Abstract
Background
Asthma is a prominent non-communicable inflammatory disease that affects both children and the elderly. Younger people are more prone to asthma, and most prescribed anti-asthmatic medicines relieve symptoms but do not cure the condition completely. We investigated the ability of a phytochemical plumbagin to alleviate ovalbumin (OVA)-induced asthma in BALB/c mice.
Materials and Methods
The allergic asthma-induced mice were treated with two different doses of 25 and 50 mg/kg bwt plumbagin, and to compare the efficacy of plumbagin, a standard drug dexamethasone treatment was given. OVA-specific IgE and eotaxin were quantified to determine the induction of asthma and the inhibitory role of plumbagin. Total leukocyte and differential count were done to assess the effect of plumbagin on inflammatory cells. Inflammatory cytokines inducing both atopic and non-atopic asthma were quantified to examine the efficacy of plumbagin against allergic and non-allergic-induced asthma. Nitric oxide (NO) and myeloperoxidase activity were measured to investigate the anti-asthmatic potential of plumbagin. The antioxidant potency of plumbagin was assessed by quantifying the levels of antioxidants and the oxidative stress marker malondialdehyde. Lung weight index and histopathological analysis of lung tissue were done to confirm the ameliorative potency of plumbagin against OVA allergen-induced asthma.
Results
Plumbagin treatment significantly decreased the status of OVA-specific IgE and eotaxin, thereby prevented the eosinophilic infiltration. It also inhibited the synthesis of both atopic and non-atopic inducing inflammatory cytokines. Plumbagin treatment also increased the levels of antioxidants and prevented the lung tissue damage, which was evidenced with our histopathology study of lung tissue.
Conclusion
Overall, our finding confirms that plumbagin is persuasively alleviated OVA allergen-induced asthma complications in mice model and may be an alternative for currently available anti-asthmatic drugs.
Introduction
Allergic asthma is a non-communicable inflammatory disease that affects all the age group (WHO, 2021). According to the WHO data, approximately about 339 million individuals were affected with asthma and prevalence all over the globe differs from 1% to 18%. Comparatively, the mortality rate was increased in the geriatric population and the prevalence rate was drastically increasing around the globe (Cazzola et al., 2022; Global initiative for asthma, 2020; Reddel et al., 2022). Asthma is occurs mainly due to pollution of industrial smoke, tobacco smoking, irritants both in outdoor and indoor, and so on. The other causative agents are infections, obesity, lifestyle, and diet intake. Genetics, immunity, and socio-economic status play critical roles in preventing and treating asthma patients (Alomary et al., 2022; Nappi et al., 2022).
Asthma are classified into allergic asthma and non-allergic asthma in which allergic asthma accounts to be predominant (Noureddine et al., 2022). Allergic asthma is a multifaceted heterogeneous inflammatory disease with diverse clinical pathologies. The clinical presentations of asthma are airway hyperresponsiveness, over-activation of eosinophils, increased mucus secretion, pulmonary edema, bronchial obstruction, and fibrosis (Olin & Wechsler, 2014; Park et al., 2016). Immune cells are key mediators in the activation of immune system with subsequent inflammatory cells infiltration which leads to induction of asthma (Postma & Rabe, 2015). Among the inflammatory cells, eosinophils are believed to perform a remarkable role in causing damage of the pulmonary epithelial cells. About 60% of asthma patients are eosinophilic phenotype, whereas only 25%−30% are non-atopic eosinophilic (Froidure et al., 2016).
Environmental factors such as duration and dose of allergen exposure along with genetic susceptibility influence the severity of asthma (Maspero et al., 2022). The genetic predisposition in susceptible patients triggers IgE antibodies, which tend to be major risk factor of asthma (Holgate et al., 2007). Allergic reactions induced during asthma elicit the penetration of leukocytes such as eosinophils, T helper 2 cells, and neutrophils (Cohn et al., 2004; Kay, 2001). At present, the most prescribed drug for asthma is corticosteroid inhalers, which provide immediate relief by ailing the symptoms (Papi et al., 2020). Short-acting beta-agonists are one of the most commonly used corticosteroid that effectively relaxes the bronchoconstriction. The major drawback of this therapy is its usage; most patients prefers to use it during critical condition that results in over usage and this increased the risk of asthma-associated mortalities (Suissa et al., 2000; Suissa et al., 2002). Hence, it is necessary to detect a drug that alleviates the disease and not the symptoms.
