Abstract
Background
Traditional Chinese Medicine (TCM) has been used in the treatment of anthracycline (ANTs)-induced cardiotoxicity. This meta-analysis aims to evaluate systematically the efficacy and safety of TCM combined with dexrazoxane (DRZ) on cardiotoxicity caused by ANTs.
Materials and Methods
Retrieval of the China National Knowledge Infrastructure (CNKI), WAN FANG DATA, Chinese Biomedical Database (CBM), Cochrane Library, and PubMed was conducted through various methods. The meta-analysis was registered on PROSPERO (Registration ID: CRD42023435324). A randomized controlled trial (RCT) of TCM in combination with DRZ to avert cardiotoxicity caused by ANTs was compiled. The efficacy and safety of TCM Combined with DRZ, the cardiotoxicity induced by ANTs was synthesized and discussed, along with the outcomes of cardiac troponin T (cTnT), cardiac troponin I (cTnI), brain natriuretic peptide (BNP), creatine kinase isoenzymes (CK-MB), creatine kinase (CK), left ventricular ejection fraction (LVEF), electrocardiogram (ECG) abnormalities, and the incidence of adverse reactions. RevMan 5.3 was used for meta-analysis.
Results
A total of 10 RCTs with 746 patients were included. The pooled results showed that TCM combined with DRZ treatment decreased significantly the incidence of ECG abnormalities (RR = 0.47, 95% CI [0.32,0.70], p = 0.0001), the level of cTnT (MD = −7.16, 95% CI [−8.31, −6.02], p<0.001), CK (MD = −14.94, 95% CI [−22.91, −6.97], p<0.001), cTnI (MD = −0.10, 95% CI [−0.14, −0.06], p<0.001), BNP (MD = −33.66, 95% CI [−36.95, −30.37], p<0.001), CK-MB (MD = −5.93, 95% CI [−8.16, −3.70], p<0.001). However, there was no significant change in LVEF in the experimental group compared with the control group (MD = 4.81, 95% CI [−02.99, 12.21], p = 0.23). Moreover, adverse events have no significant difference between the two groups as well (RR = 0.60, 95% CI [0.31, 1.15], p = 0.13).
Conclusion
This meta-analysis revealed TCM combined with DRZ appears to be able to improve the efficacy of cardiotoxicity caused by ANTs, but the included studies were of poor quality, and high-quality RCTs would be preferred for future studies.
Keywords
Introduction
Anthracyclines (ANTs) have been reported as potent anti-tumor drugs capable of inducing cardiotoxicity and heart failure since the 1970s (Middleman et al., 1971). Furthermore, cardiotoxicity induced by ANTs at the end of treatment is usually irreversible (Swain et al., 2003; Yeh & Bickford, 2009). Cardiovascular disease is the primary factor that results in death for malignant tumor survivors, except for recurrence, progression, and metastasis of malignant tumors (Johnson et al., 2016; Okwuosa et al., 2017). Dexrazoxane (DRZ) is the only medicine capable of effectively preventing cardiotoxicity caused by ANTs; however, it has various side effects, including myelosuppression (Langer, 2014; Tebbi et al., 2007). The US Food and Drug Administration approved DRZ limited to women carrying metastatic breast cancer, who should be administered a cumulative dose of more than 300 mg/m2 doxorubicin and be required additional doses to maintain tumor control (Food and Drug Administration, 2020). In recent decades, Traditional Chinese medicine (TCM) has played an important complementary role in the prevention and treatment of chronic cardiotoxicity diseases in China (Jiang et al., 2011). According to the principles of TCM, symptoms such as chest tightness and palpitations resulting from ANTs are related to Qi-Yin deficiency (Yu RC, 1983; Xq, 2003). Zhi-Gan-Cao-Tang is a herbal formula recorded in the Chinese medical classic “Shanghan Lun,” which nourishes Yin-blood, supplements Yang-Qi, and strengthens the heart spirit when used as a complementary medicine to relieve heart failure-related symptoms (Wang et al., 2018). In a randomized controlled trial, Platycodon grandiflorus reduced the incidence of subclinical heart failure and cardiac troponin T (cTnT) levels resulting from ANT-induced cardiotoxicity (Hao et al., 2020). Presently, there are many clinical trials of TCM combined with DRZ for ANT-induced cardiotoxicity that have been reported with positive results. Nevertheless, there is no minutely evaluated evidence or meta-analysis on the potential benefit of TCM combined with DRZ for ANT-induced cardiotoxicity to justify their recommendation for clinical treatment. This study aims to conduct a meta-analysis to evaluate the efficacy of TCM combined with DRZ to treat cardiotoxicity caused by ANTs.
