Abstract
Background
Astragalus membranaceus (Fisch.) Bge., as a Traditional Chinese Medicine, not only has ornamental and medicinal value but also plays a role in the effect of sand fixation. At present, the resources of wild A. membranaceus are exhausted with the deterioration of the ecological environment and the increase in market demand. Moreover, there are no excellent varieties for the artificial cultivation of A. membranaceus to meet the market demand.
Objectives
To establish a method to create an efficient induction of tetraploids by colchicine technique in A. membranaceus.
Materials and Methods
The germinating seeds were soaked in different concentrations of colchicine solution and exposure times to induce tetraploids in A. membranaceus, and the induced plants were identified by morphological characteristics comparison and root-tip chromosome determination. The contents of polysaccharides, saponins, and flavonoids and the activity of superoxide dismutase (SOD), peroxidase (POD), and catalase (CAT) were measured in diploid and tetraploid plants.
Results
The results showed that the highest tetraploid induction ratio of germinating seeds in A. membranaceus is obtained from the 0.2% (w/v) colchicine treatment for 36 h, and the chromosome number of tetraploid seedlings was 2n = 4x = 32. The morphological traits and anatomical structure characteristics of leaves from tetraploid plants exhibited significant differences from the diploid plants. The contents of polysaccharides, saponins, and flavonoids and the activity of SOD, POD, and CAT are also increased in tetraploid plants.
Conclusion
In summary, our study provided an effective method to induce A. membranaceus polyploidy, which could be used to develop new germplasm innovations for improving the medicinal value of A. membranaceus.
Introduction
Astragalus membranaceus (Fisch.) Bge., a member of the genus Astragalus, is a perennial herbal plant mainly produced in Inner Mongolia, Shanxi, Heilongjiang, and other places in China (Li et al., 2014). Previous studies (Huang et al., 2015; Zhang et al., 2019) have shown that polysaccharides, saponins, and flavonoids are the main active ingredients in Astragalus. As a Traditional Chinese Medicinal material, A. membranaceus has the functions of enhancing immunity, protecting the liver, and acting as a diuretic, antiaging, and antihypertensive agent (Balakrishnan et al., 2021; Block & Mead, 2003; Fu et al., 2014). Normally, the roots of mature Astragalus are used as medicine, and the stems and leaves are discarded after excavation. However, the yield of the stems and leaves of Astragalus is several times that of the roots, which causes a serious waste of many resources. In addition, the resources of wild A. membranaceus are gradually being reduced with the deterioration of the ecological environment, and the phytochemicals of cultivated A. membranaceus are too low to limit utilization. Therefore, new germplasm resources with high medical components need to be created to meet market demand.
Polyploidy, which has an occurrence rate in the range of 30%–70% in angiosperms (Wolfe, 2001), is responsible for the generation of new species during plant evolution. Polyploids usually possess tolerance to environmental stress and characteristics different from diploids, such as larger leaves, stems, roots, and flowers, because their cells contain more than two complete sets of chromosomes (Sattler et al., 2016). The creation of polyploid germplasm is often considered an effective strategy to improve plant resistance, yield, and components (Cheng & Korban 2011). At present, polyploidy induction with chemical mutagens is the most effective and economical method. Among the more than 200 chemical reagents that induce polyploidy in plants, the colchicine application to increase the number of chromosomes has been successfully employed in many plant species. For example, chromosome doubling was induced by colchicines in Populus hopeiensis (Wu et al., 2022) and Bletilla striata (Thunb.) Reichb. F (Li et al., 2018). Several medicinal plant polyploids have been successfully induced, resulting in varieties with increased biomass or higher content of phytochemicals (Gao et al., 2002; Huang et al., 2008; Mishra et al., 2010; Wei et al., 2018; Yan et al., 2016). On this basis, we speculate that the yield and phytochemical content were also improved in the polyploidy of A. membranaceus. However, there is no report on polyploid induction of A. membranaceus. Therefore, it is necessary to establish a method for generating polyploidy, which should help to solve the problems in A. membranaceus production.
In this work, a method for inducing polyploids of A. membranaceus using colchicine was established by the observation of the influences of different colchicine concentrations and treatment times. Tetraploids of A. membranaceus were identified by the morphological traits and leaf structure characteristics and root-tip chromosome determination. Additionally, the contents of polysaccharides, saponins, and flavonoids and the activity of SOD, POD, and CAT in A. membranaceus tetraploids were higher than those in diploids. The results provide theoretical support for creating new germplasm and improving the main contents of the main phytochemicals of A. membranaceus.
