Abstract
Background
Biosynthesized copper nanoparticles (CuNPs) are eco-friendly, cost-effective, and biocompatible agents reported for extensive biomedical and bioengineering applications. Different chemical synthesis approaches have been established recently, with challenges of higher toxicity and high cost involved in the synthesis process. Green synthesized nanoparticles emerged and was extensively reported to address the challenges faced by traditional chemical synthesis processes. However, the high toxicity remains a significant challenge for translating the green synthesized nanoparticles into clinically valuable products.
Purpose
To synthesize, characterize, and evaluate the citrus extract-based CuNps cytotoxicity against JURKAT cell lines.
Materials and Methods
An aqueous extract of the citrus fruit was used as a reducing agent, and the CuNps were synthesized. Fourier Transform Infra-Red (FTIR), scanning electron microscopy (SEM), dynamic light scattering (DLS), Ultraviolet-visible (UV-vis) spectrophotometry, and X-ray diffraction (XRD) were used to confirm the synthesis of CuNp and its structure. Furthermore, the effect of CuNPs on cell viability and toxicity was evaluated by mitochondrial toxicity tests (MTT) and LDH assays against the JURKAT cell lines.
Results and Conclusion
The synthesized nanoparticle’s size ranged from 40 to 70 nm, as confirmed through nanoparticle tracking analysis (NTA) and SEM. The synthesized nanoparticles were confirmed to be anti-proliferative with a high percent of cytotoxicity as found from MTT and LDH leakage assays. The size and shape of the synthesized CuNPs as studied by SEM were found to be 30–70 nm and more or less spherical. MTT reported 64.87% inhibition at 320 µg/mL with an IC50 value of 80.78 µg/mL (p < 0.05). Cytotoxicity as measured by the LDH assay was found to be 53.12 ± 0.89% at 320 µg/mL with an IC50 of 23.12 ± 0.39 when compared to the positive control (11.21 ± 0.15 µg/mL). Green-synthesized CuNPs exhibited potential anti-cancerous activity in JURKAT cell lines, as evidenced by the LDH and MTT assays.
Introduction
Over the past two decades, the material science domain, in conjunction with biotechnology, has emphasized nanoparticles due to their tunable size, shape, and optical properties, thus diversifying their functionalities and applicability in the biomedical field (Abdullah et al., 2015). Numerous studies have been done on nanoparticles and their numerous uses in biosensing and chemotherapy, energy-saving devices, drug delivery mechanisms, electrochemistry and catalysis, optoelectronics, optics, and pharmaceutical therapeutics such as antibacterial, anticancer, and antimicrobial agents (Aruoma et al., 2012). Advantages of their unique catalytic properties include their nanoscale size, distinctive shape, and large surface area. Additional surface modifications for the materials are made using post-functionalization methods to produce distinctive physical properties (Boyd et al., 2018). It is being evaluated whether to use top-down or bottom-up ways to synthesize NPs using a variety of physical and chemical techniques (Campos et al., 2015). However, the majority of synthetic methods call for extreme temperatures, pressures, and powerful reducing agents, which generate harmful byproducts in situ and are forbidden by green protocols (Dobrucka et al., 2021; Elemike et al., 2020). Several situations call for expensive and difficult methods. As a result, the need for environmentally benign nanotechnology is growing to enable its advantages (Boyd et al., 2018).
Green synthesis is a reliable, eco-friendly, and sustainable method that avoids the generation of harmful by-products. Among all these, plant extract is mainly used to synthesize metal oxide nanoparticles because it requires less time, is a straightforward procedure, and is cost-effective (Srimurugan et al., 2020). The nanoparticles synthesized using plant extract by the green method are also known as biogenic nanoparticles (Kong et al., 2011).