Materials and Methods
The meta-analysis was registered on PROSPERO (Registration ID: CRD42023435324).
Search Strategy
An independent review of the literature was conducted by the China National Knowledge Infrastructure (CNKI), WAN FANG DATA, the Chinese Biomedical Database (CBM), the Cochrane Library, and PubMed. Literature was limited to those in English and Chinese and published from the built library on February 27, 2020. Medical Subject Heading (MeSH) terms were applied in the databases, as were keyword search terms for “Dexrazoxane”[Mesh], “ANTs”[Mesh], and cardiotoxicity.
Study Selection
Pieces of literature were selected by two review authors independently, and then all titles and abstracts were screened to identify the systematic search. The full text of potentially eligible literature with relevant abstracts and titles was retrieved and independently assessed for eligibility according to the set inclusion and exclusion criteria. All excluded studies were recorded, and the reasons for exclusion were properly explained.
Inclusion and exclusion criteria were included if they satisfied the following criteria: (1) The search was also restricted to literature concerning randomized controlled trials (RCTs) and published in English or Chinese; (2) Participants were malignant tumor patients with a history of the first diagnosis; and (3) The intervention measure of the control group complies with the use of ANTs + DRZ, and the experimental group uses TCM + ANTs + DRZ. Studies were excluded if they assessed: (1) patients used other drugs to prevent cardiotoxicity; (2) animal experiments; (3) conference papers; (4) reviews and case reports; (5) unclear outcome indicators; and (6) duplicate literature.
Data Extraction
Two reviewers independently conducted data extraction, utilizing a standardized, pilot-tested form. Disagreements between reviewers were resolved by consensus or by a third reviewer. For the respective RCTs, several data were extracted (e.g., first author, year of publication, country, type of disease, number of cases, interventions, as well as clinical endpoint). The clinical endpoint covers the following parameters: (1) electrocardiogram (ECG) abnormality rate; (2) biochemical marker signal after treatment [e.g., cTnT, cardiac troponin I (cTnI), brain natriuretic peptide (BNP), creatine kinase isoenzymes (CK-MB), creatine kinase (CK), left ventricular ejection fraction (LVEF)]; and (3) adverse events (incidence of myelosuppressive response).
Quality Assessment
The Cochrane Collaboration tool was used for assessing the quality of each included RCT (Higgins et al., 2011). The items included in this tool were random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other biases. Two reviewers assigned a “high,” “low,” or “unclear” risk of bias to each domain, while the third reviewer adjudicated a disagreement.
Statistical Analysis
Microsoft Office Excel 2010 (Microsoft, Redmond, Washington) was employed as the program for data extraction. RevMan 5.3 software was exploited for a meta-analysis. Relative ratios (RR) with 95% confidence intervals (95% CI) were calculated for the ECG abnormality rate and incidence of myelosuppression. Median differences (MD) with 95% CI were used to report continuous data, such as cTnT, CK, cTnI, BNP, CK-MB, and LVEF.
Statistical heterogeneity was quantified using the inconsistency index (I2), where I2 > 50% is considered large heterogeneity, I2 = 25%–50% is moderate heterogeneity, and I2 < 25% is absent of heterogeneity. I2 > 50% or p-value < 0.1 indicates large heterogeneity; a random-effects model was used. Otherwise, a fixed-effects model was applied. The sensitivity analysis is used to evaluate the stability of the results when the heterogeneity is considered statistically significant, and the funnel plot is used to analyze the potential publication bias of the included research.