Materials and Methods
Plant Material
Mature seeds were collected from the fruit of A. membranaceus (Fisch.) Bge. (2x = 16), which was cultured in the Chabei Management Area, Zhangjiakou City, China.
Seed Treatment and Germination Test
Three methods were used to treat seeds from broken capsules of A. membranaceus before germination. First, the seeds were submerged in boiling water and stirred with a glass rod for approximately 2 min, and then, the cold water was added to lower the water temperature to approximately 40 for 5 h. Second, the seeds were soaked in 90% sulfuric acid solution for 2 min, and then immediately rinsed in sterile water five times for 3 min each time. Third, the seeds were ground with abrasive paper until the seed coat was white.
Three hundred seeds from each treatment were randomized into three groups and placed in a petri dish with filter paper in a 25 incubator (3,000 lux). The humidity of the Petri dish was maintained, and the seed numbers of germination in each Petri dish were counted for 7 days. The average value of the three groups was taken to count the germination potential, germination rate, and germination index.
Colchicine Treatment for Tetraploid Induction
Further, 2,160 seeds germinating with a radicle length of approximately 0.5 cm were randomly and evenly divided into 24 groups and each group has 90 seeds. Then, 24 groups were evenly divided into six parts and submerged in one of six colchicine concentrations (0, 0.1, 0.2, 0.3, 0.4, and 0.5% w/v), respectively. The changes in roots were observed at 12, 24, 36, or 48 h after treatment to select the optimum treatment time and concentrations of polyploidy induction. The swollen root tips were used for the preliminary identification of a mutagenic polyploid. All seeds of swollen roots were then transferred to culture bowls with vermiculite and cultured in an illuminated incubator under a 16-h photoperiod of 5,000 lux light intensity at 25°C. The lateral roots of all the surviving materials were used to identify chromosomes.
Chromosome Determination
Lateral root tips about 0.5 cm in length were cut from the seedlings, soaked in 0.1% colchicine at 25°C for 4 h, washed with distilled water, and fixed with Carnoy’s solution (containing 3:1 ethanol and glacial acetic acid) at 4°C for 24 h. The fixed root tips were hydrolyzed in 1N HCl for 10–15 min at 65°C and rinsed with tap water for 10 min. These treated root tips were placed on a slide, chopped up, and stained with one drop of improved carbol-fuchsin solution (1.8 g of sorbitol dissolved in 10 mL of carbol-fuchsin and then mixed with 90 mL of 45% v/v acetic acid) for 1–2 min, squashed under cover glass, and observed for chromosome numbers under a microscope (Leica DM2000).
Analysis of Morphological and Stomatal Characteristics
The morphological traits of diploid and tetraploid plants including the plant growth state and hypocotyl and leaf morphology, were observed. For stomatal analysis, approximately 0.1 cm2 on the lower epidermis was torn from leaves of tetraploids and diploids obtained from the same internode (Leaf no. 4 or 5 from the top of the shoot, fifth internode), respectively, using the nail polish smear and tear method. Then these epidermal layers were spread onto a glass microscope slide with one drop of distilled water. After covering the cover glass, measure the length and width of at least 30 stomatal apparatuses with reference to scales under a photomicroscope (Leica DM2000). The stomatal density was calculated from 10 randomly sampled microscopic fields of each leaf.
Observation and Determination of the Anatomical Structures
Cross-sections of leaves obtained from the same internode in tetraploid and diploid plants were made by paraffin sectioning. Small squares of 1 cm × 1 cm from both sides of the major vein of the leaf were cut and placed into FAA fixing solution (mixed with 70% ethanol, glacial acetic acid, and formaldehyde at a volume ratio of 18:1:1) for fixation. Permanent slices with a thickness of 8–12 mm were made by the conventional paraffin slicing method (Miao et al., 2014). Olympus BX53 biomicroscope was used to photograph the anatomical structures of leaves. The anatomical parameters of leaves including the thickness of the upper epidermis polysaccharides, lower epidermis, palisade tissue, and spongy tissue were measured using TopView software.