The way to expand a green procedure is to modify a gentle synthesis proposal that uses innocuous reactions to create a wide variety of progressive nanostructures matching the refinement of ongoing engineered materials. Plant bioactives serve as an easy, natural, cost-effective, and comprehensive scale manufacturing method for bioabsorbable nanoparticles (Eppert et al., 2011). Plant biodiversity contains many phytochemicals or secondary metabolites, mainly in leaves. These are phenols, flavonoids, terpenoids, amides, aldehydes, and ketones (Fafal et al., 2017).
Leukemia is the sixth most prominent cause of death in humans and causes high mortality all over the world. T-cell acute lymphoblastic leukemia (T-ALL) is an aggressive malignancy that does not respond well to chemotherapy. One study said it depends biologically on the protein degrading process in the mucosa-associated lymphoma translocation protein 1 (MALT1) protease (Gopinath et al., 2008). A therapeutic approach like intense chemotherapy did not get enough success in treating leukemia as there is leukemic cell resistance due to significant doses of chemical drug therapy, a big tumor, and threats imposing biological impediments (He et al., 2011). Especially in children and adults, the development of leukemia may be acute or chronic, and it is most commonly observed in the blood near the marrow and organs that produce blood (Elemike et al., 2020). Generally, myeloid leukemia involves granulocytes (basophils, neutrophils, and eosinophils) and myeloid cells, whereas lymphocytic leukemia includes B and T lymphocytes (Hu et al., 2016). Jurkat cells are an immortalized line of human T lymphocyte cells that are used to study acute T cell leukemia, T cell signaling, and the expression of various chemokine receptors susceptible to viral entry, particularly HIV. Jurkat cells can produce interleukin 2 and are used in research involving the susceptibility of cancers to drugs and radiation (Abraham & Weiss, 2004).
Recently, many researchers tried treating cancer using various anti-proliferative compounds like antimetabolites, plant derivatives, alkylating agents, protein kinase inhibitors, and cytotoxic antibiotics, inhibiting the proliferation of tumor cells or destroying them (Guo et al., 2014). Chemotherapy is a combination of many drugs that provides a significant reduction in many cancer cells (Jan et al., 2014). However, these approaches have yet to fully address the cancer menace.
Nanoparticles synthesized with green plant extracts are fascinating materials that have significantly built the therapeutic class of nanoparticles and nanomedicines (Jin et al., 2020). The green synthesis approach gained significant focus because it includes three crucial green elements: Ag, ZnO, and CuO, which are environmentally compatible with water systems without requiring solvents, naturally present high reducing agents, and possess safety over other elements (Kanase et al., 2020).
There are numerous possible uses for green nanoparticle synthesis in the biomedical and environmental sciences. Green synthesis specifically seeks to reduce the use of harmful chemicals. For instance, it is typically acceptable to use biological materials like plants. These plant compounds act as caps and reducing agents. Many medicinal plants and spices were used for the green synthesis. Gold, silver, copper, palladium, platinum, zinc oxide, and titanium dioxide are all examples of nanoparticles (Jadoun et al., 2021).
Nanoparticles with superior efficacy and selective targeting capabilities are attracting significant interest for targeted drug delivery for many diseases (Karade et al., 2018). Nanoparticles with metal oxides functionalized with plant biomolecules have numerous applications as drug portage in the medical field. The biological synthesis of copper nanoparticles (CuNPs) has become one of the options for chemical and physical synthesis of nanoparticles (Khashan et al., 2018). CuNPs may act as promising agents due to their properties like synthesis, high selectivity, biocompatibility, and enhanced cytotoxicity. Copper is one of the co-factors for many enzymes, and it plays an essential role in many cellular mechanisms (Laurent et al., 2008).
