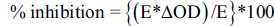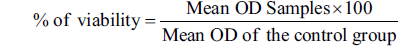Abstract
Background
Alzheimer’s disease (AD) is considered to be the most common form of dementia. The drugs that are available for the treatment of AD provide only partial improvement of the condition. In recent years, natural ingredients have received increased attention in generating novel medications for treating various neurological disorders, including AD. Vigna radiata and Vigna pilosa are plants that are routinely included in many Ayurvedic formulations used to manage memory impairment.
Objectives
The present study was conducted to evaluate the in vitro anticholinesterase and neuroprotective activities of the plants V. radiata and V. pilosa.
Materials and Methods
A modified 96-well microplate assay according to Ellman’s method was employed to measure acetyl choline esterase (AChE) inhibitory activity. In vitro neuroprotective effect was measured by the 3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide (MTT) assay using the SHSY-5Y (neuroblastoma cells) cell line against β-amyloid induced neurotoxicity. The ethyl acetate extract of V. radiata and the ethanolic extract of V. pilosa, which showed maximum AChE inhibitory activity, were selected for the MTT assay. The cell viability was evaluated by direct observation of cells with the help of an inverted phase contrast microscope, followed by the MTT assay method.
Results
Maximum AChE inhibitory activity was exhibited by an ethyl acetate extract of V. radiata. It showed AChE activity with a half-maximal inhibitory concentration (IC50) value of 286.40 µg/mL, while the ethanol extract of V. pilosa showed an IC50 value of 160.19 µg/mL. Extracts of both plants, V. radiata and V. pilosa, significantly (p ≤ 0.001) improved the percentage of cell viability in a dose-dependent manner.
Conclusion
The plants V. radiata and V. pilosa showed potent anti-Alzheimer activity when tested using in vitro AChE inhibitory activity and by MTT assay using an β-amyloid-induced in vivo cytotoxicity model. These findings demand the need for further studies using in vivo models.
Introduction
The most prevalent type of dementia is Alzheimer’s disease (AD), a neurodegenerative condition that was initially identified by German psychiatrist Alois Alzheimer in 1907. AD is characterized by three primary neuropathological symptoms: senile plaques, neurofibrillary tangles (NFTs), and neuronal death (Razgonova et al., 2019). Extracellular amyloid-protein A and apolipoprotein E deposits are seen in senile plaques. NFTs are apolipoprotein E and hyperphosphorylated protein aggregates that are found within and outside neurons (Masondo et al., 2019). The loss of intellectual capacity enough to hinder the social or professional activities of an individual is the main feature of dementia (Donev et al., 2009). Only a few medicines are available to treat AD. By elevating neurotransmitter molecules, these medications temporarily reduce AD symptoms. These medicines have several disadvantages, including poor bioavailability, high cost, and a need for periodic blood testing (Tuzimski & Petruczynik, 2022). Some of the medications are hepatotoxic and increase the risk of urinary incontinence and bradycardia, as well as other cardiovascular complications, weight loss, and pulmonary disorders (Ferreira et al., 2020). In recent years, natural ingredients have received increased attention for generating novel medications for treating various illnesses, including AD (Ali-Shtayeh et al., 2014; Poliseno et al., 2021). Traditional medicinal herbs have been used as a source of bioactive components and as the foundation for the development of novel drugs. Plants have been employed in ancient medical practices to improve cognitive performance and lessen symptoms of dementia (Tuzimski & Petruczynik, 2022).
Vigna radiata and Vigna pilosa are plants belonging to the Fabaceae family. V. radiata is a low-altitude crop and a climbing herb that grows from sea level to about 2000 m above sea level. V. pilosa is a climbing herb, 1–3 m long, with tender branches. V. radiata has been reported to have antidiabetic (Amare et al., 2022) and antiviral (Lo et al., 2020) activities. These plants are routinely included in many Ayurvedic formulations used for the treatment of central nervous system disorders, including memory impairment (Dinesh, 2009; Ministry of Health and Family Welfare, 2000; Vagbhatta, 2011). But, from the literature review, it was evident that there is a scarcity of research publications that establish the neuroprotective activity of these plants. Hence, this study aims to evaluate the possible in vitro anti-Alzheimer’s activity of the plants V. radiata and V. pilosa. The methods to be employed are in vitro acetyl choline esterase (AChE) inhibitory activity evaluation and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay using the SHSY-5Y neuroblastoma cell lines.
