Abstract
Background
One of the key factors limiting the effectiveness of chemotherapy treatment for malignancies is multidrug resistance (MDR). The MDR phenotype is related to P-glycoprotein (P-gp) expression and function. The main triterpenoid saponins generated from Bupleurum chinense DC (BCDC), saikosaponin A (SSa), has been found to have anti-tumor potential. Saikosaponin B (SSb) has the potential for utility in combination with anticancer drugs as the secondary saikosaponins.
Objective
In this study, we looked into the impact of SSa and SSb on doxorubicin (Dox)-resistant breast cancer cells and its underlying mechanisms.
Materials and Methods
Dox-resistant breast cancer cells (MCF-7ADR) and MCF-7 cells were used in the study. The experimental cells were divided into a different concentration SSa administration group, a different concentration SSb administration group, and a control group, and the related biochemical parameters of MCF-7 and MCF-7ADR cells were detected.
Results
We discovered that SSa and SSb both suppressed MCF-7 and MCF-7ADR cell proliferation in a dose-dependent manner. Additionally, SSa at 2.5 and 5.0 µg/mL and SSb at 3.0 and 7.0 µg/mL could significantly enhance the cytotoxicity of Dox and reverse MDR in MCF-7ADR cells. The combination of Dox and SSa or SSb induced obvious synergistic effects. SSa and SSb could increase the sensitivity of MCF-7ADR cells to Dox by decreasing P-gp expression, increasing intracellular accumulation, and delaying the drug efflux of rhodamine 123 (Rh123, a P-gp substrate). Additionally, SSa and SSb both induced G1-phase arrest in MCF-7ADR cells in the presence of Dox.
Conclusion
According to the study, SSa and SSb may be novel MDR reversal medicines for breast cancer chemotherapy and have significant therapeutic significance for MDR during tumor therapy.
Introduction
The second-leading cause of cancer-related death in women is breast cancer, a cancer in which women are most frequently diagnosed (Ullah, 2019). One of the main treatments for people with breast cancer has been chemotherapy. Doxorubicin (Dox) is an anthracycline antibiotic that has long been used to treat breast cancer. It is known to intercalate with DNA base pairs, bind to DNA-associated enzymes, and target several molecules to cause various cytotoxic effects (Tacar et al., 2013). However, harmful side effects, such as cardiotoxicity that might be fatal, severely restrict the use of Dox. Especially among the most significant obstacles to successful breast cancer chemotherapy is multidrug resistance (MDR) (Reis et al., 2020; Wang et al., 2016).
MDR refers to the cancer cells’ resistance to various unrelated cytotoxic substances, is the most significant barrier to effective treatment (Palko-Łabuz et al., 2019). Multiple processes, such as reduced drug uptake, increased drug efflux, activation of detoxification systems, activation of DNA repair pathways, and evasion of drug-induced apoptosis, can lead to the development of MDR (Li et al., 2017). The expression of a family of energy-dependent efflux pumps known as adenosine triphosphate (ATP)-binding cassette (ABC) transporters is one of the most prevalent mechanisms for cancer MDR (Wu et al., 2020). P-glycoprotein (P-gp) is the most characterized ABC transporter encoded by MDR1 (Gottesman & Ling, 2006). Several substances have been reported to successfully combat MDR when used as P-gp inhibitors in conjunction with other anticancer substances for treating tumors (Palmeira et al., 2012). However, the side effects of these medications limit their use in clinical settings. Therefore, searching for novel effective agents with low toxicity for overcoming MDR is required, especially research on finding P-gp inhibitors.
One of the main triterpenoid saponins obtained from Bupleurum chinense DC (BCDC) is called saikosaponin A (SSa), which exhibits the ability to induce cell apoptosis, inhibit cell proliferation, and enhance anti-tumor immunity in cancer cells (Kim & Hong, 2011; Wen-Sheng, 2003; Zhao et al., 2019). Contrarily, initially discovered as a minor saponin in BCDC, saikosaponin B (SSb) was known to be produced during the extraction of other components (Lin et al., 2013). It was discovered that when taken orally, saikosaponin A or D may be converted into Saikosaponin B1 or B2, respectively (Nose et al., 1989). So far, there is little known information about the chemo-sensitizing effect of SSa and SSb on tumor cells. The mechanisms underlying the chemo-sensitization effect of SSa and SSb on breast cancer cells need further research. In this study, we examined the ability of SSa and SSb to enhance the therapeutic efficacy of Dox. We explored the mechanism of sensitization to Dox by SSa and SSb in human breast cancer.