Citrus fruits possess innumerable bioactive compounds, including essential oils, flavonoids, vitamins, and phenolics, which have anti-oxidative, anti-inflammatory, anti-tumor, and anti-microbial properties (Lv et al., 2015). The total isolates, non-polar and polar extracts, and their formulations in combination showed immuno-pharmacological activity in the cancer research domain (Mosmann 1983). Citrus fruits possess polyphenolic compounds, including coumarins, limonoids, terpenoids, flavonoids, phenolic acids, tannins, carotenoids, and stilbenes (Guo et al., 2014). Citrus species were used in the green synthesis of silver, gold, and CuNPs. These synthesized nanoparticles were further evaluated for antioxidant, antibacterial, antifungal, and antiproliferative properties (Alkhulaifi et al., 2020). Green synthesized TeNPs with citrus exhibited significant antibacterial activity against gram-negative and gram-positive bacteria and also showed potential anti-cancerous effects against human melanoma cells with an IC50 of 50 µg/mL (Kelebek et al., 2009).
Thus, to address the existing challenges and develop a cost-effective nanoparticle against leukemia, we used a simple green synthesis method to create CuNPs. We tested their anti-cancer properties against JURKAT cell lines. The research regarding citrus-mediated NPs and their biomedical applications is limited, especially on copper-based NPs. This study aims to elaborate and understand the cytotoxicity against JURKAT lines with mitochondrial toxicity tests (MTT) and LDH leakage assays. Various analytical techniques such as the Fourier Transform Infra-Red (FTIR), X-ray diffraction (XRD), scanning electron microscopy (SEM), and nanoparticle tracking analysis (NTA) were employed to characterize the green-developed nanoparticle.
Materials and Methods
Materials and Reagents
Fresh citrus fruits (Citrus limonum) were collected from the local market in Bangalore, Karnataka, India. Copper oxide, fetal bovine serum (FBS), trypsin, antibiotics (penicillin/streptomycin), and RPMI-1640 medium were procured from Sigma-Aldrich, India. The human acute T-cell leukemic cell line (JURKAT; accession no. TIB-152) was purchased from the American Type Culture Collection (ATCC). MTT (3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide) reagent was bought from Sigma Aldrich, USA.
Preparation of C. limonum Extract
Fruits of C. limonum) were washed thoroughly, peeled, and cut into fine, small pieces. The samples were air-dried at room temperature and pulverized into a fine powder using a blender (Bakhshi et al., 2022). The powder suspension was mixed with distilled water (in a 1:10 ratio) and agitated at 37°C for 1 h in a shaker incubator at 150 rpm. Following filtration, the filtrate was stored at room temperature in the dark and used for green synthesis with CuNPs (CuO).
Green Synthesis of C. limonum CuNPs
Each 50 mL of citrus filtrate and copper oxide solution (20 mM) were mixed in a conical flask and agitated on a magnetic stirrer for about 24 h at 40℃ (Bakhshi et al., 2022). The suspension was checked for a color change to brown. Following the color change, the contents were centrifuged at 12,000 rpm for 15 min, and the pellet obtained was washed twice with deionized water and then lyophilized to a fine powder and used for further characterization by FTIR, SEM, dynamic light scattering (DLS), Ultraviolet-visible (UV-vis) spectrophotometry, and XRD to study the structure, composition, and morphology of the produced nanoparticles.
UV-vis Spectroscopic Analysis
Green synthesized CuNPs were observed for the color change, from colorless to dark brown. The particles were diluted with deionized water and screened for the absorbance peak using UV-vis spectroscopy (NanoDrop ND2000, Thermo Scientific, Waltham, MA, United States; 200–800 nm, resolution of 1 nm).
FTIR
To validate any effects of various phytochemicals present in the citrus fruit extract on the surface modification of the green synthesized CuNPs, FTIR (JASCO Ltd., Tokyo, Japan) was done in the wavelength range of 4,000–400 cm−1. A powdered sample was added to potassium bromide (to run the full mid-IR region down to 400 wave numbers and avoid split mulls) before evaluation.27
XRD
XRD analysis was used to identify the compound’s chemical composition and crystalline structure. Crystallographic analyses of the green synthesized CuNPs were performed using X-ray powder diffraction (XRD D8 Advance, Bruker, Madison, WI, USA) equipped with Cu Kα (λ = 1.540Å) as a radiation source and with a Ni filter. The diffraction pattern was observed over the 2θ range (10–700) at a scan speed of 0.4°/min. The diffractogram was later compared to the standard database of the International Center for Diffraction Data (ICDD).