Materials and Methods
Plant Materials
The whole plants of V. radiata and V. pilosa belonging to the family Fabaceae were collected in December 2021 from Varam village, Kannur district, Kerala. The plants were authenticated by Dr Sreelakshmi. P, Taxonomist, M/s. Arogya Medicare, a GMP-certified Ayurvedic drug manufacturing company based in Kannur, Kerala. The voucher specimens of the plants were deposited at the herbarium (AHAMS 172 dated 21/12/2021 and AHAMS196 dated 21/12/2021). The taxonomic authentication was done using morphological and molecular methods.
Instruments
Dulbecco’s Modified Eagles medium (DMEM) (Sigma Aldrich, USA D5648), fetal bovine serum (FBS, Gibco, US origin), micropipettes, phase contrast microscope with Optika Pro 5 Camera, CO2 incubator (Eppendorf), culture plates and flasks (Thermo Scientific), MTT (Sigma Aldrich M5655), ELISA Reader.
Chemicals
5,5′-Dithiobis-(2-nitrobenzoic acid) (DNTB, Ellman’s reagent), 50 Mm Tris-HCl (pH 8.0), 0.1 M NaCl, 0.02 M MgCl2, acetylthiocholine iodide (ATCI), AChE, 0.25% trypsin, bis(trimethylsilyl)acetamide (BSA), and FBS.
Preparation of Extract
The whole plants were shade-dried for 2 weeks, ground, sieved through sieve number 22, and then subjected to successive extraction using the solvents hexane, chloroform, ethyl acetate, ethanol, and water. A rotary flash evaporator was employed to concentrate the extracts under reduced pressure. The resulting residue is kept in a desiccator for further use.
Phytochemical Screening
For the qualitative detection of phytoconstituents present, different extracts of the plants V. radiata and V. pilosa were subjected to evaluation for phytochemical constituents. Tests to determine the presence of alkaloids, glycosides, tannins, saponins, steroids, and triterpenes were conducted (Harborne 1984; Trease & Evans, 2002).
Evaluation of AChE Inhibitory Action
The serine hydrolase enzyme, AChE, hydrolyzes acetylcholine into acetic acid and choline. A modified version of Ellman’s assay, using a 96-well microplate, was employed to assess the AChE inhibitory activity. The enzyme hydrolyzes the substrate, acetylthiocholine to thiocholine, which combines with Ellman’s reagent to form 2-nitrobenzoate-5-mercaptothiocholine and 5-thio-2-nitrobenzoate. The absorbance was measured at 412 nm. A buffer of 50 mM Tris–HCl, with pH 8.0 was used for the assay. AChE from Electrophorus electricus (electric eel) was utilized in this experiment; it was a Type VI-S lyophilized powder with 222 U/mg solid and 268 U/mg protein from Sigma Aldrich. The 222 U/mL enzyme stock solution was maintained at −80°C. Enzyme dilution was carried out in a buffer containing 0.1% BSA. This buffer, which contains 0.02 MgCl2 and 0.1 M NaCl, was used to dissolve DNTB.
Deionized water was used to dissolve ATCI; 100 µL of 3 mM DNTB, 20 µL of 0.26 U/mL of AChE, and 40 µL of buffer (50 mM Tris pH 8.0); 20 µL of each extract of the plants V. radiata and V. pilosa (25, 50, 100, 200, and 400 µg/mL) were dissolved in buffer and then added to a 96-well plate. Proper mixing was done, and then the plate was subjected to incubation for 15 min at 25°C. The absorbance was measured at 412 nm. The reaction was then started by the addition of 20 µL of ATCI. After mixing, absorbance was measured at two time intervals (5 and 20 min) using a double-beam UV-vis spectrophotometer (Ingkaninan et al., 2003; Mathew & Subramanian, 2014).