Materials and Methods
Drugs and Reagents
SSa and SSb powder (purity both >98%, Nanjing Jingzhu Biotechnology Co., Ltd, Jiangsu, China); Purified Dox (Hisun Pharmaceutical Co., Ltd, Zhejiang, China); Verapamil (Ver) (Hefeng Pharmaceutical Co., Ltd, Shanghai, China); MTT and Rhodamine123 (Rh123) (Sigma-Aldrich, St. Louis, MO, USA); cell cycle analysis kit (Nanjing KeyGen Biotech. Co, Ltd, Jiangsu, China); and fluorescein isothiocyanate (FITC)-labeled anti-human P-gp monoclonal antibody (eBioscience, San Diego, CA, USA).
Cell Culture
The Bogoo Biotechnology Co., Ltd. provided the human breast cancer cell line MCF-7 and its Dox-resistant equivalent, Dox-resistant breast cancer cell (MCF-7ADR) (Shanghai, China). Cells were cultured in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA), 100 U/mL penicillin, and 100 µg/mL streptomycin at 37°C in an incubator with humidified 5% CO2. To retain the MDR phenotype, the MCF-7ADR cells were grown with 1.0 µg/mL of Dox and eliminated 1 week before the experiment.
MTT Assay
Cell cytotoxicity and the MDR reversal assay were performed using the MTT method. In brief, cells were seeded into 96-well plates (8 × 103/well) overnight. After treatment with different concentrations of drugs for 48 h, 15 µL of MTT solution (5 mg/mL) was added to each well. To dissolve the blue formazan crystals, 150 µL of dimethyl sulphoxide (DMSO) was added after 4 h of incubation, and the supernatant was discarded. All tests were run in triplicate, and the optical density (OD) was measured at 570 nm using an enzyme immunoassay device. Inhibition rate (%) = [(OD of control group – OD of treated group)/OD of control group] × 100%. The medication concentration necessary to reduce cell survival to 50% is known as the IC50 value. MDR’s reversal fold (RF) was calculated by dividing the IC50 values for cells to Dox in the absence of modulators by those in the presence of modulators (Fu et al., 2004). By computing the combination index (CI) using the Chou-Talalay method, the combined effect was analyzed (Chou, 2010).
Real-Time qPCR Assay
Using the RNAiso Plus kit and the PrimeScriptTM (both from TaKaRa Biotechnology Co., Dalian, China) RT reagent kit, RNA isolation and reverse transcription were performed. SYBR Premix Ex TaqTM II (Takara) and a PikoReal 24 Real-Time PCR system were used to perform real-time PCR (Thermo Fisher Scientific, America). The MDR1 and GAPDH primer sequences (synthesized by Shanghai Sangon Biotechnology Co., Ltd.) were as follows: MDR1 forward primer (CCCATCATTGCAATAGCAGG) and MDR1 reverse primer (GTTCAAACTTCTGCTCCTGA); GAPDH forward primer (ATGGGGAAGGTGAAGGTCG) and GAPDH reverse primer (GGGTCA TTGATGGCA-ACAATATC). In brief, 0.4 µM of the indicated primers and 10 µL of SYBR Premix Ex Taq II were added to a 5 µL aliquot of 1:10 diluted cDNA. The reaction conditions were as follows: 95°C for 15 min, 40 cycles of 95°C for 10 s, and 60°C for 10 s. Melting curves were used to analyze aliquots of the PCR products. MDR1 mRNA levels were normalized to GAPDH expression, and the amplification results for RT-qPCR were analyzed using the 2–∇∇Ct method. Each experiment was performed three times, and triplicate samples of each were tested.
