Abstract
Background
The aggregation of tau hyperphosphorylation (p-tau) into neurofibrillary tangles (NFT) is a hallmark in the histopathology of Alzheimer’s disease (AD). Our previous experiments found that β-asarone could prevent injury of PC12 cells induced by A 1–42, but could it fight cell damage of p-tau induced by okadaic acid (OA) is poorly understood.
Objectives
The emphasis of this study lies in β-asarone’s therapeutical effect on p-tau inhibition stimulated by OA.
Materials and Methods
175 nmol OA was used to establish AD cells. Cell viability rate and cell toxicity were evaluated by the CCK-8 kit and LDH kit, respectively. The p-tau, Aβ42, β-secretase, and protein phosphatase 2A (PP2A) were examined by ELISA. Proteins closely related to the pathogenesis of AD are involved p-tau, Beclin-1, p-Akt, and p-mTOR were analyzed by western-blotting and immunofluorescence detection.
Results
The results revealed that β-asarone enhanced cell viability induced by OA in a dose-dependent manner. Moreover, compared to the OA model, p-tau, Aβ42, β-secretase, and Beclin-1 were reduced, while PP2A, p-Akt, and p-mTOR increased after treatment with β-asarone.
Conclusion
All data suggested that β-asarone decreased p-tau, Aβ42, and β-secretase levels, and activated PP2A levels by inhibiting Beclin-1-dependent autophagy in OA model cells, involving Akt/mTOR/Beclin-1 pathway.
Introduction
Alzheimer’s disease (AD) is becoming increasingly global in the elderly and is one of the most common causes of dementia (Mangialasche et al., 2010). The recognized pathologic features of AD are widely described: deposition of abnormal proteins in nerve cells, involving senile plaques, amyloid β (Aβ), and the aggregation of tau hyperphosphorylation (p-tau) (Lane et al., 2018). Aβ1–40 and Aβ1–42 are common intracellular Aβ, produced by the β-secretase decomposition of amyloid precursor protein (APP) (Imbimbo & Watling, 2019). Although the cause of AD remains unknown, autophagy is regarded as a central player in AD (Salminen et al., 2013). Autophagy is the primary mode of clearance for reducing abnormally aggregated proteins, and increasing autophagy is a reasonable therapeutic strategy to contract the relative overproduction of neurotoxic Aβ (Li et al., 2017). Studies confirm a dual role for autophagy in the extracellular transport of Aβ.
β-asarone is a natural volatile oil, which is extracted from Acorus tatarinowii Schott. Our previous research found that the learning and memory abilities of APP/PS1 transgenic mice were improved after β-asarone administration, and the mechanism may be related to the inhibition of autophagy in APP/PS1 transgenic mice through the regulation of PI3K/Akt/mTOR pathway (Deng et al., 2016). β-asarone ameliorates the learning behavior ability of Aβ-induced AD rats by modulating the PINK1-Parkin-mediated mitophagy (Han et al., 2020). β-asarone against oxidative stress and neuronal damage in rats induced by Aβ (Saki et al., 2020). Its anti-Aβ neurotoxic effects were manifested as improved learning memory in APP/PS1 mice, increased expression of SYP and GluR1 (Liu et al., 2016), promoted autophagy in AD cells (Wang et al., 2019), and attenuated autophagy by activating the Akt-mTOR pathway (Xue et al., 2014). However, the effects of β-asarone on resisting its induction p-tau by okadaic acid (OA) in cells were still unknown.
To elucidate the therapeutic effect of β-asarone on AD further, this experiment was carried out using OA on AD. OA can lead to p-tau emerging by suppressing the activity of protein phosphatase 2A (PP2A) (Ma et al., 2018). Here, our initial experiments suggest that β-asarone was capable to alleviate the cytotoxicity caused by OA. In the meantime, we adopted an autophagy activator-rapamycin and an autophagy inhibitor 3-methyladenine (3MA) in the follow-up experiment. Here we assume that β-asarone was able to suppress autophagy of OA-induced PC12 cells and this process should occur by regulating an Akt/mTOR/Beclin-1 pathway.