DLS
DLS method was used to screen for the hydrodynamic size (Z average), polydispersity index (PDI), and surface charge (zeta potential) of the green synthesized CuNPs (Horiba SZ-100 analyzer, Kyoto, Japan). Particle size analysis was done at a scattering angle of 90° with a medium count rate of 210 kCPS at about 25°C.
The morphological investigation was done using SEM. The morphology of the green synthesized CuNPs was investigated for any formation of aggregates or agglomerates.
Cell Culture and MTT Assay
Human acute T-cell leukemic cell lines (JURKAT) were maintained in RPMI-1640 medium supplemented with 10% FBS, penicillin (100 µg/mL), streptomycin (100 µg/ml), and 2 mM L-glutamine. They were cultured in a humidified atmosphere with 5% CO2 at 37℃.
Mitochondrial Cytotoxicity Assay Was Done Using MTT Reagent
Cell viability was screened with an MTT assay. It works on the reduction principle of tetrazolium salt (MTT) to soluble blue color formazan crystals in active cells. This implicates the possible rate of metabolism within the treated and control cells. In this assay, 5 × 104 cells were seeded onto 96-well plates with 180 µL of RPMI-1640 medium and incubated in a CO2 incubator. Following 24 h of incubation, the medium was replaced with 100 µL of fresh medium, and 10 µL of MTT reagent (0.5 mg/ml) was added to each well and further incubated for 4 h. Cells of green synthesized CuNPs were treated with varying concentrations (10, 20, 40, 80, 160, and 320 µg/mL). Vinblastine (25 µM) was used as the positive control. Following incubation, the solution from the wells was discarded and added to 100 µL of dimethyl sulfoxide (DMSO) (Jin et al., 2020). The absorbance was recorded at 590 nm in a microplate reader (Spectramax I3X Plate Reader, Biorad).
LDH Assay for Testing Cell Cytotoxicity
The LDH leakage from cells following membrane damage was estimated using an LDH reagent cytotoxicity assay kit (HiMedia, India). In this assay, the enzyme released converts into a tetrazolium salt (iodonitrotetrazolium violet; INT), a red formazan product (Dobrucka et al., 2019). The amount of color formed is directly proportional to the number of lysed cells. Cells were cultured with varying concentrations (10, 20, 40, 80, 160, and 320 µg/mL) of green synthesized CuNPs as in the previous section. Vinblastine (25 µM) was used as the positive control. Following 24 h of incubation, the medium was replaced with 50 µL of fresh medium and 50 µL of LDH reagent (0.5 mg/ml) and further incubated for 30 min in the dark at room temperature. Following incubation, the absorbance was recorded at 490 nm using the ELISA plate reader (Genetix). The CuNPs-mediated cytotoxicity was calculated with the formula: % cytotoxicity = Experimental LDH release/Maximum LDH release. Cultural media background noise was nullified, and the half inhibitory concentrations (IC50) of green synthesized CuNPs were determined using the SPSS software.
Statistical Analysis
All the experiments were performed in triplicate, and the data were expressed as the mean ± SD of the triplicates (n = 3) and analyzed using the SPPS software.
Results
Characterization Assays for the Green Synthesized CuNPs
UV-vis Spectrophotometry
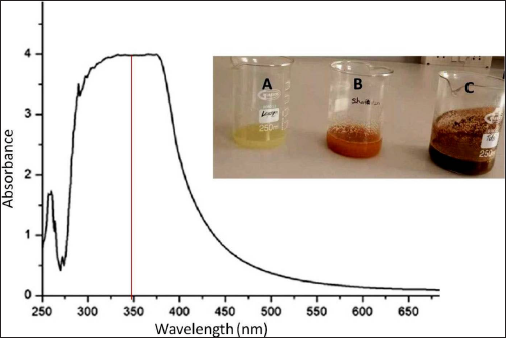
The process of CuNP synthesis was monitored by the change in color and UV-vis spectra. The color of the mixture changed from light brown to dark brown after adding the citrus extract for 24 h. The findings show that the intensity of the absorbance peaks steadily increased when reaction time was extended up to 24 h. CuNPs are well known for their reddish-brown hue in water, which results from activating the CuNPs’ excitation of the surface plasmon resonances (SPR) of CuNP.
