Abstract
Background
Osteosarcoma (OS) is an extremely aggressive primary bone cancer (BC) malignancy. A variety of malignancies can develop in the bones, including BC. Primary BCs are tumors that start in the bone. Bones can also become affected by tumors that start in the body’s organs or other tissues.
Objectives
Several reports suggested that corilagin (CL) exerts anticancer properties on various kinds of tumor cells; however, its molecular action on OS cells remains undefined.
Materials and Methods
The CL activity of OS cells’ cytotoxicity, intracellular reactive oxygen species (ROS), mitochondrial membrane potential (MMP), apoptosis, and cell-cycle distribution was evaluated by 3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide (MTT) assay, dichloro-dihydro-fluorescein diacetate (DCFH-DA), Rh-123, acridine orange and ethidium bromide (AO/EB), 4′,6-diamidino-2-phenylindole (DAPI), and flow cytometry analysis.
Results
Results revealed that CL could suppress OS cell proliferation via enhanced intracellular ROS, and MMP loss, and triggered apoptosis in a dose-dependent has an inferior manner. Our findings demonstrated that CL alleviates U2OS and MG-63 cell proliferation by the ROS-mediated apoptosis, which triggers G0/G1 cell-cycle arrest.
Conclusion
Thus, CL might be a protective therapeutic agent against BC.
Introduction
Osteosarcoma (OS) is a very uncommon bone tumor, occurring in 3.4 instances per million persons each year globally (Damron et al., 2007; Kager et al., 2010). OS generally instigates from lengthy bones with hasty growth, comprising the femur, the tibia, and the humerus (Li et al., 2015). Bone tumors are relatively uncommon, constituting only 0.5% of global cancer incidence among all types of cancers (Kager et al., 2010). A retrospective study conducted in 2007 by Gulia et al. (2016) to ascertain the prevalence of bone and soft-tissue tumors in India revealed that almost 60% of all musculoskeletal neoplasms were bone tumors and 36% were soft-tissue tumors. However, 66% of bone tumors were cancerous, 15% were benign, and 19% were not cancerous at all (Li et al., 2015). The most frequent benign tumor was a giant cell tumor, while the most frequent malignant tumor was OS, which is described by immature osteoid development and atypical osteoblastic disparity (Tang et al., 2015). OS displays bimodal age dissemination with a high peak in young adults and a minor peak in the elderly (Tang et al., 2008). Amongst people below 20 years, the OS prevalence rate is 8.7/million, and males have a higher risk (Mirabello et al., 2009). The modern treatment for OS is neoadjuvant chemotherapy before surgical resection and chemotherapy over ensuing surgery. By these controlling approaches, the 10-year disease-free survival is achieved in almost 60% of patients with non-metastatic ailment; however, it has an inferior effect for patients diagnosed with metastatic cancer (Longhi et al., 2006). Hence, the development of new remedies has chiefly been crucial to exploring the mechanisms underlying the occurrence and development of OS with illuminating the overall survival rate.
Numerous plant-derived natural ingredients are unveiling promising anti-tumor beneficial properties and are being assessed in clinical experiments (Cragg & Newman, 2005). Corilagin (CL) is a gallotannin; a polyphenol tannic acid that is mainly isolated from Phyllanthus urinaria possesses anti-tumor, anti-inflammatory, antioxidant, and anti-coagulation activities (Rangkadilok et al., 2007; Yeo et al., 2015). Moreover, recent research exposed that CL has been beneficial in tumor therapy, making it a potential candidate for diverse tumor cells such as ovarian cancer (Attar et al., 2017), hepatoma (Ming et al., 2013), glioblastoma (Milani et al., 2018), and cholangiocarcinoma (Gu et al., 2016). Earlier studies demonstrated that CL could regulate malignant cells’ growth through switch several cell signaling pathways in various tumors. In a previous study, it was explained that CL prevents ovarian malignant cells’ growth in vitro and in vivo by enhancing cell-cycle arrest at the G2/M stage, thereby augmenting apoptosis (Li et al., 2018). Additionally, CL exhibits ovarian cancer cells’ reluctance to proliferate when the Smad and ERK/AKT pathways are blocked. Although CL contains anti-tumor and anti-inflammatory properties, its potential use or even the mechanism preventing BC is unclear. The goal of the current study was to determine how CL affected the anticancer and apoptotic activities of BC cells from the U2OS and MG63 strains.