Determination of Astragalus Polysaccharides in A. membranaceus
Ground samples 2.0 g) of A. membranaceus were placed into 50 mL conical flasks with 50 mL of distilled water. After incubation in a water bath for 2 h at 68°C, the filtrate was collected by filtration with gauze, and the above operation was repeated for the filter residue. The two filtered solutions were combined and centrifuged at 3,500 revolution per minute for 10 min. The supernatant was concentrated to 10 mL by heating evaporation to obtain the polysaccharide sample solution. The extracted samples were analyzed by the phenol-concentrated sulfuric acid method (Nielsen, 2010).
Determination of Saponin Content in A. membranaceus
Ground samples (2.0 g) of A. membranaceus were accurately weighed and placed in a 50-mL centrifugal pipe with 40 mL of 80% ethanol. The samples were reflux extracted at 85°C for 2.5 h until the liquid had no color. The extract was recovered, and water was added to 10 mL. The mixture was extracted with 20 mL n-butanol to recover the n-butanol layer, and the above operation was repeated three times. Recyclables were injected into the D101-type large Porous adsorption resin column (inner diameter of 1.5 cm and column height of 12 cm). After waiting for 2 min, the piston was opened, eluted with 50 mL of water, and the flow rate was adjusted to approximately 2 drops/sec. The column was eluted with 30 mL of 40% ethanol, and the eluent of these two washes was discarded. Finally, the column was eluted with 80 mL of 70% ethanol, and the eluent was collected. The collected eluent was evaporated to dryness, methanol was added to dissolve the residue, and the compound was transferred to a 5-mL volumetric flask. After adding methanol to a constant volume, the extracts were identified by reaction with trichloroacetic acid.
Determination of the Flavonoid Content in A. membranaceus
Ground samples (2.0 g) of A. membranaceus were accurately weighed, placed in a 50-mL centrifugal pipe with 40 mL of 80% ethanol for 12 h and extracted with an ultrasonic bath (4,000 Hz) at 75°C for 40 min. The mixture was centrifuged for 5 min at 4,000 rpm, and then, the supernatant was identified by an aluminum trichloride reaction.
Analysis of SOD, POD, and CAT Activities
To determine SOD, POD, and CAT enzyme activities, 0.4 g fresh leave was homogenized using 5 mL 0.1 M PBS (phosphate-buffered saline, pH 7.5). The homogenates were centrifuged at 12,000×g for 15 min at 4°C, and the supernatants were collected for enzyme assays. SOD and POD activities were analyzed according to Giannopolitis and Ries (1977) and Doerge et al. (1997), respectively. CAT activity was measured by monitoring the consumption of hydrogen peroxide (H2O2) at 240 nm (Larrigaudiere et al., 2004). The enzyme activities were expressed as U/g fresh weight (FW).
Data Analysis
The results are presented as the mean values ± standard deviation (SD) from three biological replicates. Treatment results were compared by a one-sample t-test using Origin software (Version 8.5).
Results
Effects of Different Treatment Methods on the Germination of A. membranaceus Seeds
The seed coat of A. membranaceus (Fisch.) Bge. Contains a cuticle of pectin and endogenous inhibitors, which greatly reduces the seed germination rate. To increase the germination rate, the A. membranaceus seeds were treated with mechanical damage, sulfuric acid soaking, and warm soup soaking. As shown in Table 1, the germination potential, germination rate, and germination index of seeds treated with mechanical damage were 73.33%, 92.33%, and 34.35%, respectively, which were significantly higher than the other two treatments. The three indices of sulfuric acid soaking and soaking seeds in the warm soup were 50.67%, 79%, 27.1%, and 42.67%, 62%, and 21.58%, respectively. The significance analysis results showed that the germination rate and germination index of the sulfuric acid soaking treatment were significantly higher than those of the warm soup soaking. This result indicates that certain mechanical damage can eliminate the constraints of the cuticle on the germination of A. membranaceus seeds to some extent.
Effects of Different Treatment Methods on the Seed Germination of A. membranaceus.

Germination potential (%) = (maximum germination number of seeds ÷ number of tested seeds) × 100%;
Germination rate (%) = (number of germinated seeds in 5 days ÷ number of tested seeds) × 100%; Germination index = ƩGt/Dt, Gt: number of germinated seeds on the 7th day; Dt: germination days.