Flow Cytometry Analysis for P-gp
MCF-7ADR cells were seeded into 6-well plates (5 × 105 cells/well) overnight and then treated with drugs at non-toxic concentrations. At different time point, cells were collected and washed with ice-cold PBS twice, then incubated at 4°C for 30 min with 500 µL of the FITC-labeled P-gp antibody (dilution: 1:100). Following incubation, cells were once again washed and resuspended in icy PBS before being promptly analyzed using flow cytometry (FCM).
Rh123 Accumulation and Efflux Assay
In the Rh123 accumulation assay, MCF-7ADR cells (5 × 105 cells/well) were cultured in 6-well plates overnight and then treated with 5.0 µg/mL of SSa, 7.0 µg/mL of SSb, and 5.0 µg/mL of Ver (a P-gp inhibitor), respectively. After pre-treatment for 48 h, each well added a new medium containing 5 g/mL of Rh123, which was then incubated for 0, 30, 60, 90, 120, and 150 min. At each time point, cells were gathered and subjected to FCM to determine the mean fluorescence intensity (MFI).
In the Rh123 efflux assay, MCF-7ADR cells (5 × 105 cells/well) were cultured in 6-well plates overnight and then treated with 5.0 µg/mL of SSa, 7.0 µg/mL of SSb, and 5.0 µg/mL of Ver, respectively. Each well received a 5 g/mL addition of Rh123 after a 48-h pre-treatment period, followed by a 120-min incubation period. Cells were resuspended in the media for 0, 5, 15, 30, and 60 min, after which they were collected, and MFI was determined by FCM.
Cell Cycle Analysis
Cells were seeded into 6-well plates (5 × 105/well) overnight and then treated with drugs for 48 hr. After treatment, cells were washed in PBS and resuspended in 70% ethanol at 4°C overnight for cell fixation. Before analysis, cells were washed once with PBS, suspended in 100 µL of RNaseA for 30 min, and then mixed with 400 µL of propidium iodide at 4°C for 30 min. Staining cells were measured using the FACScan flow cytometer from BD Biosciences in the United States, and the findings were analyzed using CellQuest software.
Statistical Analysis
The data were displayed as means ± SD. In SPSS 17.0, all statistical analyses were performed. For two groups, the two-tailed Student’s t test was used to determine statistical significance, and the one-way analysis of variance (ANOVA) was used for groups larger than two. A p-value < 0.05 was considered significant.
Results
Cytotoxicity of SSa and SSb in MCF-7 and MCF-7ADR Cells
To ascertain the cytotoxic effect of SSa and SSb on MCF-7 and MCF-7ADR cells, the different concentrations of SSa and SSb were added to cells for 48 h, and MTT detected cell growth. We discovered that following SSa and SSb treatment, the growth of MCF-7 and MCF-7ADR cells was inhibited in a dose-dependent manner (Figure 1A and B). The dose-effect curve showed that both SSa and SSb had obvious anticancer effects. The cell inhibition rate less than 10% was defined as the non-toxic concentration; SSa at 1.0, 2.5, and 5.0 µg/mL and SSb at 3.0, 5.0, and 7.0 µg/mL were used as the dose for reversing MDR.