Apoptosis is an involuntary cell death function, and has a crucial role in the chemotherapy of various types of cancer (Zimmermann et al., 2001). Reactive oxygen species (ROS) are the oxygen active forms that are by-products of cellular metabolism activities (Fruehauf & Meyskens, 2007). Cell propagation and differentiation can be stimulated by a modest upsurge in ROS, while extreme quantities of ROS are capable to restrict cellular signaling pathways owing to proteins, lipids, and DNA oxidative damage (Trachootham et al., 2009). Remarkably, collecting data proposes that tumor cells are under elevated oxidative stress, which specifies that they are further probable to be destroyed with more ROS prevailing persuaded by exogenous agents (Pelicano et al., 2004). Largely, the anti-carcinogenic characteristic of phytochemicals is supposed to be allied with their capacity to subdue intracellular ROS (Palit et al., 2015). Conversely, the pro-oxidant action of phytochemicals, rather than their antioxidant mechanism in malignant cells, has been described to be a central tool for arbitrating their anti-tumor activities (Galati & O’Brien, 2004). Accumulating suggestion designates that ROS are intricate in several signaling pathways and serve a vital role in triggering cell apoptosis (Wang et al., 2017). Undue ROS quantities can alter numerous signaling pathways, such as MAPK family members, including p38, ERK, and JNK (Smolensky et al., 2018). Dysregulation of NF-κB signaling is accompanied by extreme cellular explosion and developmental signals in carcinogenesis. Certainly, the NF-κB signaling pathway has been stated to be intricate in inflammatory proliferation and differentiation of OS cells (Chang et al., 2009). It has been previously suggested that NF-κB could aid a causative role in the progression of OS (Mongre et al., 2014). CL diminishes acetaminophen induced hepatotoxicity over MAPK, and NF-κB pathway in the mice model (Liu et al., 2020). As a result, in this study, we assessed the anticancer and apoptotic effectiveness of CL on U2OS and MG63 human OS cells using ROS-mediated apoptosis, as well as cell-cycle regulation through the MAPK/NF-κB signaling pathway.
Materials and Methods
Chemicals
Corilagin (CL), DMEM, FBS, antibiotics, PBS, 3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide (MTT), DCFH‐DA, Rh-123, acridine orange and ethidium bromide (AO/EB), 4′,6-diamidino-2-phenylindole (DAPI), propidium iodide (PI), SDS, DMSO, and further biochemicals were obtained from Merck, Germany.
Cell Culture
U2OS and MG63 cell lines were procured from Shanghai Aiyan Biotechnology Co., Ltd (Shanghai, China). These cells were grown in DMEM media and were added with FBS (10%), 1% antibiotics (penicillin/streptomycin) at 37°C, and 5% CO2 atmosphere; less than 95% humidity.
MTT Cell Viability Assay
Human OS cell viability was assessed by using the MTT assay (Vijayalakshmi & Sindhu, 2017). Briefly, U2OS and MG63 cells were sowed into 96 wells (1 × 105 cells/well) and cultivated at 37°C ensuring 5% CO2 in a wet incubator for 24 h. Once incubated overnight, the culture medium was separated, and the cells were dipped with PBS and preserved with diverse dosages of CL (10, 20, 30, 40, and 50 µM/ml) for one day. Successively, the treated cells were added with MTT (10 µl) solution and kept for a further 4 h to let the transformation of MTT into insoluble formazan crystals by the mitochondrial dehydrogenase. The subsequent formazan crystals were liquefied by adding150 µl of DMSO. The optical density was determined at 490 nm by employing an ELISA plate reader (Bio-Tek Instruments, Winooski, VT, USA). Cell viability was considered as a percentage of proliferation versus untreated U2OS and MG63 control cells (100%). The IC50 value has calculated the formula as shown below:
Cell viability inhibition (%) = (Optical density of control – Optical density of test) × 100
Assessment of Intracellular ROS
Human OS cells (U2OS and MG63) were sowed in 6-well plates each and preserved for one day; the concentration of CL (30 µM/ml) was added. Then, the control and treated cells were stained with 10 µM of DCFH‐DA, and subsequently, they were incubated at 37°C for half an hour. These cells were collected and then splashed with ice‐cold PBS twice to abolish further dye. The fluorescence was determined by a multimode reader (Tecan, Austria) (excitation at 485 ± 10 and emission at 530 ± 12.50 nm, correspondingly).
