Abstract
Background
Glycitin, generated from the seeds of the legume soybean, has an essential function in antioxidant, obesity inhibition, and wound healing promotion. Furthermore, recent research has shown that it has anti-inflammatory and cartilage-protective properties.
Objectives
This study was designed to investigate the osteogenic effects of glycitin and the transforming growth factor-β (TGF-β) signaling pathway on bone marrow mesenchymal stem cells (BMSCs), as well as their interaction.
Materials and Methods
We isolated rabbit BMSCs by whole bone marrow adherent method and cultured, identified, and induced differentiation. The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method was used to determine the proliferation ability of BMSCs with different concentrations of glycitin at different times; reverse transcription-polymerase chain reaction (RT-PCR) was used to detect the expression levels of osteogenesis-related genes runt-related transcription factor 2 (Runx2), collagen type I (Col-1), and osteocalcin (OC) mRNA; and a kit was used to determine the activities of alkaline phosphatase and triglycerides (TG) in BMSCs. A Western blot was used to detect the expression of TGF-β protein in each group.
Results
On the fifth day of BMSC primary culture, the morphology of long spindle-shaped cells might be plentiful, dominated by a daisy-like or whirling arrangement. They proliferated rapidly and fused to 70%–80% on the eighth day of culture; the third generation of BMSCs was taken for phenotyping, and the detection results of flow cytometry analysis showed that the surface antigenic markers of BMSCs, CD44, and CD90 were positive, whereas CD34 and CD45 were negative; after osteogenic induction of BMSCs, visible calcified nodules were evident. There was cartilage matrix formation after osteogenic induction; glycitin promoted BMSC proliferation in a time-dependent and dose-dependent manner, and BMSC proliferation increased significantly at 20 µM glycitin for 3–5 days; glycitin increased the expression of Runx2, Col-1, and OC mRNA in BMSCs and increased the activity of alkaline phosphatase (ALP) while decreasing the activity of TG. Meanwhile, 20 µM glycitin dramatically increased the expression of TGF-β.
Conclusion
These findings imply that glycitin increases BMSCs proliferation and osteogenic differentiation. By regulating the TGF-β signaling pathway, glycitin modulates the differentiation of BMSCs into osteoblasts.
Introduction
In recent years, the incidence of steroid-induced avascular necrosis of the femoral head (SANFH) has been progressively rising. This increase can be attributed to the erroneous administration of hormones in clinical practice, coupled with prolonged high-dosage utilization. SANFH has emerged as the leading cause of non-traumatic femoral head necrosis (Chen et al., 2022a). Due to the atypical clinical manifestations of early SANFH, it is not easy to detect, often resulting in treatment delays. Therefore, as the condition progresses, patients in the late stage typically experience pain, femoral head collapse, and the onset of osteoarthritis. Consequently, a range of impairments manifests, including abnormal gait, restricted mobility, and even reliance on assistive devices like crutches or wheelchairs in daily life. Ultimately, patients necessitate hip replacement surgery (Chen et al., 2022b). Regrettably, post-hip replacement, certain patients may encounter complications such as prosthesis loosening, infections, and dislocation. These complications give rise to considerable discomfort and, in severe instances, compel the requirement for revision hip arthroplasty. Consequently, SANFH is still a clinically intractable disease (Dagneaux et al., 2021). However, the specific pathophysiology of SANFH is yet unknown, which brings some difficulties to the treatment of SANFH. Adult bone marrow mesenchymal stem cells (BMSCs) represent a versatile cell population characterized by their capacity for multidirectional differentiation. The potential applications of BMSCs are broad-ranging and encompass critical-sized defect regeneration, fracture nonunion therapy, mitigation of cartilage and tendon injuries, as well as interventions targeted towards managing metabolic bone disorders and osteonecrosis (Shenaq et al., 2010). Several investigations have revealed that one of the key pathogenic mechanisms of SANFH is a decrease in the number of BMSCs differentiating toward osteoblasts and an increase in the number differentiating toward adipocytes (Wang et al., 2018). Hormone treatment reduces the osteogenic differentiation of the femoral head as well as the quantity or activity of BMSCs, which fails to supply enough osteoblasts to fulfill the need for bone rebuilding, resulting in femoral head collapse (Hardy et al., 2018; Zhao et al., 2019). As a result, it is critical to investigate the proliferation and directed differentiation of BMSCs in order to understand the mechanism of SANFH.