Adherence is the crucial factor in allopathic treatment since the asthma symptoms worsen if the treatment is not taken regularly. Phytomedicines are the potent alternative for these drugs as adherence is not required and it does not have any side effects (Bender, 2002). Plumbagin (5-hydroxy-2-methyl-1, 4-naphthoquinone) is the major phytoconstituent that exists in the roots of Plumbago zeylanica L, which belongs to the family of Plumbaginaceae family (Arunachalam et al., 2010; Yin et al., 2020). Plumbagin possesses numerous pharmacological properties such as antibacterial (Kaewbumrung & Panichayupakaranant, 2014), antifungal (Gwee et al., 2014), anti-inflammatory (Luo et al., 2010), anticancer, antimalarial, antihyperglycemic, antioxidant (Tan et al., 2011), and anti-artherosclerosis (Shukla et al., 2021). However, the salutary properties of the plumbagin against the allergic asthma was not scientifically disclosed yet. Therefore, in this study, we assessed the potency of plumbagin against the OVA allergen-induced asthma mouse model via inhibiting inflammatory response.
Materials and Methods
Chemicals
Plumbagin, ovalbumin (OVA), aluminum hydroxide were procured from Sigma Aldrich Chemicals, USA.
Experiment Animals
The 6–8-week-aged female BALB/c mice were acclimatized in a standard animal laboratory condition, as prescribed by the ethical committee, for a week before the initiation of experiment and were fed with laboratory rat chow and drinking water ad libitum. The whole experiment protocol followed in this work was reviewed by the ethical committee and it was approved. The animals were treated with utmost care and concern.
Induction of Allergic Asthma
The induction of allergic asthma was done in mice according to the protocol of Nader et al. (2012). The mice were treated with a 200-µl suspension consisting of 100 µg/mL of ovalbumin and 10 mg/mL aluminum hydroxide Al(OH)3 dissolved in 0.9% sterile buffered saline solution through intraperitoneal injection on day 1 and day 10. The mice were placed in 25 × 13 × 13 cm closed chamber and challenged with 3% aerosolized ovalbumin for a period of 20 min daily from day 19 to day 24 of the experimental period.
Animal Grouping
The acclimatized healthy female BALB/c mice were distributed into five groups as each group consists of six mice.
Group I (Control) – The control rats were treated with 2% DMSO and sensitized with phosphate buffered saline from 19th to 24th day of treatment.
Group II (Asthma induced) – The asthma was induced as per the protocol specified in the above section.
Group III (PB25) – The asthma was induced and then treated with 25 mg/kg of plumbagin dissolved in DMSO through oral gavage from 19th to 24th day of treatment.
Group IV (PB50) – The asthma was induced and treated with 50 mg/kg of plumbagin from 19th to 24th day of treatment.
Group V (Standard drug control) – The asthma was induced and treated with 50 mg/kg dexamethasone from 19th to 24th day of treatment.
The mice were weighed before and on completion of the treatment period, the changes in the body weight and lung tissue were measured to calculate the lung index. The mice were subjected to collection of bronchoalveolar lavage fluid (BALF) and then euthanized for the dissection of lung tissue. Blood samples were collected for the analysis of biochemical parameters.
BALF Collection
On 25th day, 24 h after the last treatment, the mice were anesthetized with 10% chloral hydrate and subjected to collection of BALF. The entire procedure was performed in an animal surgical room ensuring hygienic condition. The mice were fixed on to the surgical table and an incision was made on the neck skin. The muscles layers were detached and the trachea was exposed carefully. To the exposed trachea, 1 mL of ice-cold PBS was instilled and the BALF was collected. It was repeated thrice and the BALF collected was subjected to centrifugation at 2,000 rpm for 5 min. Supernatant was collected and analyzed for eotaxin and OVA-specific IgE antibody.