Mitochondrial Membrane Potential Assay
Mitochondrial membrane potential (MMP) was evaluated to recognize early apoptotic stages by performing Rh-123 staining. OS cells (U2OS and MG63) were sowed in 6 wells and preserved with CO2 (5%) at 37°C in a CO2-humidified incubator for one day. Afterwards, U2OS and MG63 cells were exposed over 30 µM/ml CL for 24 h, ensuring wet two-fold with cold PBS, static with paraformaldehyde (4%) for 20 min and washed. Sequentially, control and treated cells were stained with Rh-123 (10 µg/ml) in dark conditions for 30 min at 37°C. Then, control and treated cells were washed twice with washing buffer to eradicate the further stains and examined the MMP variation over fluorescent microscopy (Olympus, Tokyo, Japan) using a blue filter (485–530 nm).
Apoptosis Evaluation by AO/EB Staining
The apoptotic morphology in OS cells visible to CL at a concentration of 30 µM/mL was distinguished by AO/EB staining (Kasibhatla et al., 2006). U2OS and MG63 cells were exposed to 30 µM/mL CL and kept for 24 h. The control and treated cells were added to the dye mixture comprising AO/EB (100 µg/ml of each dye). Then, these cells were maintained for 20 min in the dark at room temperature to ensure the untying dye was eradicated by PBS washing and perceived through a fluorescence microscope (Olympus, Tokyo, Japan).
Estimation of Rh-123 and DAPI Staining
To measure the apoptosis efficacy of CL on U2OS and MG63 cells, DAPI and Rh-123 staining were achieved. U2OS and MG63 cells were sowed in well plates and conserved at 37°C with CO2 (5%) in a humidified incubator for one day. Then, these cells were added over CL at IC50 dose (30 µM/ml) for one day. Subsequently, the control and treated cells were two-fold dipped with PBS, static with paraformaldehyde (4%) for 20 min, rinsed, stained with DAPI, and incubated for 20 min. DAPI-exposed cells were stained with Rh-123 for 30 min at 37°C. The cells were cleaned two-fold with methanol to eradicate further stains and rinsed with PBS and photographed the MMP variation by using a fluorescent microscope (Olympus, Tokyo, Japan).
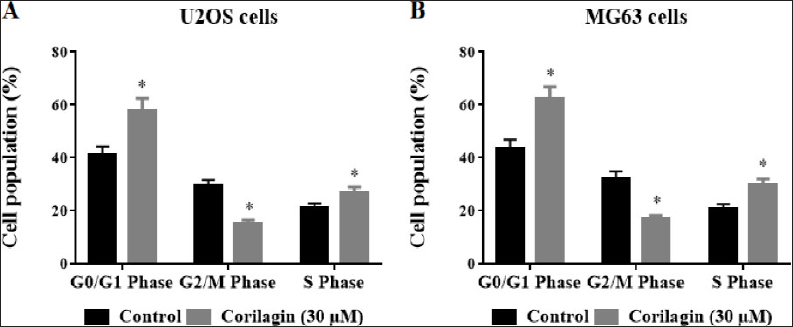
Detection of Cell-Cycle Distribution by PI Staining
Evaluation of PI staining was achieved to explore the cell-cycle distribution of OS cells. U2OS and MG63 BC cells were added with 30 µM/ml of CL incubated at 48 h. These treated cells were reaped by trypsinization and the cells in each group were dipped twice with cold PBS. Ethanol (70%) was castoff overnight fixation and centrifuged; cells were resuspended in 500 µl of PBS. Later, 20 µl of TritonX-100 (0.05%) and RNase (1 mg/ml) were added and kept at room temperature for 30 min; the next 5 µl PI (1 mg/ml) solutions were added and re-preserved for 30 min at room temperature. Flow cytometry analysis on a FACScan was then accomplished to detect the cell-cycle distribution.
Statistical Analysis
A statistical study was achieved by Graphpad Prism software version 8.0.1 with comparison attained by ANOVA and subsequently by Duncan’s test. The results were considered statistically significant values p < 0.05.