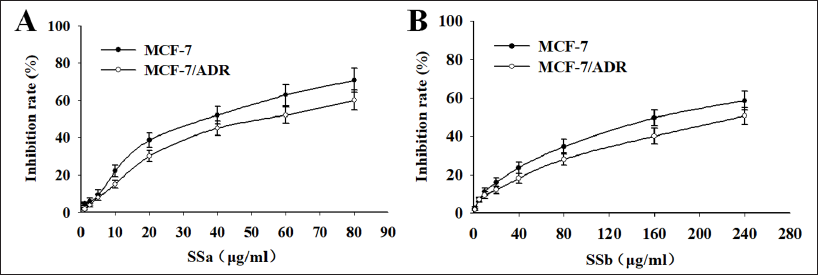
SSa and SSb Enhance Dox Sensitivity and Reverse MDR in MCF-7ADR Cells
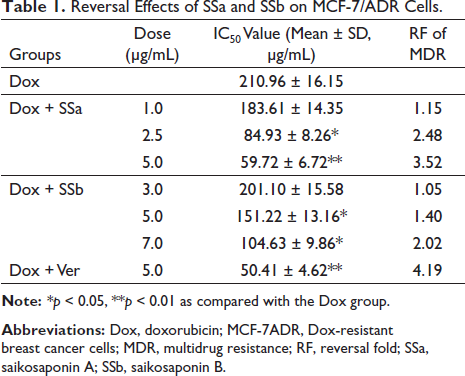
To determine whether exposure to SSa and SSb could restore the sensitivity of Dox and reverse MDR in MCF-7ADR cells, cells were treated with a broad range of concentrations of Dox in combination with non-toxic concentrations of SSa or SSb for 48 h. Ver at 5.0 µg/mL (a non-toxic concentration) was used as positive control. The data of Table 1 show that SSa at 2.5 µg/mL and 5.0 µg/mL significantly lowered the IC50 value of Dox from 210.96 ± 16.15 µg/mL to 84.93 ± 8.26 µg/mL and 59.72 ± 6.72 µg/mL (p < 0.05, p < 0.01), about a 2.48-fold and 3.52-fold reversal of MDR, respectively. Interestingly, SSb at 5.0 µg/mL and 7.0 µg/mL also significantly lowered the IC50 value of Dox (p < 0.05, p < 0.05), and the reverse folds were 1.40-fold and 2.02-fold, respectively. These findings demonstrate that SSa and SSb can enhance Dox sensitivity and reverse MDR in MCF-7ADR cells.
Reversal Effects of SSa and SSb on MCF-7/ADR Cells.
SSa and SSb Synergistically Sensitized to Dox in MCF-7ADR Cells
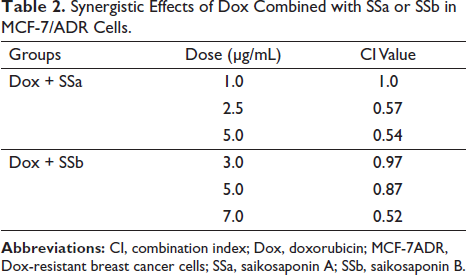
Using a CI, the effects of Dox and SSa or SSb in combination were assessed. As shown in Table 2, the CI values of SSa at 1.0, 2.5, and 5.0 µg/mL combining with Dox were 1.0, 0.57, and 0.54 µg/mL, the CI value of SSb at 3.0, 5.0, and 7.0 µg/mL mixing with Dox were 0.97, 0.87, and 0.52 µg/mL, respectively. These results indicate that SSa and SSb both have synergistic effects with Dox in MCF-7ADR cells.
Synergistic Effects of Dox Combined with SSa or SSb in MCF-7/ADR Cells.
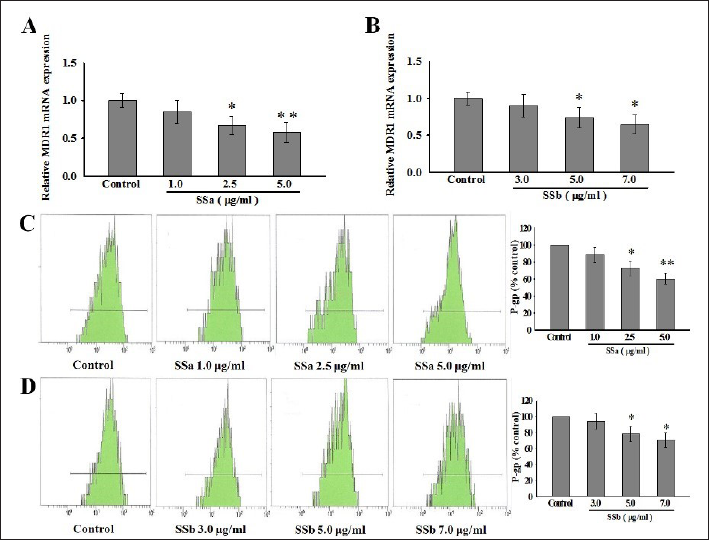
SSa and SSb Downregulated MDR1 mRNA and P-gp Expression