The percentage inhibition of AChE was assessed by the following formula:
Calculations were done using ED50 PLUS V 1.0 Software.
In vitro Neuroprotective Activity
Neuroblastoma cell lines were purchased from the National Centre for Cell Science (NCCS) in Pune and cultured in DMEM. The cell line was grown in a 25-cm tissue culture flask with an antibiotic solution containing the following antibiotics: penicillin (100 U/mL), streptomycin (100 g/mL), and amphotericin B (2.5 mg/mL). The DMEM was supplemented with 10% FBS,
Cytotoxicity Assay by Direct Microscopic Observation
With an inverted phase contrast tissue culture microscope (Olympus CKX41 with an Optika Pro5 CCD camera), the entire plate was viewed every 24 h for up to 72 h, and microscopic observations were captured as photographs. Morphological alterations of the cells, including rounding, granulation, shrinking, and vacuolization in the cytoplasm, were all considered indications of cytotoxicity.
Cytotoxicity Assay by MTT Method
In 3 mL of PBS, 15 mg of MTT (Sigma, M-5655) was reconstituted and well dissolved before being filter sterilized. After 24-h incubation, the sample content in the wells was removed, and 30 µL of reconstituted MTT solution was added to each test and cell control well. The plate was gently shaken, and the incubation period was extended by 4 h at 37°C in a humidified 5% CO2 incubator. Immediately after the incubation, the supernatant was taken out, and 100 µL of MTT solubilization solution (DMSO) was added. After that, the wells were subjected to gentle agitation by pipetting up and down to solubilize the formazan crystals. Absorbance was measured at a wavelength of 540 nm. The absorbance values were further calculated with a microplate reader (Talarico et al., 2004).
The growth inhibition (percentage) was calculated using the formula:
Statistical Analysis
All values are expressed as the mean ± standard deviation. The significance level was analyzed by one-way ANOVA with Dunnett’s post hoc test performed using GraphPad Prism version 5.00 for Windows (Graph Pad Software, San Diego, California, USA).
Results
The percentage yield of extracts for V. radiata is as follows: ethanol 3.06%, ethyl acetate 0.70%, water 3.82%, hexane 0.37%, and chloroform 0.78%. V. pilosa: ethanol 4.08%, ethyl acetate 0.72%, water 3.76%, hexane 0.70%, and chloroform 1.11%.
A preliminary phytochemical examination revealed a high content of alkaloids in the hexane and ethyl acetate extracts of V. radiata. While the ethanol and chloroform extracts showed a moderate content of alkaloids. Flavonoid content was high in the water and chloroform extracts and moderate in the ethyl acetate extract. Various extracts also showed the presence of other constituents, including phenols, saponins, steroids, tannins, and triterpenes. Ethyl acetate, ethanol, and chloroform extracts of V. pilosa showed a high content of alkaloids. A high flavonoid content was observed in the ethanol extract. Phenolic content was high in ethanol and chloroform extract. Other phytoconstituents, including glycosides, saponins, steroids, tannins, and triterpenes, were also present in different extracts.
