In this study, through the research of network pharmacology, the relevant targets of active components of HL in the treatment of LUAD were selected. The “compound target pathway” network and “PPI” network were established, and the top five key compounds were screened according to the target degree value, which were PIK3CD, SRC, MAPK1, BRAF, and MET, respectively. SRC had the highest clustering degree in the PPI network. The results of the GO enrichment and KEGG enrichment analysis showed that the core target treatment of LUAD was mainly enriched in a variety of cancer pathways and phosphatidylinositol 3-kinase protein kinase B signaling pathway (PI3K/Akt signaling pathway), which provided a way for us to further verify the experiment. The results of the molecular docking experiment showed that SRC has stronger binding ability with seven compounds, among which the binding energy with OPB is the highest, which was consistent with the results of the previous MTT experiment. It was again confirmed that OPB is the most important compound in the active components of HL, and SRC may be the key target of OPB in the treatment of LUAD.
Abnormal expression of SRC has been implicated in the development of numerous types of cancer. As a member of the tyrosine kinase family, it changes the conformation of SRC by dephosphorylating amino acids, leading to the autophosphorylation of another tyrosine residue in the protein activation loop. This enhances its activity, upregulating many signaling cascades associated with tumor progression and resulting in increased cell growth, migration, and invasion (Bagnato et al., 2020). SRC has been identified as a critical target for cancer treatment. Its activation is associated with the proliferation and migration of cancerous cells (Lai et al., 2017). It has been demonstrated that SRC is an essential target for anti-LUAD, and its correlated pathways warrant attention.
Zhang et al. have demonstrated in vitro that OPB reduces SRC activity, leading to the inhibition of tumor cell proliferation and migration. Our study utilized tumor-bearing mice as the research model to investigate the in vivo anti-LUAD effect of OPB (Zhang et al., 2021b). In comparison with the normal group, the group given Dasatinib exhibited more signs of depression, while the group given OPB displayed normal behavior. This implies that Dasatinib has significant toxic and side effects, while OPB has minimal to no side effects on mice. During the experiment, both the high-dose OPB group and the Dasatinib group had a significant impact on preventing tumor growth, with Dasatinib having the highest inhibition rate. The tumor volume change curve of mice indicated this result. Furthermore, the pathological sections demonstrated a reduction in the number of cells and destruction of the nuclear structure in both the OPB and Dasatinib groups when compared to the model group. Immunohistochemical staining revealed that OPB and Dasatinib regulated the expression of apoptosis, migration-related proteins, and SRC proteins in tumor cells, which corresponds to in vitro experimental results. It was preliminarily confirmed that OPB enhanced tumor cell apoptosis and inhibited tumor cell migration through the SRC protein pathway, resulting in an anti-LUAD effect.
Conclusion
The active components of HL were identified using HPLC and MTT in this study, followed by determining the targets and pathways of HL. Subsequently, in vitro experiments demonstrated that OPB can induce tumor cell apoptosis and prevent tumor cell migration via the SRC protein pathway to enable the anti-LUAD effect. This study establishes a theoretical understanding of the potential of HL for treating LUAD.
Availability of Data and Materials
The datasets used and/or analyzed during the current study were available from the author upon reasonable request. The main supporting data can be found in the supplementary material of the article.
Abbreviations
OPB: Oxyepiberberine; LUAD: Lung adenocarcinoma; HL: Huanglian; TCM: Traditional Chinese Medicine; WHO: World Health Organization; HPLC: High performance liquid chromatography; C-T-P: Compounds targets pathways; PPI: Protein-protein interactions; GO: Gene ontology; KEGG: Kyoto encyclopedia of genes and genomes; BP: Biological process; MF: Molecular function; CC: Cellular component; H&E staining: Hematoxylin eosin staining; EGFR: Epidermal growth factor receptor; OD: Optical density.
Footnotes
Acknowledgments
We extend our gratitude to the Provincial Key Laboratory of Hubei University of Traditional Chinese Medicine and the National Key Research and Development Plan Fund for their valuable support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal experimental procedures and animal care were approved by the Hubei University of Chinese Medicine Ethics Committee and were conducted in accordance with the guidelines of the National Institutes of Health on the care and use of animals. Approval No. HUCMS 202006011.
Funding
This work was supported by National Key R & D Plan of TCM Modernization Research (2017YFC1701000).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