Glycitin, a compound derived from the seeds of legumes, is the main component of the free aglycones of soybean isoflavone. Because its molecular structure is similar to estrogen, it is a naturally active substance with an estrogen-like effect and beneficial to the human body, so it is also called phytoestrogen (Seo et al., 2017a). Since the 1980s, many studies and animal experiments have shown that glycitin has multiple biological activities, including antibacterial and antiviral, antioxidant, obesity inhibition, and wound healing (Choi et al., 2007; Kang et al., 2007). In addition, glycitin can prevent and treat osteoporosis and protect cartilage by regulating the dynamic balance process of osteoclasts and osteoblasts (Wang et al., 2020). Transforming growth factor-β (TGF-β) expression has also been observed to be increased by glycitin (Kim et al., 2015). And TGF-β is one of the favored growth factors for controlling BMSCs because it plays a significant role in regulating their proliferation and directed differentiation, particularly in osteogenic differentiation, bone matrix formation, and bone repair and rebuilding. TGF-β is regarded as a preeminent growth factor choice for orchestrating the regulation of BMSCs (Sun et al., 2018). Glycitin may plausibly engage in an interaction with the TGF-β signaling pathway, potentially manifesting a reinforcing or synergistic effect with TGF-β to potentiate the promotion of osteogenesis within BMSCs.
Glycitin may be an advantageous choice for the prevention and treatment of a number of chronic bone loss-related disorders, including osteomalacia and osteoporosis, by stimulating the proliferation and differentiation of osteoblasts. If this theory is confirmed, we may think about using glycitin in BMSCs to overcome the difficulty of obtaining the huge number of seed cells and stable transformation into osteoblasts needed for SANFH therapy. In order to provide an experimental foundation for the study of the pathogenesis of SANFH and, at the same time, to provide a new pharmacological strategy for the early treatment of this disease, the goal of this experiment was to ascertain how glycitin regulates the TGF-β signaling pathway to affect the proliferation, osteogenic and lipogenic differentiation of BMSCs.
Materials and Methods
Materials
Main Reagents and Instruments
Nucleic acid protein detector (Thermo Fisher Scientific Inc.,), TH4-200 inverted fluorescence microscope (Olympus Company, Japan), 1300 real-time quantitative PCR (RT-PCR) instrument (AB Company, USA), Western Blot electrophoresis/transposition instrument (Bio-Rad Company, USA), and chemiluminescence gel imager (General Electric Company, USA). Alkaline phosphatase (ALP) kit; 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) detection kit; TGF-β monoclonal antibody (Biyuntian Biotechnology Co., China); Alcian blue staining solution (Sigma Company, USA); triglyceride (TG) kit (Nanjing Jiancheng Bioengineering Institute, China); Trizol (Invitrogen Company, USA); runt-related transcription factor 2 (Runx2), collagen type I (Col-1), and osteocalcin (OC) gene primers (Shanghai Bioengineering Company, China); and β-actin monoclonal antibody (Beijing Tiandeyue Company, CN).
Methods
Isolation, Culture, Passaging, and Characterization of BMSCs
Following the euthanasia of experimental rabbits via air embolism, the hind limbs were longitudinally incised along the femoral shaft, and the soft tissue was bluntly separated from the femoral shaft. The medullary cavity of the rabbit femoral shaft was exposed under aseptic operation, and serum-free Dulbecco’s Modified Eagle Medium (DMEM)/F12 was rinsed 3–5 times until no obvious bone marrow rushed out. Tissue pieces were collected using a 70-µm filter into a 15-mL centrifuge tube and centrifuged for 5 min at 1200 rpm. The cells were resuspended in DMEM/F12 media containing 10% fetal bovine serum and 1% penicillin/streptomycin before being seeded into Petri dishes and then put in a 37°C, 5% CO2 cell incubator. The initial half-volume medium exchange is scheduled for 3 days post-initiation, followed by subsequent exchanges every 3 days thereafter. In the event of inadequate nutrient provision, observable yellowing of the culture medium indicative of a lowered pH, or an accumulation of metabolites, it is recommended to perform a medium change. When the cells were 70%–80% merged, they were digested with 0.25% trypsin and passaged in a 1:2 ratio. The third-generation BMSCs were taken into cell suspension, and then representative cell surface antigen markers CD34, CD44, CD45, and CD90 were selected for detection on flow cytometry.