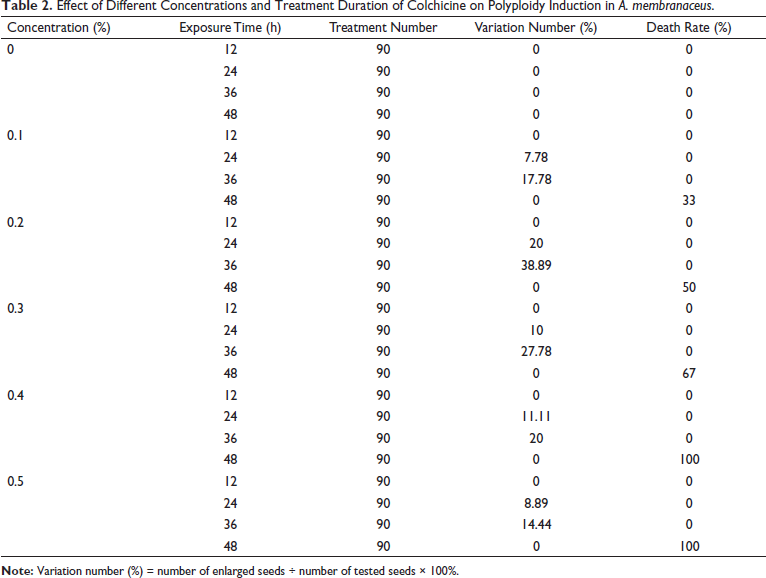
Effects of the Colchicine Concentration and Incubation Time on the Seeds of A. membranaceus
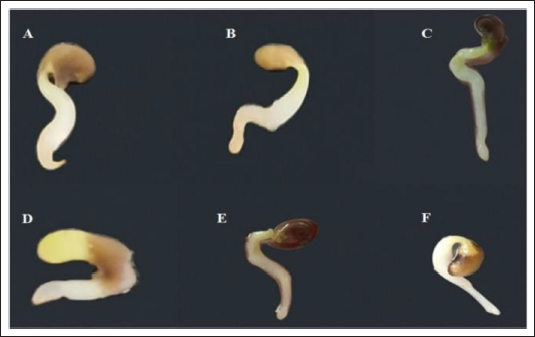
Germinating seeds were incubated in different concentrations of colchicine solutions at different times. Root meristem enlargement is considered to be a morphological variation of doubling the number of chromosomes in cells (Figure 1). The results indicated that there was no significant change after treatment with different colchicine concentrations solutions for 12 h. However, morphological variation increased with increasing incubation time from 24 to 36 h at different concentrations. In addition, the roots became transparent and rotted with the extension of the processing time, which resulted in a high mortality rate and a decrease in the variation rate after 48 h treatment. Finally, the variation efficiencies of the 0.2% colchicine treatment were much higher than those under all other concentrations, and the variation efficiencies reached 38.89% at 36 h (Table 2).
Root Meristem Characteristics of A. membranaceus After 36 h Treated with H2O (A) and 0.1% Colchicine (B), 0.2% Colchicine (C), 0.3% Colchicine (D), 0.4% Colchicine (E), and 0.5% Colchicine (F).
Effect of Different Concentrations and Treatment Duration of Colchicine on Polyploidy Induction in A. membranaceus.
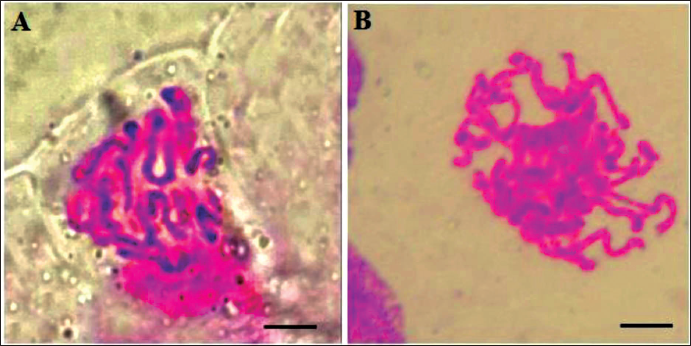
Identification of Chromosome Number
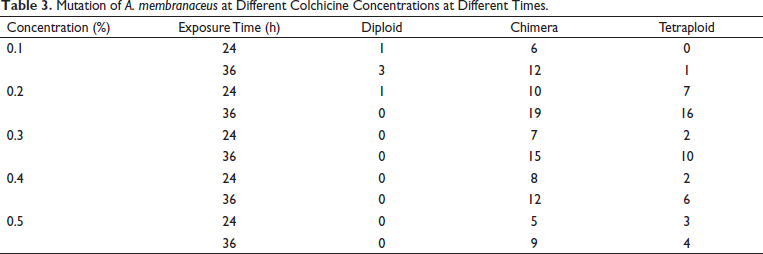
The chromosome of the lateral root tips was used to identify the ploidy of A. membranaceus plantlets treated with colchicine. The chromosome number of the diploid seedlings was 2n = 2x = 16, whereas the tetraploid had 2n = 4x = 32 (Figure 2). A plant with all of the root tip cells containing 16 chromosomes was identified as diploid, with some cells containing 16 chromosomes and the other cells containing 32 chromosomes being classified as a chimera, and that with all of the cells containing 32 chromosomes was identified as tetraploid. Finally, the chromosome counts indicated that 51 tetraploid plantlets were obtained (Table 3).