Detection of Eotaxin and OVA-IgE Concentrations
The level of eosinophilic specific chemoattractant and OVAspecific IgE concentrations in the BALF were estimated using the ELISA kits(MyBiosource, USA). Samples were diluted and the standards were prepared according to the kit manual instruction. Then, 100 µl of standard and samples were mixed to the precoated ELISA plates for 2 h at 37°C and 100 µl of 1X biotin antibody was mixed to the ELISA plates for 1 h at 37°C. After the incubation period, the solutions were agitated and the wells were washed gently thrice using buffer. Then, 100 µl of 1X HRP-avidin was mixed for 1 h at 37°C and the solution was aspirated and washed thrice with buffer. Further, 90 µl of TMB substrate was mixed to the wells and sustained for 15−30 min at 37°C. Finally, 50 µl of termination buffer was mixed to the plates and the absorbance was measured at 450 nm.
Assessment of Total and Differential Cell Count
The cell pellet obtained from the centrifuged BALF was assessed for the differential and total cell count in experimental animals. The cell pellet were re-suspended with 200 µl of phosphate buffered saline and loaded on to the hemocytometer to count the total leukocyte count. The cells were stained with Wright-Giemsa stain to assess the differential count of leukocytes. For each slide, 200 cells were counted and recorded.
Quantification Inflammatory Markers
The level of tumor necrosis factor (TNF-α), IL-12, IL-6, and thromboxane B2 (TBX2) were estimated in serum of experimental animals. The contents were determined using the assay kits procured from MyBiosource, USA. The procedure was performed as per the guidelines provided in the kit and the experiment was performed in triplicates.
Quantification of Nitric Oxide
The levels of nitric oxide (NO) were investigated by measuring the levels of nitrite and nitrate concentration. The lung tissue homogenate was prepared with PBS buffer and the supernatant was used for the estimation of NO. The NO was measured using the NO and nitrate/nitrite kits were purchased from R&D systems. The NO concentration was measured by converting nitrate to nitrite using nitrate reductase and the nitrite further reacts with sulfanilic acid that produces diazonium ion. The final product was measured at 540−570 nm.
Quantification of Myeloperoxidase Activity
Myeloperoxidase, which is a marker of neutrophil infiltration, was quantified by the method of Goldblum et al. (1985). The lung tissue homogenate was prepared using hexadecyltrimethylammonium bromide (HTAB) buffer and subjected to centrifugation. The hydrogen peroxide-dependent oxidation of supernatant was dianisidine hydrochloride and was measured at 460 nm to determine the myeloperoxidase activity.
Estimation of Oxidative Stress Markers
Oxidative stress marker malondialdehyde, antioxidants glutathione, and superoxide dismutase were measured in the experimental animals. Lung tissue homogenate was prepared using 150 mM KCl and the supernatant were utilized for the estimation of oxidative stress.
Lipid Peroxidation Estimation
The levels of lipid peroxidation in lung tissue of experimental animals were estimating by measuring the levels of malondialdehyde using the procedure of Ohkawa et al. (1979). To 100 µl of homogenate, 200 µl of 8.1% SDS, 1.5 mL 20% acetic acid, and 1.5 mL of 0.8% TBA were mixed and the pH was adjusted to 3.5. The 4 mL volume was made with deionized water and 5 mL of n-butanol and pyridine (15:1, v/v) mixture. The solution was vigorously shaken and subjected to centrifugation at 5000 rpm for 10 min. The supernatant was collected and the absorbance was measured at 532 nm.
Glutathione Estimation
Glutathione levels were estimated in the lung tissue homogenate of experimental animals using the Ellman’s technique prescribed by Ali et al. (2021). The supernatant was added to DTNB Ellman’s reagent and maintained for 10 min at 37°C. The final absorbance of the mixture was measured at 412 nm.
Superoxide Dismutase Estimation
Superoxide dismutase activity was estimated using the protocol of Fridovich (1983). The red formazan crystals formed by superoxide radicals reacting with p-iodonitrotetrazolium violet were measured to detect the SOD activity. The absorbance was measured at 505 nm.
Quantification of Proinflammatory Cytokines
The status of proinflammatory cytokines IL-4, IL-5, IL-13, and INF-γ was quantified in the lung tissue homogenate using commercially available ELISA kit procured from MyBiosource, USA. The tests were done using the protocol of manufacturer and the experiment was done in triplicates.
Lung Histology
Lung tissues dissected from the experimental mice were gently cleaned with PBS and fixed using 4% paraformaldehyde for 24 h. The formaldehyde-fixed lung tissues were dehydrated using gradient concentration of alcohol. The dehydrated tissue was transparentized with xylene and embedded in paraffin wax. The tissue blocks were sectioned into sections of 4-micron thickness using microtome. The sections were deparaffinized and stained with hematoxylin and eosin stains. The stained lung tissue sections were observed under light microscope.