BMSCs Osteogenic and Chondrogenic-induced Differentiation
Passage 3(P3)–Passage 4(P4)-generation BMSCs were harvested for osteogenic induction, and BMSCTXTs were planted at a density of 5 × 104 per well on six-well culture plates containing slides. Subjected to osteogenic induction medium for a duration of 21 days. The osteogenesis induction medium consisted of 50 µg/mL vitamin C, 10% fetal bovine serum, 10−4 µmol/L retinoic acid, and 1 µmol/L dexamethasone in DMEM basal medium, and the culture medium was changed once every 2–3 days. After staining for osteoblast-specific expression with ALP, the samples were air-dried and sealed with neutral gum, and the calcium salt deposition was seen and photos were recorded using an inverted microscope.
Take P3–P4-generation BMSCs that have been treated as described above and induced for 21 days using chondrogenic induction media. This chondrogenic induction medium formulation encompassed 50 µg/mL of vitamin C, 10% fetal bovine serum, 10−4 µmol/L of retinoic acid, and 1 µmol/L of dexamethasone. These constituents were incorporated into a DMEM basal medium. The culture medium was refreshed at intervals of 2–3 days during this phase. Chondrogenesis was examined under an inverted microscope in acidic pH tissue stained with Alcian Blue and treated with paraformaldehyde, and pictures were recorded to detect type II collagen exclusively expressed by chondrogenic cells.
MTT Technique for Determining BMSCs Proliferation Rate in Glycitin Group
BMSCs in the logarithmic growth phase were cultured in DMEM medium in 96-well cell culture plates (8000 cells per well, 200 µL), and we obtained the concentration gradient of glycitin for cell proliferation (Li et al., 2005) and mapped the conditions of the treatment concentration in the current experiments, which were divided into the control group and groups of 1, 5, 10, 20, and 40 µM glycitin. After 48 h, 20 µL of MTT solution was added to each well, the wavelength of the enzyme labeling equipment was adjusted to 490 nm, and the corresponding absorbance values were calculated and recorded. According to the findings of BMSC proliferation by varied concentrations of glycitin, they were separated into control and 5, 10, and 20 µM glycitin groups, which were treated for 1, 3, and 5 days, respectively. Cell proliferation rate = (A1 − A0)/(A2 − A0) × 100%, A1 represents the experimental group’s A value, A2 represents the control group’s A value, and A0 represents the blank control group’s A value.
Determination of ALP, TG Expression Levels in BMSCs
The cells were counted using ALP and TG kits. BMSCs in the logarithmic growth phase were taken and inoculated in 96-well plates. BMSCs were divided into control, 5, 10, and 20 µM glycitin groups, and the fluids were changed every 2–3 days, and the BMSCs were lysed using cell lysis solution after 7 days, and ALP and TG expression levels were detected in strict accordance with the kit’s instructions.
Detection of Osteogenesis-Related Gene Expression Levels in BMSCs
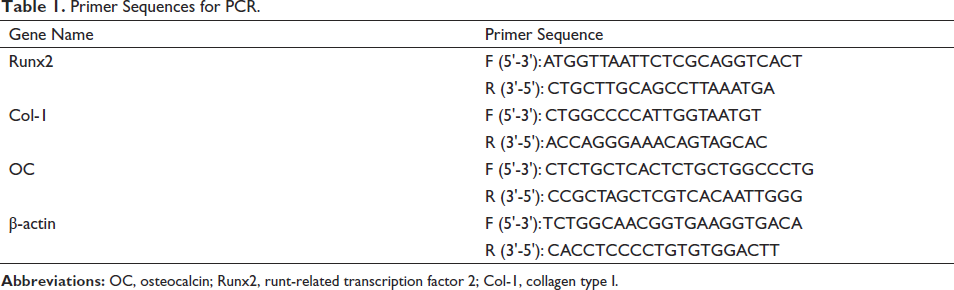
BMSCs were grown in groups of control, 5, 10, and 20 µM glycitin. Cellular RNA was extracted using the kit’s instructions; reverse transcription was used to generate cDNA, and amplification was carried out. The reverse transcription-polymerase chain reaction (RT-PCR) amplification procedure was as follows: 3 min of pre-denaturation at 95°C, 3 s of denaturation at 95°C, and 20 s of annealing at 60°C. After 35 cycles, the relative expression levels of the genes in the cells were calculated using 2−△△CT with β-actin as the internal reference gene. The primer sequences of the Runx2, Col-1, and OC mRNA genes of BMSCs are shown in Table 1.