Materials and Methods
Drugs and Reagents
β-asarone was extracted from a commonly used Chinese medicine A. tatarinowii Schott as reported previously (Fang et al., 2012). Its purity was found to be as high as 99.55% by the three spectral purity identification, including gas chromatography-mass spectrometry (GC-MS), infrared spectrum (IR), and nuclear magnetic resonance (NMR) detection. OA, 3MA, and Rapamycin were purchased by the following three companies respectively: Adamas Reagent Co. Ltd (cat. no. P112508); Sigma (USA; cat. no. M9281); Sigma (USA; cat. no. K0103). DMEM (cat. no. 12800017), PBS (cat. no. C10010500BT), and FBS (cat. no. 10099141) were obtained from Gibco (Thermo Fisher Scientific, Inc., Waltham, MA, USA). Cell Counting Kit-8 (CCK-8) was obtained from Dojindo Molecular Technologies, Inc. (Tokyo, Japan). Cytotoxicity LDH Assay Kit-WST (LDH) was supplied by Nanjing Jiancheng Bioengineering Institute (Nanjing, China). Anti-P-akt, Anti-P-mTOR, Anti-Beclin-1, Anti-p-tau, and Anti-GAPDH were obtained from Abcam (Cambridge, UK).
Cell Culture and Experimental Design
The highly differentiated PC12 cells used in this study were provided by the Chinese Academy of Sciences in Shanghai (Shanghai, China). The cells were cultivated in DMEM containing 10% FBS and 1% penicillin/streptomycin at 37°C in a humidified 5% CO2 atmosphere. Change the culture medium daily. We adopted 10 µg/mL OA cultured PC12 cells for 48 h under the same conditions (DMEM containing 10% FBS and 1% penicillin/streptomycin at 37°C in a humidified 5% CO2 atmosphere) to set up the AD cell model as before (Huang et al., 2019). To further substantiate the role of β-asarone in AD treatment, PC12 cells were divided into 8 treated groups: normal control group, model group (175 nmol OA), 4 different doses (0.25, 0.5, 1, and 2 µg/mL) of β-asarone treated groups, autophagy inhibitor (5 nmol 3MA) group and autophagy activator (100 nmol rapamycin) group. Cells in all groups except the normal control group were incubated in a medium containing 175 nmol OA and then incubated again with β-asarone, 3MA, or rapamycin, respectively.
Cell Viability and Cytotoxicity
Cell viability and cytotoxicity of PC12 in this study were determined according to the instructions of CCK-8 (Dojindo Molecular Technologies, Inc., Japan) and LDH kit (Nanjing Jiancheng Bioengineering Institute, China), respectively. For determining the OA concentration and action time for AD model cells, the PC12 cells with different concentrations of OA (0, 100, 150, 200, 250, 300, 350, 400, 450, and 500 nmol) were treated at a density of 5 × 103 cells/well in 96-well plates and were cultured at 37°C in an atmosphere containing 5% CO2 for 12, 24, and 48 h. In addition, the PC12 cell groups treated with 175 nmol OA plus β-asarone (0.25, 0.50, 1, and 2 µg/mL) were seeded at a density of 5 × 103 cells/well in 96-well plates and cultured at the same cell culture conditions. CCK8 was used to detect cell viability as follows: 100 µ L of serum-free DMEM (including 10% CCK8) was added and the cells were incubated for 2 h at the above cell culture conditions. The absorbance (OD) values were used to measure cell viability. Meanwhile, LDH is used to evaluate cytotoxicity as follows: PC12 cells were inoculated into a 96-well cell culture plate with a number of 5 × 103 cells/well, and then cultured for 24 h. After treatment with OA at different concentrations (12–48 h), the LDH activity released in each group of cell media was determined according to the experimental steps in the manual.