Statistics
Statistical analysis of the obtained data was done with statistical software GraphPad Prism. Analysis of variance and Tukey’s post hoc assay were performed to estimate the statistical difference of individual groups. The values were represented as mean ± SEM. p < 0.05 and p < 0.01 was set as significant.
Results
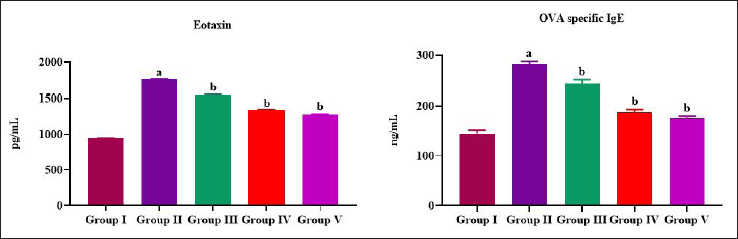
Plumbagin Attenuates the Eosinophilic Attractant and Allergen-specific Antibody in Allergic Asthma-induced Mice
Eotaxin, an eosinophilic chemoattractant protein, was quantified in the BALF of allergic asthma-induced untreated mice and plumbagin-treated mice (Figure 1A). Asthma induction with ovalbumin and AlCl3 increased the levels of eotaxin drastically to 1520 ± 2.5 pg/mL, which was only 980 ± 1.8 pg/mL in control rats. Plumbagin treatment significantly decreased the levels of eotaxin in allergic asthma-induced rats in a dose-dependent manner (1240 ± 3.2, 1120 ± 2.8 pg/mL simultaneously). Figure 1B depicts the levels of ovalbumin allergen-specific IgE antibody in the BALF of asthma-induced untreated mice and plumbagin-treated mice. Plumbagin treatment significantly inhibited the ovalbumin-induced IgE antibodies production in mice. The concentration of 25 mg/kg and 50 mg/kg of plumbagin-treated mice showed 185 ± 1.4 and 170 ± 1.7 ng/mL, respectively whereas it 235 ± 2.1 ng/mL of OVA-specific IgE antibodies was quantified in allergic asthma-induced untreated rats. No significant variations were noted on the status of eotaxin and OVA-specific IgE antibodies between the standard drug dexamethasone treated and the control mice.
Plumbagin Prevents Inflammatory Cell Infiltration in BALF of Allergic Asthma-induced Mice
Figure 2A depicts the total leukocytes count in BALF of allergic asthma-induced untreated and plumbagin-treated mice. Ovalbumin treatment significantly doubled the number of leukocytes count when compared to control mice. Asthma-induced untreated mice showed 44 ± 0.3 × 10−5 cells/mL whereas it is only 18 ± 0.4 × 10−5 cells/mL of leukocytes in control mice. Plumbagin treatment significantly decreased the leukocyte infiltration in BALF. Further, 25 mg/kg plumbagin-treated mice showed 38 ± 0.5 × 10−5 cells/mL and 50 mg/kg plumbagin-treated mice showed 34 ± 0.5 × 10−5 cells/mL.
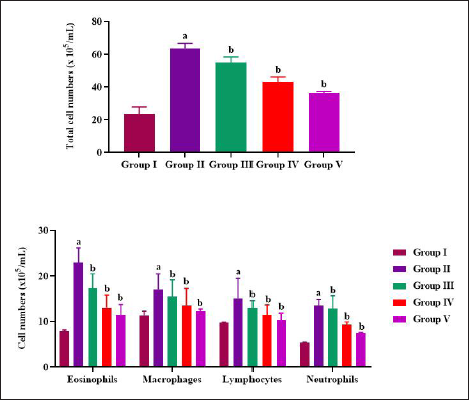
Differential count of leukocytes were counted in BALF of allergic asthma-induced untreated and plumbagin-treated mice and the results were illustrated in Figure 2B. In comparison to macrophages, it was observed that neutrophils and lymphocytes drastically increased in eosinophil count in the asthma-induced untreated rats. Ovalbumin significantly doubled the eosinophils count in BALF in comparison to control mice. Plumbagin substantially depleted the infiltration of all the inflammatory cells, eosinophils, neutrophils, macrophages, and lymphocytes.