Microscopic Analysis of Chromosomes from Root Tip Cells of (A) Diploid (2n = 2x = 16) and (B) Tetraploid (2n = 4x = 32) A. membranaceus (10 × 100) (Bar = 2 µm).
Mutation of A. membranaceus at Different Colchicine Concentrations at Different Times.
Morphological Differences Between Diploid and Tetraploid Plants
The phenotypic characteristics of tetraploid plants are often different from those of diploid plants, such as deeper green leaf color and larger leaf area. After culture for 7 days, the leaf color was deeper green, and the A. membranaceus hypocotyl was shorter and sturdier in tetraploids than in diploids (Figure 3A). The hypocotyl perimeter of the tetraploid was 0.06 cm larger than that of the diploid (0.03 cm), whereas the hypocotyl length of the tetraploid was 0.02 cm shorter than that of the diploid (0.09 cm) (p < 0.05) (Table 4). The leaves of tetraploid plants shrank and grew slowly from half a month to one month, and then, the leaves gradually expanded with increasing growth and development (Figures 3B–D). The average leaf length in tetraploid plants was 0.4 cm, which was lower than that in diploid plants (0.7 cm), whereas there was no significant difference in leaf width between diploid and tetraploid plants after culture for 7 days. When the plant grew for approximately 60 days, the average leaf lengths and widths of tetraploid plants were 1.5 and 0.9 cm, respectively, which were larger than those of diploid plants (1.0 and 0.5 cm) (p < 0.05) (Table 5).
Plant Morphology of Diploid and Tetraploid A. membranaceus. (A–D) Seedlings Cultivated for 7, 15, 30, and 60 Days.

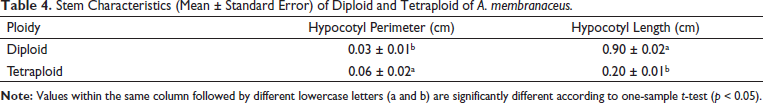
Stem Characteristics (Mean ± Standard Error) of Diploid and Tetraploid of A. membranaceus.
Leaf Characteristics (Mean ± Standard Error) of Diploid and Tetraploid A. membranaceus.
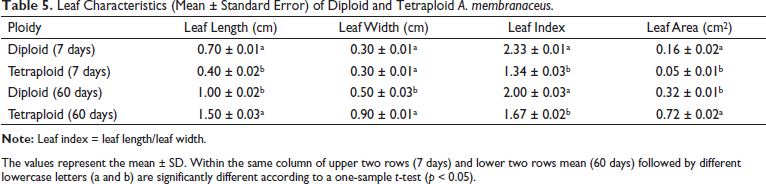
The values represent the mean ± SD. Within the same column of upper two rows (7 days) and lower two rows mean (60 days) followed by different lowercase letters (a and b) are significantly different according to a one-sample t-test (p < 0.05).