AChE Inhibition
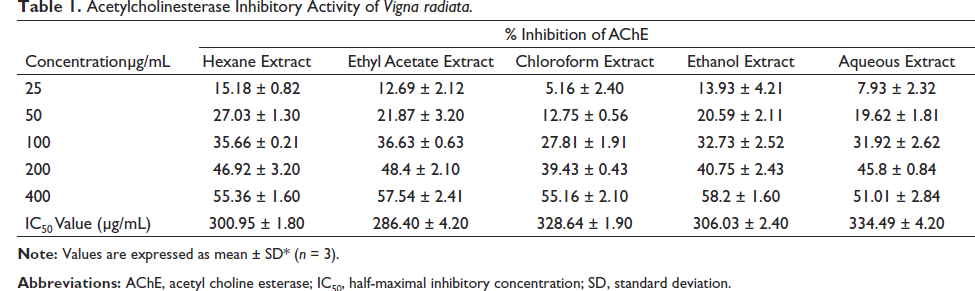
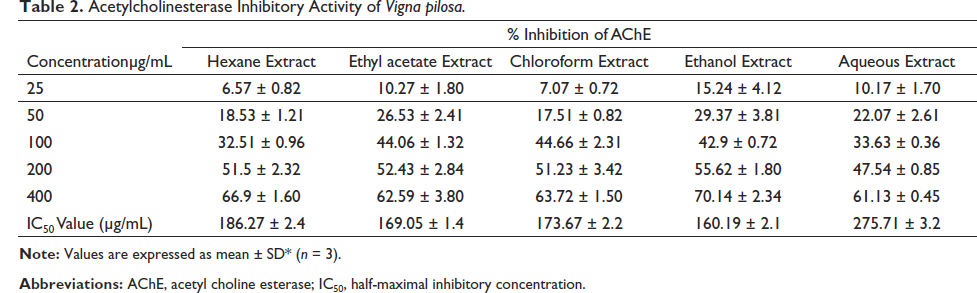
AChE inhibitory activity of various extracts was estimated by a modified 96-well microplate assay based on Ellman’s method. The hexane, chloroform, water, ethyl acetate, and ethanolic extracts of V. radiata and V. pilosa were subjected to the assay. Table 1 depicts the AChE inhibitory effect of various extracts of V. radiata at concentrations of 25, 50, 100, 200, and 400 µg/mL. Ethyl acetate extract of the plant V. radiata exhibited maximum activity with a half-maximal inhibitory concentration (IC50) value of 286.40 µg/mL. Table 2 shows the AChE inhibitory activity of different extracts of V. pilosa at concentrations of 25, 50, 100, 200, and 400 µg/mL. The ethanolic extract of V. pilosa showed maximum inhibitory action with an IC50 value of 160.19 µg/mL. So, these extracts were selected for evaluation of neuroprotective activity by MTT assay using SHSY-5Y neuroblastoma cell lines. The use of SH-SY5Y cells has been included in the investigation of Parkinson’s disease, AD, and the pathogenesis of viral infections (Agholme et al., 2010; Shipley et al., 2016; Xie et al., 2010).
Acetylcholinesterase Inhibitory Activity of Vigna radiata.
Acetylcholinesterase Inhibitory Activity of Vigna pilosa.
Neuroprotective Activity
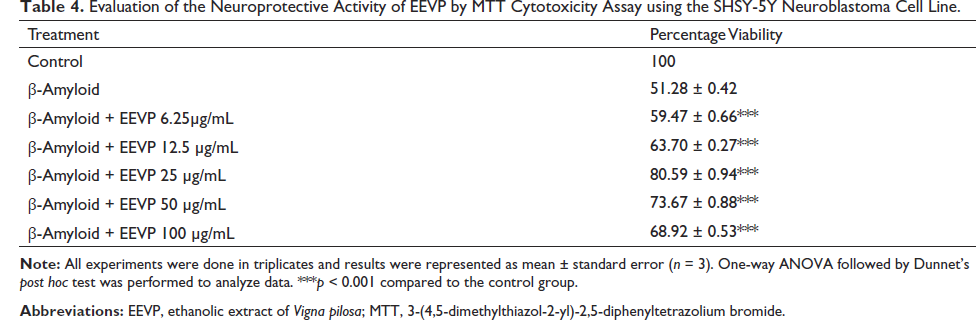
The neuroprotective activity of the selected extracts of V. radiata and V. pilosa was evaluated by MTT cytotoxicity assay using beta-amyloid-induced cytotoxicity on the SHSY-5Y neuroblastoma cell line. Table 3 shows the effect of different concentrations of ethyl acetate extract of V. radiata. The concentrations of extracts used were 6.25, 12.5, 25, 50, and 100 µg/mL. The percentage viability of the cells showed a significant increase (p < 0.001) in comparison with the control. It was observed that the viability was 77.27 ± 1.49 for the ethyl acetate extract of V. radiata, and with further increases in the concentration of the extract, the cell viability was found to be decreasing. Table 4 depicts the effect of the ethanolic extract of V. pilosa on cell viability. Here also, the percentage viability of the cells was significantly increased (p < 0.001) compared to the control. The extract exhibited maximum activity at a dose of 25 µg/mL, with a cell viability of 80.59 ± 0.94. With further increases in the concentration, the cell viability was found to be decreasing.