We next investigate the reversal mechanism of P-gp-mediated MDR by SSa and SSb. MCF-7ADR cells were treated with SSa and SSb for 48 h, and MDR1 mRNA and P-gp expression were evaluated by real-time PCR and FCM, respectively. We observed a dose-dependent reduction of MDR1 mRNA and P-gp expression after SSa and SSb treatment by comparing the results with the control group. MDR1 mRNA expression was significantly down-regulated by SSa at 2.5 and 5.0 µg/mL (p < 0.05, p < 0.01) (Figure 2A). Treatment of MCF-7ADR with SSb at 5.0 and 7.0 µg/mL also led to the downregulation of MDR1 expression (p < 0.05, p < 0.05) (Figure 2B). The P-gp levels were decreased in a good correlation with the decrease in MDR1 mRNA levels. SSa at 2.5 and 5.0 µg/mL significantly reduced P-gp expression by 27.5% and 39.8% (p < 0.05, p < 0.01; Figure 2C). According to Figure 2D, SSb at 5.0 and 7.0 µg/mL significantly decreased P-gp expression by 21.48% and 29.50% (p < 0.05, p < 0.05). According to these results, SSa and SSb downregulate the expression of P-gp and MDR1 mRNA in MCF-7ADR cells, increasing their sensitivity to Dox.
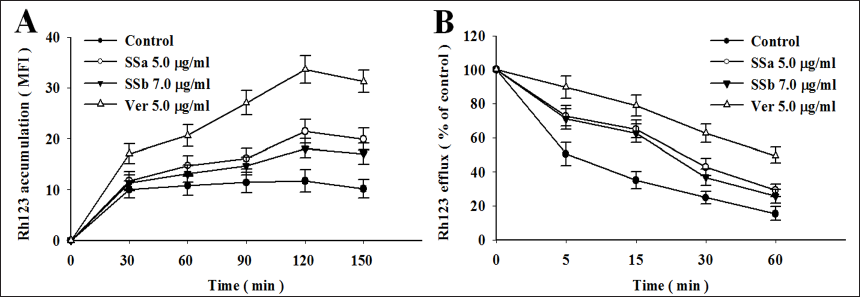
In MCF-7ADR cells, SSa and SSb boosted Rh123 accumulation while reducing its efflux. Using the P-gp substrate Rh123, it was examined whether SSa and SSb could prevent P-gp-mediated transport. Based on FCM, we detected Rh123 accumulation and efflux in MCF-7ADR cells exposed to SSa at 5.0 µg/mL and SSb at 7.0 µg/mL. Data showed that the accumulation levels of Rh123 in MCF-7ADR cells pre-incubated with SSa and SSb were much higher at each time point than in the control group (Figure 3A). In addition, SSa were more productive than SSb as shown in Figure 3A. MCF-7ADR cells pre-incubated with SSa and SSb significantly slowed down the rate of Rh123 release in the Rh123 efflux assay compared to the control group. The Rh123 efflux rate was reduced by 13.68%–29.92% after SSa pre-incubation and decreased by 10.26%–20.51% after SSb pre-incubation (Figure 3B). These findings demonstrate that SSa and SSb can restore intracellular Dox accumulation and decrease Dox efflux.