Plumbagin Inhibited Synthesis of Allergic Inducers in Allergic Asthma-induced Mice
Figure 3A illustrates the status of TNF-α in serum of allergic asthma-induced untreated and plumbagin-treated mice. In comparison to control mice, the TNF-α (32 ± 0.08 pg/mL) was significantly increased in asthma-induced untreated mice (66 ± 0.05 pg/mL). In comparison to 25 mg/kg bwt plumbagin-treated mice (58 ± 0.1 pg/mL), 50 mg/kg plumbagin-treated mice (38 ± 0.07 pg/mL) showed substantial diminution in the TNF-α level.
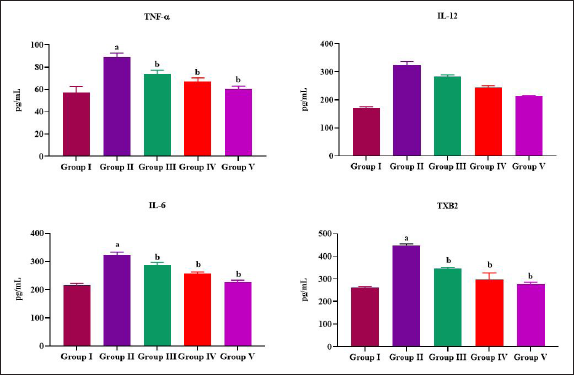
Plumbagin treatment significantly decreased both proinflammatory cytokines IL-12 and IL-6 in allergic asthma-induced rats (Figure 3B and C). The reduction of proinflammatory cytokines was in dose-dependent manner. Further, 25 mg/kg bwt plumbagin-treated mice showed 172 ± 0.9 pg/mL of IL-12 and 174 ± 0.5 of IL-6, whereas it was 160 ± 0.07 pg/mL and 162 ± 0.07 in 50 mg/kg bwt plumbagin-treated mice. In comparison to control mice, both IL-12 and IL-6 (156 ± 0.7 and 152 ± 1.2 pg/mL) levels were significantly increased in allergic asthma-induced untreated mice (245 ± 1.2 and 218 ± 1.5 pg/mL, respectively). Dexamethasone remarkably diminished the status of IL-12 and IL-6 (153 ± 0.9 and 148 ± 0.8 pg/mL) when compared to the control mice.
TBX2 was quantified in the serum of allergic asthma-induced untreated and plumbagin-treated mice and the results were depicted in Figure 3D. Ovalbumin treatment significantly increased the levels of TBX2 (452 ± 0.3 pg/mL) in the serum of mice when compared to the control (286 ± 0.5 pg/mL) mice. Plumbagin treatment significantly decreased the levels of TBX2 in both the treatment doses of 25 and 50 mg/kg bwt (393 ± 0.4, 340 ± 0.07 pg/mL). In comparison to control mice, dexamethasone-administered mice showed remarkable suppression in the levels of TBX2 (245 ± 0.06 pg/mL).
Plumbagin Treatment Prevented Lung Edema in Allergic Asthma-induced Mice
Figure 4A depicts the lung weight index of allergic asthma-induced untreated and plumbagin-treated mice. Ovalbumin treatment showed significant increase in lung weight index of 0.46 ± 0.009 when compared to the control mice whose lung weight index was 0.29 ± 0.005. Plumbagin treatment significantly decreased the lung weight index to 0.36 ± 0.006 in 25 mg/kg bwt dose and 0.32 ± 0.008 in 50 mg/kg bwt dose. Dexamethasone treated showed decreased lung weight index of 0.24 ± 0.01, which is significantly lesser than the all other groups.