Evaluation of the Neuroprotective Activity of EAEVR by MTT Cytotoxicity Assay using the SHSY-5Y Neuroblastoma Cell Line.

***p < 0.001 compared to the control group.
Evaluation of the Neuroprotective Activity of EEVP by MTT Cytotoxicity Assay using the SHSY-5Y Neuroblastoma Cell Line.
Cytotoxicity Assay by Microscopical Observation
In direct microscopic observation, cell morphological alterations such as rounding or shrinking, granulation, and vacuolization in the cytoplasm were all considered indications of cytotoxicity as a result of the addition of beta-amyloid. Figure 1 shows the microscopy of SHSY-5Y cell lines after treatment with ethyl acetate extract of V. radiata at concentrations of 6.25, 12.5, 25, 50, and 100 µg/mL. It is evident that the cytotoxicity was a result of the addition of an ethyl acetate extract of V. radiata.
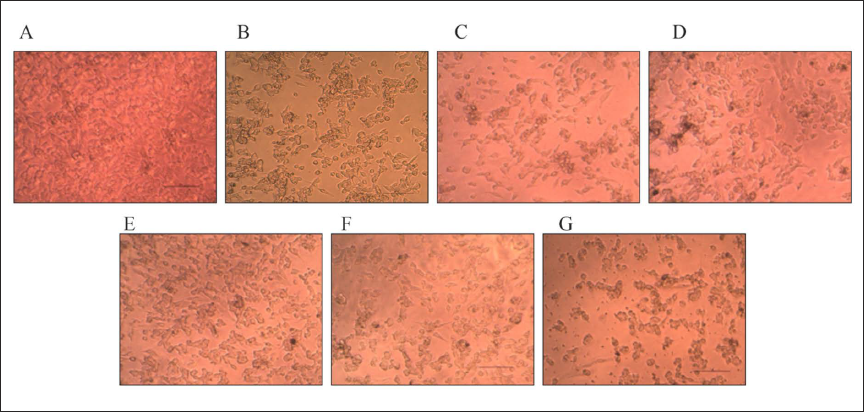
Figure 2 displays the microscopy of SHSY-5Y cell lines after treatment with an ethanolic extract of V. pilosa at concentrations of 6.25, 12.5, 25, 50, and 100 µg/mL. The indicators of cytotoxicity were less prominent in the microscopic examination, indicating reduced cytotoxicity.

Discussion
AD is a disorder due to neurodegeneration that mainly affects old people and is characterized by early cognitive impairment and later social and behavioral decline (Jiang et al., 2018). The development of neurodegeneration is influenced by the formation of free radicals, reactive oxygen species, and oxidative damage (Dumont et al., 2010; Feng & Wang, 2012). Amyloid beta peptide (Aβ25−35) addition produces an accumulation of this peptide within and outside the neurons that causes the release of mediators of inflammation, including interleukin-6 (IL-6), interleukin-1β (IL-1β), and tumor necrosis factor-α (TNF-α). This mediates an inflammatory response that leads to neuronal damage (Park et al., 2021).
Many international health organizations have advocated a range of plant-based functional foods in order to improve health, reduce risk, or prevent certain chronic diseases (Hou et al., 2018; Kumar et al., 2018). The mung bean (Vigna) contains many nutrients and bioactive compounds, especially polyphenols, polysaccharides, and polypeptides (Kabir et al., 1998). It has many health benefits, including hypoglycemic and hypolipidemic activities (Lopes et al., 2018), hepatoprotective (Xie et al., 2019), antihypertensive (Li et al., 2006), anticancer (Hafidh et al., 2012), and immunomodulatory (Dai et al., 2014) activities.