Primer Sequences for PCR.
Western Blot Method to Detect TGF-β Protein Expression in Each Group
Cells from each group were collected; radio immunoprecipitation assay lysate and phenyl methane sulfonyl fluoride were added in a 100:1 ratio to washed phosphate-buffered saline (PBS) cells, and the washed cells were put on ice for lysis to extract the proteins in the cells, and the extracted proteins were measured. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis was done sequentially, then transferred to polyvinylidene fluoride (PVDF) membranes, which were sealed with a 5% skim milk powder sealing solution using PBS Tween-20 (PBST) and skim milk powder for 1 h. After overnight incubation at 4°C, the PVDF membranes were washed with PBST three times, and the membranes were treated with horseradish peroxidase-labeled secondary antibody at room temperature for 1 h. The membranes were then incubated for 1 h at room temperature with PBS and PBST, followed by 1 h with a HORSERAPY peroxidase-labeled secondary antibody. Finally, using an ultrasensitive enhanced chemiluminescence kit, develop the membrane, measure the bands created by the Western blot with a gel imager, and save the pictures.
Statistical Analysis
The data presented in this study were derived from a minimum of three independent replicates for each experiment. Statistical analysis and data processing were conducted utilizing GraphPad Prism 8 (GraphPad Software Inc., La Jolla, California, USA). The measurement results were expressed as the mean ± standard error of the mean. For numerous comparisons, a one-way ANOVA was performed, and the least significant difference test t-test was utilized for two-way comparisons between groups. A statistically significant difference was defined as p < 0.05.
Results
In vitro Primary Culture and Passaging of BMSCs
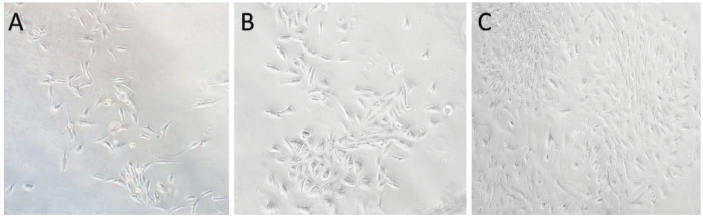
After a 2-day in vitro culture using the whole bone marrow adherent method, a small number of spindle-shaped cells develop, along with a substantial number of spherical cells. When the culture time is extended, the wall cells gradually expand while the red blood cells gradually disappear with the fluid change. After around 5 days, the adhering cells take the form of long spindle-shaped cells that may multiply in a clonal way, and the cells are mostly grouped in a daisy-like or vortex-like pattern. The cells are subsequently digested with trypsin and grow quickly following a 1:2 passage. After 8 days of union, the cells retain their long shuttle form (Figure 1).
Characterization of the Cellular Immunophenotype of BMSCs
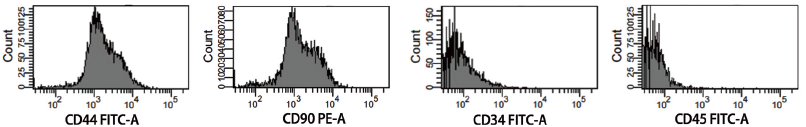
For phenotyping, P3–P4 BMSCs were used; typical cell surface antigen markers of CD34, CD44, CD45, and CD90 were chosen. Flow cytometry examination of the test findings revealed that BMSCs were positive for the surface antigen markers CD44 and CD90, but negative for CD34 and CD45 (Figure 2). It confirmed that the previously separated cells were BMSCs and ruled out contamination by fibroblasts and other cells.