Results
Search Results
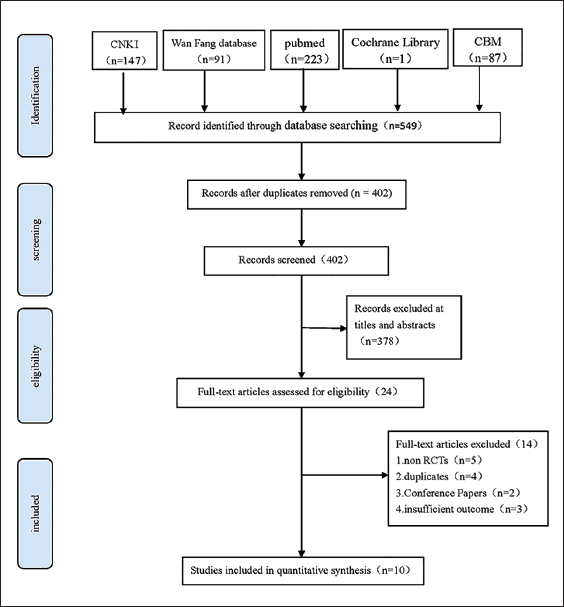
A total of 549 studies were identified from the search in the selected databases. After the duplicates were removed, 402 studies screened the titles and abstracts. Based on the exclusion criteria, 378 citations were ruled out of the database. Lastly, 10 studies were contained after full-text assessment, including 746 patients (i.e., 397 in the experimental group and 349 in the control group) (Figure 1 and Table 1).
PRISMA Flow Diagram Showing the Selection Process and Criteria of the Included Studies.
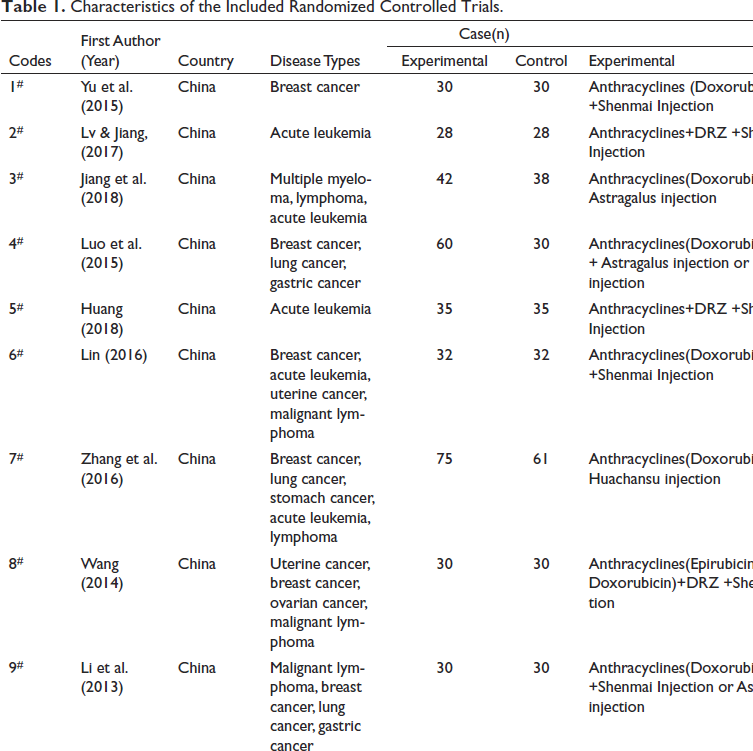
Characteristics of the Included Randomized Controlled Trials.