Stomatal Feature Analysis
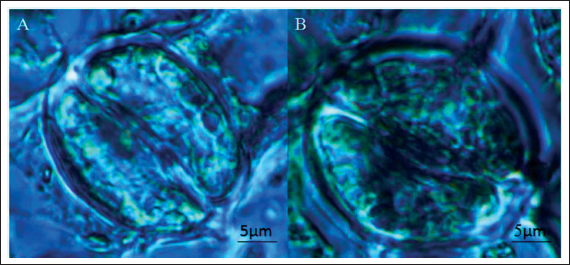
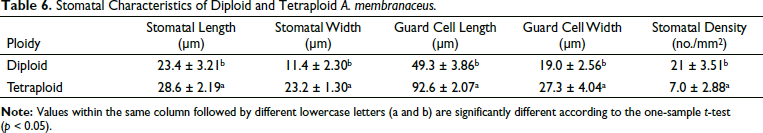
Generally, larger stomata are regarded as one of the hallmark characteristics of polyploidy. To analyze how ploidy variation affected stomata changes, the stomatal density, length, and width of the leaves of diploid and tetraploid plants for 60 days in vitro cultures were measured (Figure 4 and Table 6). The average length and width (28.6 and 23.4 µm, respectively) of stomata in tetraploid plants were significantly higher than those in diploid plants (23.4 and 11.4 µm, respectively). The average length and width of the guard cells of the tetraploid plants were 92.60 and 27.60 µm, which were significantly larger than those of the diploid plant with values of 49.30 and 29.04 µm, respectively. However, the stomatal density of diploid plants was approximately triple that of tetraploid plants.
Stomata Characteristics of Diploid (A) and Tetraploid (B) A. membranaceus.
Stomatal Characteristics of Diploid and Tetraploid A. membranaceus.
Anatomic Structure of Diploid and Tetraploid Leaves
To explore the effect of ploidy level-affected changes on leaf anatomical structures, the anatomical structures and structure parameters of 60 days diploid and tetraploid leaves were examined. The results showed that the mean upper epidermis and lower epidermis cells of tetraploid plants (37.35 ± 2.22 and 52.22 ± 2.14 µm, respectively) were significantly thicker than those of diploid plants (28.24 ± 1.72 and 40.21 ± 2.18 µm, respectively). Similarly, the thicknesses of the palisade tissue and spongy tissue of the tetraploid (103.23 ± 3.25 and 32.2 ± 2.21 µm, respectively) were also significantly superior to those of the diploid (46.9 ± 2.35 and 22.74 ± 1.44 µm, respectively) (Figure 5 and Table 7). Overall, we proved that ploidy change could increase the thicknesses of the upper and lower epidermis, palisade, and spongy tissue of leaves.
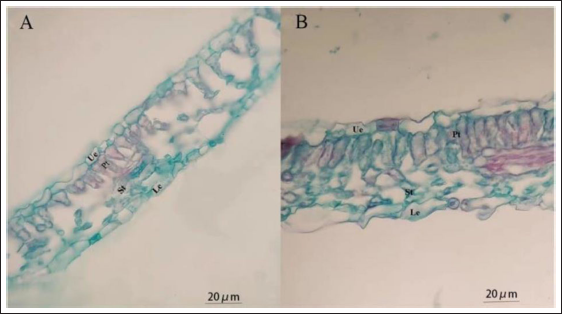
Leaf Cross-section Characteristics (Mean ± Standard Error) of Diploid and Tetraploid A. membranaceus.

Determination of the Medicinal Content
The contents of the effective constituents of tetraploid and diploid A. membranaceus plants at 60 and 90 days were extracted and analyzed. Compared with the A. membranaceus diploids, the contents of polysaccharides, saponins, and flavonoids in the tetraploids increased (Figure 6). After culture for 60 days, the polysaccharide, saponin, and flavonoid contents in the aerial parts (stem and leaf) of tetraploids (0.002, 0.1815, and 0.0222 mg/g, respectively) were significantly higher than those in diploids (0.0015, 0.064, and 0.0107 mg/g, respectively). The polysaccharide and saponin contents in tetraploid roots (0.0027 and 0.2385 mg/g) were significantly higher than those in diploid roots (0.0021 and 0.0954 mg/g). The polysaccharide, saponin, and flavonoid contents all increased with the growth of tetraploid and diploid plants. After culture for 90 days, the polysaccharide, saponin, and flavonoid contents in tetraploids were higher than those in diploids. The polysaccharide, saponin, and flavonoid contents of aerial parts and roots in tetraploids (0.959, 0.310, 1.378, and 2.393, 0.534, 1.019 mg/g, respectively) were significantly higher than those in diploids (0.640, 0.197, 0.755, and 1.348, 0.344, 0.561 mg/g, respectively). In summary, artificial chromosome doubling can increase the polysaccharide, saponin, and flavonoid contents in A. membranaceus.