Characterizing the Osteogenic and Chondrogenic Differentiation Capacity of BMSCs
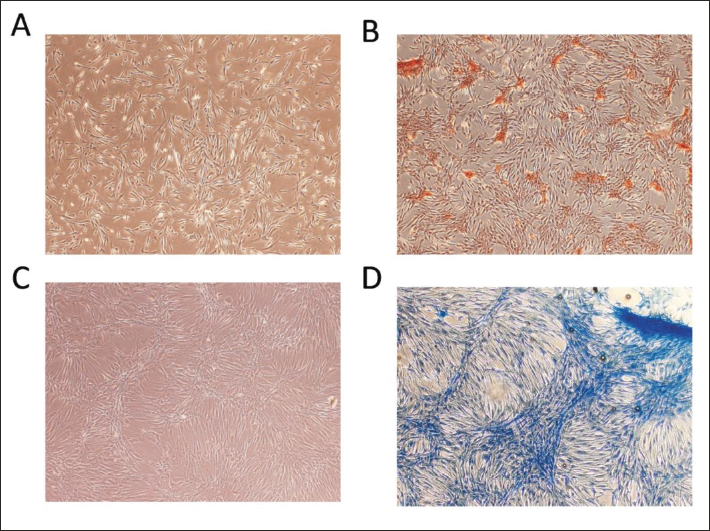
After 21 days of osteogenic induction, BMSCs revealed localized calcium salt deposition, which was stained with ALP as gray granules or lumpy and streaky precipitates, and evident calcified nodules were seen. After 21 days of chondrogenic induction, cartilage-like tissue with a harder texture was created, and Alcian blue-stained type II collagen was evident, indicating extracellular cartilage matrix production (Figure 3).
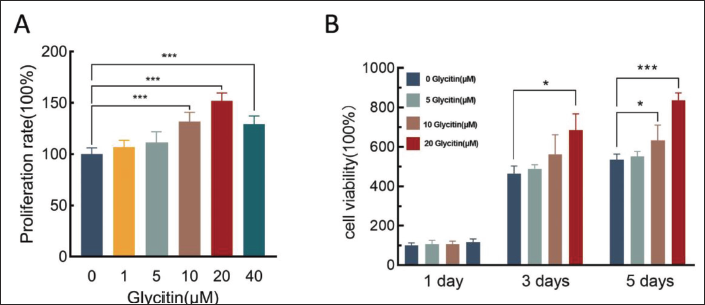
Effect of Glycitin on the Proliferative Capacity of BMSCs
MTT was used to assess the influence of glycitin on the proliferation potential of BMSCs. The results showed a significantly increased in the proliferation of BMSCs within the glycitin-treated groups at 10, 20, and 40 µM concentrations after a 48 h culture period, in contrast to the control group (p < 0.001). Considering the cytotoxic effect of the 5, 10, and 20 µM glycitin group for time-dependent assay, the results showed that there was a time-dependent effect of glycitin on the cell proliferation of BMSCs (Figure 4). Compared with the control group, on the third and fifth days of culture, 20 µM glycitin significantly increased the proliferation of BMSCs (p < 0.05, p < 0.001). According to the above results, we conclude that 20 µM glycitin is one of the best concentrations for BMSCs proliferation, and is time-dependent.
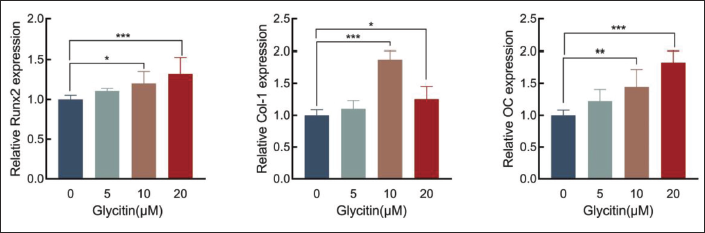
Effect of Glycitin on the Expression of Osteogenesis-related Gene
RT-PCR was used to study the effects of glycitin on the osteogenic markers Runx2, Col-1, and OC mRNA in BMSCs. Compared with the control group, the expression of Runx2 and OC increased in the 20 µM glycitin group significantly increased (p < 0.01, p < 0.001), while the expression of Col-1 in the 10 µM glycitin group was the most obvious (p < 0.001). These observations collectively indicate that glycitin exerts a stimulatory effect on the osteogenic differentiation of BMSCs (Figure 5).