Elisa Analysis
The PC12 cells were grouped as follows: normal control group, model group, 0.25 µg/mL β-asarone group, 0.5 µg/mL β-asarone group, and 1 µg/mL β-asarone group. The cell supernatant of each treatment group was collected and the contents of p-tau, Aβ42, β-secretase, and PP2A in the cell supernatant were detected according to the corresponding ELISA kit instructions (Hermes Criterion Biotechnology; Elixir Canada Medicine Company Ltd.).
Western Blot Analysis
The cells are grouped as follows: control group (only medium), model group (175 nmol OA), 3MA group (5 nmol 3MA), rapamycin group (100 nmol rapamycin), and β-asarone group (1 µg/mL β-asarone + 175 nmol OA). The cells were collected and lysed with 100 nmol/L phenylmethanesulfonyl fluoride lysis buffer (Sigma-Aldrich; Merck KGaA) and the total proteins were extracted according to the instructions of the protein extraction kit (Wuhan Boster Biological Technology, Ltd., Wuhan, China). 40 µg protein was collected, separated by 10% SDS-PAGE, and transferred to the PVDF membrane. Then, the designated primary antibodies against GAPDH (cat. no. ab8245; Abcam), p-Akt (cat. no. 9275S; Cell Signaling Technology, Inc., Danvers, MA, USA), p-mTOR (cat. no. ab109268; Abcam), and Beclin-1 (cat. no. ab62557; Abcam) were used to incubating the membrane overnight at 4°C (all 1:1,000). The membrane was washed 3 times and incubated for 2 h with the appropriate secondary antibodies (1:2,000; cat nos. 7076S and 7074S, respectively; Cell Signaling Technology, Inc.). The immune complexes were visualized with a chemiluminescence detection system (Bio-Rad Laboratories).
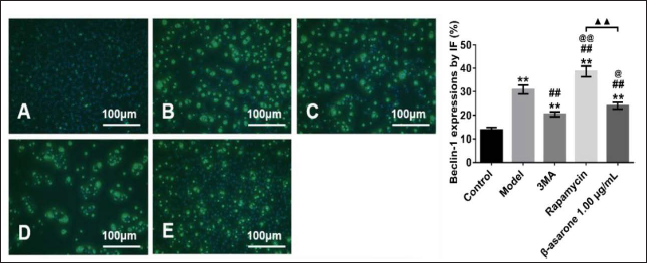
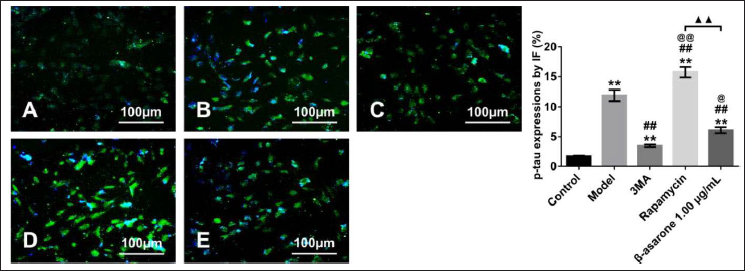
Immunofluorescence Analysis
PC12 cell grouping is the same as western blot analysis preprocessing. Collected cells and fixed with 4% paraformaldehyde for 60 min and then washed cells with PBS and sealed with 5% BSA for 20 min at 37°C. Cells were then incubated with rabbit anti-Beclin-1 and anti-p-tau antibodies (1:50 dilution; Abcam) for 1 h at 37°C, cultured using Alexa 488-conjugated anti-rabbit IgG (1:200; cat no. 4412S; Cell Signaling Technology, Inc.) for 30 min at 37°C and DAPI was re-dyed for 5 min to avoid light, observed and photographed under an inverted microscope (U-SPT; Olympus, Tokyo, Japan). Data were analyzed by MIAS.