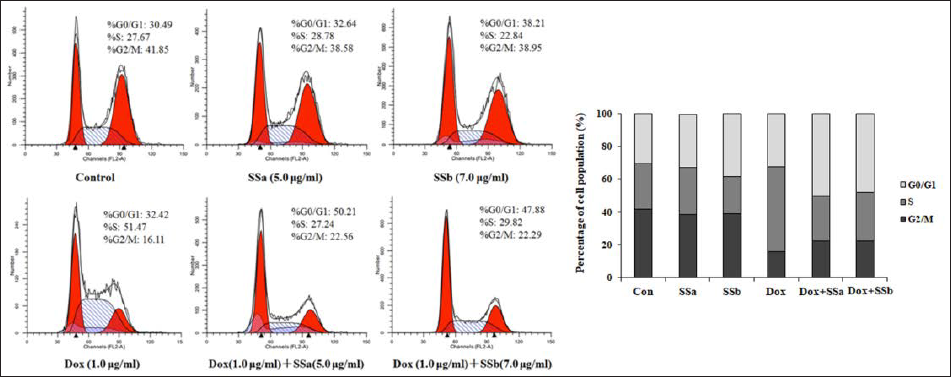
SSa and SSb-induced G1-Phase Cell Cycle Arrest in the Presence of Dox
MCF-7ADR cells were treated for 48 h with Dox (1.0 µg/mL), either alone or in combination with SSa (5.0 µg/mL) or SSb (7.0 µg/mL), to perform a cell cycle assay. As shown in Figure 4, treatment with Dox alone led to a more significant cell distribution in the S phase (from 27.6% to 51.47%) compared with the control group. Treatment with SSa or SSb alone did not result in cell accumulation at each stage of the cell cycle, whereas the combination with Dox altered the cell cycle profile in MCF-7ADR cells (Figure 4). The combination of SSa and Dox led to an obvious accumulation of cells in the G0/G1 phase (from 32.42% to 50.21%) with a concomitant decrease in the S phase (from 51.47% to 27.24%) compared with the Dox group. Additionally, SSb and Dox combination also enhanced the percentage of cells in the G0/G1 phase (from 32.42% to 47.88%) while decreased in the S phase (from 51.47% to 29.82%) compared with the Dox group. These data demonstrate that SSa and SSb may induce G1-phase arrest in MCF-7ADR cells in the presence of Dox.
Discussion
One of the most prevalent tumors in women, breast carcinoma is the main reason for cancer-related mortality globally. The main treatment plan for breast cancer has been thought to be adjuvant chemotherapy following surgery. However, the biggest issue impeding chemotherapy’s effectiveness is the emergence of intrinsic or acquired MDR (Germano & O’Driscoll, 2009; Kuo, 2007). To combat drug resistance, a combination therapy based on the synergistic interaction between chemotherapeutic agents and MDR reversal agents have been employed. Herbal medicines are valuable agents in treatment of tumors for their synergistic combinations with chemotherapy (Hu et al., 2016). Pharmacologists worldwide have made great progress by applying herbal medicines, particularly in cancer patients who display MDR (Li et al., 2014). In consideration of the side effects of chemotherapy drugs, synergistic effects may enhance efficacy, overcome MDR, and optimize safety.
Recently, many groups have reported several new compounds from herbal medicines that can reverse MDRin vitro and in vivo cancer models (Dai et al., 2007; Kwon et al., 2008; Liu et al., 2012; Liu et al., 2013). One of the main bioactive triterpene saponins that come from the root of BCDC is called SSa. It was reported that the administration of SSa could inhibit the proliferation of many tumor cells, including those in the colon, breast, and liver tumors (Kim & Hong, 2011; Wen-Sheng, 2003; Zhao et al., 2019). Therefore, SSa may be a medication with the potential to successfully treat tumors. However, SSb, the secondary saikosaponins, has not attracted much attention, despite being the component transformed from the major active saikosaponins (Lin et al., 2013). SSb is supposed to be the effective substance for BCDC decoction and has otential utility in combination with anticancer drugs (Ma et al., 2017). There has not been much research on the effects of SSa and SSb on the reversal of MDR yet. The MDR of MCF-7ADR cells in response to Dox was shown in the present work to be reversed by SSa and SSb, which could increase the cytotoxicity of anticancer medicines. The reversal effect of SSa and SSb is dose-dependent, as evidenced by the fact that the IC50 of Dox considerably decreased with the concentration of SSa and SSb when compared to the control group. We used 5.0 µg/ml SSa and 7.0 µg/ml SSb as the maximal non-toxic concentrations to perform our study in this manuscript. The reverse folds of MDR for SSa and SSb are 3.52 and 2.02, respectively. The MDR reversal effect of SSa appears to be more effective than SSb. However, further investigations into the underlying mechanisms are needed.
Combination drugs are one of the most widespread approaches to treating tumors. The scientific term CI was developed to describe the impact of drug combinations (Chou & Talalay, 1984). Our findings demonstrated that SSa, SSb, and Dox combinations were synergistic at non-toxic concentrations in MCF-7ADR cells.