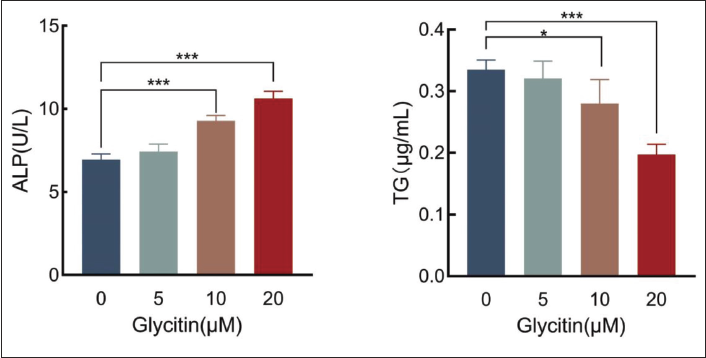
Effects of Glycitin on the Activities of the Bone-related Marker ALP and the Marker of Lipidogenic Differentiation TG in BMSCs
An ALP/TG kit was used to determine the influence of glycitin on the activity of bone-related markers ALP and TG in BMSCs. In contrast to the control group, the application of glycitin at concentrations of 10 and 20 µM induced a significant augmentation in the expression of ALP in BMSCs (p < 0.001). Additionally, in comparison to the control group, both the 10 and 20 µM glycitin-treated groups exhibited a significant reduction in TG expression levels within BMSCs (p < 0.05, p < 0.001). These findings collectively imply that the presence of 20 µM glycitin actively promotes the differentiation of BMSCs into osteoblasts while concurrently inhibiting the process of adipogenesis (Figure 6).
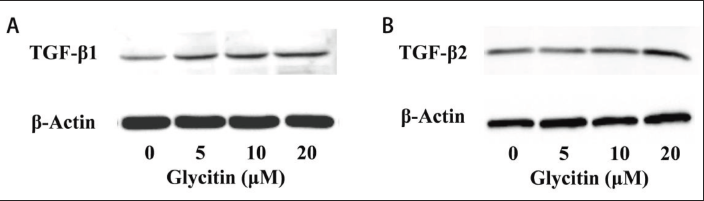
Effect of Glycitin on TGF-β Protein in BMSCs
A Western blot assay was used to detect the effect of glycitin on TGF-β1 and TGF-β2 protein expression in BMSCs. The results showed that 20 µM glycitin significantly promoted the expression of TGF-β1 and TGF-β2 (Figure 7).
Discussion
BMSCs are a kind of pluripotent, self-renewing non-hematopoietic stem cell that is frequently employed in clinical and experimental research (Zhang et al., 2023). However, the isolation and culture of BMSCs is influenced by a number of parameters that have a direct impact on cell purity, viability, and functional performance (Hou et al., 2021). This experiment covers the important elements influencing BMSCs isolation and culture. (1) In the initial culture of BMSCs, there are other cells such as fibroblasts and hematopoietic cells, etc., and these impurity cells may be efficiently eliminated by flushing the bone marrow to protect the purity and uniqueness of BMSCs. (2) Reasonable serum concentration management, with fetal bovine serum at 10% often chosen as a reasonable dosage to successfully preserve the viability and functional stability of BMSCs. (3) Cell inoculation density has a significant impact on the development and functional performance of BMSCs. It is advised that 5 × 105 cells/mL per milliliter of media be used to enable moderate BMSC communication and proper production of local secretory cytokines. As a result, correct handling and optimum circumstances will aid in the production of high-purity and healthy BMSCs.
Glycitin is extensively found in a number of plants as one of the key components of soybean isoflavones (Johnson et al., 2016). Because of its structural similarities to estrogen, it has been classified as a phytoestrogen, which has natural estrogen-like properties and is thought to be advantageous to the human body (Azam et al., 2020). Glycitin’s influence on BMSC proliferation was shown to be time-dependent and dose-dependent. Incubating BMSCs with glycitin at a concentration of 20 µM for 3–5 days dramatically boosted their proliferation. This proliferative impact was consistent with earlier findings, suggesting that glycitin may have a proliferation-promoting action (Zhang et al., 2016). Runx2, Col-1, and OC are all involved in controlling the production of extracellular matrix proteins throughout the differentiation process of osteoblasts (Moser & van der Eerden, 2018; Zhang et al., 2020). In this study, we discovered that glycitin increased the expression of Runx2, Col-1, and OC mRNA in BMSCs. It is suggested that glycitin possesses the capacity to stimulate osteoblast differentiation, thereby bolstering bone tissue regeneration and fortifying the bone formation process. Consequently, these attributes are anticipated to contribute to the maintenance of bone health. Moreover, glycitin’s role in enhancing osteogenic potential in BMSCs is evidenced by its capacity to augment osteoblast quantity and activity. Such effects, in turn, hold promise for facilitating bone rejuvenation and repair processes, reducing fracture susceptibility, aiding in fracture healing, and serving as a preventive measure against osteoporosis. The implications of this study further underscore the affirmative impact of glycitin on bone tissue growth and regeneration.