Results
Effect of CL on OS Cells’ Morphology
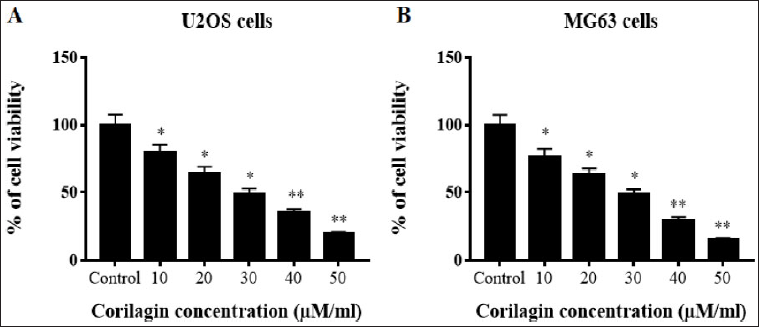
The human OS cells’ U2OS and MG63 cytotoxicity are evaluated by MTT test with diverse concentrations (10, 20, 30, 40, and 50 µM/ml) of CL. The results revealed that CL had cytotoxic and anti-proliferative effects on U2OS and MG63 cells in a concentration-reliant mode. CL treatment at a concentration below 10 µM could not expressively affect the viability of these OS cells. However, at high concentrations of 30, 40, and 50 µM/ml of CL exhibited the inhibition of U2OS and MG63 cell proliferation. The IC50 value of CL was calculated and 30 µM/ml concentrations were selected for further studies (Figure 1).
Effect of CL on Intracellular ROS Production in OS Cells
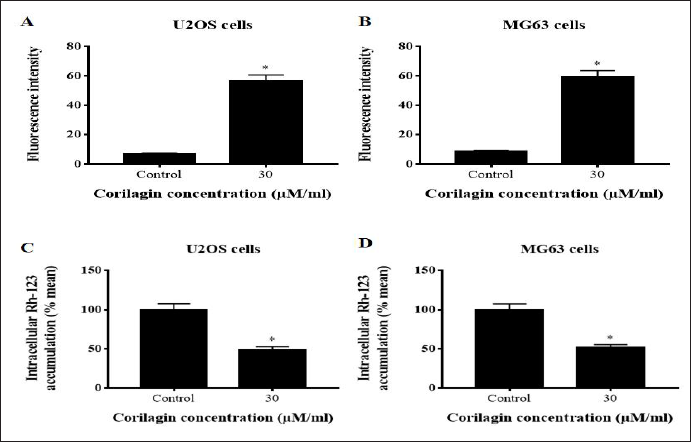
Intracellular ROS formation is linked to various stimuli and can trigger apoptosis and cell-cycle arrest. The intracellular ROS levels were enhanced in U2OS and MG63 cells after being added with 30 µM/mL CL for 24 h. To directly observe the accumulation of ROS, dichloro-dihydro-fluorescein diacetate (DCFH-DA)-labeled cells were viewed under a fluorescence microscope. The fluorescence intensity of ROS was considerably higher (p < 0.05) with 30 µM of CL in comparison to untreated U2OS and MG63 control cells (Figure 2A and B).
Effect of CL on MMP Loss in OS Cells
Mitochondrial membrane depolarization was noticed by the cells capability to acquire Rh-123 dye. The control U2OS and MG63 cells presented high Rh‐123 intensity nearly 99% of MMP. The accumulation of Rh-123 fluorescence was depleted in CL-treated (30 µM/mL) U2OS and MG63 cells in a concentration-reliant mode. CL-treated U2OS and MG63 cells (30 µM) revealed significantly high (p < 0.05) loss of MMP due to intensified mitochondrial depolarization. These data confirmed that CL-treated U2OS and MG63 cells reduced the MMP, thereby augmenting the mitochondrial apoptosis (Figure 2C and D).