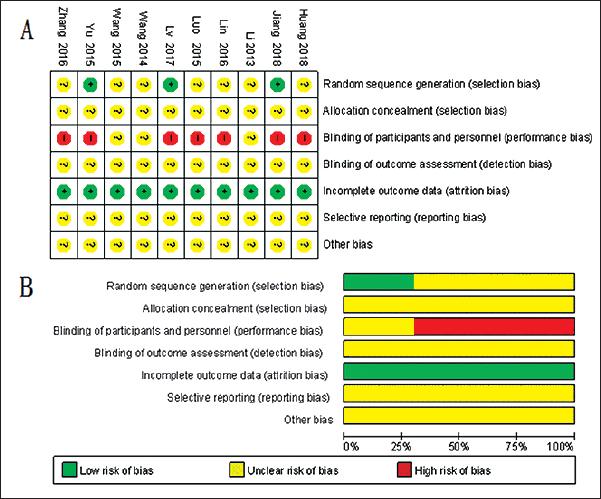
Risk of Bias Assessment
The Cochrane risk of bias tool was exploited to assess the risk of bias in the involved studies. All involved studies mentioned “random,” whereas only three studies (Jiang et al., 2018; Lv & Jiang, 2017; Yu et al., 2015) described the specific randomization methods. None of the involved RCTs reported allocation concealment. Seven studies (Huang, 2018; Jiang et al., 2018; Lin, 2016; Luo et al., 2015; Lv & Jiang, 2017; Yu et al., 2015; Zhang, 2016) did not implement blinding of participants and personnel for ethical approval required, whereas there was unclear blinding of outcome assessment. All studies (Huang, 2018; Jiang et al., 2018; Li et al., 2013; Lin, 2016; Luo et al., 2015; Lv & Jiang, 2017; Wang et al., 2015; X, 2014; Yu et al., 2015; Zhang, 2016) reported incomplete outcome data, whereas unclear risk bias of selective reporting and other biases were reported. However, the vast majority of the included studies had a low risk of bias (Figure 2).
Risk of Bias Summary and Diagram. (A) Risk of Bias Summary: Review Authors’ Judgments about Each Risk of Bias Item for each Included Study. (B) Risk of Bias Graph.
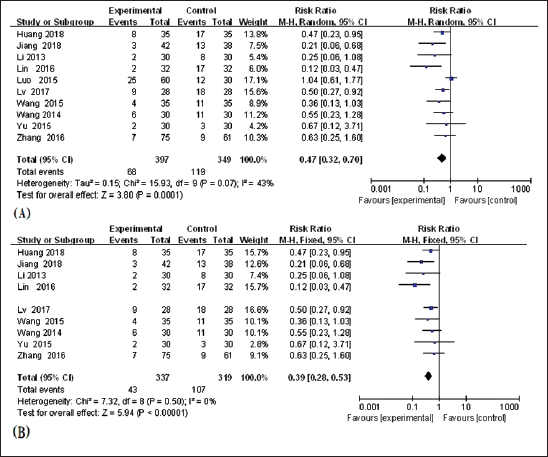
Meta-Analysis of the Outcomes
Incidence of ECG Abnormalities
Ten studies (Huang, 2018; Jiang et al., 2018; Li et al., 2013; Lin, 2016; Luo et al., 2015; Lv & Jiang, 2017; Wang et al., 2015; Wang X, 2014 Yu et al., 2015; Zhang, 2016) reported the incidence of ECG abnormalities, and moderate heterogeneity was identified in the involved studies (I2 = 43%, p = 0.07). A meta-analysis was conducted with a random-effects model. As revealed from the results, the incidence of ECG abnormalities was significantly lower in experimental studies compared to controls (RR = 0.47, 95% CI [0.32, 0.70], p = 0.0001, Figure 3A). Sensitivity analysis was applied to test heterogeneity, and it was found that (Luo et al., 2015) was removed for the analysis of the ECG abnormalities. The heterogeneity had a significant impact on the final results, indicating that the heterogeneity stemmed from Luo’s study after removing the study one by one (I2 = 0%, p = 0.50). After reading the full text, it was found that this may be caused by an asymmetric sample size between the experimental group and the control group. Meta-analysis was conducted with the fixed-effects model, and there was a statistically significant difference between the two groups (RR = 0.39, 95% CI [0.28,0.53], p = 0.0001, Figure 3B), indicating that TCM combined with DRZ in the treatment of cardiotoxicity induced by ANT is more effective than DRZ based on the incidence of ECG abnormalities.
Biochemical Marker Levels
No heterogeneity was identified between the two studies (Huang, 2018; Lv & Jiang, 2017) reporting the cTnT levels after treatment (I2 = 0%, p = 0.79). Compared with the control, the cTnT level of the experimental group decreased significantly with the use of a fixed-effect model (MD = −7.16, 95% CI [−8.31, −6.02], p <0.001, Figure 4A).