ALP is a key indication in the differentiation of BMSCs into osteoblasts, and its activity is linked to osteoblast activity and osteogenesis (Liu et al., 2021). According to research on the differentiation of BMSCs into osteoblasts, the process primarily involves cell proliferation, aggregation, and extracellular matrix calcification. The maturity of BMSCs differentiation into osteoblasts is characterized by increased ALP activity and extracellular matrix calcification (Guo et al., 2021). According to research, higher osteoblast differentiation correlates with increased ALP activity in BMSCs. TG are adipocyte indicators (Witztum et al., 2019), and in vitro studies have demonstrated that the TG content of BMSCs from SANFH is dramatically enhanced, resulting in an increase in adipocytes. Glycitin substantially boosted ALP activity in BMSCs while lowering TG activity in this experiment. This shows that glycitin promoted BMSCs development and maturation into osteoblasts. Simultaneously, glycitin exhibits the ability to impede the differentiation of adipocytes. Nonetheless, certain bone-related conditions, including femoral head necrosis and osteoarthritis, have revealed that adiponectin, a product of adipocytes, can potentially impairment the osteoblast function through alterations in the expression of adiponectin receptor I (Savopoulos et al., 2011). Consequently, the application of glycitin may hold the potential to mitigate inflammation and enhance joint functionality to a certain extent, achieved by mitigating the adipocyte count.
TGF-β is a polypeptide protein with multiple activities that is widely expressed as a growth factor in human tissues, including four subtypes, including TGF-β1 and TGF-β2, with the highest levels in bone tissue and platelets (Jann et al., 2020; Seo et al., 2017b). TGF-β engages in the creation of a signaling pathway via interactions with receptors situated on the cell membrane and intracellular signaling molecules (Garcia et al., 2021). The initiation of TGF-β’s signal transduction transpires through serine or threonine receptor kinases (Zhang et al., 2014). This leads to the phosphorylation of intracellular transducers Smad2/3, which subsequently form a complex with Smad4 (Bertrand-Chapel et al., 2022). This intricate assemblage traverses from the cell membrane into the nucleus, thereby eliciting a transcriptional response to TGF-β within the nucleus. This process activates the expression of the downstream pivotal transcription factor Runx2 (Wu et al., 2016). Simultaneously, Runx2 assumes the role of triggering the synthesis of bone matrix proteins, including Col-1 and OC (Huang et al., 2014). This orchestration further governs the regulation of osteogenesis. In this work, we discovered that 20 µM glycitin dramatically increased TGF-β expression. This suggests that glycitin may influence BMSCs osteogenic development via the TGF-β signaling pathway, hence improving BMSCs osteogenic capacity.
Conclusion
Glycitin increased the number of BMSCs in rabbits via the TGF-β pathway, as well as the expression of osteogenesis-related genes such as Runx2, Col-1, and OC, as well as the activity of ALP and the content of TG. These findings imply that glycitin increased osteoblast formation while reducing adipocyte differentiation and number. However, there are still some limitations to this experiment. This study focuses on in vitro experiments and needs further verification by animal experiments. There is still a certain distance between the application of glycitin and clinical treatment. In the future, it is necessary to establish an animal model of SANFH, study the interaction of various links in the process of SANFH bone repair and pay attention to the dosage, route, and time of glycitin so as to lay the foundation for clinical treatment. With the continuous, in-depth study of SANFH, we believe that SANFH disease will eventually be cured in the future.
Abbreviations
TGF-β: Transforming growth factor-β; BMSCs: Bone marrow mesenchymal stem cells; Runx2: Runt-related transcription factor 2; Col-1: Collagen type I; OC: Osteocalcin; ALP: Alkaline phosphatase; TG: Triglycerides.
Experimental Animals
Experimental rabbits, clean grade, male or female, 6 months old, weighing 1−1.5 kg, provided by Beihua University Animal Experiment Center. Animal specimens are collected in accordance with the institution’s relevant ethical regulations.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the 13th Five-Year Plan Science and Technology Research Project of Jilin Province Department of Education (JJKH20200066KJ); the Jilin Province Science and Technology Department Project (20230401103YY) and Jilin City key medical and health innovation special fund (20190403193).
Statement of Ethical Approval and Informed Consent
We have completed the animal experimental based on the animal ethical guidelines and same experimental procedures were approved by the committee.