Ethical Statement
This manuscript is a cell experiment, does not involve animal ethics.
Statistical Analysis
The data analysis was performed with SPSS 13.0 (SPSS, Inc., Chicago, IL, USA). The significance of differences between groups was used one-way analysis of variance with a Bonferroni post hoc test. Values are expressed as the means ± standard deviation and a p < 0.05 represents a statistically significant difference.
Results
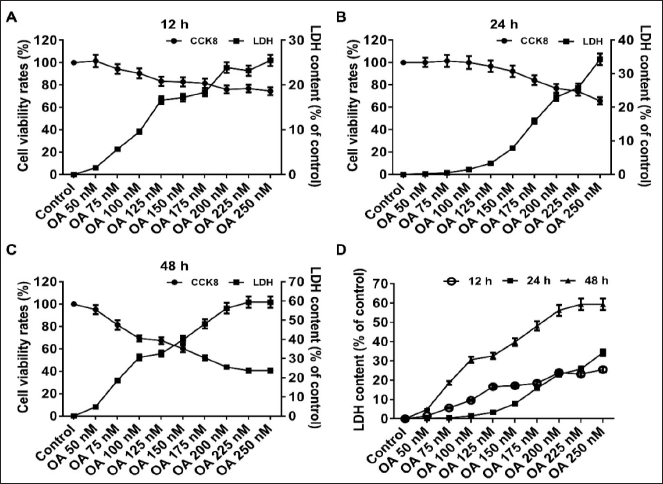
Effects of Different Concentrations and Different Times of OA on PC12 Cells
In this study, OA was used to construct AD cells. With the increase of OA concentration and duration of action, AD cell cytotoxicity LDH increased in a dose-effect manner; meanwhile, the cell viability of PC12 cells decreased as shown in Figures 1A–D. During the 12 h, the cell activity decreased sharply at 0–250 nmol OA, decreased slowly at 250–400 nmol OA, and remained flat at 400–500 nmol OA; however, the trend in the cytotoxicity LDH was just the opposite (Figure 1A). During the 24 h, the cell activity kept stable at 0–250 nmol OA and then decreased dramatically at 250–500 nmol OA; and the trend in the cytotoxicity LDH was going in the opposite direction (Figure 1B). During the 48 h, the cell activity showed a precipitous decline at 0–500 nmol OA; meanwhile, the trend in the cytotoxicity LDH presented rapidly rising (Figure 1C). We found that the LDH contents were not significantly different during 12 h and 24 h, while that of 48 h was significantly higher than that of the other time points (Figure 1D). We extrapolated IC50 with an SPSS calculator and chose 175 nmol at 48 h as the experiment condition.
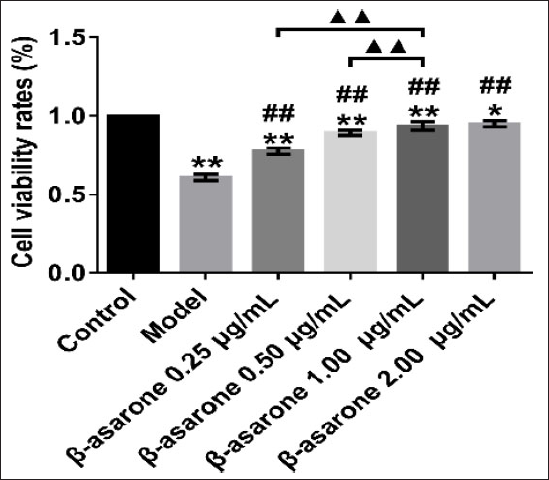
β-asarone Suppressed Cell Injury Induced by OA
To investigate the anti-OA toxicity of the β-asarone, different concentration gradients of β-asarone (0.25, 0.5, 1.00, and 2.00 µg/mL) were added to the OA cell for 48 h and then the cell supernatant was collected for CCK-8 detection. We found that β-asarone can resist the toxicity of OA, promote the growth of cells and it’s dose-dependent. The viability of OA-induced PC12 cells was higher after the administration of β-asarone compared with the model cell (p < 0.01). This result inhibits that β-asarone may alleviate cellular damage caused by OA (Figure 2).