The UV-vis spectra of synthesized C. limonum CuNPs were recorded at room temperature between 250 and 900 nm against distilled water as a reference. Due to SPR, CuNPs showed characteristics peaking at 350 nm (Figure 1).
FTIR Spectroscopy
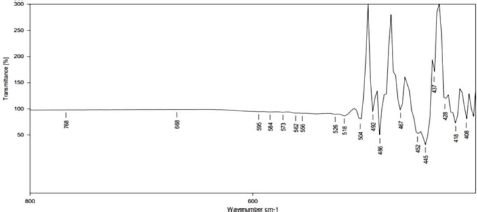
FTIR analysis determined the chemical change due to the interaction between the copper sulfate and functional groups found in the citrus extract. FTIR of the green synthesized CuNPs revealed the presence of nine bands at 492, 486, 467, 452, 445, 437, 428, 418, and 408 cm−1. The FTIR spectra of synthesized C. limonum nanoparticles were recorded between 500 and 400 cm−1 (Figure 2). This spectroscopy analyzed the different functional groups attached to nanoparticles. The peaks at 455.81 and 428.81 cm−1 correspond to the O–H bond.
XRD
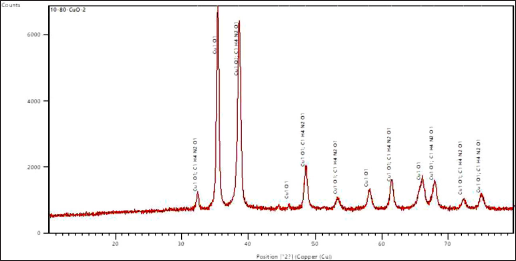
The crystalline phase and orientation of the created nanoparticles were examined using the XRD technique. The magnetite phase of the produced CuNPs was deduced from the XRD pattern. From the highest peak, the size of the nanoparticles was estimated. The peaks obtained are due to crystallized organic compounds on the nanoparticles (Bakhshi et al., 2022). The XRD pattern of green synthesized CuNPs is presented in Figure 3. The pattern clearly shows the prominent peaks at (2θ) = 32.4039 (342.83), 35.4024 (4487.21), 38.6348 (3826.77), 44.5811 (99.71), 46.1724 (79.23), 48.6342 (1055.76), 53.3641 (214.6), 58.0957 (441.58), 61.4685 (606.31), 65.9057 (649.75), 67.8711 (640.27), 72.2614 (212.09), and 75.017 (286.28). The planes of the respective peaks are shown in brackets. Based on the XRD spectra shown in Figure 3, the shape of the CuNPs was determined to be of face-centered cubic geometry.
DLS
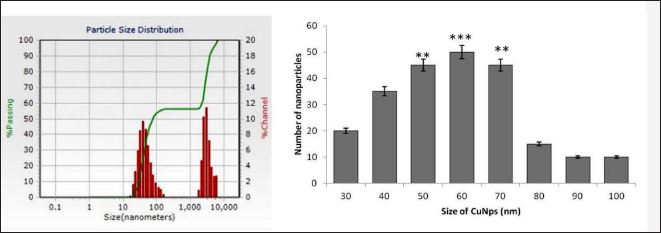
The NTA images showed that most of the synthesized nanoparticles have an average size diameter of 60 nm, which was calculated based on the Brownian motion of particles (Figure 4). The size and shape of the produced CuNPs were also investigated using SEM. The CuNPs were primarily scattered and more or less spherical. The particles ranged in size from 30 to 70 nm (Figure 4).