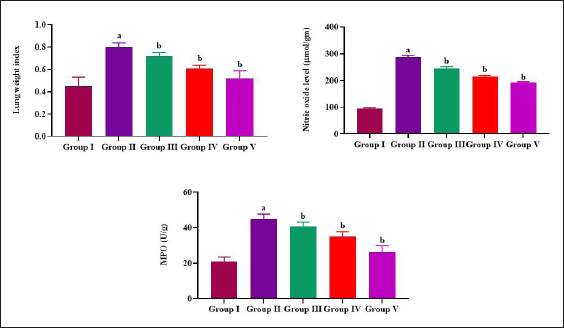
Plumbagin Treatment Decreased NO and MPO Levels in Allergic Asthma-induced Mice
Ovalbumin treatment significantly increased the NO and MPO activity (238 ± 1.3 µmol/g and 24 ± 0.03 U/g respectively) when compared to the control mice (100 µmol/g and 8.7 ± 0.009 U/g respectively). Plumbagin treatment significantly decreased the NO levels and MPO activity to 178 ± 0.8 µmol/g and 18 ± 0.05 U/g, respectively, in 25 mg/kg bwt treated and 162 ± 1.1 µmol/g and 15 ± 0.07 U/g in 50 mg/kg bwt-treated mice. In comparison to plumbagin-treated mice, the standard drug dexamethasone-treated mice show significant reduction in both NO levels and MPO activity (Figure 4B and C).
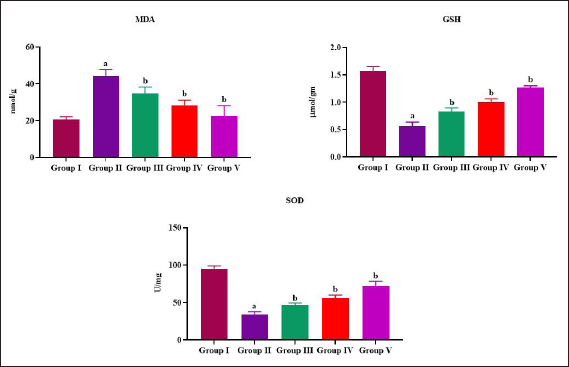
Plumbagin Treatment Scavenged Oxidative Stress in Allergic Asthma-induced Mice
The mice challenged and sensitized with ovalbumin significantly decreased the levels of antioxidants glutathione and superoxide dismutase when compared to the control mice. It also increased the lipid peroxidation that was evidenced by increased levels of MDA in allergic asthma-induced mice. Plumbagin appreciably elevated the status of both glutathione and SOD and decreased the MDA in asthma-induced mice (Figure 5).
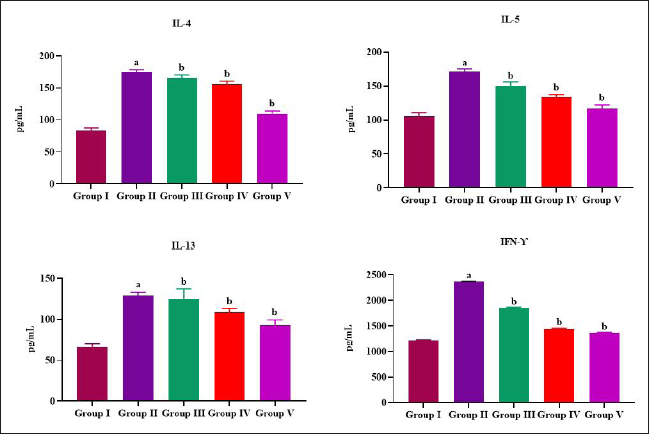
Plumbagin Treatment Reduced Proinflammatory Cytokines in Allergic Asthma-induced Mice
Figure 6 represents the status of proinflammatory cytokines IL-4, IL-5, IL-13, and INF-γ in the lung tissue of allergic asthma-induced mice. In comparison to control mice (84 ± 0.008, 98 ± 0.009, 56 ± 0.007 pg/mL, respectively), the untreated allergic asthma-induces mice showed substantial elevation in the status of IL-4, IL-5, and IL-13 (134 ± 0.04, 152 ± 0.05, and 94 ± 0.02 pg/mL, respectively). Plumbagin remarkably depleted the status of IL-4, IL-5, and IL-13 in both 25 mg/kg (128 ± 0.02, 137 ± 0.03, and 81 ± 0.04 pg/mL, respectively) and 50 mg/kg treatment (98 ± 0.01, 122 ± 0.04, and 75 ± 0.06 pg/mL, respectively). Plumbagin treatment also significantly reduced the levels of INF-γ to 1750 ± 2.4 pg/mL in 25 mg/kg bwt and 1220 ± 1.8 pg/mL in 50 mg/kg bwt-treated mice when compared to the allergic asthma-induced untreated mice (2195 ± 2.2 pg/mL).