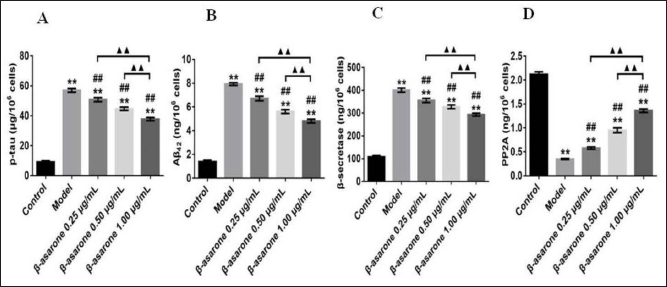
Effects of β-asarone on Abnormal Proteins and Related Enzymes
To further clarify β-asarone’s function in removing the abnormal proteins, we measured the levels of abnormal proteins and related enzymes in the OA cells. Significantly increased β-secretase, p-tau, and Aβ42 contents were observed in AD cells compared with those in the normal cell (p < 0.01). Conversely, significantly decreased β-secretase, p-tau, and Aβ42 levels were observed in β-asarone intervention groups compared with those in AD cells (p < 0.01). Moreover, a significantly increased in PP2A was observed between β-asarone-treated groups and the model cell (p < 0.01). The results revealed that the decrease of Aβ42 and p-tau may be related to the decrease of β-secretase or the increase of PP2A after β-asarone intervention in OA cells (Figure 3).
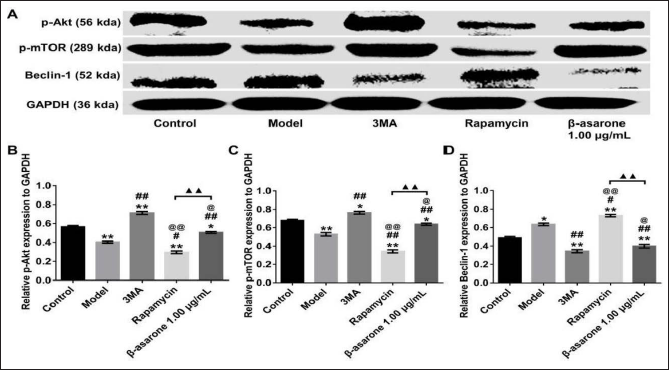
β-asarone Promoted the Expression of p-Akt and p-mTOR While it Inhibited Beclin-1
To explore the possible mechanism of its anti-OA toxicity, we set up the autophagy-activated group (100 nmol rapamycin) and the autophagy-inhibited group (5 nmol 3MA). Experimental results show that p-Akt or p-mTOR proteins both decreased, whereas Beclin-1 expression was enhanced in AD cells compared to normal cells (p < 0.05). Notably increased of p-Akt and p-mTOR and lessened Beclin-1 were observed in the 3-MA or β-asarone intervention groups compared to AD cells (p < 0.05). However, an opposite trend of p-Akt, p-mTOR, and Beclin-1 was discovered in the rapamycin or β-asarone intervention groups compared with those in the 3MA intervention group (p < 0.05). In addition, a significant increase of p-Akt and p-mTOR but a significant decrease of Beclin-1 were observed in the β-asarone intervention group compared with those in the rapamycin intervention group (p < 0.05). We could declare from these consequences that β-asarone could suppress Beclin-1-dependent autophagy by regulating Akt/mTOR pathway (Figure 4).