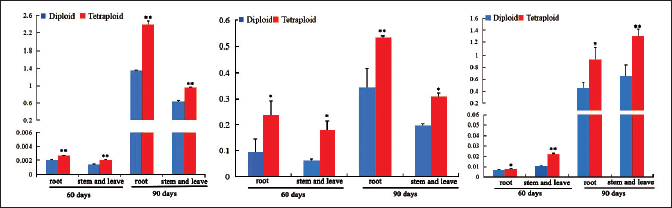
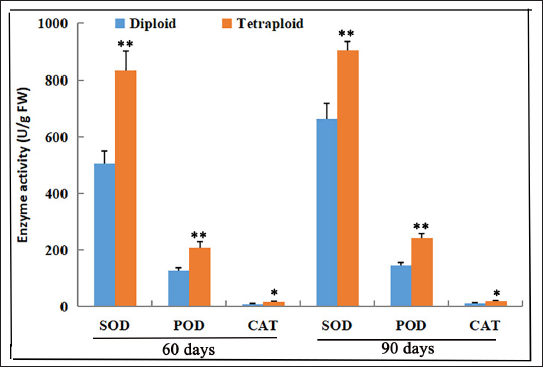
Genome Duplication Improved Reactive Oxygen Species System
The SOD, POD, and CAT are important antioxidant enzymes, and their activity levels can reflect the ability of plants to resist stress. We discovered that the activities of SOD, POD, and CAT were increased in tetraploid plants by 64.47%, 65.07%, and 91.45% compared with those in diploids, respectively (Figure 7). The significance analysis results showed that the SOD, POD, and CAT activities of tetraploid plants were significantly higher than those of the diploid plants (p < 0.01). The result indicated that tetraploid plants possessed great potential for stress resistance.
Discussion
With the modernization of Traditional Chinese Medicine development, the demand for Traditional Chinese Medicine is growing. However, due to the serious destruction of the ecological environment, the wild resources are gradually reduced, and the photochemical properties of cultivated varieties are too low to meet the market demand. Therefore, it is urgent to create high-quality and high-yield herbal varieties. Polyploid plants have not only large nutritional organs but also strong resistance. Hence, the polyploidy breeding of medicinal plants can not only improve the quality and yield but also enhance their ability to resist adverse ecological environments.
Colchicine is the most widely used chemical mutagen to induce polyploidy, and many polyploids of medicinal plants have been obtained by colchicine treatment (Clarindo et al., 2008; Liu et al., 2007; Pintos et al., 2007; Takamura & Miyajima, 1996). This paper is the first to describe the induction of A. membranaceus by colchicine treatment. In our study, the highest induction rate of tetraploid was observed in the presence of 0.2% colchicine treatment for 36 h. It is noteworthy that the induction efficiency of different plants or different parts of the same plant is dissimilar. For example, the tuber segment and hybrid ovules of cyclamen obtained the highest percentage of polyploidy after treatment with 100 mg/L and 0.05% colchicine for 4 days and 10–15 days, respectively (Ishizaka & Uematsu, 1994; Takamura & Miyajima, 1996). Therefore, the concentration of colchicine and exposure time need to be explored when polyploidy is induced using colchicine. The mortality of A. membranaceus increased with increasing colchicine concentration or exposure time during induction, which reduced the induction rate. The toxic effect of high-concentration colchicine on cells has been described in previous studies (De et al., 2010; Gu et al., 2005). Another problem is that polyploid plants induced by colchicine are chimeras. It is necessary to select homozygous polyploid plants from the offspring of chimeras. However, the probability of polyploid homozygous can be enhanced by single-cell culture, callus, and plant tissue culture. However, it is still a difficult problem and worthwhile to further study the screening of polyploid homozygotes from chimeras.