The development of AChE inhibitors for the treatment of AD was based on the discovery that cholinergic pathways in the cerebral cortex and basal forebrain are significantly compromised in AD, and the cholinergic deficit caused by this contributes to the impairment of cognitive functions (McGleenon et al., 1999).
Medicinal plants, which are used traditionally, have always been considered a base for new drug research. Many natural products from plants with neuroprotective activity, such as alkaloids, terpenoids, and phenolic compounds, are currently considered useful in the treatment of many neurodegenerative diseases, such as AD and Parkinson’s disease. The use of herbal medicine for the management of symptoms of dementia and cognitive impairment has a long history, especially in East Asian countries. A lot of products of herbal origin have routinely been tested and used in the treatment of AD, with varying clinical responses (Santos-Neto et al., 2006, Lee et al., 2020).
This study aimed to examine the in vitro anti-Alzheimer’s activity of the plants V. radiata and V. pilosa belonging to the family Fabaceae. In vitro AChE inhibitory activities of various extracts of V. radiata and V. pilosa were estimated. Various extracts of the plants exhibited potent AChE inhibitory activity. The extracts that showed maximum activity (ethyl acetate extract of V. radiata and ethanolic extract of V. pilosa) were selected for in vitro neuroprotective activity evaluation by MTT assay. The results of the study showed significant neuroprotection against amyloid beta-induced neurotoxicity in SHSY-5Y neuroblastoma cells.
The phytochemical evaluation of the plant extracts revealed a high content of flavonoids. It has been well established that flavonoids are exogenous antioxidants, and they are effective in reducing the formation of reactive species by different mechanisms, including the inhibition of enzymes such as nitric oxide synthase and xanthine oxide synthase or by regulating ion channels (Al-Khayri et al., 2022). They are also involved in the modulation of certain enzymes, which are likely to be involved in oxidative processes. Flavonoids exert multiple neuroprotective actions within the brain, including protecting neurons against injury induced by certain neurotoxins, reducing neuroinflammation, and improving cognitive function (Ayaz et al., 2019; Vauzour et al., 2008). The flavonoid content of the plants could have contributed to the neuroprotective activity of the plants in the MTT assay, probably by reducing the inflammatory response.
The peptides obtained from mung bean (V. radiata) were reported to have potent antioxidant activity when tested using ferrous ion chelating activity (1400–1500 µg EDTA equivalent/mL). The chelation of ferrous ions was enzyme- and hydrolysis time-dependent (Kusumah et al., 2020). The antioxidant potency of the plants could be a contributing factor to the neuroprotective activity that they have exhibited. Since oxidative stress is a major cause of neuroinflammation, it is rational to use antioxidants to manage neuroinflammation and the resulting neurodegeneration. The results of this study strongly support the beneficial effects of the plants V. radiata and V. pilosa in the management of AD. But to confirm it, more advanced studies are needed.
Conclusion
The plants V. radiata and V. pilosa are included in many Ayurvedic medicaments that are routinely employed in the management of memory-related problems. These plants were subjected to successive extractions, and the extracts were screened for in vitro AChE inhibitory activity. Ethyl acetate extracts of V. radiata and ethanolic extracts of V. pilosa showed maximum activity. Hence, these extracts were selected for in vitro neuroprotective activity against beta-amyloid-induced neurotoxicity using SHY neuroblastoma cell lines. Both of these extracts were found to have improved the percentage viability of the cells, indicating significant neuroprotective activity. The findings of this study demand further studies on the neuroprotective potency of these plants.
Footnotes
Acknowledgments
The authors are thankful to the management of Al Shifa College of Pharmacy, Malappuram, Kerala, for providing the facilities for the study and to the Centre for Molecular Biology and Applied Science, Thiruvananthapuram, for their support and guidance for cell line studies.
Abbreviations
AD: alzheimer’s disease; AChE: acetyl choline esterase; ATCI: acetyl thio choline iodide; BSA: bis (trimethylsilyl) acetamide; DTNB-5,5′: dithiobis-(2-nitrobenzoic acid); DMEM: dulbecco’s modified eagles medium; FBS: fetal bovine serum; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.