β-asarone Inhibited by Decreasing p-tau and Beclin-1 Expression
In the AD model cells, we observed that autophagy was suppressed. Moreover, the immunofluorescence result found that β-asarone inhibited Beclin-1-dependent autophagy (Figure 5), which was consistent with the western blot result (Figure 4). In addition, a similar trend was observed in p-tau expression after the administration of β-asarone compared with the model cells (p < 0.05) (Figure 6). This result suggested that β-asarone could remove p-tau from OA-induced cells by inhibiting Beclin-1-interrelated autophagy.
Discussion
AD is a progressive and fatal neurodegenerative disorder. Currently, the pathogenesis of AD involves several hypotheses, mainly involving Aβ toxicity, oxidative stress, and abnormal p-tau accumulation (Silva et al., 2019). The aggregation of p-tau into neurofibrillary tangles (NFT) is a hallmark in the histopathology of AD. Despite extensive research into AD, no treatment has been able to reverse its progression. Therefore, we need suitable models to study the pathogenesis of AD and to find effective treatments. Depending on the purpose of the study, the cell model of AD mainly adopts Aβ1–42 or Aβ25–35 to induce neurotoxic injury of neurons (Malishev et al., 2019), Hydrogen peroxide is used to study oxidative stress injury (Tabner et al., 2002), and OA can be used to prepare AD cell models with p-tau (Jiang et al., 2016).
Numerous studies have shown that p-tau plays a crucial role in the pathogenesis of AD and that a reduction in tau protein, Aβ or β-secretase may contribute to recovery from AD (Foidl & Humpel, 2018; Fu et al., 2017; Gao et al., 2018). OA, an inhibitor of protein phosphatase PP2A, induces excessive accumulation of p-tau, mimicking abnormal phosphorylation changes of tau protein, which is consistent with AD pathology (Rami, 2009). In this study, different concentrations of OA (50, 75, 100, 125, 150, 175, 200, 225, and 250 nmol) treatment were used to establish the AD model in PC12 cells. Cell viability or toxicity was evaluated by detecting supernatant. The results showed that 175 nmol OA had obvious toxicity on PC12 cells and significantly reduced the survival rate of cells.
β-asarone is the most abundant component in the volatile oil of A. tatarinowii and can be quickly distributed into the brain rapidly. A growing number of studies have shown the pharmacological effects of β-asarone in a variety of diseases, including neurodegenerative and cerebrovascular diseases. Preliminary studies have revealed that β-asarone has a bidirectional autophagic effect on PC12 cells induced by Aβ1–42, manifesting as inhibition and activation of autophagy (Ma et al., 2018; Xue et al., 2014). These results suggest that the possible mechanisms of β-asarone and autophagy in anti-AD neuro cytotoxicity have not been evaluated. Our research group is dedicated to the study of the efficacy and mechanism of β-asarone against AD. Previous in vivo experiments revealed that β-asarone has significant anti-AD activity, the mechanism of which was mainly related to autophagy, but the experimental subject was Aβ42-induced AD cell model. However, the etiology of AD is complex, and single-cell models are not sufficient to explain it. Therefore, this study was complemented with an OA-induced cellular model to improve the persuasive effect of β-asarone on AD. Subgroups were designed with different concentrations of β-asarone intervention.
Cell proliferation assays showed that β-asarone increased OA-induced cell viability in a dose-dependent manner. Furthermore, protein phosphatase PP2A catalyzes the dephosphorylation of tau protein and plays a key role in the inhibition of p-tau in the brain of AD patients. Our current research demonstrates that β-asarone inhibits p-tau production, which is closely associated with PP2A after OA intervention. Although there is evidence that β-asarone may affect p-tau, Aβ, PP2A, or β-secretase levels, whether it may be an effective scavenger of abnormal proteins remains to be further tested. Our results suggest that β-asarone can inhibit the deposition of Aβ42 and p-tau proteins in the OA-induced AD cell model.