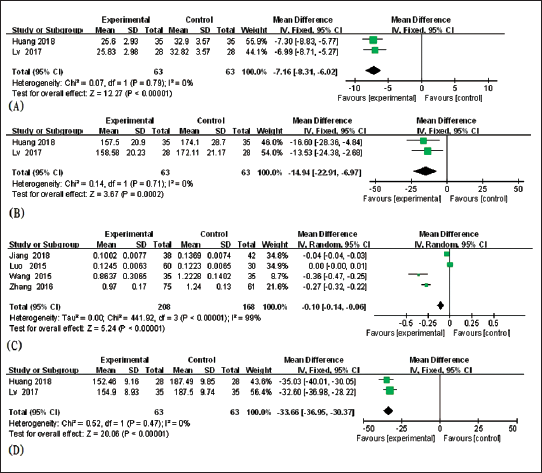
Two pieces of literature (Huang, 2018; Lv & Jiang, 2017) reported the CK levels after treatment, and no heterogeneity was identified (I2 = 0%, p = 0.71). As suggested from the results, the CK level of the control group was higher than that of the control group with a fixed-effects model (MD = −14.94, 95% CI [−22.91, −6.97], p<0.001, Figure 4B).
Large heterogeneity was observed when testing heterogeneity in four pieces of literature (Jiang et al., 2018; Luo et al., 2015; X, 2014; Zhang, 2016) (I2 = 99%, p<0.001). Sensitivity analysis was carried out one by one, and it was found that there was no significant change, thus the random-effects model was used as well. As indicated by the results, the cTnI level of the experimental treatment decreased significantly compared with the control (MD = −0.10, 95% CI [−0.14, −0.06], p<0.001, Figure 4C). After reading the full text, it was found that this may be caused by different indicator units of cTnI, such as the unit in the Jiang et al. (2018) study, which is × 102ng/mL−1, Luo et al. (2015) ng·mL−1(× 10−2), Zhang (2016), and Wang et al. (2015), which is ng·mL−1.
Two studies (Huang, 2018; Lv & Jiang, 2017) reported the level of BNP after treatment and no heterogeneity in each other (I2 = 0%, p = 0.47). A meta-analysis was conducted with a fixed-effects model. As suggested from the results, the BNP level of the experimental group was decreased compared to the control group, and the difference was considered statistically significant (MD = −33.66, 95% CI [−36.95, −30.37], p<0.001, Figure 4D).
Seven studies (Huang, 2018; Jiang et al., 2018; Lin, 2016; Luo et al., 2015; Lv & Jiang, 2017; X, 2014; Zhang, 2016) compared CK-MB after TCM. Combined with DRZ treatment in the control group, heterogeneity analysis demonstrated that there was heterogeneity (I2 = 90%, p <0.001). The random-effects model was used. Compared with the experimental group, the level of CK-MB in the control group decreased significantly (MD = −5.93, 95% CI [−8.16, −3.70], p<0.001) (Figure 5A).
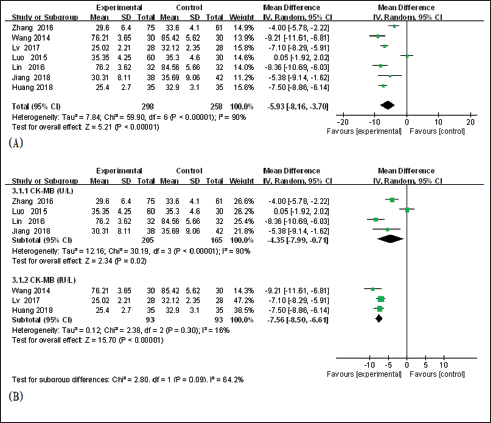
Subgroup analysis based on different indicators of CK-MB levels (IU/L vs. U/L), showed that TCM combined with DRZ could significantly decrease the level of CK-MB more than DRZ alone (Figure 5B). However, group U/L has high heterogeneity (I2 = 90%, p <0.001), but group IU/L has no heterogeneity (I2 = 16%, p = 0.30), which may be induced by the different indicators of CK-MB levels.