CL-induced Apoptosis on OS Cells is Evidenced by AO/EB Staining
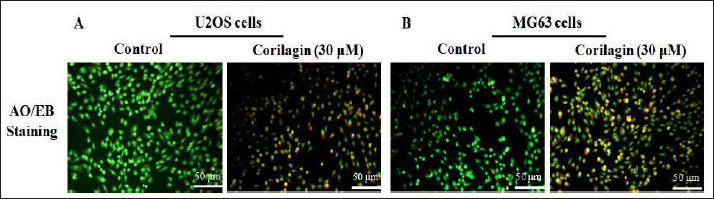
Apoptotic cells imagined the distinctive morphological alterations performed by the dual staining of AO/EB. Untreated OS control cells (U2OS and MG63) displayed uniformly stained green live cells (Figure 3A and B). CL (30 µM/ml) administration revealed intensified cell apoptosis in a concentration-reliant manner. Initial apoptotic cells presented condensed chromatin and membrane blebbing in 30-µM CL treatment, which exhibited as light greenish-yellow dots. Late apoptotic cells showed orange color that has lost their membrane integrity due to EB co-stain in 30-µM CL added groups. Chromatin condensation, membrane blebbing, fragmented nuclei, and late-stage apoptotic effect were perceived in 30-µM CL added U2OS and MG63 cells.
Effect of CL-induced Apoptosis in OS Cells
The assessment of CL (30 µM/mL) on OS cells’ (U2OS and MG63) morphological apoptotic features were investigated by DAPI staining (Figure 4A). DAPI staining is distinguished to form adducts over double-stranded DNA. The CL-prompted apoptosis on U2OS and MG63 cells were recognized to be greater morphology of condensed nuclei, nuclear body disintegration, and membrane integrity harm when compared to untreated OS cells in a dosage-reliant way. These results exposed that CL unveils anti-proliferative and apoptotic activity towards U2OS and MG63 cells.
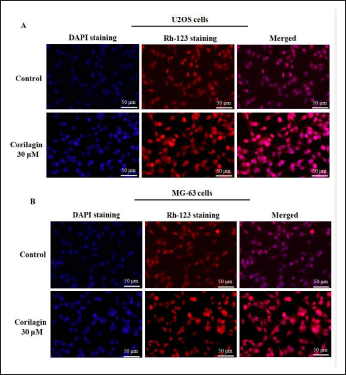
The Rh-123 staining has produced a mitochondrial membrane depolarization, which was visualized in primary apoptosis. Rh-123 stain is cationic lipophilic fluorescent intricate proximate functional mitochondria with an amplified MMP and confirmations mitochondrial control in the apoptotic model. The high strength of fluorescence (488/525 nm) was noticed in the U2OS- and MG63-untreated control cells, which have augmented MMP and the accumulation of Rh-123 fluorescence seemed red in colour. The accumulation of Rh-123 fluorescence declined in CL-treated (30 µM/mL) U2OS and MG63 cells in a concentration-reliant way. These effects are documented that CL-treated OS cells attenuated the MMP, thereby eliciting mitochondrial apoptosis (Figure 4B).
Effect of CL on Cell-Cycle Arrest in BC Cells
The cell-cycle distribution studies demonstrated that the cycle of OS cells (U2OS and MG63) was re-distributed after the treatment with 30-µM/mL CL in a dosage-depended manner (Figure 5A and B). The administration of CL expressively (p < 0.05) amplified G0/G1 phase cells; further, the amount of G2/M phase cells markedly (p < 0.05) abridged, and percentage of S-phase cells had no substantial alteration against OS control cells.
Discussion
OS is an aggressive bone tumor malignancy with a great prevalence of lung metastasis. Although there are substantial signs of progress in OS management, the existing treatments still have major confines; the long-term survival and death rates endured unaffected (Kager et al., 2010; Li et al., 2007; Longhi et al., 2006). As a result, innovative beneficial tactics are instantly requisite that can efficiently act through innumerable anticancer tools. Experimental proof in the last two decades points out that dietary constituents, predominantly medicinal plant isolated components own the ability to restrain the multifaceted occurrence of tumorigenesis over variation of gene expression and apoptosis induction (Cragg & Newman, 2005; Kuno et al., 2012). The use of herbal or plant-derived constituents, namely polyphenols and flavonoids, have been increased considerably in recent centuries; they act as a novel substitute for selectively prompting apoptosis in malignant cells (Cragg & Newman, 2005). CL is a well-known polyphenol tannic acid that has been documented beneficial in several tumor cells, such as ovarian cancer (Attar et al., 2017), hepatoma (Ming et al., 2013), glioblastoma (Milani et al., 2018), and cholangiocarcinoma (Gu et al., 2016). Thus, in this current work, we established the anti-proliferative and apoptotic mechanism of CL against OS U2OS and MG63 cells. The current research intended to explore the anticancer and anti-proliferative molecular action of CL (30 µM/mL), particularly its apoptosis induction against OS U2OS and MG63 cells.