SEM
SEM was used to study the surface morphology of synthesized C. limonum CuNP. SEM analysis confirmed that synthesized C. limonum CuNP showed a stabilized nanocube-like structure (Figure 5). SEM analysis confirmed that the green synthesized CuNPs were cubical and polydispersed with a size range of 30–80 nm (mean size = 55.4 nm).

Evaluation of the Effect of Synthesized Nanoparticles in the JURKAT Cell Line
MTT was carried out with MTT reagent. The green synthesized CuNPs remarkably inhibited the cell viability of the cell lines in the study at concentrations of 25 and 80 µg/mL when compared to the positive control. The green synthesized CuNPs inhibited the cell lines from 46.33% to 64.87% at 80 and 320 µg/mL, respectively. On the contrary, positive control showed inhibition of the cell lines from 56.58 to 76.9 at 25 and 100 µg/mL, respectively (Figure 6). It is observed from Figure 6 that citrus-synthesized CuNPs demonstrated a reduction in viability with an IC50 value of 80.78 µg/mL when compared to control cells (p < 0.05) by MTT assay, indicating anti-proliferating activity of synthesized nanoparticles compared to the positive control that showed an IC50 value of 16.26 µg/mL (p < 0.05).
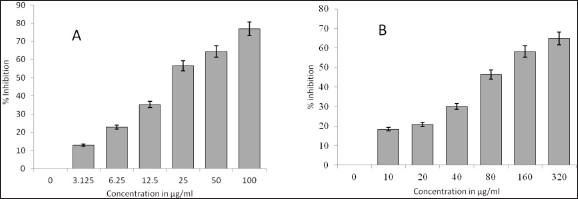
LDH Assay
Our findings from the LDH assay support the findings of the MTT assay. Total cytotoxicity was estimated by comparing the LDH levels secreted from the CuNPs-treated cells with the cellular LDH levels observed from cells treated with the positive control (100%). Our green-synthesized CuNPs exhibited an increase in the leakage of LDH in a dose-dependent manner. The cytotoxicity measured for the JURKAT lines was from 21.34 ± 0.56% to 53.12 ± 0.89% at 10 and 320 µg/mL (Figure 7). The IC50 was also calculated to estimate the cytotoxic effect of green-synthesized CuNPs. As calculated, the IC50 growth inhibition values were 23.12 ± 0.39 and 11.21 ± 0.15 µg/mL, respectively, for CuNPs and the positive control.
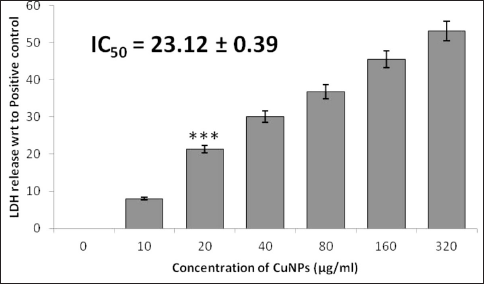
Discussion
Leukemia-related illnesses involve the proliferation and malignant cloning of blood progenitor cells. These cells predominantly form in the bone marrow (BM), from where they are spread to hematopoietic organs. Leukemia is said to increase the risk of infections that endanger human life and create some systemic symptoms, including anemia and bleeding (Khani et al., 2018).
In the present study, we developed a biosynthesis method for CuNP using citrus extract. The developed CuNPs were of size 30−70 nm and cubical shaped, as characterized by NTA and SEM analysis. The cytotoxicity evaluation by MTT and LDH assays demonstrated enhanced cytotoxic effects against leukemia cell lines. The citrus extract developed into CuNPs must be further explored against leukemia patient samples to explore cytotoxic activity and development as a potential anti-cancer agent.