Echocardiogram
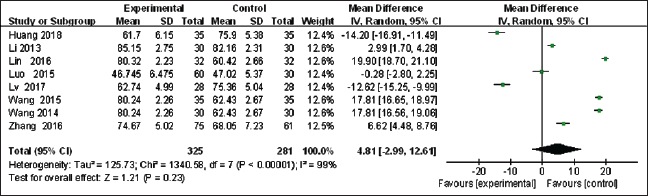
Absent heterogeneity was identified in eight studies (Huang, 2018; Li et al., 2013; Lin, 2016; Luo et al., 2015; Lv & Jiang, 2017; Wang et al., 2015; X, 2014; Zhang, 2016) (I2 = 99%, p<0.0001). Sensitivity analysis was carried out one by one, and it was found that there was no significant change. The random-effects model was adopted for meta-analysis. Meta-analysis demonstrated that there was no significant change in LVEF in the experimental group compared with the control group (MD = 4.81, 95% CI [−2.99, 12.21], p = 0.23, Figure 6).
Adverse Events
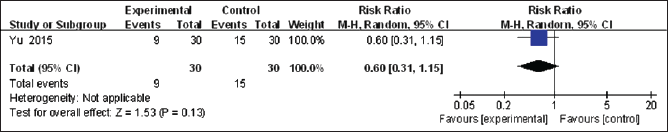
One study (Yu et al., 2015) reported adverse events (the incidence of adverse reactions). A total of 60 patients were included in the study, and there was no significant decrease between the two groups (MD = 0.60, 95% CI [0.31, 1.15], p = 0.13, Figure 7).
Forest Plot of the Incidence of Adverse Reactions.
Publication Bias
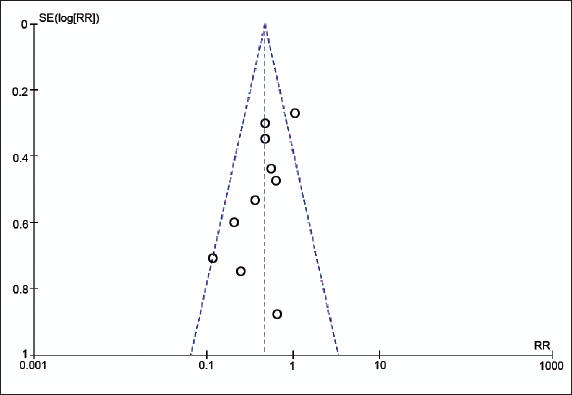
Funnel plots of the 10 studies that reported the incidence of ECG abnormalities were used to assess publication bias (Figure 8). The funnel plot, which was asymmetrically distributed on both sides, suggested that there was a potential publication bias in the included studies. The heterogeneity probably stemmed from the different sample sizes and timing of the research.
Discussion
Cardiotoxicity, particularly heart failure, is the leading cause of morbidity and mortality in cancer survivors and is related to the overall dose of ANTs administered (Sallustio & Boddy, 2021; Zhang, 2016). Fogarassy et al. (2020) analyzed the risk factors of doxorubicin-associated heart failure via a retrospective study of integrated Hungarian databases, and the results indicated that a cumulative dose of doxorubicin of more than 400 mg/m2 increased the risk of heart failure among the 3,288 patients treated with doxorubicin. The ANTs with a mean cumulative dose of 228.2 ± 42.3 mg/m2 also induced cardiotoxicity in pediatric patients with acute lymphoblastic leukemia after ending therapy at 12.6 ± 4.3 years (Amigoni et al., 2010). The LVEF (58 ± 1% to 53 ± 1%) decreased within 6 months even with low to moderate doses of ANTs, which were associated with the early development of subclinical abnormalities of cardiac and vascular functioning (Drafts et al., 2013). Thus, pharmacological interventions should be performed in advance to minimize the cardiotoxic effects of ANTs on patients.
This study systematically reviewed the efficacy and safety of TCM combined with DRZ on cardiotoxicity induced by ANTs. A total of 10 studies have been adopted, including Shenmai injection, Astragalus injection, and Huachansu (cinobufagin) injection. In TCM theory, Shenmai injection contains extracts of the traditional Chinese remedies Ginseng Rubra (Araliaceae) and Ophiopogon japonicus (Liliaceae), where G. Rubra reinforces Qi and prostration while O. japonicus replenishes Yin and promotes the production of fluids, complementing each other, which is beneficial for Qi-Yin deficiency and coronary heart disease (Yu et al., 2015). Astragalus, also known as Huangqi in Chinese, is a perennial herbaceous plant of the Leguminosae family that can supplement Qi and nourish Yin, as well as strengthen healthy Qi to eliminate pathogens. Huachansu, one of the most widely studied traditional Chinese medicines, is a water-soluble extract from Bufo toad skin that can remove blood stasis and lumps. Both Astragalus and Huachansu can prevent Qi-Yin deficiency caused by the cardiotoxicity of ANTs (Jiang et al., 2018; Zhang, 2016). TCM combined with DRZ is a likely and novel treatment for cardiotoxicity induced by ANTs.