Cell proliferation is a vital indicator for tumor expansion. Therefore, constraining malignant development by stimulating apoptosis of cancer cells is the central objective in averting tumor progression (Normile, 2002). A large-scale bioactive compound for anti-tumor drug screening was extensively used by the MTT cytotoxicity assay (Hu et al., 2018). In the existing study, MTT assay results exposed that CL expressively subdued U2OS and MG63 cells viability in a dosage-reliant way. The OS cells’ morphological observations and apoptosis findings further directed that U2OS and MG63 cells’ proliferation reduction was triggered by CL-prompted apoptosis. Gu et al. (2016) have investigated the anticancer prospective of CL against cholangiocarcinoma cell lines and suggested that CL repressed proliferation, invasion, migration, cell-cycle progress, and triggered CCA cells’ apoptosis. Therefore, the main goal of stopping tumor progression is to limit malignant development by encouraging cancer cell death (Qiu et al., 2019). Xu et al. (2019) also evaluated the efficacy of CL on SGC7901 and BGC823 gastric cancer cells and stated that it intentionally suppressed cell propagation, apoptosis, and autophagy. In this study, we demonstrated that CL persuades ROS-mediated apoptosis in OS cells.
ROS and mitochondrial oxidative metabolism have a crucial role in the induction of apoptosis under various physiological and pathological environments (Trachootham et al., 2009). ROS can tempt diverse apoptotic signaling cascades, intracellular redox homeostasis distraction, and oxidative alterations of DNA, lipids, or protein (Circu & Aw, 2010). Malignant cells have augmented intrinsic ROS levels owing to an imbalanced mitochondrial oxidative metabolic process in disparity with normal cells (Trachootham et al., 2009). Intracellular ROS levels play critical roles in triggering apoptotic signaling pathways and cell proliferation control of malignant cells (Liou & Storz 2010). Thus, based on the above findings, high intracellular ROS levels are employed in several chemotherapeutics to persuade cancer cell apoptosis (Trachootham et al., 2009). Our documents exhibited that CL- induced intracellular ROS production in OS cells, which may be interconnected with the loss of MMP, and subsequently leading to cell death. Tong and coworkers (Zhao et al., 2008) described that CL stimulated ROS-mediated apoptosis in MDA-MB-231 and MCF-7 breast cancer cells. CL prompted ROS build-up stimulated apoptosis, which is crucial for restricting the growth of human gastric cancer cells (Xu et al., 2019). These data suggest the critical role of ROS in CL-induced anticancer and anti-proliferative effects.
Conclusion
We concluded that CL (30 µM/ml) could effectively arrest the G0/G1 phase and induce apoptosis in U2OS and MG63 OS cells. Intracellular ROS generation, which may be caused by MMP collapse due to the down-regulation of MAPKs/NF-κB signaling, revealed the cytotoxic effects of CL. We hypothesized that CL might act as an agonist to induce apoptosis and cause OS cells to arrest at the G0/G1 phase, and hence, we proposed a different treatment strategy for treating OS.
Footnotes
Summary
CL increased ROS generation in bone cancer cells.
CL induced DNA damage in osteosarcoma cells.
CL inhibits cell proliferation of U2OS and MG63 cells.
Abbreviations
OS: Osteosarcoma; ROS: reactive oxygen species; CL: corilagin; BC: bone cancer; MMP: mitochondrial membrane potential; AO/EB: acridine orange and ethidium bromide; DAPI: 4′,6-diamidino-2-phenylindole; DCFH-DA: dichloro-dihydro-fluorescein diacetate; MTT: 3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide; PI: propidium iodide.
Declaration of Conflicting Interests
The authors declare no conflict of interest.
Statement of Ethical Approval and Informed Consent
This research was approved by Shandong Second Provincial General Hospital on May 4, 2022. Informed consent is not applicable since this study does not involve animal and human experiments.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