Consequently, this experiment may provide some research basis for the treatment of AD with β-asarone. The main role of autophagy is to remove intracellular abnormal proteins and impaired organelles. Research has shown that autophagy is closely related to the etiopathogenesis of AD (Hamano et al., 2018). Beclin-1 associated with autophagy is the promoter of autophagy (Xu & Qin, 2019). As one of the important markers of autophagy, Beclin-1 is now usually used to monitor autophagy (Liang et al., 1999). Here we still used Beclin-1 as an indicator to detect autophagy in OA-induced PC12 cells. In addition, the PI3K-Akt-mTOR pathway regulates the autophagy of non-mammalian cells (Xue et al., 2017). The autophagy process is closely associated with the progress of AD and involves the PI3K-Akt-mTOR pathway (Heras-Sandoval et al., 2014). The statistical significance of this study postulates a strong correlation between Beclin-1 and the PI3K-Akt-mTOR signaling pathway. Our experimental results reveal that β-asarone can inhibit the production of Aβ42 and p-tau by β-secretase or PP2A in OA-intervened cells, this mechanism may involve Akt/mTOR/Beclin-1 pathway regulation. These results suggest that the Akt/mTOR/Beclin-1 pathway modulated autophagy in OA-interfering cells after β-asarone treatment and is involved in the clearance of abnormal proteins, providing a new mechanism for the treatment of AD.
Conclusion
From the above discussion, it is clear that β-asarone activates PP2A via the Akt/mTOR/Beclin-1 pathway, thereby removing p-tau protein from OA cells. β-asarone promoted Akt and mTOR phosphorylation and inhibited Beclin-1-related autophagy in OA cells. In future studies, we will use Akt and mTOR pathway inhibitors as positive controls to further clarify the efficacy and related mechanisms of β-asarone on p-tau-induced AD animal models, providing an experimental basis for β-asarone as a new clinical drug for AD treatment.
Footnotes
Summary
OA can lead to p-tau, the enhancement of β-secretase activity, and the aggregation of β-amyloid protein in PC12 cells, inducing cell damage. β-asarone, an active component of Acorus tatarus, can improve cell viability after OA intervention and play its neuroprotective role by regulating Akt/mTOR/Beclin-1 Pathway, involving β-asarone could be decreased p-tau, Aβ42, β-secretase levels, and activated PP2A level.
Abbreviations
Aβ: Amyloid β; APP: Amyloid precursor protein; AD: Alzheimer’s disease; GC-MS: Gas chromatography-mass spectrometry; IR: Infrared spectrum; 3MA: 3-methyladenine; NMR: Nuclear magnetic resonance; NFT: Neurofibrillary tangles; OA: Okadaic acid; PP2A: Protein phosphatase 2A; p-tau: The aggregation of tau hyperphosphorylation.
Acknowledgments
We thank all the researchers from the Guangzhou University of Chinese Medicine for their assistance in completing this experiment.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was financed by the Lingnan Normal University-level talent project (grant no. ZL1801); The Natural Science Foundation of Guangdong province of China (grant no. 2018A030307037); The National Natural Science Foundation of China (grant nos. 31670363, 81904104 and 31900297); The Science and Technology Planning Project of Guangdong Province, China (grant no. 2014A020221057); The key discipline research project of Guangdong Province (2019 GDXK-0025 and 2021ZDJS035); The Yanling excellent young teacher program of Lingnan Normal University (YL20200210) and National Training Program of Innovation and Entrepreneurship for Undergraduates (202010579006); The foundation of Education Bureau of Guangdong Province (2015KTSCX087); The Postdoctoral Foundation of China, (2021M690759) and The Administration of Traditional Chinese Medicine of Guangdong Province, China (20211203), the Open Project of Mangrove Institute, Lingnan Normal University (grant no. YBXM01); the Open Project of Western Guangdong Characteristic Biology and Medicine Engineering and Research Center (grant no. 2022K03).
Statement of Informed Consent and Ethical Approval
Not applicable.