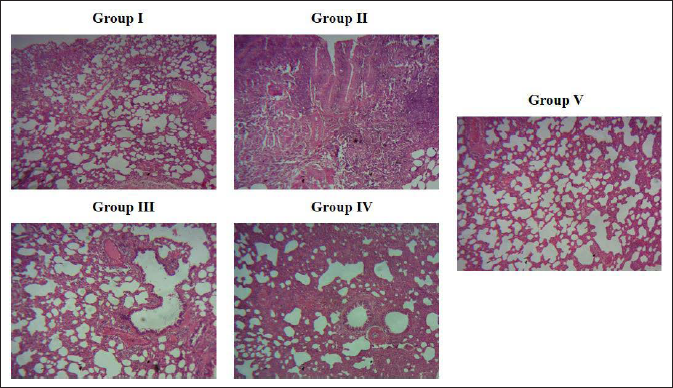
Plumbagin Ameliorated Allergic Asthma-induced Lung Tissue Injury in Mice
Figure 7A depicts the representative image of H&E stained lung tissue section of control group, which shows normal lung tissue histology with normal pulmonary alveoli; whereas allergic asthma-induced untreated mice showed thickened alveoli wall with increased inflammatory cells infiltration and perivascular edema (Figure 7B). Both 25 and 50 mg/kg bwt of plumbagin-treated mice lung tissue sections show decreased number of inflammatory cells infiltration (Figure 7C and D). In comparison to 25 mg/kg bwt of plumbagin-treated mice, the 50 mg/kg bwt plumbagin and standard drug dexamethasone-treated mice lung section show decreased alveoli septal wall thickness and considerably decreased perivascular edema. (Figure 7D and E).
Plumbagin Ameliorated Allergic Asthma-induced Lung Tissue Injury in Mice. H&E Stained Lung Tissue Representative Images of Group I: Control, Group II: Asthma induced, Group III: Asthma + 25 mg/kg Plumbagin-treated, Group IV: Asthma + 50 mg/kg of Plumbagin Treated, Group V: Asthma + Dexamethasone-treated. Control – Normal Control Mice, Asthma-induced – Mice-induced with Allergic Asthma with Ovalbumin + Aluminum Hydroxide, PB25 – Asthma-induced Treated with Plumbagin 25 mg/kg bwt, PB50 – Asthma-induced Treated with Plumbagin 50 mg/kg bwt, and SDG – Standard Drug Control Mice Asthma-induced Treated with Dexamethasone.
Discussion
Asthma is a non-communicable chronic inflammatory disease with evident heterogeneity in pathophysiology, etiology, and symptoms; it is classified as atopic and non-atopic asthma (Lötvall et al., 2011). The pathophysiology of asthma was assorted and multifaceted and it depends on the host and environment (Papi et al., 2018). Bronchoconstriction causing wheezing, cough, shortness of breath, and chest tightness that occurs in episodes (WHO, 2017). Ongoing treatments aim to alleviate these symptoms and it requires huge expenditure on the treatment. Even though enormous amount was spent to treat asthma, the control of disease is still questionable and there is a steady increase in asthma-related morbidity (Ishizaka et al., 1966). Therefore, we analyzed the potency of a phytoconstituent plumbagin against treating allergic asthma in mice model.
IgE immunoglobulins possess a distinctive property when compared to other isotypes that act rapidly and induce the immunological cells. IgE status was reported to be significantly increased in the atopic asthma patients (Ishizaka et al., 1966; Palomares et al., 2017). The binding of IgE on to the FCεRI receptors of dendritic cells increases the antigen presenting ability of dendritic cells (Matucci et al., 2018; Schroeder et al., 2010). In our study, the allergen ovalbumin treatment significantly increased the status of OVA-specific IgE antibodies and the plumbagin treatment depleted the status of OVA-specific IgE. This confirms the inhibitory property of plumbagin against IgE production induced by the allergen ovalbumin. Plumbagin treatment had also significantly decreased the status of eotaxin in the BALF of allergen ovalbumin-challenged mice. Eotaxins are chemotactic cytokine attractants that are produced by the epithelial cells of airway passage upon cytokine stimulation (Vatrella et al., 2022). These eotaxins present in asthmatic airways induce the chemotaxis of eosinophils, which cause bronchi constriction and leads to pulmonary damage (Nakamura et al., 1999). Plumbagin significantly inhibited the synthesis of eotaxin, and thereby decreased the eosinophilic cells infiltration in ovalbumin allergen-treated mice.