According to the theory of Western medicine, almost 30% of patients had cardiotoxicity after ANT treatment, which is characterized by ECG abnormalities, including tachycardia, atypical ST changes, and badly compromising cancer treatment (Murabito et al., 2020). Furthermore, numerous studies (Cardinale et al., 2020; Feng & Yang, 2015; Hachey, 2017; Herrmann et al., 2014; Lenihan et al., 2016; Mastro et al., 2015) found that the levels of cTnT, cTnI, BNP, CK-MB, and CK significantly increased and the LVEF decreased conversely when cardiac function injury appeared. LVEF, one of the indexes evaluating cardiac function, has a 1-year cardiotoxicity risk of 6.8% for those without an early LVEF decline and a baseline LVEF ≥60%, a 15.7% risk for those without an early LVEF decline and a baseline LVEF < 60%, and a 66.7% risk for those with an early LVEF decline and a baseline LVEF < 60% (Posch et al., 2019). In this meta-analysis, the pooled results showed that the incidence of ECG abnormalities, cTnT, cTnI, BNP, CK-MB, and CK levels of patients administered DRZ with TCM significantly decreased compared with those administered DRZ alone. However, there was no significant change in LVEF or adverse events in the experimental group compared with the control group. The meta-analysis result shows that TCM combined with DRZ has advantages in the efficacy of treating the cardiotoxicity caused by ANT chemotherapy; in the meantime, there is not sufficient evidence to prove it is safe.
Our research has several limitations. First, there was a lack of large, multicenter, standardized RCTs, probably leading to a risk of bias from incomplete data that could constrain the reliability of our results. Second, all the included studies were performed and published in China, which limited the evaluation of the reliability of the conclusion when this integrated therapy was applied to other populations. Third, only one piece of literature reported the incidence of adverse reactions, and this lack of reporting affected the limitations and reliability of the results of the TCM safety evaluation. Fourth, the disease type, time of the research, indicator units, and sample size were not consistent among the included studies, which could result in large heterogeneity in this meta-analysis, which should be more concerned with in future research.
Conclusion
As revealed from the results of the meta-analysis, TCM combined with DRZ appears to be able to improve the efficacy of cardiotoxicity caused by ANTs, but in the meantime, there is not sufficient evidence to prove it is safe. Nevertheless, the included studies were of generally poor quality and limited the evaluation of the reliability of the conclusion. A multicenter, larger sample, especially one containing safety assessments and high-quality RCTs, is needed in future research to provide more reliable evidence for clinical treatment.
Abbreviations
TCM: Traditional Chinese medicine; ANT: Anthracycline; DRZ: Dexrazoxane; CNKI: China National Knowledge Infrastructure; CBM: Chinese Biomedical Database; MeSH: Medical subject heading; cTnT: cardiac troponin T; cTnI: cardiac troponin I; BNP: Brain natriuretic peptide; CK-MB: Creatine kinase isoenzymes; CK: Creatine kinase; LVEF: Left ventricular ejection fraction; RR: Relative ratios; ECG: Electrocardiogram; IV: Inverse variance; MD: Mean difference; SMD: Standardized mean difference; CI: Confidence intervals; I2: Inconsistency index.
Footnotes
Acknowledgments
The authors appreciate Musi Liu and Muxiang Liu for their support in this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The research was funded by the self-financing scientific research project of the Guangxi Province Health and Health Committee (No. Z20180156), the Natural Science Foundation Project of Guangxi Province (No. 2018GXNSFAA281056), and the self-financing scientific research project of the Administration of Traditional Chinese Medicine of Guangxi Province (No. GXZYJ20220628).