Generally, the external morphology of plants usually changes as the number of chromosomes increases (Miguel & Leonhardt, 2011; Mo, 2020). Morphological characteristics are commonly used as an indicator of the ploidy level of plants (Borrino & Powell, 1988; Cui et al., 2017; Thomas et al., 2000). In this study, the tetraploid A. membranaceus had more enlarged thick and dark green leaves than the diploid plants, and subsequently, the leaves turned shrinking in the first month of plant growth (Figure 3), which is consistent with previous studies (Liu et al., 2007; Paniel et al., 2011). Moreover, the characteristics of stomata are also used as indicators for polyploid plant identification (Duren et al., 1996). The stomatal length and width of A. membranaceus tetraploid plants were significantly larger than those of diploid plants (Figure 4 and Table 6). Morphological changes, especially in leaves and stomata, have been observed in many polyploids of herbal plants (Rao et al., 2019; Yan et al., 2016). Therefore, observing the characteristics of leaves and stomata can be used as a very convenient and reliable method to identify tetraploid A. membranaceus. At present, few studies have focused on the change in leaf tissue structure in polyploids. In this study, a thicker upper epidermis along with a thinner epidermis, palisade, and spongy tissue of tetraploid leaves were observed (Figure 5 and Table 7), which indicated that leaf tissue structure may also be used as an indicator of polyploids in addition to leaf and stomatal characteristics.
A high active compound content is important for herbal medicine to use in clinical and industrial extraction. Our study indicated that the content of active compounds in tetraploid plants, including saponins, polysaccharides, and flavonoids, was higher than those in diploid plants. The same result also occurred in medicinal materials such as Sophora tonkinensis (Wei et al., 2018) and Salvia miltiorrhiza (Chen et al., 2018). The reasons for the increases in active compound contents in polyploids may be related to changes in gene number, genetic transcription, and expression differences in polyploids, which result in an increase in secondary metabolites. In addition, thicker palisade tissue can store more water and chloroplasts, which improves photosynthetic efficiency (Guo & Wu, 2018; Tang et al., 2014). Meanwhile, the thicker upper epidermis of leaves can prevent the loss of water by reducing transpiration, and improve plant drought resistance (Zhang et al., 2017). SOD, POD, and CAT are the main enzymes in the plant antioxidant system, and the level of their activity is an important indicator of plant stress resistance. The results of previous studies showed that the physiological indicators related to stress resistance of tetraploid and diploid plants changed significantly under the same planting conditions. For example, the activities of SOD, POD, and CAT in tetraploid pear plants were significantly higher than those of diploid plants (Yang et al., 2017). The higher activity levels of SOD, CAT, and POD and thicker upper epidermis and palisade in A. membranaceus tetraploid leaves indicate that tetraploid plants had a strong material basis for stress resistance. However, the resistance in tetraploid A. membranaceus plants still needs to be further studied under various stress conditions. Moreover, the active compound contents of tetraploid A. membranaceus not only improved in the roots used as medicine but also in the stems and leaves, which are usually abandoned. The contents of flavonoids in stems and leaves were even higher than those in roots (Figure 6). The results suggest that the medicinal components in the stems and leaves of A. membranaceus can be used reasonably in the future.
Conclusion
An efficient colchicine-mediated technique for tetraploid induction in A. membranaceus had been established. The most suitable protocol for tetraploid induction of A. membranaceus was 0.2% colchicine treatment for 36 h after the seed’s germination. The stomatal length, width, and density of leaf blades significantly differed between diploid and tetraploid plants. The tetraploid plants with a higher content of polysaccharides, saponin, and flavonoids were obtained to be used in further breeding programs aimed at increasing the production of medicinal compounds.
Summary
Colchicine can efficiently induce the tetraploid plants of A. membranaceus.
The morphological characteristics of tetraploid plants were different from diploid plants.
The contents of polysaccharides, saponins, and flavonoids and the activities of SOD, POD, and CAT of tetraploid plants were higher than those of diploid.
Footnotes
Abbreviations
SOD: superoxide dismutase; POD: peroxidase; CAT: catalase.
Acknowledgments
The authors wish to thank for providing financial support from the Natural Science Foundation of Hebei Province, China (No. C2021201043) and the Key R&D Program of Hebei Province Special Project for Tackling Key Common Agricultural Technologies (No. 20327508D).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Statement of Informed Consent and Ethical Statement
The seeds of Astragalus membranaceus (Fisch.) Bge. were collected in full compliance with the guidelines and regulations of Hebei Province, China. Meanwhile, the collection was carried out in accordance with relevant legislation and with the permission of regulatory authorities.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Natural Science Foundation of Hebei Province, China (No. C2021201043) and the Key R&D Program of Hebei Province Special Project for Tackling Key Common Agricultural Technologies (No. 20327508D).