MDR can have several causes, but one major cause of resistance to chemotherapy has been correlated with the presence of the over-expression of P-gp, an ATP-dependent membrane transporter protein encoded by the MDR1 gene (Aller et al., 2009). P-gp can efflux various xenobiotics out of cells by using ATP, which is not limited to anticancer drugs (Choi, 2005). Breast cancers, solid tumors, hematological malignancies, lymphomas, and other cancers have all been linked to P-gp overexpression (Leighton & Goldstein, 1995; Marie, 1995; Verrelle et al., 1991; Yagi et al., 2013). Therefore, in this study, we examined the P-gp expression of a protein related to drug resistance. Our research showed an obvious decrease in mRNA and protein levels of MDR1 with SSa and SSb treatment in MCF-7ADR cells, indicating that SSa and SSb enhanced Dox cytotoxicity by downregulating P-gp expression. Furthermore, while both SSa and SSb could reduce the mRNA and protein levels of MDR1, SSa appears to be much more effective than SSb. These results may partially explain why SSa has a better reversal effect on MDR than SSb.
The fluorescent dye Rh123 has been extensively used as an index to study the inhibition of P-gp function and expression in MDR cells (Perloff et al., 2003). In the accumulation study, MCF-7ADR cells treated with SSa and SSb could accumulate more Rh123 than untreated cells. The efflux experiment further showed that they significantly slowed down the rate at which Rh123 was released from MCF-7ADR cells. Moreover, Rh123 accumulation and efflux with SSa treatment appeared to be more significant than the effects of SSb treatment. As a result, it was further proven that SSa and SSb may inhibit the P-gp efflux pump through their ability to restore Rh123 accumulation and delay its efflux.
Cell cycle checkpoints are used to monitor and regulate the progress of the cell cycle. Altered cell cycle checkpoints are also described as one of the mechanisms of MDR (Kebsa et al., 2018; Wang et al., 2019). By using FCM, we measured the cell cycle arrest of Dox combined with SSa and SSb compared with Dox alone. When co-cultured with SSa and SSb, cell-cycle arrest in G1/G0 phase by Dox increased from 32.42% to 50.21% and from 32.42% to 47.88%, respectively. These findings suggested that the capacity of SSa and SSb to induce cell cycle arrest may play a role in their anti-MDR effects in cancer cells.
Conclusion
Our findings showed that SSa and SSb dramatically increased the chemosensitivity of MCF-7ADR cells to ADR and downregulated MDR1/P-gp expression. SSa and SSb enhanced the sensitivity to ADR, for which cell cycle arrest was also an important determinant. Therefore, SSa and SSb may be novel MDR reversal medicines for chemotherapy for breast cancer and are of significant clinical significance for MDR during tumor therapy.
Footnotes
Summary
SSa and SSb are potentially new MDR reversal drugs for chemotherapy for breast cancer and are highly clinically significant for MDR during tumor therapy.
Abbreviations
MDR: multidrug resistance; Pgp: P-glycoprotein; BCDC: Bupleurum chinense DC: SSa, saikosaponin A; SSb: saikosaponin B; Dox: doxorubicin; MCF-7ADR: Dox-resistant breast cancer cells; MCF: 7 breast cancer cells; Rh123: rhodamine 123; ATP: adenosine triphosphate; ABC: ATP-binding cassette; Ver: Verapamil; DMSO: dimethyl sulphoxide; OD: optical density; RF: reversal fold; CI: combination index; FCM: flow cytometry; FITC: fluorescein isothiocyanate; MFI: mean fluorescence intensity.
Acknowledgments
We thank Professor Chun Li from the College of Basic Medicine and Professor Chunyan Yu from the College of Basic Medicine for their critical comments on the manuscript and Duochun Ji for his English editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by the Jilin Provincial Department of Science and Technology (Grant no. 20190201220JC, YDZJ202101ZYTS089, YDZJ202101ZYTS090, and YDZJ20230-1ZYTS112), Jilin Provincial Health, and the Family Planning Commission (Grant no. 2021JC084).
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