In severe asthma patients, eosinophilic-induced airway inflammation was reported and this signifies the association between the eosinophils and the asthma severity (Bousquet et al., 1990; Garcia et al., 2013; Price et al., 2015). Activated eosinophils cause hyperresponsiveness in the airways, which increase the mucus production causing persistent inflammation. These eosinophils are activated by Th2 cytokines such as IL-4, IL-5, and IL-13, which attract the eosinophils and thereby induces the eosinophilic infiltration (Lee et al., 2019; Peebles & Aronica, 2019; Schatz & Rosenwasser, 2014). Therefore, at present, researches were focused on suppressing the Th2 cytokines to treat allergic asthma (Gandhi et al., 2018; Leόn, 2017). Plumbagin treatment significantly decreased the status of IL-4, IL-5, and IL-13, and thereby diminished the eosinophilic infiltration in ovalbumin-treated mice. Plumbagin treatment decreased levels atopic inducing cytokines but also reduced the status of INF-γ which is associated with non-atopic asthma (Farrar & Schreiber, 1993).
Reports suggests that asthma patients show increased levels of serum Il-6 when compared to the active non-smoker control individuals (Tillie-Leblond et al., 1999; Yokoyama et al., 1995). IL-6 along with IL-1β and TNF-α were considered as biomarkers for various inflammatory diseases (Dejager et al., 2015; Kishimoto, 2010). Plumbagin treatment remarkably diminished the IL-6, IL-12, and TNF-α levels and this proves the anti-inflammatory effect of plumbagin against ovalbumin allergen (Dogné et al., 2002).
Plumbagin, a potent antioxidant, significantly increased the levels of antioxidants glutathione and superoxide, and thereby prevented the lung tissue from lipid peroxidation. It also decreased the levels of NO and myeloperoxidase activity in ovalbumin-treated mice lung tissue. Myeloperoxidase is a leukocyte-derived enzyme and it catalyzes the generation of reactive oxygen species (Ndrepepa, 2019). Plumbagin significantly decreased the levels of leukocytes in ovalbumin-treated mice and this may be reason for reduced myeloperoxidase activity in ovalbumin-treated lung tissue. The lung weight index and the histopathological analysis of plumbagin-treated allergic asthma-induced mice lung tissue confirm the ameliorative effect of plumbagin against allergen-induced asthma.
Conclusion
In this study, we effectively induced allergic asthma in BALB/c mice using the allergen ovalbumin. The phytochemical plumbagin was tested for its ability to alleviate allergic asthma in mice. Plumbagin treatment significantly decreased the levels of OVA-specific IgE and eotaxin, thereby preventing the synthesis of inflammatory cytokines and alveolar damage, which are responsible for the bronchoconstriction and other clinical manifestations of atopic asthma. It also reduced non-atopic asthma markers, demonstrating plumbagin’s protective role in both allergic and non-allergic-induced asthma. Plumbagin may be a potent alternative drug to treat asthma patients.
Summary
Asthma is a major non-communicable chronic inflammatory disease that affects the population of all age groups.
Environmental factors like duration and dose of allergen exposure along with genetic susceptibility influence the severity of asthma.
Plumbagin possesses numerous pharmacological properties such as antibacterial, antifungal, anti-inflammatory, anticancer, antimalarial, antihyperglycemic, antioxidant, and anti-artherosclerosis.
Plumbagin treatment significantly decreased the levels of OVA-specific IgE and eotaxin, and thereby prevented the synthesis of inflammatory cytokines and alveolar damage that are responsible for the bronchoconstriction and other clinical manifestations of atopic asthma.
Footnotes
Abbreviations
TBX2: Thromboxane B2; HTAB: Hexadecyltrimethylammonium bromide; OVA: Ovalbumin; BALF: Bronchoalveolar lavage fluid; TNF-α: Tumor necrosis factor.
Acknowledgments
This work was supported by the Respiratory Intensive Care Unit II, Weifang Peoples Hospital, 151, Guangwen Street Weifang City, Shandong Province, 261000, China.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The entire experimental protocol followed in this work was reviewed by the ethical committee
Funding
The author received no financial support for the research, authorship and/or publication of this article.