In our present study, the C. limonum-conjugated CuNPs showed promising anti-tumor activity against JURKAT cell lines. The green-synthesized CuNPs exhibited efficient and potent inhibition of cell growth. The effective capping activity of bioactive compounds from citrus extract is indicated by the homogenous spherical form of the CuNPs produced by utilizing SEM. The creation of a single-phase cubic fluorite structure is confirmed by the fact that all of the peaks in the XRD pattern are well-indexed. No peaks are attributable to any contaminant, such as Cu and Cu2O. Tang et al. (2020) stated that, in order to comprehensively study the pure phase of the generated CuNPs sample, stretching and vibrational modes of molecular structures in the FTIR spectrum are studied, and the values of the FTIR spectrum confirm purely monoclinic morphology (Rajeshkumar, 2016; Tang et al., 2020). SPR peaks around 350 nm indicated the bio-reduction of copper oxide CuNPs. This is consistent with other investigations that discovered CuNP’s UV-vis absorbance ranges from 280 to 360 nm (Shende et al., 2015). This work studied the anti-cancer effect of green synthesized CuNPs from citrus extracts. The cytotoxicity of CuNPs was measured both by MTT and LDH assays.
The impact of the synthesized nanoparticles on the viability of the JURKAT cell line was estimated, and the green-synthesized CuNPs showed inhibition of the cell lines, indicating a reduction in viability of cells by up to 80.78% when compared to control cells (p < 0.05) by MTT assay. Green-synthesized CuNPs also exhibited an increase in the leakage of LDH in a dose-dependent manner. Our data also showed that the synthesized material showed anti-proliferative activity against JURKAT cells. The LDH assay also ensured the inhibition of cell viability in a concise way. This investigation showed that C. limonum-conjugated nanoparticles could be used as a preventative measure or a treatment for T-lymphocytic carcinoma. Commercial-scale methods that use the synthesis of an environmentally friendly method of producing nanoparticles should be supported because they can be employed in various industries such as foods, pharmaceuticals, and medical applications.
The regulation of different classes of histone deacetylases (HDACs) may be the cause of copper oxide nanoparticles (CuO NPs) promoting anticancer action (Kalaiarasi et al., 2018). Kalairasi A (2018) confirmed the potential toxicity of the CuNPs in the human chronic myelogenous leukemia cell-line K562. Their study also reported the possible effect of the NPs at the gene level, which coordinate apoptosis. Our study could thus possibly attack the cell lines and downregulate the proteins required for cancer cell proliferation.
Conclusion
Green chemistry is the most reliable, eco-friendly, and preferably biocompatible technique to synthesize extremely fine-size CuNPs. The current study exploited an eco-friendly safe and biogenic way of synthesizing CuNPs from citrus extract. Due to their small size of 50 ± 5 nm with a large surface area, the effect of NPs on MTT and LDH assays concludes positive anti-proliferative activity. We have experimentally proven that these synthesized CuNPs are potential anti-cancer agents, as verified by MTT and LDH assays. Therefore, in conclusion, the C. limonum-conjugated silver nanoparticles may exhibit considerable anti-carcinogenic activity and may act as valuable therapy options in treating leukemia-like cancers. The use of these biosynthesized CuO NPs in the pharmacological, clinical, and biotechnological fields may result from adequate reproduction of our results. It may be easier to understand how these NPs work in various areas if one is aware of their precise mechanism of action.
Footnotes
Abbreviations
CuNPs: copper nanoparticles; FTIR: Fourier Transform Infra-Red; SEM: scanning electron microscopy; XRD: X-ray diffraction; DLS: dynamic light scattering; NTA: nanoparticle tracking analysis; MTT: mitochondrial toxicity test; T-ALL: T-cell acute lymphoblastic leukemia; MALT1: mucosa-associated lymphoma translocation protein 1; FBS: fetal bovine serum; PDI: polydispersity index; DMSO: dimethyl sulphoxide; SPR: surface plasmon resonances; BM: bone marrow.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Ethical Approval and Informed Consent
This work does not involve animal testing and does not require ethical approval. Informed consent of all authors has been obtained for this work.
